Abstract
In this review we discuss the analytical inadequacies of oestradiol assays in relation to the clinical requirements for performing them, and make recommendations for their improvement. The measurement of oestradiol can be requested in a number of clinical scenarios (precocious puberty, infertility, assisted conception, hormone replacement therapy). The very wide dynamic range of oestradiol concentrations is a huge challenge for routine assays, which they are unlikely to meet on theoretical as well as practical grounds. The EQA performance of oestradiol assays in terms of trueness, comparability, recovery and analytical sensitivity leaves much to be desired and indicates that calibration is compromised by poor analytical specificity. To make oestradiol assays fit for purpose requires concerted action by all stakeholders to define analytical quality specifications for the various clinical scenarios involved, and then to encourage concerted action by the diagnostic industry to use the steroid reference measurement system to improve specificity, trueness and traceability.
Introduction
Oestradiol physiology in health and disease
Oestradiol production and regulation at the key stages of life
17β Oestradiol is a C18 steroid derived from cholesterol and is the oestrogen present in the highest concentration in the serum of premenopausal women (Figure 1). The main site of oestradiol synthesis in premenopausal women is the ovary. However, throughout life the ovaries and adrenals secrete various precursor steroids (i.e. androstenedione, testosterone, dihydroepiandrosterone) that can be converted to oestradiol and other oestrogens by aromatization in the peripheral tissues (e.g. adipose tissue. brain and bone). 1 In men, direct secretion of oestradiol by the testes accounts for about 20% of circulating hormone, the remainder being derived from peripheral aromatization by androgens. 2 Serum oestradiol concentrations are generally low (<100 pmol/L) in both sexes during infancy although, as stated later in this article, this can only be an estimate due to the inability of many oestradiol assays to accurately measure the hormone at these low concentrations. Concentrations rise in women during puberty reflecting increased ovarian activity. Smaller increases are also seen in men due to the increased conversion of testosterone by peripheral tissues. 1,3 In women, oestradiol production varies during normal menstrual cycles with the highest serum concentrations (typically about 1000 pmol/L) being preovulatory with a second peak in the luteal phase due to secretion by the corpus luteum. In postmenopausal women, oestradiol concentrations are low (typically <100 pmol/L) with the primary oestrogen in the plasma being oestrone produced from the conversion of androstenedione in adipose tissue. 1,4 In men, oestradiol concentration in plasma is typically <200 pmol/L. Oestrone is also present in the serum of men at a slightly higher concentration than oestradiol. 2
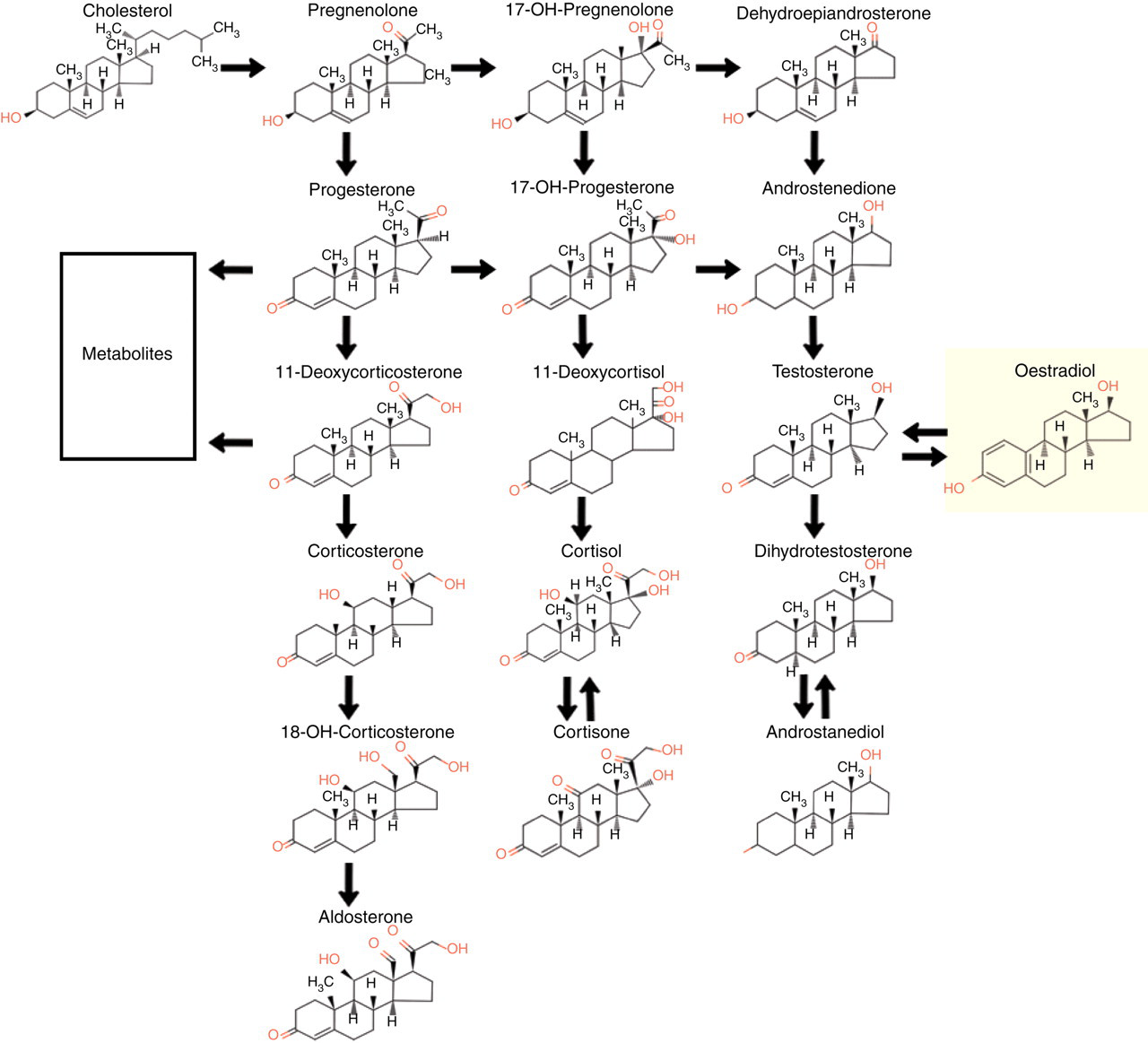
Synthetic pathway for oestradiol (from
Secretion of oestradiol is under the control of the trophic hormones, luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which are secreted in a pulsatile fashion under the influence of gonadotrophin releasing hormone (GnRH) from the hypothalamus. The regulation of serum oestradiol concentrations is primarily by negative feedback on gonadotrophin release from the pituitary, although gonadotrophin release is also controlled by a positive feedback mechanism in the late stage of the follicular phase of the menstrual cycle.
‘Bound’ and ‘free’ oestradiol in the circulation
Over 97% of oestradiol in the serum is bound reversibly to either sex hormone binding globulin (SHBG) in a high affinity reaction or with lower affinity to albumin. Only 1–3% of plasma oestradiol is in the non-protein bound (or free) state. It has been considered that the free hormone is the metabolically active fraction able to diffuse across cell membranes and interact with receptors and exert a feedback effect. However for many years it has been suggested that the albumin and even the SHBG bound fraction may also be biologically active. 5,6
Signal transduction and end organ effects
In women the major action of oestradiol is on the reproductive tract. It promotes development of the female reproductive organs and stimulates the growth of the epithelial lining of the uterus in the follicular phase of the menstrual cycle. Oestradiol also has other wide range of roles not related to reproduction. These include effects on the bone, skin and muscle, liver, brain, vascular function and lipid metabolism. Good reviews of the various functions of oestradiol have been written by Ruggerio et al. 4 and Gruber et al. 1 The physiological role of oestrogens in men is less clear. A major role that has been proposed for oestrogens in men is that they are required for bone maturation. Men with oestrogen deficiency have osteopenia with low bone density, increased bone turnover and delayed epiphyseal closure. 2 Men with oestrogen deficiency also have increased serum total cholesterol and low-density lipoprotein cholesterol and reduced high-density lipoprotein cholesterol. 2 In a recent article, Simpson et al. reviewed the concept of ‘local oestrogen biosynthesis’ where they suggested that in men and postmenopausal women, oestrogens do not function as circulating hormones but have a paracrine function at the local level. Circulating oestrogen concentrations in these groups simply reflect metabolism of oestrogens at peripheral sites. 7
The effect oestradiol has on the cell depends on the response that occurs after binding to the oestrogen receptor within the target cell. The exact location of oestrogen receptors within the cell is unknown. It is thought that they are in equilibrium between the cytoplasm and the nucleus. Binding of oestradiol to the receptor releases the receptor from its receptor-associated proteins and allows the diffusion of the complex to the cell nucleus. The complex then binds to specific estrogen response elements on the DNA, thereby modifying protein synthesis within the cell. 1 Two types of oestrogen receptor (ERα and ERβ) have been described in both men and women. These two receptor types are present in many organs of the body with a complex pattern of distribution. A major area of research in the pharmaceutical industry is in designing synthetic oestrogen like molecules (selective oestrogen receptor modulators – SERMs), which can bind to either of these receptors and cause agonistic or antagonistic effects. 8,9
Metabolism, conjugation and excretion
Oestradiol is metabolized into a variety of less potent or inactive compounds by a number of different mechanisms:
Interconversion of oestradiol and oestrone by 17β hydroxysteroid dehydrogenase; Sulphation or glucuronidation; Hydroxylation and subsequent methylation.
The water soluble metabolites are excreted in the bile or urine. Subsequent reabsorption of these oestrogens after hydrolysis by intestinal flora results in an enterohepatic circulation. An important metabolite of oestradiol is oestrone sulphate, which due to its long half-life in blood is a major oestrogen component in the blood of men and women.
2,10
Oestradiol measurement
History
Oestradiol is a low molecular weight, non-immunogenic molecule, present in very low concentrations in association with very similar metabolites. These factors render this analyte one of the most difficult to measure by immunoassay in the laboratory repertoire, yet it is available on all the major assay platforms as a widely performed ‘routine’ assay and its analytical problems seem not to be high priority. There is little evidence for improvement in UK NEQAS performance over the years.
In the early days of oestrogen quantitative estimation (oestradiol was not distinguished at that time), bioassay was the only method available, but this was replaced in the 1950s and 1960s with colorimetric and fluorimetric assays, and then gas liquid chromatography (GLC) and high performance liquid chromatography (HPLC) techniques, which enabled the various oestrogens to be quantitated separately. 11 Radioimmunoassays (RIA) became available as in-house systems in the early 1970s, with commercial assays following quite soon after. Other types of ligand-binding assay and even non-competitive format ‘idiometric’ assays 12 have been investigated, but none of these has been a serious contender to the basic competitive, limited reagent, labelled antigen, assay format.
Because of the nature of the oestradiol molecule, antibody specificity is crucial for the development of assays with sufficient analytical sensitivity at picomolar concentrations. To circumvent lack of specificity, solvent extraction has been successfully employed for 30 years, with some in-house extraction assays being capable of exquisite (<10 pmol/L) analytical sensitivity. 13 In past UK NEQAS for Steroid Hormones reference method exercises, the few remaining in-house extraction RIA users have often performed better than market-leading immunoassay platforms in terms of comparability (see Figure 2) and recovery. Only three laboratories in the UK maintain these assays.
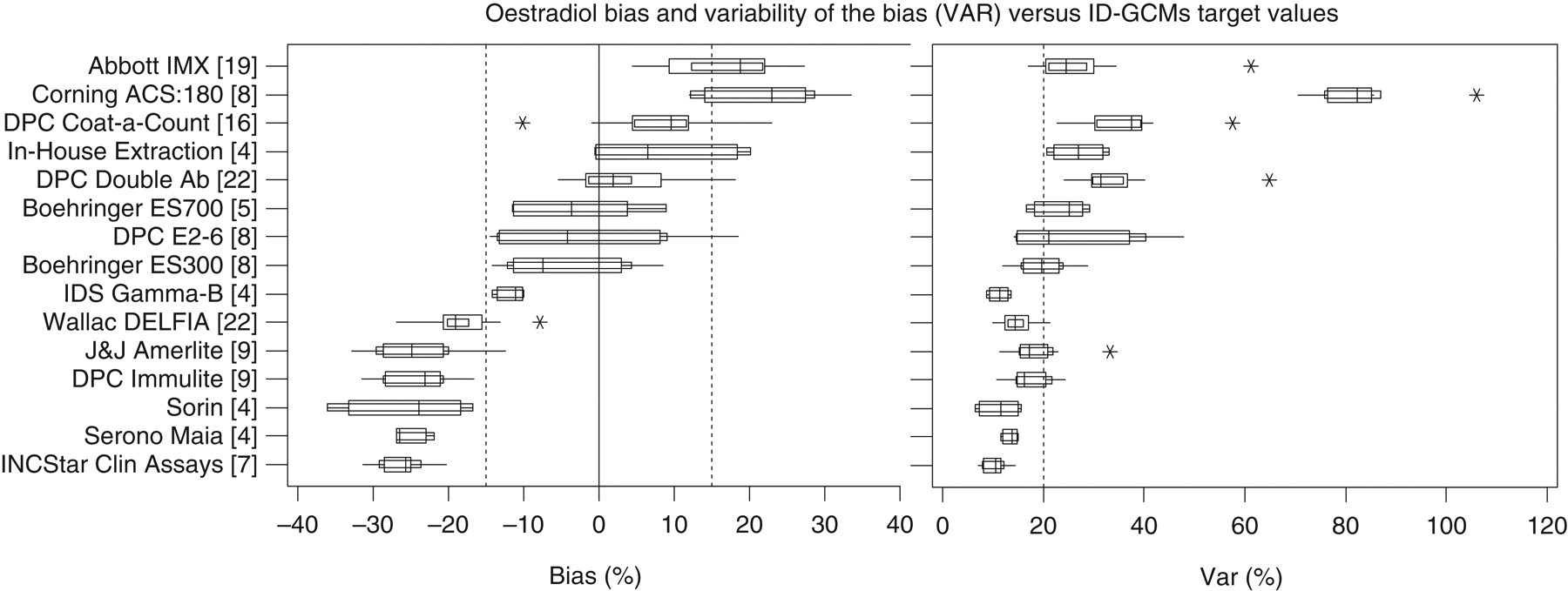
1995 ID-GCMS (isotope dilution gas-chromatography, mass spectrometry) exercise. Historical data extracted from the UK NEQAS report of September 1995, showing the relationship between UK NEQAS six-month cumulative bias (equivalent to the current B score), and the variability of the bias (VAR – equivalent to the current C score) and GCMS target values. The vertical dotted lines indicate the limits of acceptable performance agreed by the National Quality Assurance Advisory Panel at that time. For each method, the name and number of users in parentheses is given in the left-hand column. The spread of BIAS and VAR data for each method is indicated by a box and whisker graphic. The short vertical line within the box indicates the median, the outer box indicates the interquartile range and the whiskers the range of values represented by 95% of the data. The asterisks are considered to be outliers. The Corning ACS:180, Boehringer ES300 & 700, IDS Gamma-B, J&J Amerlite and Serono Maia assays are either no longer in use or have been superceded. © UK NEQAS for Steroid Hormones – reproduced with permission from the Organizer
Direct (non-extraction) RIAs were developed in response to workload and health and safety pressures, but marketing considerations placed these in the assisted conception area of application, where concern for low-end sensitivity was secondary to a wide dynamic range. Attempts have been made by some manufacturers to improve specificity by switching from 3-position linked immunogens to 6-position linked. This moves the point of attachment away from the part of the oestradiol molecule that is needed to confer antibody specificity.
The advent of multianalyte immunoassay analysers in the 1980s created a highly competitive market where analyte repertoire and speed became more important considerations. Highly ambitious claims were made that very short (<10 min) incubation times were practical for oestradiol and other steroid hormones. During the 1980s and 1990s the UK NEQAS for Steroid Hormones repeatedly showed that automated immunoassay platform oestradiol assays were diverging in results produced, had poor recovery of added pure steroid and poor low-level performance (see UK NEQAS for Steroid Hormones Annual Reviews available from JGM at UK NEQAS, PO Box 3909, Birmingham B15 2UE).
Although oestradiol reference methods based on isotope dilution gas-chromatography, mass spectrometry (ID-GCMS) had been developed from the pioneering work of Siekman 14 in the late 1970s, interest in using these to underpin the calibration of routine oestradiol assays did not come to the fore until the early 1990s, when Thienpont et al. 15 attempted to bring all manufacturers together in a series of workshops to discuss this, and point the way forward for the systematic and coherent use of the steroid reference laboratory network. Progress has been slow with this, although manufacturers seem much more open now to discuss calibration and traceability, no doubt as a result of the publication of the EU In Vitro Diagnostic Devices (IVDD) Directive and its traceability element. It is gradually becoming accepted that the only way properly to assess calibration is with a panel of unprocessed single donation serum sample with a wide range of oestradiol concentrations, with values assigned by ID-GCMS by a member of a recognized reference laboratory network. 16 A fully metrologically traceable result has been possible for oestradiol for decades, but the implementation of this concept across the diagnostic industry has not occurred.
State-of-the-art of present immunoassays for oestradiol
Current UK NEQAS performance
Figure 3a shows a ‘box and whisker’ plot of six-month time window B score (mean percentage bias from the All-Laboratory Trimmed Mean (ALTM)) for the main method groups in the UK NEQAS for oestradiol (main scheme – normal cycle concentrations) at distribution 333 in September 2007. The horizontal line within the box indicates the median bias, the box indicates the 25th to 75th centile of the bias (interquartile range) and the whiskers indicate the 5th to 95th centile of the bias. The light shading (±10–15%) indicates a ‘warning area’ and the dark shading (±15%) indicates bias in excess of the National Quality Assurance Advisory Panel (NQAAP) for Chemical Pathology acceptable limits of performance for this analyte. Within the time window at this distribution (333) were 30 specimens with ALTMs ranging from 49 to 1424 pmol/L. It can be seen that there are large differences in bias (>50%) which are characteristic of each method.
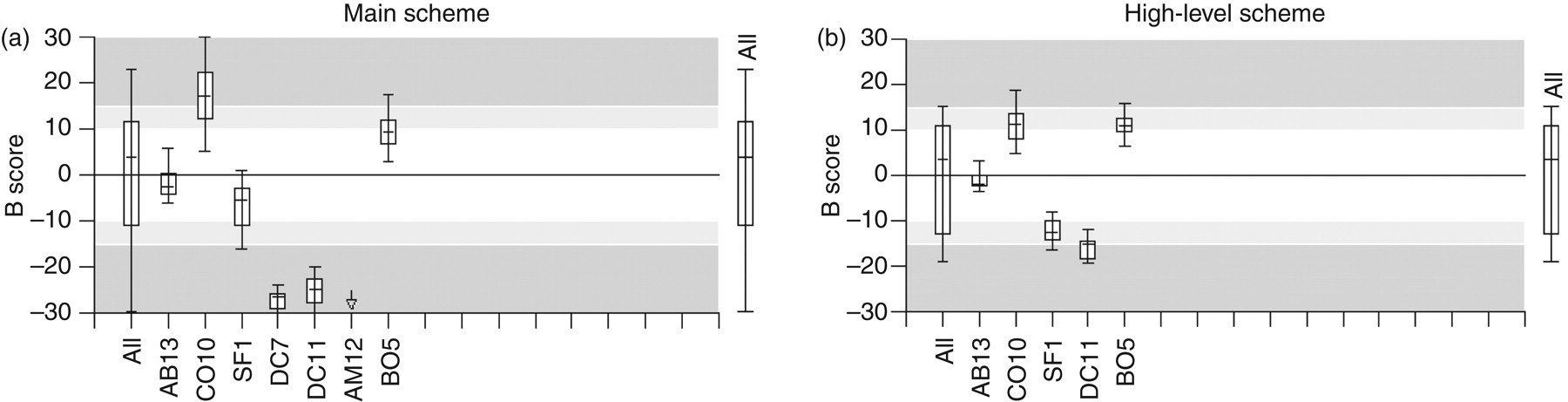
Box and whisker plots of B score – average percentage bias from the all-laboratory trimmed mean (ALTM) for all samples in the six-distribution period in the UK NEQAS for oestradiol (main and high-level schemes) at distribution 333. Main scheme methods and numbers of users: AB13 is Abbott Architect (47 users), CO10 is Siemens Advia Centaur (57 users), SF1 is Beckman Access (23 users), DC7 is Siemens Immulite (6 users), DC11 is DPC Immulite 2000 (42 users), BO5 is Roche Elecsys/E170 Modular (80 users) and AM12 is Ortho Vitros ECi (4 users) (off the plot at –36%). High-level scheme methods and numbers of users: AB13 is Abbott Architect (19 users), CO10 is Siemens Centaur (20 users), SF1 is Beckman Access (8 users), DC11 is Siemens Immulite 2000 (15 users) and BO5 is Roche Elecsys/E170 Modular (31 users). © UK NEQAS for Steroid Hormones – reproduced with permission from the Organizer
Figure 3b shows the same plot for the separate high-level oestradiol scheme (concentrations associated with the stimulation phase of assisted conception). Within the time window at this distribution (312) were 24 specimens with ALTMs ranging from 204 to 9045 pmol/L. It can be seen that the pattern of bias between methods is similar, but the overall dispersion is less (method medians within ±15%).
Figure 4 displays individual plots of performance scores (B score is average bias from the ALTM in the current six-month time window; C score is the standard deviation of this average bias and is an index of bias variability with concentration, within or between distributions; it is not ‘imprecision’) for each method. The dotted line shows the limits of acceptable performance set by the NQAAP; the unshaded rectangle shows the 5th to 95th centile of all-laboratory performance; the shaded rectangle shows the 5th to 95th centile of method group performance; each open circle is one individual user of the method and the cross is the method mean B score versus method mean C score coordinate. These plots show the characteristic variability of scores between users for each method. Such variability is an indication of combined instrument, reagent and operator performance effects.
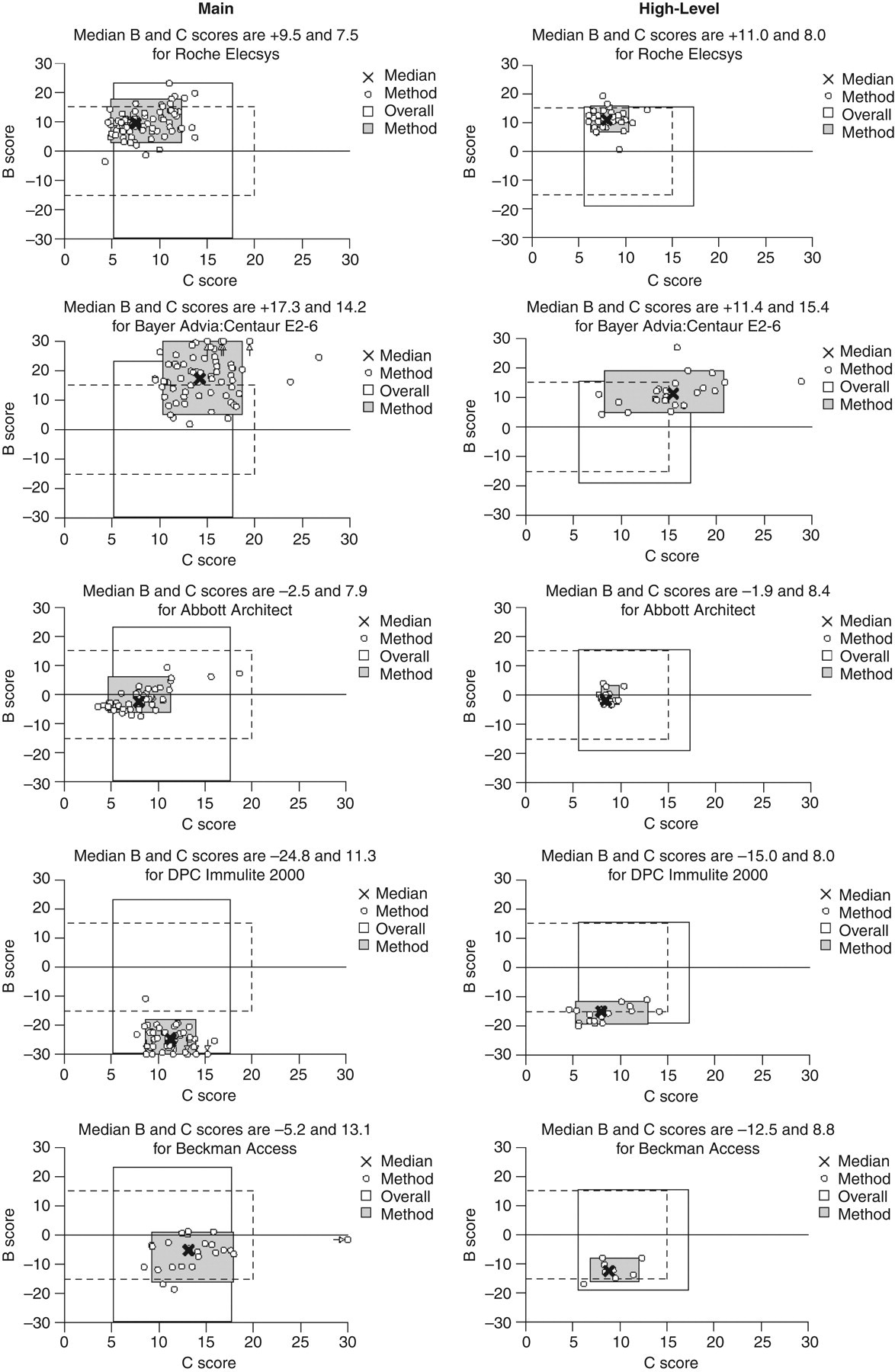
Plots of B score (trimmed mean of all the sample percentage biases from target value in the six-distribution scoring time window) versus C score (standard deviation of the B score) for the main method groups in the UK NEQAS for oestradiol (main and high-level schemes) at distribution 333. Note that the Bayer and DPC methods are now Siemens Healthcare Diagnostics Ltd. © UK NEQAS for Steroid Hormones – reproduced with permission from the Organizer
Low concentration performance special analysis
Table 1 shows the statistical analysis of results for one low-concentration sample (331A) distributed in the summer of 2007. Some large variation in results was seen, with Siemens Centaur and Beckman Access being particularly variable. The majority of Siemens Immulite 2000 users reported ‘less than’ results. The range of ‘less than’ cut-off values which labs used was rather wide, but this demonstrates that many laboratories recognize assay limitations.
Low-level oestradiol between-laboratory comparability for all laboratories and selected methods for UK NEQAS sample 331A. Oestradiol values are in pmol/L
numeric, number of numeric results received; <, number of ‘less than’ results received; <, low lowest value below which a less than result is given; <, high highest value below which a less than result is given; max:min, ratio of minimum to maximum value
AB13 = Abbott Architect; AM12 = Ortho Vitros Eci; BO5 = Roche Elecsys; E170 = Roche E170 modular; CO10 = Siemens Centaur; DC11= Siemens Immulite 2000/2500; SF1 = Beckman Access/Dxi
© UK NEQAS for Steroid Hormones – reproduced with permission from the Organizer
Trueness and comparability
But which (if any) of these methods is/are correct? Steroid hormones exist as defined chemical entities that can be purified and obtained ‘off the shelf’ 99% pure. As such they can be weighed into standard solutions with very low uncertainty. Furthermore, reference methods have been available for decades, which enable the content of serum calibrants and controls to be established with negligible error. Networks of steroid reference laboratories also exist whose members exchange protocols and results so as to maintain a trueness base. The newly formed Joint Committee for Traceability in Laboratory Medicine (JCTLM) 17 is compiling databases of reference methods and reference materials which can be used to assign values to calibrants and controls and thus meet the traceability requirements of the EU IVD Directive. 18 Materials and methods for steroid hormones were among the first to be included in the JCTLM database. All assays should therefore be accurately calibrated and their relationship with the reference method is well established and understood. Why then, is comparability so poor?
To establish metrological traceability, there must be an unbroken chain of comparisons from the end result backup through the traceability chain of increasingly higher order reference methods and materials, to the SI definition of the analyte. It is critically important to understand that metrological traceability can only be established if the assay is specific for the analyte, as any interference by related compounds break the chain.
Calibration begins with establishing a working standard in a suitable matrix, usually based on adding a known amount of certified reference material into stripped serum. In an ideal world, where the antibody is exquisitely specific for the analyte, the assay would be matrix independent, and would achieve results that agreed well with weighed-in values. In reality, however, antibodies are not specific – they react to cross-reacting and interfering substances. The initial calibration process may therefore not yield correct results (in terms of the calibrant used) on a clinical matrix because this contains other reacting species. Furthermore, assay conditions – ionic strength, temperature, timing, etc. – also affect antibody specificity. The manufacturer has to balance all these factors, as well as platform-related constraints, in constructing an assay. In the past, solvent extraction was the strategy used to remove interfering substances, but this is no longer acceptable in the market place.
The only choice manufacturers may have to bring their assays into line with expected values is to adjust the calibration curve so that results lie in a range which is perceived to be acceptable. In the case of oestradiol, where clinical applications lie over a wide range of concentrations, manufactures should ideally choose where to target their assay and optimize for that concentration range, e.g. paediatric, menopausal, normal cycle, assisted conception. In the main, they have not done this and have marketed ‘broad brush’ assays that attempt to cover a very wide range of concentrations, sometimes exceeding the limits of competitive immunoassay designs.
Such attempts to circumvent non-specificity may be revealed by the standard approach we recommend of assaying panels of single donation, unprocessed human serum materials with a wide range of concentrations established by the reference method, and by recovery exercises. The recovery data presented in this review and discussed below review demonstrate this. At low analyte levels, a non-specific assay may have its calibration ‘adjusted’ so that it produces lower results. This will be exposed by under-recovery at this level – which is what is observed. At higher levels of analyte, where the signal noise ratio is higher, recovery may be more quantitative.
In the UK NEQAS for Steroid Hormones, trueness (accuracy) is assessed in two ways. Firstly, recovery exercises are undertaken on an annual basis, and results for the main and high-level oestradiol scheme exercises at distribution 330 are shown in Table 2. Five pools were distributed for the main scheme: a basal pool and four others spiked with doubling amounts of pure oestradiol in ethanolic solution. Four pools were distributed for the high-level scheme, a different basal pool and three others with much higher doubling amounts of oestradiol. The table shows the percentage recovery for each spiked sample and the average recovery overall. (It should be noted that recovery tends to be a stable characteristic of methods when exercises are repeated over time.)
Recovery exercises for oestradiol (main scheme and high-level scheme) at UK NEQAS distribution 330
For the four columns on the left side of the table, ‘sample’ = UK NEQAS specimen identifier; ‘ALTM’ = All-Laboratory Trimmed Mean; ‘added’ = amount of oestradiol added to the spiked pools; ‘recovery’ = % recovery of the amount spiked
Under each method code is the number of users returning results, method mean results for each sample and the % recovery of the amount spiked.
The average recovery is the mean of the individual recovery figures obtained for the separate samples.
AB13 = Abbott Architect; AM12 = Ortho Vitros Eci; BO5 = Roche Elecsys/E170 modular; CO10 = Siemens Centaur; DC11 = Siemens Immulite 2000/2500, DC7 = Siemens Immulite 1; PH2 = Wallac Delfia; SF1 = Beckman Access/Dxi; TO1 = Tosoh AIA
© UK NEQAS for Steroid Hormones – reproduced with permission from the Organizer
For the main scheme, it can be seen that two methods achieved near quantitative overall average recovery (Roche Elecsys/E170 modular and Siemens ADVIA Centaur) but the former had marked non-linearity of recovery with concentration. Other methods had very low recovery figures (especially Ortho Vitros Eci). There were marked differences in the basal pool estimate.
For the high-level scheme, overall recovery of the ALTM was much nearer quantitative, and some low recovering methods in the main scheme had improved figures. It was interesting that the non-linearity of the Roche Elecsys/E170 method was in the other direction for the high-level exercise.
Secondly, periodic exercises are undertaken using pools that have had reference method (ID-GCMS) values assigned by the steroid reference laboratory of Professor Linda Thienpont (University of Gent, Faculty of Pharma- ceutical Sciences, Laboratory for Analytical Chemistry, Harelbekestraat 72, B-9000 Gent, Belgium), which is listed in the JCTLM database and participates in RELA – the IFCC External Quality assessment scheme for Reference Laboratories in Laboratory Medicine (see
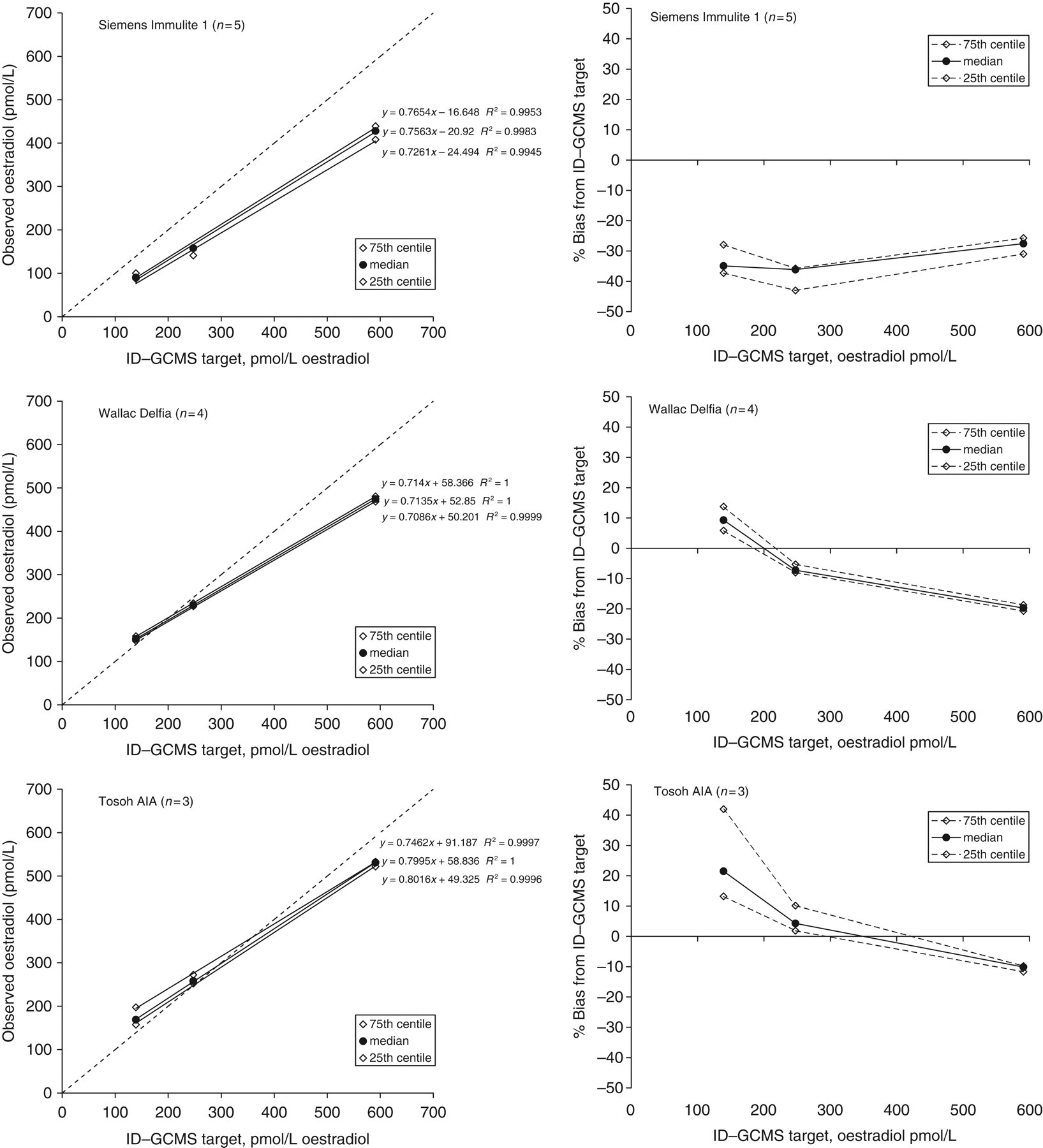
Regression and difference plots for the oestradiol ID-GCMS (isotope dilution gas-chromatography, mass spectrometry) targeting exercise conducted at distribution 332. Note that the Bayer and DPC methods are now Siemens Healthcare Diagnostics Ltd. © UK NEQAS for Steroid Hormones – reproduced with permission from the Organizer
Clinical applications of oestradiol assays – suitability of current assays
The measurement of serum oestradiol in isolation is often not useful and results are usually interpreted in conjunction with those of gonadotrophins, human chorionic gonadotrophin (hCG) prolactin, testosterone and progesterone. The instances in which a single estimation of oestradiol may be of use are in the monitoring of ovulation induction regimens and in vitro fertilization (IVF) treatment cycles, in the diagnosis of oestrogen secreting tumours and in the monitoring of patients on oestradiol implants.
In normal and abnormal development
The measurement of serum oestradiol is sometimes requested in prepubertal children being investigated for developmental problems and precocious puberty. Current assays still do not have the sensitivity to accurately measure the concentration of oestradiol present in normal children. A highly sensitive recombinant cell bioassay developed over 10 years ago 19 was used to measure the serum oestradiol concentrations in healthy children. The mean oestradiol concentration in normal girls was <2.2 pmol/L and <0.3 pmol/L for apparently healthy boys. In Table 1, it can be seen that at an oestradiol concentration of 50 pmol/L five out of seven methods studied had a coefficient of variation of >20% with one other >13%. These results reflect the inability of current immunoassays accurately to measure oestradiol at the very low concentrations (<10 pmol/L) required in children.
Precocious puberty is encountered more in the female than male child. In the female child precocious puberty is defined as the onset of puberty before the age of 8 years. The most common cause of this condition is premature activation of the hypothalamic–pituitary–gonadal axis. The gonadotrophins rise to pubertal concentrations although due to the reasons stated above many assays may fail to detect the rise in oestradiol which will still fall below their detection limit. This could potentially lead to the diagnosis not being made in these patients. Signs of precocious puberty can also arise as a result of excessive oestrogen secretion by an ovarian or adrenal tumour. In this situation, the serum gonadotrophins are low and serum oestradiol concentrations can be in the postpubertal range.
Serum oestradiol measurement is also sometimes requested in females with primary amenorrhoea (absence of the menarche by the age of 16 years) or in children during the investigation of short stature. In these cases, the serum oestradiol concentration is again often undetectable with current immunoassays and results must be interpreted in conjunction with gonadotrophins, prolactin, thyroid function tests and sometimes karyotyping studies.
In endocrine and malignant disease
Malignant disease
Serum oestradiol measurements are used in the diagnosis of oestrogen-secreting tumours. These tumours are very rare, occur in both sexes and arise in the gonads (testes or ovary) or in the adrenals. Tumours secreting hCG may also cause excessive production of oestradiol by stimulation of the testes or ovaries. In these cases the serum oestradiol is usually elevated with suppression of gonadotrophins.
Newer treatments for breast cancer involve the administration of aromatase inhibitors to inhibit the production of oestrogens from androgen precursors. Klein et al. 20 showed, using a recombinant cell bioassay, that the oestradiol concentration suppressed to less than 1 pmol/L six weeks after the commencement of treatment with an aromatase inhibitor. They concluded that in research studies into the potency and efficacy of aromatase inhibitors was being inhibited by the lack of sensitivity of oestradiol immunoassays and that assays with a sensitivity of less than 1 pmol/L were required for this type of study. Although clinical requests for oestradiol measurement in this group of patients are rare, it is obvious that current assays are unsuitable for monitoring the efficacy of aromatase inhibitor therapy.
Gynaecomastia
The endocrinology of gynaecomastia has recently been well reviewed by Ismail and Barth. 2 They concluded that, although the measurement of oestradiol is commonly requested in the assessment of gynaecomastia, the quality of assays available was a major limitation of its use. In particular, they noted that the imprecision of most assays in the range of oestradiol concentrations in males (<150 pmol/L) was inadequate for the investigation of adult men and that ‘hyperoestrogenism may exist despite an apparently normal serum oestradiol concentration.’ They recommended that ‘all men, and particularly those with borderline oestradiol concentrations, should be investigated with an assay that employs organic extraction and is designed for the measurement of low concentrations’. Ideally, an oestradiol assay that is to be used for the investigation of gynaecomastia in men should have adequate precision (a coefficient of variation of less than 15%) between 50 and 100 pmol/L.
In assessing fertility
The measurement of serum oestradiol is often requested in the investigation of female infertility with or without amenorrhoea. There have been a number of reviews on the causes and treatment of female infertility in which the role of laboratory tests have been discussed. 21,22 In the investigation of female infertility, an isolated measurement of oestradiol is rarely of any benefit and results should always be interpreted in the light of other hormonal investigations. In patients with regular menstrual cycles, results are often difficult to interpret due to the variability of oestradiol concentrations during the cycle and the lack of relevant clinical information on most request forms. Measurement of oestradiol can sometimes be useful in the investigation of patients with amenorrhoea or oligomenorrhoea and raised levels of gonadotrophins. In this situation, a low concentration of oestradiol would indicate a menopausal pattern whereas a high concentration would suggest the pattern of results found at mid cycle. It should be noted however that many commonly used oestradiol immunoassays lack the analytical sensitivity (coefficient of variation [CV] <20% below 100 pmol/L) to reliably measure the low concentrations of hormone found during the menopause (<100 pmol/L) with the potential for misclassification in this group of patients. 13
During assisted conception
The measurement of serum oestradiol can be of use in patients undergoing ovulation induction treatment prior to either natural conception or IVF treatment. Serum oestradiol concentrations and follicular size/number detected by ultrasound are closely related in both spontaneous and induced ovulation. Oestradiol concentrations can therefore be used to predict when optimal maturity of the follicles has been attained and to avoid hyperstimulation of the ovary.
In patients undergoing ovulation induction with natural conception, the aim of the treatment is to stimulate one or at most two follicles in order to avoid the risks of hyperstimulation and multiple pregnancy. Daily measurements of oestradiol coupled with ovarian ultrasound are performed to predict the optimum timing of hCG administration to trigger ovulation. As an approximate guide an oestradiol concentration of 1000 pmol/L is usually used as a marker of one fully developed follicle (>15 mm diameter). 3
In an IVF treatment cycle the aim is to stimulate multiple follicular development. Large numbers of oocytes can then be recovered and frozen for use in subsequent cycles of treatment if necessary. Previously, it was important to carefully monitor oestradiol concentrations during these stimulation procedures as there was an unpredictable response to the ovulation induction regimen requiring close observation so that egg recovery could be carefully timed. Introduction of GnRH agonists allowed downregulation of pituitary LH and FSH and reduction of oestradiol concentrations to low concentrations prior to ovulation induction. This allowed far more regulation of the induction procedure such that some IVF units now do not perform any endocrine monitoring during IVF stimulation cycles relying totally on ultrasound assessment. 23
If used in an ovulation induction setting, the major requirement for an oestradiol assay is that the working range should be extended so that concentrations up to 10–12,000 pmol/L can be measured with good precision. 3 In addition, it is important that the results are available within 2–3 h to allow time for decisions on further treatment to be made. Some manufacturers offer an alternative protocol to their standard method to allow these higher concentrations to be measured. If however an IVF unit wishes to monitor its patients during the downregulation phase of their treatment, a highly sensitive assay, with good precision at <100 pmol/L, will be required. As discussed previously, many commercial assays do not meet the analytical sensitivity requirements for use in this context.
In the perimenopause and during hormone replacement therapy
Postmenopausal hormone replacement therapy (HRT) is widely used to relieve menopausal symptoms and is one of a number of treatments for osteoporosis. Biochemical monitoring of HRT is needed in some patients, depending on the purpose of treatment and its route of administration.
Confirmation of the menopause
FSH is the investigation of choice to confirm the postmenopausal state. 24 In most cases, no other hormonal measurement is necessary. Serum oestradiol should not be routinely requested unless there is a specific indication but may be useful in selected patients, for example, in the peri-menopause, in deciding the correct time to initiate treatment and to exclude sampling in mid cycle as a cause of raised gonadotrophins. As stated previously, a recent study has shown that some direct methods for the measurement of serum oestradiol lack the sensitivity to accurately measure oestradiol in postmenopausal women further limiting their use in these patients. 13
Monitoring HRT
Serum oestradiol is the investigation of choice, but its value depends on the route of administration and the nature of the HRT preparation used. Serum FSH is unhelpful because replacement with physiological levels of oestradiol alone cannot suppress serum FSH to premenopausal levels. 24
Oral HRT
The most commonly used oral oestrogen preparations are mixtures of water-soluble, oestrogen conjugates, which are converted to a variety of compounds with differing biological activity. 4 Circulating oestrogen conjugates can cross-react in the direct serum oestradiol assays that most laboratories now use. Therefore, the measured value may not reflect bioactive oestrogen or correlate with treatment efficacy. Micronized pure oestradiol is also used orally but there is little published data on the metabolism of these preparations and whether measurement of serum oestradiol concentrations is useful.
Parenteral HRT
Serum oestradiol concentrations measured in most assays give accurate results in this group of patients because the preparations are formulated as pure oestradiol and are absorbed directly into the bloodstream from the site of application. This avoids the problem with circulating oestrogen conjugates from metabolism in the liver of differing preparations used in oral HRT.
Percutaneous oestrogen patches
Serum oestradiol concentrations peak approximately 2–8 h post-application and are said to plateau before beginning to fall after 72 h. They return to baseline values within 24 h after removal of the patch. The best time to sample is just before applying a new patch (trough level), the purpose being to confirm the adequacy of oestradiol replacement. 25
Oestradiol gels and nasal spray
The value of monitoring oestradiol concentrations in these patients is still unclear.
Subcutaneous oestrogen implants
The main problem with this mode of therapy is the risk of accumulation if implants are given too frequently. The decision to re-implant should not be based on symptom recurrence alone. Serum oestradiol measurement is therefore essential for determining the need for re-implantation. It is recommended that concentrations are measured three-monthly over the 12 months following the first implant and annually following subsequent implant. 25
Interpretation of serum oestradiol concentrations
To relieve menopausal symptoms, adequate oestrogen replacement is that which achieves the therapeutic goal! On present knowledge, maintenance of serum oestradiol between 200–300 pmol/L is believed to be adequate to protect against osteoporosis. Equally, there is concern to avoid prolonged exposure to excessively high oestrogen concentrations (>1000 pmol/L). A study conducted at Hope Hospital by Buckler and colleagues using the Wallac DELFIA oestradiol assay recommended that implants should not be repeated if serum oestradiol concentrations are >400 pmol/L. If the concentrations are between 250 and 400 pmol/L, the decision to re-implant must be based on clinical grounds in the individual case. 26 The data we have presented show that there are large differences in bias between the various methods that could potentially lead to mistreatment of patients if not taken into account when setting target concentrations. 25
Analytical system specification and tendering by laboratories
Since it is the laboratory which must take responsibility for the quality of its results, it is critically important that laboratories specify parameters for trueness and traceability in their tender documents. Only by doing this en masse in a systematic way, might all manufacturers be encouraged to use the steroid reference measurement system to underpin the calibration of their assays.
Laboratories should request two graphs for each analyte:
(1) A regression plot of system result versus ID-GCMS target value for a series of single donation, unprocessed, patient's serum sample with analyte concentrations across the range of clinical application and target values assigned by a member of the steroid reference laboratory network.
(2) A difference plot of the same data.
The slope of the regression line will indicate overall trueness of calibration; the intercept will indicate the degree of uncertainty at low concentrations; the scatter (Syx ) about the regression line will indicate specificity and the shape of the difference plot will indicate any concentration-related bias. All manufacturers should be encouraged to employ the same materials in the same way and display their data in the same format for easy comparison between systems.
It is also imperative that laboratories emphasize to instrument producers that they need properly optimized assays for the concentration range of interest. To cover all possible clinical uses of oestradiol assays, three separate assays would be required: (1) ultra-low (for paediatric, prepubertal and postmenopausal), (2) normal cycle and (3) stimulation phase. One assay cannot cover the whole 10 pmol/L–12,000 pmol/L range with adequate imprecision.
However, the authors fully recognize the potential conflict of offering a wide repertoire of assays on a single analytical platform and ensuring that each is properly optimized. Compromises are inevitable in this process, but they should be limited through an effective partnership with the diagnostic industry as described below.
Action by manufacturers and industry regulators
Manufacturers: Diagnostic companies should cease the undesirable practice of ‘calibrating’ assays to produce the same results as other systems, EQA schemes or ‘expected values’, especially where the origin of the latter may be uncertain. The established reference measurement system must be used to calibrate according to the scientific principles of metrological traceability. Manufacturers must collaborate on the underpinning scientific basis of assay standardization so that the customer can be assured that they are fulfilling both the spirit and the letter of the IVD Directive and that the process is independent and scientific. If all manufacturers use the same trueness benchmark, this should improve assay comparability;
Regulators: In the UK, the diagnostic industry is not regulated for trueness or traceability except through the EU IVDD. The Medicines and Healthcare Regulatory Agency (MHRA) has a strong role as the Notified Body under the IVDD to monitor and take action where assay systems might cause patient harm. Trade organizations (e.g. British In Vitro Diagnostics Association – BIVDA – in the UK) have a role in ensuring that all its members sign up to and comply with the IVDD and could also be instrumental in encouraging co-operation and collaboration on the scientific basis of standardization. In particular, they could help coordinate production of the single donation panels described above. This should be a collaborative exercise between all stakeholders: laboratories, EQA services, reference laboratories and manufacturers, with the latter accepting the major burden for funding of collection, value assignment and storage. The exemplar here would be the reference panel produced for cortisol in response to the Bergmeyer Conference. 27
Action by professional bodies, accrediting organizations, government
Professional bodies have strong educational and leadership roles in promulgating a thorough understanding of metrological traceability, and in helping clinicians and patients come to terms with revised analyte concentrations and reference range values when improvements in assays generate new data. This is particularly important in respect of the production of guidelines that have defined analyte concentrations incorporated in them;
The use of methods underpinned by or incorporating mass spectrometry technology may lead to a different understanding of hormone concentrations that are used in the assessment of patients. Improvements in the analytical specificity of field methods have always led to evolutionary change in laboratory medicine, so care must be taken no longer to fossilize ‘expected values’. Nothing should be more important for a clinical laboratory than to produce an analytically valid result. Arguments that it is in the interest of patients that long established ‘numbers’ used by clinicians (often based on superceded non-specific methods) must not be changed because it will cause confusion, are spurious. There will have to be re-education about the meaning of new reference values and guideline limits, but such issues are inevitable as the technology of measurement evolves;
Accrediting organizations generally assess quality systems, but analytical quality should be an integral part of quality assurance;
Government/Department of Health/National Health Service must take great care to ensure that scientists with a detailed understanding of the relevant analytical and metrological issues are included in the advisory panels considering National Service Frameworks or Guidelines for investigation, screening and monitoring of treatment. The effects of non-specificity, calibration and method variability must be estimated as part of an uncertainty budget around any hard numerical values that are recommended. It is also imperative that those involved in large-scale health-care computing and electronic patient record management also understand the effect of variable assay bias on result comparability over geography and time.
Improving oestradiol assays – who needs to get involved?
There is a role for the research scientist in developing new immunoassay designs that might re-configure the assay in a labelled antibody format, or tandem mass spectrometry applications for routine use. With respect to the latter, the assays for oestradiol developed by Nelson et al. 28 and Kushnir et al. 29 are of great interest. Their method comparisons with routine immunoassays broadly reflect the UK NEQAS experience discussed in this review;
There is a role for the clinical scientist in assessing clinical needs and creating analytical and quality specifications that meet them, as well as understanding the limitations of current assays and giving appropriate advice;
EQA organizers must continue to monitor trueness and comparability using materials targeted by the reference measurement system so as to provide a clear benchmark for manufacturers to improve assays and laboratories to gain appropriate objective information when tendering for equipment;
Manufacturers must produce assays that match analytical and quality specifications and meet EU IVDD requirements for metrological traceability. They should collaborate together to improve comparability and fully engage with the reference measurement system in place;
Clinicians must understand the limitations of oestradiol assays in different clinical applications, re-evaluating clinical need as technology develops and adjusting oestradiol requesting patterns and interpretation in accordance with the evidence-base.
Summary of clinical requirements, state-of-the-art and action points
Table 3 summarizes our conclusions for oestradiol. In many cases, the answer to the question ‘Are oestradiol assays fit for purpose?’ is ‘No’, yet we have the apparent paradox that very many laboratories offer this test and many thousands of tests are regularly requested of them annually. All stakeholders should address this issue and consider our recommendations.
Summary of clinical requirements, state-of-the-art and action points
*‘Ultra-low’ means capable of measuring below 10 pmol/L with acceptable imprecision
DECLARATIONS
