Abstract
There is considerable variation in the methods used to diagnose and investigate adrenal insufficiency in clinical practice. These include a range of adrenocorticotropin (ACTH) stimulation and other dynamic testing protocols, serum cortisol cut-off values for diagnosis and tests used for differential diagnosis. With the introduction of modern cortisol and ACTH assays, the interpretation of tests used for diagnosis and differential diagnosis has become more complex and requires local validation. This review examines the basis of normal hypothalamic–pituitary–adrenal axis function and adrenal insufficiency states based upon an evidence base accumulated over the past four decades. The role of the laboratory in the differential diagnosis and interpretation based upon assay methodology is discussed. The accurate identification of patients who may benefit from corticosteroid replacement in special settings such as critical illness is challenging and will be explored.
Introduction
Adrenal insufficiency is a clinical syndrome arising from disruption of the normal hypothalamic–pituitary–adrenal (HPA) axis regulation of steroidogenesis. Symptoms are due to deficient production of cortisol and in some aetiologies mineralocorticoids and sex steroids also. Adrenal insufficiency presents insidiously or acutely in clinical practice and responds to steroid replacement therapy. In this context the role of corticosteroids in physiological and pathophysiological states is discussed. The clinical presentation of adrenal insufficiency and biochemical testing strategies for diagnosis will also be reviewed based upon published evidence identified from a literature search of clinical trials over the past four decades.
Overview of adrenal physiology
The adrenal glands are comprised of two functionally distinct areas: an outer cortex and an inner medulla. The three zones of the adrenal cortex that account for approximately 90% of gland mass are identified on the basis of microscopic differences. The mineralocorticoid aldosterone is produced in the outer zona glomerulosa, glucocorticoids are produced predominantly within the central zona fasciculata and sex steroids arise mainly within the inner zona reticularis.
Corticosteroid synthesis
Adrenal steroids are produced from the common cholesterol substrate low-density lipoprotein-derived cholesterol arising from plasma, intracellular cholesterol ester stores or via synthesis from acetate. 1 The adrenals may also use high-density lipoprotein (HDL)-derived cholesterol following uptake through a putative HDL receptor, SR-B1. 2 Steroidogenesis is facilitated through expression of enzymes depicted (see Figure 1). Transport of intracellular cholesterol from the outer to the inner mitochondrial membrane, the rate-limiting step of steroid synthesis, is regulated by the steroidogenic acute regulatory protein (StAR). 1 P450c17 is an enzyme with both 17α-hydroxylase and 17,20 lyase activity and is localized to the endoplasmic reticulum. It exists as a complex containing P450 oxidoreductase (which facilitates electron transfer from nicotinamide adenosine dinucleotide phosphate [NADPH]) and the flavoprotein cytochrome b5, which acts as an allosteric facilitator of P450 oxidoreductase. It is involved in the conversion of pregnenolone to 17-OH-pregnenolone and dehydroepiandrosterone and of progesterone to 17-OH-progesterone and androstenedione. 1 Adrenocorticotropin (ACTH) stimulates intracellular cyclic adenosine monophosphate and induction of StAR. 4 The zona glomerulosa is the only region of the adrenal cortex to contain the enzyme CYP11B2, and thereby limits the synthesis of mineralocorticoids to this site.
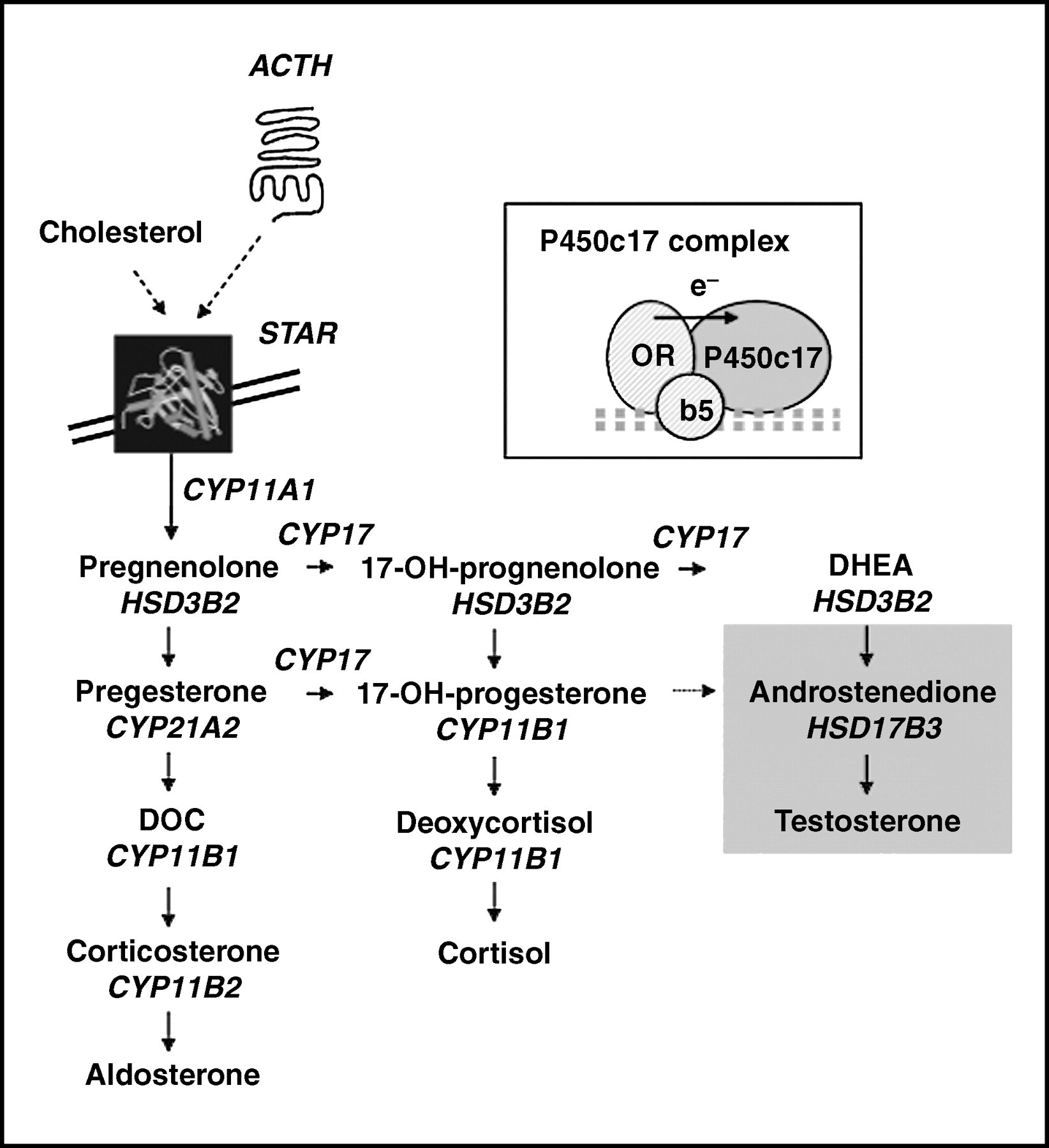
Overview of steroidogenesis. ACTH, adrenocorticotropic hormone; STAR, steroidogenic acute regulatory protein; DOC, deoxycorticosterone; DHEA, dehydroepiandrosterone. Modified and reprinted with permission from Lin L, Achermann JC. Horm Res 2004;
The HPA axis
Adrenal glucocorticoid production is tightly regulated by a negative feedback loop in response to pituitary-derived ACTH. The HPA axis exerts control over the system via corticotropin-releasing hormone (CRH) from the hypothalamus that in turn regulates ACTH production from the anterior pituitary (see Figure 2). CRH, a 41-amino acid peptide, binds to type-1 CRH cell surface receptors on the anterior pituitary corticotrophic cells leading, within seconds, to the release of ACTH and other pro-opiomelanocortin (POMC)-related peptides. 6 Arginine-vasopressin (AVP) and oxytocin are weak secretagogues of ACTH on their own, but they potentiate the action of CRH. Glucocorticoid feedback to the hypothalamus down-regulates CRH secretion, via reductions in CRH and AVP mRNA and peptide concentrations in the paraventricular nucleus. Other effects include diminished pituitary ACTH secretion via suppression of POMC gene transcription. 7
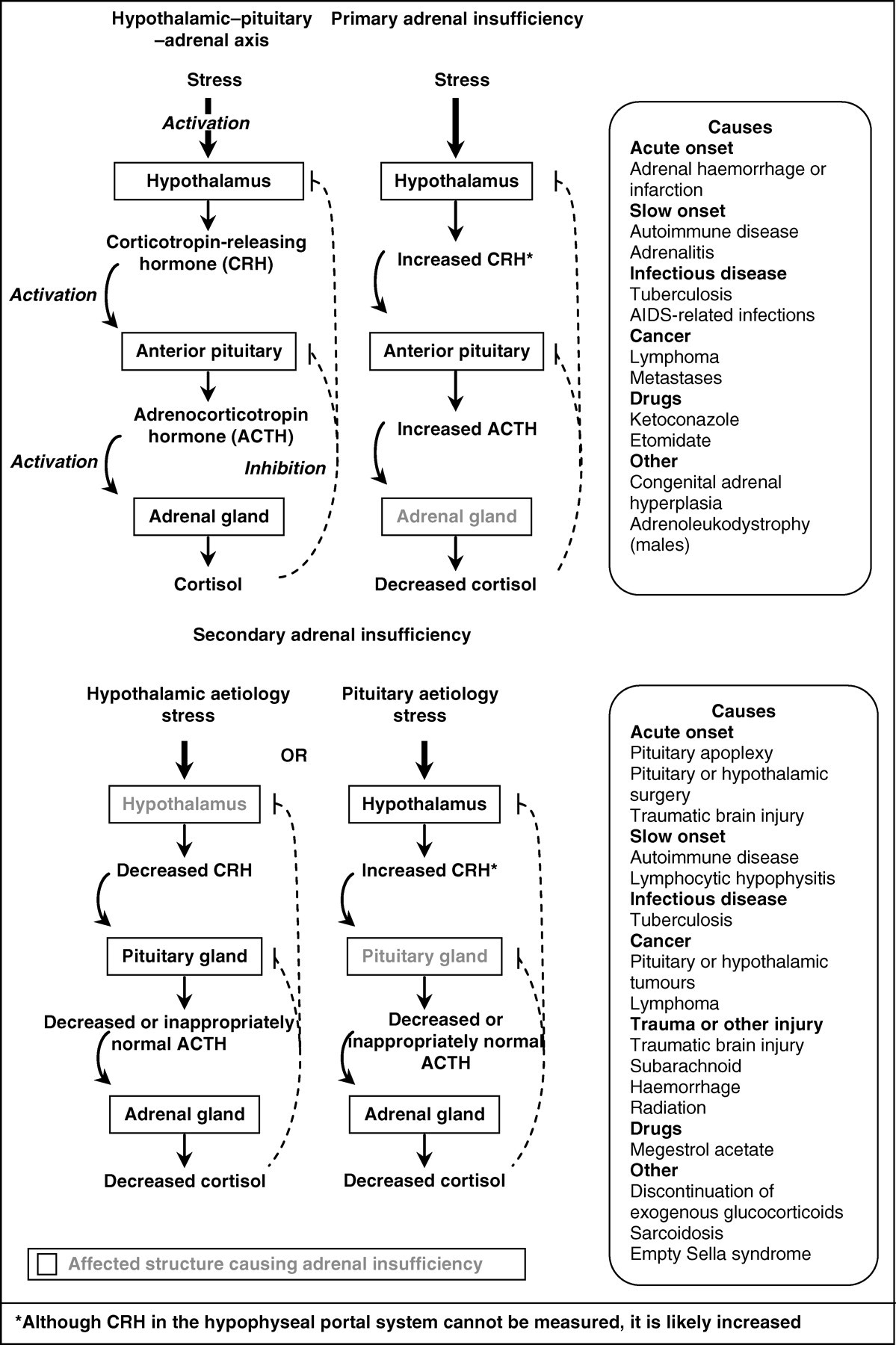
Hypothalamic–pituitary–adrenal axis and causes of primary and secondary adrenal insufficiency. Reprinted with permission from Salvatori R. Adrenal insufficiency. JAMA 2005;
ACTH in turn is synthesized, along with lipotropins, N-terminal peptide and β-endorphin, as part of the precursor POMC. ACTH has a half-life of 4–8 min and binds to the melanocortin-2 receptor at the adrenal cortex leading to adrenal StAR induction and increased conversion of cholesterol to pregnenolone. Later effects are up-regulated enzyme activity of the steroidogenic pathway and corticosteroid production. ACTH stimulates increased size, cell number and secretory capacity of the zona fasciculata. This effect accounts for the increased adrenal size observed in pituitary-dependent Cushing's syndrome, or conversely adrenal atrophy in ACTH-deficient states. 8
Circadian rhythm
Peak corticosteroid concentrations occur around the time of waking (275–555 nmol/L), whereas a nadir (<140 nmol/L) is observed at night. 9 Maximal cortisol concentrations arise during elevated HPA axis activity associated with increased sensitivity of the adrenal cortex to ACTH. 10 The hypothalamic suprachiasmatic nucleus stimulates peak hypothalamic activity and may act to inhibit hypothalamic activity at the onset of sleep. 11 The circadian rhythm is altered in response to changes in the activity cycle, ageing, sleep deprivation or depression. 12 In humans, nadir cortisol concentrations increase with age. 13
Corticosteroid transportation
Cortisol is approximately 90% bound to corticosteroid-binding globulin (CBG), 6% to albumin and 4% is unbound or free. 1 Only free circulating cortisol is biologically active. Circulating CBG (approximately 700 nmol/L) has negligible affinity for synthetic corticosteroids and is synthesized in the liver. Reduced CBG concentrations are observed in states such as cirrhosis, hyperthyroidism, nephrotic syndrome and protein-losing enteropathies. CBG concentrations increase 2–3 fold with oestrogen administration and pregnancy and in this scenario total plasma cortisol concentrations can be high with a normal free (unbound) cortisol concentration. 14
Corticosteroid metabolism
Cortisol has a circulating half-life of 70–120 min with metabolism occurring principally in the liver. The major steps are the interconversion of cortisol to cortisone (via 11β-hydroxysteroid dehydrogenases; 11β-HSD) or reduction via 5α-reductase (to yield 5α-tetrahydrocortisol) or 5β-reductase (to yield 5β-tetrahydrocortisol). 15 Various metabolites are rendered more water-soluble by conjugation with glucuronic acid or sulphate. Excretion occurs via kidneys with only 1% of the total cortisol excretion accounted for by free (unconjugated) cortisol. Approximately 50% appears in the urine as 5α-tetrahydrocortisol, 5β-tetrahydrocortisol and tetrahydrocortisone. There are two distinct isoenzymes of 11β-HSD, which are responsible for interconversion of cortisol and cortisone. 11β-HSD 2 is co-expressed with the mineralocorticoid receptor (MR) in various tissues and inactivates cortisol to cortisone, hence only allowing aldosterone to bind to the MR. 16 11β-HSD 1 is expressed principally in the liver and adipose tissue with the generation of cortisol from cortisone predominating. There is interest currently in the therapeutic potential of modulation of 11β-HSD for treatment of the metabolic syndrome. 1
Pathophysiology of primary, secondary and iatrogenic adrenal insufficiency
Clinical features
The clinical features of adrenal insufficiency are non-specific and therefore adrenal insufficiency may not be considered as a possible diagnosis until some time has elapsed, potentially leading to presentation with acute adrenal insufficiency. In primary adrenal insufficiency, the adrenal gland has impaired secretory function with intact hypothalamic and pituitary reserve. In secondary and tertiary adrenal insufficiency, there is impaired stimulation of the adrenals as a result of disruption to normal ACTH and CRH secretion, respectively. In primary adrenal insufficiency patients have features due to deficiency of glucocorticoids, mineralocorticoids and androgens, whereas mineralocorticoid secretion is usually preserved in secondary or iatrogenic adrenal insufficiency. Primary adrenal insufficiency is typically associated with hyperpigmentation of the skin and mucosal surfaces due to elevated plasma β-lipotropin concentrations (derived from POMC) and increased synthesis of melanin by melanocytes. 5 Mineralocorticoid deficiency can contribute to salt craving and postural hypotension. 17
Hyponatraemia may be a feature of both primary and secondary adrenal insufficiency, with different aetiologies in each case. 18 In primary hypoadrenalism mineralocorticoid deficiency leads to a hypovolaemic hyponatraemic state, whereas in secondary hypoadrenalism the mineralocorticoid axis is intact and there are raised concentrations of AVP. This leads to water retention and a dilutional or hypervolaemic hyponatraemia. 19
Clinical presentations may be associated with alopecia, vitiligo or other organ-specific autoimmune diseases. Adrenal insufficiency may present acutely in a crisis state and should be considered as a diagnosis in patients presenting with hypotension or non-specific features such as nausea, vomiting, abdominal pain, lethargy, pyrexia, myalgia or hypoglycaemia.
Primary adrenal insufficiency
The estimated prevalence of primary adrenal insufficiency is around 93–140 per million population with an incidence of 4.7–6.2 per million in white populations. 20,21 According to the original description by Thomas Addison (first presented in 1849 and later published in 1855) adrenal insufficiency occurred due to adrenal tuberculosis and while this may remain the most common cause worldwide, in Western populations autoimmune adrenal destruction accounts for 80–90% of cases. 22–24 Primary adrenal insufficiency is characterized by the absence of cortisol leading to elevated ACTH concentrations due to reduced negative feedback effects on the anterior pituitary gland. This is an important concept in the differential diagnosis and is discussed later. Causes of primary adrenal insufficiency may be classified into three broad aetiologies: adrenal destruction, adrenal dysgenesis/hypoplasia and causes of impaired steroidogenesis.
Autoimmune adrenalitis and autoimmune polyglandular syndromes
Autoimmune adrenalitis may occur as an isolated condition (approximately 40% of cases) or as part of an autoimmune polyglandular syndrome (APS). 24 Autoimmune adrenalitis of any aetiology, including APS, is more common in women. APS is a constellation of multiple endocrine glandular insufficiencies and is usually classified as type 1 or 2. Type 1 APS is inherited as an autosomal recessive condition caused by mutations in the autoimmune regulator (AIRE) gene and is characterized by development of autoimmune adrenal insufficiency, hypoparathyroidism and mucocutaneous candidiasis in childhood. Type 2 APS is typically associated with adrenal insufficiency and autoimmune thyroid disease and a range of organ-specific autoimmune diseases including type 1 diabetes, primary hypogonadism, coeliac disease and vitiligo.
Adrenal haemorrhage
Adrenal haemorrhage may occur in sepsis as Waterhouse-Friderichsen syndrome with sudden circulatory collapse, pyrexia, dyspnoea, purpura and acute adrenal insufficiency. 25,26 Other causes include anticoagulant therapy, antiphospholipid syndrome, trauma, spontaneous idiopathic haemorrhage and following surgery. 26
Infections
Adrenal tuberculosis only rarely occurs in isolation. 27 Most cases are accompanied by pulmonary tuberculosis with approximately 5% of cases of tuberculosis having adrenal involvement. 28 Non-tuberculous causes of primary adrenal infection including cryptococcus, histoplasma, cytomegalovirus, toxoplasma and pneumocystis are quite rare and tend to occur in immunosuppressed patients. 29
Infiltrative causes
The reported prevalence of adrenal insufficiency in patients with bilateral adrenal metastases ranges between 0% to 33%. 30 Clinically apparent adrenal insufficiency occurs with destruction of around 90% of the adrenal cortex in this setting. 31 Other infiltrative causes that include amyloidosis, sarcoidosis or haemochromatosis should be considered on the basis of suspicious clinical, biochemical or imaging features.
Other rare causes of adrenal insufficiency
Congenital adrenal hyperplasia (CAH) arises from several autosomal recessive mutations of genes for enzymes involved in steroidogenesis. 32 Patients with CAH due to 21-hydroxylase deficiency most commonly present with acute adrenal insufficiency in the neonatal period (salt-wasting) or with virilization. Detailed discussion of CAH is beyond the scope of this review but has recently been reviewed elsewhere. 33 Adrenoleukodystrophy is an X-linked condition that occurs due to a mutation in the ABCD1 gene located at locus Xq28. 34 The syndrome affects 1 in 20,000 men with adrenal insufficiency and is due to defective β-oxidation of very long-chain fatty acids. Patients may present with adrenal insufficiency but develop neurological symptoms many years later. It has been suggested that analysis of very long-chain fatty acids should be performed in all antibody-negative men presenting with primary adrenal insufficiency; however, this is not commonly undertaken in routine practice. 35 The classic neurological finding is of progressive central nervous system demyelination. To date only bone marrow transplants have shown obvious clinical benefit if performed early in the disease process. 36 Another rare cause of adrenal insufficiency is Triple A (Allgrove) syndrome, which is characterized by features of achalasia, alacrima and primary adrenal insufficiency. 37 Familial glucocorticoid deficiency syndrome (FGD1) results from mutations in the MC2R gene located at locus 18p11. This gene codes for the G-protein-coupled ACTH receptor. Phenotypically patients are tall and usually present with hypoglycaemia and features of adrenal insufficiency. Clinically these patients do not generally require mineralocorticoid replacement, hence identification may affect treatment choices. 3
Drug-induced adrenal insufficiency
Ketoconazole, metyrapone, mifepristone, aminoglutethimide and etomidate inhibit corticosteroid metabolism and potentially lead to adrenal insufficiency during treatment of Cushing's syndrome. 38 Iatrogenic adrenal insufficiency may occur during treatment with megestrol, a synthetic progestational agent that is used as an appetite stimulant. 39
Secondary adrenal insufficiency
Secondary adrenal insufficiency has a higher reported prevalence than primary adrenal insufficiency of around 150–280 per million population, with the most common cause arising from exogenous corticosteroid administration. 40 Secondary adrenal insufficiency is characterized by low concentrations of pituitary ACTH production. In the absence of ACTH stimulation adrenal atrophy occurs leading to inadequate corticosteroid responses to stress. 41 A range of pituitary or cranial disorders including pituitary tumour, pituitary apoplexy, subarachnoid haemorrhage, traumatic brain injury, empty sella, infiltrative disorders or rarer genetic causes of pituitary insufficiency can also result in hypopituitarism.
Iatrogenic adrenal insufficiency
Exogenous glucocorticoid administration via any route may lead to suppression of the HPA axis depending on the dose and duration of therapy. 42 Therefore slow withdrawal of corticosteroid therapy, over a period of 9–12 months, is often required for patients on long-term pharmacological corticosteroid dosing, so that the HPA axis can regain its ability to respond to appropriate stimulation. 43,44 Glucocorticoid formulations with a longer half-life including prednisolone, dexamethasone and prednisone have a higher risk of inducing adrenal insufficiency than short-acting formulations such as hydrocortisone or cortisone. Prednisolone doses, or corticosteroid equivalents, >5 mg/day correlate with inadequate responses to synacthen testing. 45
Investigation of suspected adrenal insufficiency
Cortisol assays
The usefulness of cortisol assays in the evaluation of possible adrenal insufficiency is limited by methodological aspects such as imprecision, accuracy, lack of standardization, specificity, other assay factors and choice of the appropriate decision limits for the assay used. Analytical performance criteria based on biological variation have been published for serum cortisol
46
and updated regularly on
Imprecision of cortisol methods
The imprecision stated by manufacturers in their assay data for three of the most commonly used assays in the UK are 1.4–2.8% for Roche E170, 6.8–8.2% for Siemens Immulite 2500 and 6.0–7.9% for Beckman Coulter Access DXi (total imprecision, coefficient of variation [CV%]). 47–49 However, the imprecision obtained by users may be higher when reagent and calibrator lot-to-lot variation is taken into account. Indeed, when data from two College of American Pathologists (CAP) surveys, in which the same fresh frozen serum pool (with a median cortisol concentration of 402 nmol/L) was issued, were used to assess the long-term (6 months) within-laboratory imprecision, the average within-laboratory CV for five cortisol methods ranged from 4.7% to 9.6%. 50 The within-method, between-laboratory CV% for the five principal cortisol assay methods in two recent UK NEQAS (National External Quality Assessment Service) EQA distributions (numbers 338 and 339, March and April 2008) ranged from 3.6% to 9.5% at values close to 500 nmol/L and from 5.6% to 15.7% at values close to 100 nmol/L. 51
Bias/accuracy
Cohen et al. 52 reported that two of three widely used cortisol assays examined returned results significantly higher than a high-performance liquid chromatography method in patients in an intensive care setting (Immulite, higher by an average 95% and Abbott TDx by 79%). 52 Data from a UK NEQAS exercise in 2008 shows that the average bias of commonly used methods for cortisol, when compared with gas chromatography–mass spectrometry (GCMS) values, varied from −20.6% for Beckman Access and −15.1% for Siemens Immulite to +11% for the Roche Elecsys group, at a cortisol concentration near 600 nmol/L. 51 This means that a ‘true’ cortisol value of 600 nmol/L would yield an average value of 476 nmol/L with one method and 666 nmol/L with another method. When all the laboratory (all methods) bias is examined, the range of bias values varies from −26.3% to +16.3% (5th to 95th percentiles): in this case a true cortisol value of 600 nmol/L would yield values ranging from 442 to 698 nmol/L.
The average bias of methods at low cortisol concentrations (129 nmol/L) was positive in relation to GCMS values, ranging from −4.3% for the Abbott Architect to +10.5% for the Siemens Centaur. 51 The total all-laboratory (all-method) bias at this low cortisol concentration ranged from −11.9% to +23% (5th to 95th percentiles), which means that a true cortisol value of 100 nmol/L could vary from 88 to 123 nmol/L in different laboratories.
All-laboratory, all-method imprecision
An estimate of the variability of results across a large number of laboratories using a number of different cortisol methods can be obtained from the CV% of the all laboratories trimmed mean (ALTM) for the results of each specimen analysed in the UK NEQAS. The CV% of the ALTM for various specimens sent out for analysis in 2007 ranged from 8.4% to 18.7% for specimens with values in the range of 500–600 nmol/L and from 9.5% to 13% for specimens with values circa 100 nmol/L. 51 The variability for female specimens was greater than for male specimens.
Specificity
Synthetic glucocorticoids such as prednisolone and methyl-prednisolone cross-react significantly in most cortisol assays, while prednisone cross-reacts to a smaller extent, but is converted to prednisolone in vivo. Cortisol precursors, e.g. 21-deoxycortisol and 11-deoxycortisol, and cortisol metabolites, e.g. 6β-hydroxycortisol, cross-react in some assays, but may not present a problem in clinical use (e.g. see Roche E170/Elecsys, Siemens Immulite 2500 and Coulter Access DXi reagent data sheets). 47–49
Standardization
The Institute for Reference Materials and Measurements (IRMM) and the International Federation for Clinical Chemistry and Laboratory Medicine (IFCC) have certified a reference material (CRM) for cortisol under the name and number IRMM/IFCC-451. This reference panel consists of 34 serum specimens from 18 female and 16 male donors, with cortisol values ranging from 82.7 to 764.2 nmol/L established by isotope dilution GCMS. The objective of preparing this reference panel was to make it available to manufacturers to standardize cortisol test systems. It is recommended that manufacturers use this reference panel in the verification of the metrological correctness of values obtained in their test system or to re-calibrate the test system if necessary. Despite the availability of this reference material, there is still significant assay bias for some methods compared with GCMS, as indicated above.
Other factors influencing results
As cortisol in plasma is 90% protein bound, all non-extraction assays require an agent such as 8-anilino-1-naphthalene-sulphonic acid to dissociate cortisol from binding proteins. The type and concentration of the dissociating agent may vary from one manufacturer to another and assays may cope to differing degrees with elevated CBG concentrations, such as may occur in pregnancy or on oestrogen-containing treatment. 53 It is possible that this may explain the greater all-laboratory variability in results for female specimens compared with male specimens in the UK NEQAS results referred above. In addition, cortisol results with some methods are influenced by albumin concentration of specimens, for instance, the Siemens Centaur method shows significant under-recovery of cortisol at albumin concentrations lower than 24 g/L. 54
Diagnosis of adrenal insufficiency
Basal cortisol
Serum cortisol concentrations should be interpreted in the context of existing assay methodology, which measures total cortisol concentrations. Ideally the cut-off should be determined depending on the assay method used in local practice. Increased cortisol binding globulin and total cortisol concentrations are associated with oestrogen use. For this reason it is advisable to discontinue oestrogen therapy for a period of at least six weeks prior to assessing the HPA axis. 55 Low serum albumin concentrations, as found in hypoproteinaemic states such as hepatic or renal failure, may lead to misleadingly reduced total serum cortisol concentrations despite normal biologically active free cortisol concentrations. 56
Random basal serum cortisol or ACTH measurements are of limited value for assessment of HPA axis reserve but when used are usually undertaken as an early morning sample at the time of maximal secretion (7:00–9:00). 10 Intact adrenocortical reserve can usually be confirmed with basal morning cortisol concentrations of >500 nmol/L. 41,57,58 The exclusion of adrenal insufficiency for those with intermediate serum cortisol concentrations (100–500 nmol/L) often requires further dynamic testing. These figures are quoted widely throughout the literature with reference to multiple assays and have largely been agreed by consensus.
ACTH stimulation testing (synacthen test)
The intravenous or intramuscular administration of synthetic adrenocorticotropic (ACTH 1–24) hormone 250 μg is the most widely used test in the initial assessment of adrenal insufficiency. The short synacthen test has replaced the eight-hour infusion test as it is easier to perform and achieves similar cortisol responses 58 The test was introduced in order to assess adrenal glucocorticoid reserve rapidly and conveniently. A previous survey of UK laboratories by Barth et al. found that eight different time schedules were used with 38 laboratories advocating intravenous and 135 recommending intramuscular administration. 59 Both methods of administration give equivalent results when used in the short synacthen test. 60 Baseline cortisol concentrations are obtained before and 30 or 60 min after 250 μg synacthen is administered. Testing leads to suprapharmacological ACTH concentrations >1000 times the normal physiological peak (above 60,000 pmol/L) that readily tests adrenal cortex responsiveness. 61 The standard synacthen stimulation test leads to higher peak stimulated cortisol concentrations (620–675 nmol/L) at 60 min compared with serum cortisol concentrations measured at 30 min (390–620 nmol/L) in healthy subjects 60 As the poststimulation value is used for diagnostic purposes in this test, the effects of the circadian rhythm are negated and this test may be performed at any time throughout the day. This range of cortisol values cited above is obtained via meta-analysis of multiple studies using a wide range of assays with differing biases that greatly limits the general applicability of the results. No significant gender-related differences have been consistently demonstrated and gender-specific cut-offs for the synacthen test are generally not considered to be clinically required. 53 However, it is possible that this view may alter with future assay development.
In clinical practice, by convention, patients who achieve a peak cortisol concentration >500–600 nmol/L at 30–60 min following synacthen are usually defined as having normal adrenal reserve, with a wide range of cortisol responses (between 400 and 700 nmol/L) used in the UK to define an adequate response. 58,59,62
Low-dose synacthen test
Established primary adrenal insufficiency can be readily identified using the standard synacthen test method; however, it is less useful during the early stages of ACTH deficiency due to pituitary disease. 5,63 In this setting, the low-dose synacthen test (1 μg) has been used for the evaluation of secondary adrenal insufficiency. 64–66 The low-dose synacthen test is performed by adding 250 μg synacthen (in 1 mL) to 249 mL of sterile normal saline, which is mixed thoroughly. One millilitre is then withdrawn and the test is performed as with the short synacthen test by the intravenous route. There may be inaccuracy associated with the preparation of the diluted synacthen in house. Initial studies of the low-dose synacthen test demonstrated improved sensitivity for diagnosis of secondary adrenal insufficiency using a cortisol cut-off value of >500 nmol/L to define a ‘pass’. 66,67 Nevertheless the test is challenging to perform, blood concentrations are still supraphysiological and similar diagnostic sensitivities to the standard-dose test were demonstrated by Mayenknecht et al. 61 More recently these observations were confirmed by a meta-analysis suggesting that the low-dose test is no more effective than the standard test. 60,68 Using either the low- or standard-dose test, many patients with pituitary disease with marginal responses will require further testing with the insulin tolerance or overnight metyrapone tests. 61
Timing of poststimulation cortisol measurements
There is widespread variation in the interpretation and timing of cortisol responses to ACTH stimulation. It is desirable that the definition of satisfactory response should be method related at all time points. 69 The majority of UK endocrinologists use the 30-min value for determining pass or fail. 70
Peak cortisol responses elicited during standard short synacthen testing are similar compared with the low-dose test or insulin tolerance test (ITT); however, the peak responses may occur at different times post-stimulation. 62 During the low-dose (1 μg) test, cortisol peaks early at 20–60 min postsynacthen stimulation, whereas the peak for the standard-dose (250 μg) test occurs at 60–90 min. There is some evidence suggesting that using the 60 min poststimulation cortisol concentration does not assist diagnostic decision-making. Indeed, the 60 min cortisol concentration is probably less reliable than the 30 min post-stimulation cortisol concentration. 71
Identification of a cortisol cut-off concentration for the synacthen test
A recent meta-analysis identified a cortisol cut-off for diagnosis of adrenal insufficiency of 415 nmol/L, which had high sensitivity in primary adrenal insufficiency. 60 At a specificity of 95%, sensitivities were 97%, 57% and 61%, respectively, for summary receiver-operating characteristic curves for the standard-dose (250 µg) synacthen test in primary adrenal insufficiency, standard-dose (250 µg) synacthen test in secondary adrenal insufficiency and low-dose (1 µg) synacthen test in secondary adrenal insufficiency. 60 In clinical practice, a high cortisol cut-off is usually applied to achieve reasonable sensitivity in secondary adrenal insufficiency, which is more diagnostically challenging. In the meta-analysis by Dorin et al. 60 a cortisol cut-off concentration of 415 nmol/L was associated with a high number of false-negative (i.e. false-normal) test results. 60 While raising the cortisol cut-off concentration in this setting will reduce the number of false-negative test results, this is associated with increased false-positives. One previous study, using a cortisol cut-off value of 600 nmol/L, was associated with a false-positive rate of 40% and a false-negative rate of 10% in secondary adrenal insufficiency. 71
As published cut-off values have been obtained with a variety of cortisol assays and because differences in bias still exist with the current generation of assays, it is recommended that each laboratory establish the bias of their assay and consider this bias when adopting published cut-offs. 63 As the plasma cortisol response to synacthen is non-Gaussian, it has been suggested that the results obtained in the fifth percentile in each test be defined as the plasma cortisol cut-off value. In many cases it will still be necessary to interpret synacthen responses in the light of other supporting evidence including electrolyte disturbance, plasma ACTH or on clinical grounds. 63
By convention, a baseline (prestimulation) cortisol concentration is also measured. This provides additional information which is not required to interpret the synacthen test. A low baseline value corroborates a low poststimulation value; however, there are cases where a patient will have a baseline cortisol in an intermediate range and fail to achieve an adequate cortisol response following stimulation with synacthen. This is seen particularly in autoimmune adrenal insufficiency, where an inadequate synacthen response may present before basal cortisol concentrations fall. 72 There will always be a degree of overlap between normal responses in euadrenal patients and responses seen in hypoadrenal patients as outlined above. For this reason it is important to consider alternative methods of HPA testing if the clinical suspicion of adrenal insufficiency is high.
Choice of decision limits for cortisol assays
Most laboratories will not have established their own reference range or diagnostic cut-off data for the synacthen test or ITTs, and may instead be using values taken from the literature. As assays differ in standardization, accuracy and other aspects of analytical performance, laboratories should either seek to validate their diagnostic cut-offs or verify that their assay has similar bias to the assay(s) used to establish the published decision limits. It may be argued that laboratories should adjust their diagnostic cut-off values based on the verified bias of their assay compared with assays used to establish the published values. In view of differences in bias and the imprecision of assays, it may be safer to choose a higher cut-off value for an adequate cortisol value following stimulation tests, in order to reduce false-negative responses. It is also desirable that cortisol assays and cut-offs should in future be traceable to a reference method and/or reference standard. There is a need for national or international bodies to assist in this process.
Insulin tolerance test
The ITT is widely regarded as the gold-standard assessment of HPA reserve and has a particular utility for the detection of early secondary adrenal insufficiency and growth hormone deficiency. This test is based upon the stress response to hypoglycaemia (plasma glucose <2.2 mmol/L) following intravenous insulin administration (0.15 units/kg soluble insulin). Higher doses of insulin may be required in insulin-resistant patients including those with other pituitary disease such as acromegaly. 5 The ITT is contraindicated in patients with epilepsy, cardiovascular or cerebrovascular disease and should be reserved for use in specialist centres due to the potential risks associated with hypoglycaemia. As the ITT may precipitate an acute hypoadrenal crisis, it is generally contraindicated in those with basal cortisol concentrations <100 nmol/L, although this concept has recently been questioned, provided that other contraindications are observed and adequate monitoring is undertaken. 73 The ITT has been validated against the response to surgical stress with normal function of the HPA axis indicated by a cortisol response to stress above 500–600 nmol/L. 74 The ITT will elicit a limited number of false-positives and less commonly false-negative results. 75,76 However, a number of limitations concerning the interpretation of cortisol response to the ITT have been highlighted. 77 These include questions concerning the differing responses to surgical stress and plasma cortisol concentrations, variability between the range of commercially available cortisol assays (as for synacthen tests) and the relationship between the degree of hypoglycaemia and the cortisol response. Ultimately while in many centres the ITT is not routinely used, it has particular utility for patients whose short synacthen test is borderline or following recent pituitary surgery. 41,60,70,78
Metyrapone test (overnight test, OMT)
Metyrapone inhibits the adrenal cortex enzyme 11β-hydroxylase, thus inhibiting the conversion of 11-deoxycortisol to cortisol. Metyrapone 30 mg/kg is administered at 12 midnight and cortisol and 11-deoxycortisol concentrations are measured at 8:00 the following morning. An 8:00 serum cortisol concentration <200 nmol/L confirms drug compliance and adequate enzyme inhibition. 78,79 In normal individuals, postmetyrapone cortisol concentrations are low but 11-deoxycortisol concentrations rise significantly. Postmetyrapone 11-deoxycortisol concentrations below 200 nmol/L at 8:00 are consistent with a diagnosis of adrenal insufficiency. 41,57,78 The diagnostic yield was increased in one study by combining cortisol and 11-deoxycortisol concentrations and using a value below 450 nmol/L as consistent with a diagnosis of adrenal insufficiency. 80 This test does not carry the same risks as the ITT in terms of side-effects of hypoglycaemia, and in some centres it is performed on an outpatient basis. 79,80 As there is a potential risk of precipitating an acute hypoadrenal crisis during the metyrapone test in patients with established adrenal insufficiency, it is important to ensure that the patient has an adequate waking cortisol (>200 nmol/L) prior to performing the test. 58 However in one centre, 576 metyrapone tests were performed on 398 patients with no serious side-effects encountered. 79 The metyrapone test is not widely used in the diagnosis of adrenal insufficiency due to limited availability of metyrapone, and variations in local availability and accuracy of 11-deoxycortisol assays. 57
Managing diagnostic uncertainty
When interpreting test results an inadequate cortisol response (positive test) carries a very high likelihood of a diagnosis of adrenal insufficiency. Unfortunately, there is more difficulty with the interpretation of a negative test result as the possibility of adrenal insufficiency may not be completely excluded. If the pretest probability of adrenal insufficiency is sufficiently high then it is necessary to proceed to further testing. It is important to educate the patient to seek medical advice for reassessment and treatment in the event of clinical progression. In some cases where tests are negative but the pretest probability of adrenal insufficiency is sufficiently high commencing glucocorticoid replacement empirically is an option. 57,78 The decision to commence treatment relies upon a combination of clinical judgement and results of one or more biochemical assays that include a morning cortisol, postsynacthen stimulation cortisol and additional dynamic testing as required. The risks of empirical treatment need to be balanced against potential side-effects associated with lifelong glucocorticoid replacement.
Test comparisons
The standard (250 μg) and the low (1 μg) doses of synacthen lead to maximal stimulation of the adrenals. 68 Dorin et al. 60 found the synacthen test to have high sensitivity in cases of primary adrenal insufficiency but found no statistically significant difference in sensitivity between the standard-dose (250 μg) and the low-dose (1 μg) tests in secondary adrenal insufficiency. 60 There are conflicting data reporting that the standard-dose (250 μg) synacthen test is more sensitive than the low-dose (1 μg) synacthen test in detecting cases of secondary adrenal insufficiency. 78 It has been proposed that the incremental rise in cortisol between 0 and 15 min is a more sensitive test than the total cortisol concentration at 30 min in patients with iatrogenic adrenal insufficiency. 81 A number of studies have found the metyrapone test to be more accurate in identifying cases of adrenal insufficiency than either the standard-dose (250 μg) or the low-dose (1 μg) synacthen tests. 78,82–84 Overnight metyrapone and the standard- or low-dose synacthen testing showed similar sensitivity and specificity, but were not as reliable as the ITT in a recent Italian series. 85 However, in this series the 11-deoxycortisol cut-off concentration of 144 nmol/L used for the metyrapone test was very low, which may have reduced its sensitivity in this study. 85
Synacthen and ITTs
The usefulness of the short synacthen test has been evaluated when compared with the ITT in many studies but remains controversial in secondary adrenal insufficiency. While in one study only eight discrepancies were found in 200 patients evaluated for adrenal insufficiency, others have found a number of patients who show an adequate response to synacthen testing but who later fail the ITT. 60,71,86–91 The majority of these divergent responses occur in patients who show poststimulation cortisol values of between 500 and 650 nmol/L. 92,93 In one recent study of 148 patients at risk of developing secondary adrenal insufficiency due to cranial irradiation, pituitary surgery or concomitant glucocorticoid therapy, only one patient with a normal synacthen response (30 min cortisol >550 nmol/L) and one patient with a borderline result (510–550 nmol/L at 30 min) developed clinical adrenal insufficiency. 94 Based on these observations, the false-negative rate for the synacthen test is <1%. Furthermore, as the ITT is not 100% sensitive, there may be infrequent patients who pass the ITT but who are later found to have adrenal insufficiency. 76,93 A study of patients following pituitary surgery found six of a group of 41 who failed the low-dose (1 μg) synacthen test one week after surgery subsequently passed the ITT at 4–6 weeks postsurgery. 75 In contrast, three of 40 patients had a normal response to the standard-dose synacthen test but a subnormal response to the ITT.75. These authors concluded that both forms of the synacthen test are unreliable one week after pituitary surgery and should not be used. Consequently, it may be necessary to use one or more of the available tests for diagnosis during the work-up of patients with suspected adrenal insufficiency. For patients with known pituitary disease a combination of basal cortisol and ACTH values together with insulin tolerance testing is recommended, especially during the perioperative period.
Pituitary surgery
While the ITT is recognized as the gold-standard test for ACTH deficiency following pituitary surgery, there is increasing use of synacthen testing as this is more practical. 70 Some centres have observed a good correlation between synacthen and ITTs with false-negative rates of 3% (2 out of 69 who passed short synacthen test) in postpituitary surgery patients and a predictive value of 97%. 95 Others have found higher false-negative rates during testing, 75 particularly one week after surgery and it is generally accepted that the short synacthen test should not be used during the first 4–6 weeks following pituitary surgery. 57,61,63,75,96
As discussed earlier, while basal cortisol values above 500 nmol/L are usually considered to be consistent with adequate adrenal reserve, in the first week following pituitary surgery, basal cortisol values above 393–450 nmol/L were recently demonstrated to correlate with normal cortisol responses to hypoglycaemia. 73,75 Conversely, a 9:00 basal cortisol concentration <100 nmol/L during this period suggests adrenal insufficiency and predicts the likely need for long-term replacement. 97 However, more recently others have questioned this concept as up to one-third of patients with basal cortisol concentration below 100 nmol/L achieved adequate cortisol responses during a subsequent ITT. 73 On the basis of evidence from a number of studies using various cortisol assays, it has been recommended that patients with basal cortisol concentrations <100 nmol/L should have follow-up testing at three months postoperatively to determine the need for long-term replacement therapy. 97 One practical approach suggests hydrocortisone replacement is required for patients with basal cortisol concentrations between 100 and 250 nmol/L until further testing is completed. 75 For patients with intermediate basal cortisol concentrations (250–450 nmol/L) hydrocortisone replacement may only be required at times of stress until further HPA axis testing. 97 However, the potential risks and benefits for each patient should be considered on an individual basis according to local protocols.
Glucagon testing
The glucagon-stimulation test is occasionally used when insulin hypoglycaemia is contraindicated, but is not recommended due to variable cortisol responses and associated side-effects of nausea and vomiting. 70,98,99
Special cases
Acutely ill patients
The HPA axis responds differently to an acute or a chronic illness state. Acute responses lead to elevated cortisol concentrations causing gluconeogenesis with increased energy for use in vital organs, modulation of the immune system, a delay in anabolism and effects on the vasculature that may be beneficial. 100 However, the response of the HPA axis to prolonged critical illness may be detrimental and maladaptive. Protein catabolism, immune suppression, poor tissue healing, adipose tissue deposition and insulin resistance may develop with patients exhibiting persistent hypercortisolism. 100
Critical illness is associated with loss of normal circadian variation of cortisol, elevated ACTH and CRH concentrations. 101,102 Albumin and CBG concentrations are reduced leading to a rise in the proportion of unbound biologically active free cortisol. 103 The HPA axis is less sensitive to negative feedback in this scenario compared with healthy volunteers; excessive cytokine production may contribute, in association with decreased numbers and affinity of glucocorticoid receptors. 104,105 In chronic stress states cortisol concentrations remain high but ACTH concentrations fall. Atrial natriuretic peptide, endothelin, substance P and interleukin-6 have all been proposed as possible mediators of cortisol production. 102 Serum cortisol concentrations at the extremes of upper and lower assay reference ranges are associated with a poor prognosis in critical illness and septic shock. 106
Interpretation of cortisol responses in critical illness
No consensus opinion exists on the normal cortisol response during the stress of critical illness or on the definition of adrenal insufficiency in this state. Most authors have used baseline or stimulated responses obtained from healthy populations. However due to physiological differences in critical illness, attempts have been made to define normal responses in this setting. A range of basal serum cortisol (<500, <690, up to <1000 nmol/L) and stimulated (<500 nmol/L) serum cortisol cut-off values have been proposed to define relative adrenal insufficiency. 57,107 Most have relied upon the cortisol increment to synacthen with a rise of <250 nmol/L proposed as defining an insufficient cortisol response. This is associated with greater vasopressor requirements and increased mortality. 108 However, this interpretation has recently been called into question as serum cortisol increments are negatively correlated with baseline cortisol concentrations, and in those with maximal basal values and an intact HPA axis, a significant incremental response may be less likely. 108–111
Adrenal insufficiency in critical illness
Glucocorticoid replacement therapy has potential utility in critical illness as shown by a recent randomized controlled trial of patients with vasopressor-unresponsive septic shock that demonstrated a reduction in mortality and shock reversal in patients considered to have relative adrenal insufficiency. 108 Relative adrenal insufficiency is an ill-defined entity where cortisol production may be inadequate to control the inflammatory response or to meet an elevated metabolic demand and was arbitrarily determined by a post-synacthen-stimulated cortisol concentration increase <250 nmol/L. 112 Recent consensus from the Surviving Sepsis Campaign suggest that ACTH stimulation testing should not be used to identify the subset of patients who should receive hydrocortisone. 113 These recommendations are based upon observations that trial populations seemed to benefit independently of responses to ACTH and was confirmed in the recent CORTICUS trial. 109 Hydrocortisone therapy is perhaps best reserved for septic shock patients whose blood pressure responses are poorly responsive to fluid resuscitation and vasopressor therapy. 113
Serum-free cortisol concentrations
Conventional cortisol assays measure total cortisol (bound and unbound fractions) and may measure misleadingly low cortisol concentrations in hypoproteinaemic states such as sepsis. When compared with mass-spectrometry, conventional (immuno) assays may under- or over-estimate serum total cortisol concentrations leading to misinterpretation of adrenal reserve. Serum-free cortisol represents a more accurate index of adrenal reserve and is a more relevant measurement. This is illustrated by appropriate free cortisol concentrations and incremental responses in critical illness in patients, who would otherwise have low total cortisol concentrations, compared with controls. 114 Current data suggest that a baseline serum-free cortisol of <50 nmol/L and a postsynacthen stimulation of <85 nmol/L is consistent with adrenal insufficiency in critical illness. 114,115 More research is required in this area to define the reference range for free cortisol responses to stimulation and also to develop a standardized, reproducible, economical and practical assay for free cortisol. Direct measurement of plasma-free cortisol involves equilibrium dialysis of undiluted plasma specimens for 18 h followed by sensitive immunoassay. 114 Such methods are time consuming, technically difficult and require relatively large volumes of plasma and good, convenient assays suitable for routine use are not readily available. 116 Derivation of a free cortisol index is an alternative method to direct measurement of free cortisol, where the assay is not available, and is defined as the ratio of total cortisol to cortisol binding globulin concentrations. 116,117 This measurement accounts for variations in CBG and is therefore a potentially useful parameter that correlates with serum-free cortisol. However, there are limited data to define poststimulation free cortisol indices that represent a sufficient adrenal response.
Assessment and management of patients taking long-term corticosteroids
The most common cause of adrenal insufficiency in the general population arises from suppression of hypothalamic and pituitary secretion of CRH and ACTH, respectively, due to exogenous corticosteroid treatment. 41,42 During intercurrent illness patients such as these are at risk of developing acute adrenal insufficiency with features of electrolyte disturbance and haemodynamic compromise, as they are unable to mount an appropriate stress response. A prudent clinical approach is to assume that patients on long-term steroid therapy have a degree of adrenal insufficiency and to double the dose of exogenous corticosteroid for 3–5 days to cover the duration of the concurrent illness, until definitive testing has been completed at a later date. The risk of administering corticosteroids, or not, needs to be balanced against the risk of preventing acute adrenal insufficiency. These patients are also at risk of developing signs and symptoms of adrenal insufficiency during corticosteroid withdrawal. For this reason, it is important that careful counselling is provided regarding the management of illness as detailed above, and the possibility of symptoms of adrenal insufficiency during slow withdrawal of exogenous glucocorticoids.
In this setting it is important to recognize that all common corticosteroid preparations, except dexamethasone, may cross-react with cortisol assays. 42 As a result the risk of measuring falsely high serum cortisol concentration is increased, leading to potential false-negative results and inappropriate withdrawal of treatment. One strategy to prevent this scenario is to provide corticosteroid replacement using dexamethasone for the period of testing. Another is to perform testing at 9:00, having advised the patient to omit that morning's prednisolone or hydrocortisone dose such that testing is performed 24 h after the last administration of glucocorticoid. 42 There is a lack of evidence to guide choice of cut-off values. The synacthen test is the most convenient test to perform but the limitations of this test should be borne in mind. Prior to the development of adrenal atrophy, patients may elicit an adequate cortisol response to synacthen stimulation, but in acute stress, due to suppression of hypothalamic CRH and pituitary ACTH secretion, they may not do so. In this situation one may be falsely reassured by an appropriate synacthen response and the testing of the entire HPA axis with the ITT or metyrapone test is more appropriate. 78–80
Investigation of confirmed adrenal insufficiency
Adrenocorticotropin
In primary adrenal insufficiency, the hypothalamic and pituitary components of the HPA axis are intact and ACTH concentrations >100 ng/L are usually observed. 41,57 Elevated ACTH concentrations accompanied by normal cortisol concentrations represent a state of subclinical adrenal insufficiency that could progress to overt clinical adrenal insufficiency in the future. 118 ACTH concentrations within the reference range exclude primary adrenal insufficiency (in the absence of coexisting pituitary dysfunction), which is usually associated with early morning ACTH concentrations elevated above 100 ng/L. 57 In secondary adrenal insufficiency plasma ACTH concentrations are usually undetectable (depending on assay sensitivity); however, low or normal ACTH concentrations (<5–50 ng/L) may also be found. 41 It is important to note that basal ACTH concentrations alone are not considered to be useful for the diagnosis of adrenal insufficiency; their value is in the differential diagnosis. 58
ACTH assay
In the evaluation of patients for adrenal insufficiency, ACTH assays are used mainly in the differential diagnosis of primary from secondary causes. The commercially available assays most commonly used appear to have adequate imprecision and bias to make this distinction (see below). Immunometric assays have largely replaced radioimmunoassays, and have similar diagnostic sensitivities and specificities with improved analytical sensitivity and precision. 119–121
Imprecision
Currently ACTH assays in the UK are mainly from two suppliers, Siemens (California, USA) and Roche (Manheim, Germany). The total imprecision figures (CV%) given by the suppliers for Siemens Immulite 2500 assay is in the range 4.2–6.3% and for the Roche E170/Elecsys is in the range 3.5–5.5%. 122,123 The within-method between laboratory CV% from UK NEQAS distributions 103 and 104 in early 2008 ranges from 7.0% to 9.3% (average 8.5%) for the Siemens Immulite 2000 group and from 3.5% to 5.6% (average 4.9%) for the Roche Elecsys group, but both have a higher CV at very low ACTH concentrations. The overall all-laboratory CV% is 10–12%, except at very low ACTH concentrations. 124
Standardization and bias/accuracy
There are a number of purified synthetic preparations of ACTH1−39 but there is no generally accepted international standard. Two of these preparations, from the National Hormone and Pituitary Program and the National Institute for Biological Standards and Control (code 74/555), have similar immunoreactivity but differ significantly in biological potency. 121 In the absence of an agreed international standard for ACTH, it is difficult to draw conclusions regarding assay bias. The predominant assay method in use in the UK is the Siemens Immulite. In the UK NEQAS, bias from the group mean is strongly influenced by this. 122 In the NEQAS results for early 2008, the Roche group of laboratories had a positive bias of 20–25% relative to the Siemens group.
Specificity
Certain ACTH fragments and precursors may have a small cross-reactivity in ACTH assays, but this should not be a problem in the differential diagnosis of adrenal insufficiency. In the Roche assay, ACTH1–24 in high concentrations has a negative interference in the assay, but this would not be a problem unless ACTH treatment had been given.
Reference range
The ACTH reference ranges derived from specimens taken from 7:00–10:00 in apparently healthy subjects (2.5 to 97.5 percentiles) quoted by Siemens for the Immulite is from limit of detection 5 to 46 ng/L and for the Roche E170/Elecsys it is from 5.6 to 76 ng/L. 122,123
ACTH specimen requirements and stability
ACTH in blood specimens has been widely considered to be labile and therefore stringent specimen requirements have been recommended. 122,123 Specimens for ACTH analysis should be taken into chilled blood collection tubes containing ethylenediaminetetraacetic acid as anticoagulant. Tubes with other additives such as gel separators should be validated before use. The blood specimens should then be placed on ice and centrifuged in a refrigerated centrifuge. Plasma should be stored frozen in plastic or siliconized glass tubes at −20°C or lower. ACTH is stable in specimens stored frozen for 30 days, but repeated freezing and thawing should be avoided.
Making the differential diagnosis
Prolonged ACTH stimulation
The prolonged ACTH stimulation test (250 μg synacthen given intravenously for 8 h/day over a 48-h period) can differentiate primary from secondary or tertiary adrenal insufficiency. While the test is able to recover function in atrophic adrenals, in hypothalamic–pituitary conditions it is rarely used due to the practicalities and cost of hospital admission. Usually plasma ACTH concentrations, in conjunction with clinical presentation, is sufficient to determine the differential diagnosis.
CRH stimulation test
Human or ovine CRH is administered intravenously and cortisol responses are measured at baseline and at 15, 30 and 60 min following stimulation. ACTH responses to CRH are more commonly employed in the differential diagnosis of hypercortisolism but can be used in the evaluation of adrenal insufficiency. 41,125 The CRH test has utility for differentiating pituitary from hypothalamic aetiologies of adrenal insufficiency. 5 The CRH stimulation test has limited usefulness due to its cost, availability and limited data defining responses in normal and hypoadrenal groups. 5,58 Factors such as corticosteroid administration or desmopressin therapy complicate the interpretation of testing. 126 In one study the CRH stimulation test had high sensitivity (95%) for detection of adrenal insufficiency but had low specificity at 33%. 127 Added to these difficulties there is evidence that the CRH stimulation test may not be any more reliable than a waking serum cortisol alone. 87 Therefore, in general, the CRH stimulation test is not routinely recommended in the differential diagnosis of adrenal insufficiency. 5,57,127
Renin and aldosterone concentrations
In primary adrenal insufficiency there is destruction of the adrenal cortex leading to decreased secretion of mineralocorticoids as well as glucocorticoids. Aldosterone concentrations are typically low and are associated with elevated renin concentrations in this setting. However, in secondary adrenal insufficiency the renin–angiotensin system can function normally and aldosterone concentrations usually fall within the normal range. 5
Autoantibodies
At the time of onset of clinically apparent autoimmune adrenal insufficiency adrenal antibodies are detected in more than 90% of patients. 24 The presence of autoantibodies will often predate the onset of clinically apparent adrenal insufficiency. Adrenal insufficiency is associated with a range of steroid-cell autoantibodies that include 17α-hydroxylase antibodies, P450 side-chain cleavage antibodies and 3-β hydroxysteroid dehydrogenase (3-βHSD) antibodies. 128 The presence or absence of adrenal cortex antibodies (ACA) and 21-hydroxylase antibodies (21-OH) is used to assist in the diagnosis of autoimmune adrenal insufficiency in clinical practice. ACA are measured by immunofluorescence and 21-OH by radio-immunoprecipitation assay. 72 The presence of both ACA and 21-OH makes a diagnosis of autoimmune primary adrenal insufficiency likely, with up to 99% probability. 129 In one prospective study, ACA-positive patients who were negative for 21-OH antibodies did not develop adrenal insufficiency over six years of follow-up. 72 The titre of autoantibodies is known to decline over time, which probably arises due to progressive destruction of the target tissue. In recent-onset adrenal insufficiency ACA are present in 80–90% of patients, dropping to <80% after two years and to 10% after 15 yr. 129
Imaging
In unequivocal cases of autoimmune adrenalitis, it is not usually necessary to proceed to imaging. 57 In other forms of primary adrenal insufficiency, either computerized tomography (CT) or magnetic resonance (MR) imaging may be used to image the adrenal glands. In autoimmune adrenal insufficiency, imaging reveals small or absent adrenal glands. In infiltrative causes such as infection, haemorrhage, neoplasm, haemochromatosis and sarcoidosis radiological findings may be characteristic. 5 In late stages of tuberculous adrenalitis calcifications develop and these are more readily identified on CT than on MR imaging. 130 MR imaging has the advantage of a reduced radiation exposure but local availability may limit its use. In suspected secondary adrenal insufficiency pituitary MR imaging is indicated. 5
Adrenal biopsy
CT-guided adrenal biopsy is not usually required except in the differential diagnosis of adrenal insufficiency in patients with a presentation suspicious of metastases, in whom a known primary remains unidentified and in whom phaeochromocytoma has been excluded. 41 In this setting the presence of heterogenous mass with high attenuation values is suggestive of neoplasia. 131 Adrenal biopsy may have additional diagnostic utility in patients with other infiltrative aetiologies.
Approach to investigation
Adrenal insufficiency may present with multiple non-specific symptoms and signs to clinicians working in all fields of medical practice. A delay in making the diagnosis or misdiagnosis may be complicated by potentially life-threatening consequences. As we have explored, accurate diagnosis of adrenal insufficiency in adults can be challenging as each method used for HPA axis testing is associated with variable sensitivity and specificity for detection of adrenal insufficiency. The short synacthen test is the most appropriate and practical initial investigation for suspected primary adrenal insufficiency. In inconclusive or borderline cases, the ITT or overnight metyrapone test may be considered as second-line investigations.
In the special situation of the early postoperative period following pituitary surgery, the short synacthen test should be avoided. In this setting a combination of basal cortisol concentrations, the ITT and overnight metyrapone test have more utility for determining the need for treatment. However, there is increasing evidence that the synacthen test is an effective tool for HPA testing in patients with established pituitary disease at longer-term follow-up, provided that an assessment of growth hormone reserve is not required. A higher cortisol cut-off concentration may be used in this situation, with further testing for patients with borderline responses.
The interpretation of cortisol responses in critical illness and in sepsis is particularly challenging due to measured differences in total and biologically active free cortisol. Recent consensus has called into question the utility of testing for adrenal insufficiency in this setting. Based upon current evidence, a decision to commence hydrocortisone in critical illness should be taken on clinical grounds alone, such as in patients with vasopressor unresponsive septic shock.
The interpretation of cortisol and ACTH concentrations is dependent on methodological factors, such that reference values for diagnostic cut-offs will vary between laboratories, depending on the choice of assay methodology (see Table 1). While ideally each laboratory should use cut-offs validated for their own choice of cortisol assay, in practice this is difficult.
Biochemical diagnosis of adrenal insufficiency: (a) primary adrenal insufficiency and (b) secondary adrenal insufficiency
ACTH, adrenocorticotropin; i.v., intravenous; i.m., intramuscular
In conclusion, the decision to treat a patient with suspected adrenal insufficiency should be confirmed by formal testing of the HPA axis. Clinicians should be aware of the limitations of the test performed and also of assay imprecision. We suggest that it is important to correlate the clinical situation with results of biochemical assays. Given the risks associated with failure to diagnose adrenal insufficiency, in situations of high clinical probability and inconclusive HPA axis testing, an empirical trial of treatment with glucocorticoids may be required.
DECLARATIONS
