Abstract
Background
We have previously demonstrated that heart-type fatty acid binding protein (H-FABP) is an independent prognostic marker for survival after acute coronary syndrome (ACS). This study aimed to define the 99th centile values for H-FABP as determined with two different assays, and to study the relationship with age, gender and renal function.
Methods
H-FABP was measured on redundant routine outpatient samples using the MARKIT-M (Dainippon) and the Evidence Investigator (Randox) assays.
Results
Two hundred and forty-two subjects with Siemens Ultra-TnI value <0.045 μg/L (99th centile) were studied. In all, 174 subjects had estimated glomerular filtration rate (eGFR) >60 mL/min. The 99th centile values for subjects with eGFR >60 mL/min for the Evidence Investigator H-FABP were 5.3 and 5.8 μg/L and for the MARKIT-M H-FABP were 8.3 and 9.1 μg/L in female and male subjects, respectively. There is an increase in H-FABP with age in subjects with normal renal function for both assays. Gender comparison showed no significant difference for either assay. Comparison of samples showed that subjects with eGFR <60 mL/min showed a median increase of 0.71 μg/L with Evidence Investigator assay and 1.09 μg/L with MARKIT-M assay compared with subjects with eGFR >60 mL/min. Calibration differences were confirmed by cross measurement of calibrators and recombinant H-FABP.
Conclusions
We have defined the 99th centile values for H-FABP in a population of primary and secondary care outpatients that can be used to risk stratify patients with ACS. We have confirmed that H-FABP increases with renal dysfunction and age, but have not confirmed the gender difference previously reported.
Introduction
Heart-type fatty acid binding protein (H-FABP) is a low molecular weight protein involved in the intracellular uptake and buffering of long chain fatty acids in the myocardium. It was first noted to be a marker of myocardial infarction in 1988. 1 Early polyclonal assays were relatively non-specific and until a couple of years ago, there were few published clinical studies examining the diagnostic and prognostic utility of H-FABP. However, assays have now improved and H-FABP is receiving more attention as a cardiac biomarker. We have previously demonstrated in a series of 1448 patients presenting with acute coronary syndrome (ACS) that H-FABP taken at 12–24 h after the onset of symptoms offers superior prognostic information regarding mortality than troponin I (Beckman Access). 2 Similar data on the prognostic value of H-FABP in ACS have been published by others. 3,4 H-FABP is now recognized to be the earliest available marker for ACS detection. 5
One of the requirements for clinical use of a biomarker for ACS would be determining the 99th centile value for the normal population that can act as a decision point for abnormality. We have therefore studied two commercially available H-FABP assays with good analytical performance to determine their 99th centile values in a population of primary and secondary care outpatients and, in addition, have determined the relationship of H-FABP with age, gender and renal function.
Methods
Redundant sera from routine laboratory samples were collected after the analysis was completed. Samples were excluded if they originated from inpatient locations or renal or cardiac outpatients. Samples were anonymized after collection of age, gender and clinical details. All samples were stored at −70°C prior to analysis. This study was approved by the Leeds Research Ethics Committee.
H-FABP was measured using the MARKIT-M (Dainippon Pharmaceutical Co, Osaka, Japan) and Evidence Investigator assays (Randox Laboratories Ltd, Crumlin, Co Antrim, UK). The MARKIT-M assay is a two-step, manual, non-competitive enzyme-linked immunosorbent assay using two distinct mouse anti-human H-FABP monoclonal antibodies. The inter-assay coefficient of variation (CV) at 6 μg/L was 6.3%. The Evidence Investigator assay is a non-competitive chemiluminescent assay using two mouse anti-human H-FABP monoclonal antibodies. The inter-assay CV at 5.8 μg/L was 5%.
Troponin I and creatinine were measured on all samples, in accordance with the manufacturer's protocols. Troponin was measured using TnI-Ultra™ reagent with the Advia Centaur system (Siemens Medical Diagnostics Ltd, Camberley, UK) and creatinine was measured using O'Leary reagent (Synermed Europe Ltd, West Sussex, UK) on the Advia 2400 system (Siemens Medical Diagnostics Ltd). Estimated glomerular filtration rate (eGFR) was estimated using the MDRD (Modification of Diet in Renal Disease Study) formula with appropriate correction factor for the O'Leary reagents. 6
Recombinant H-FABP was a kind gift from Professor J F Glatz, University of Maastricht.
Statistical analysis was performed using the Analyse-it add-in package (version 2.09) for Microsoft Excel (Analyse-it, Leeds, UK,
Results
Single samples from 242 individuals with sufficient serum for all analyses were included (295 samples were collected but 53 had insufficient volume). All subjects had an ultra-troponin I value <0.045 μg/L – the 99th centile value from a normal population (n = 295, unpublished data). There were 174 samples from subjects with eGFR >60 mL/min (F:M 109:65) with ages of female subjects being 51 (45) yr and male subjects 57 (21) yr (median, IQR).
The two assays correlated well for samples from subjects with eGFR <60 mL/min (Spearman regression Rs = 0.85, P < 0.0001, n = 68) and with all degrees of renal function (Rs = 0.84, P < 0.0001, n = 242). A comparison of all subjects using an Altman–Bland difference plot is shown in Figure 1.
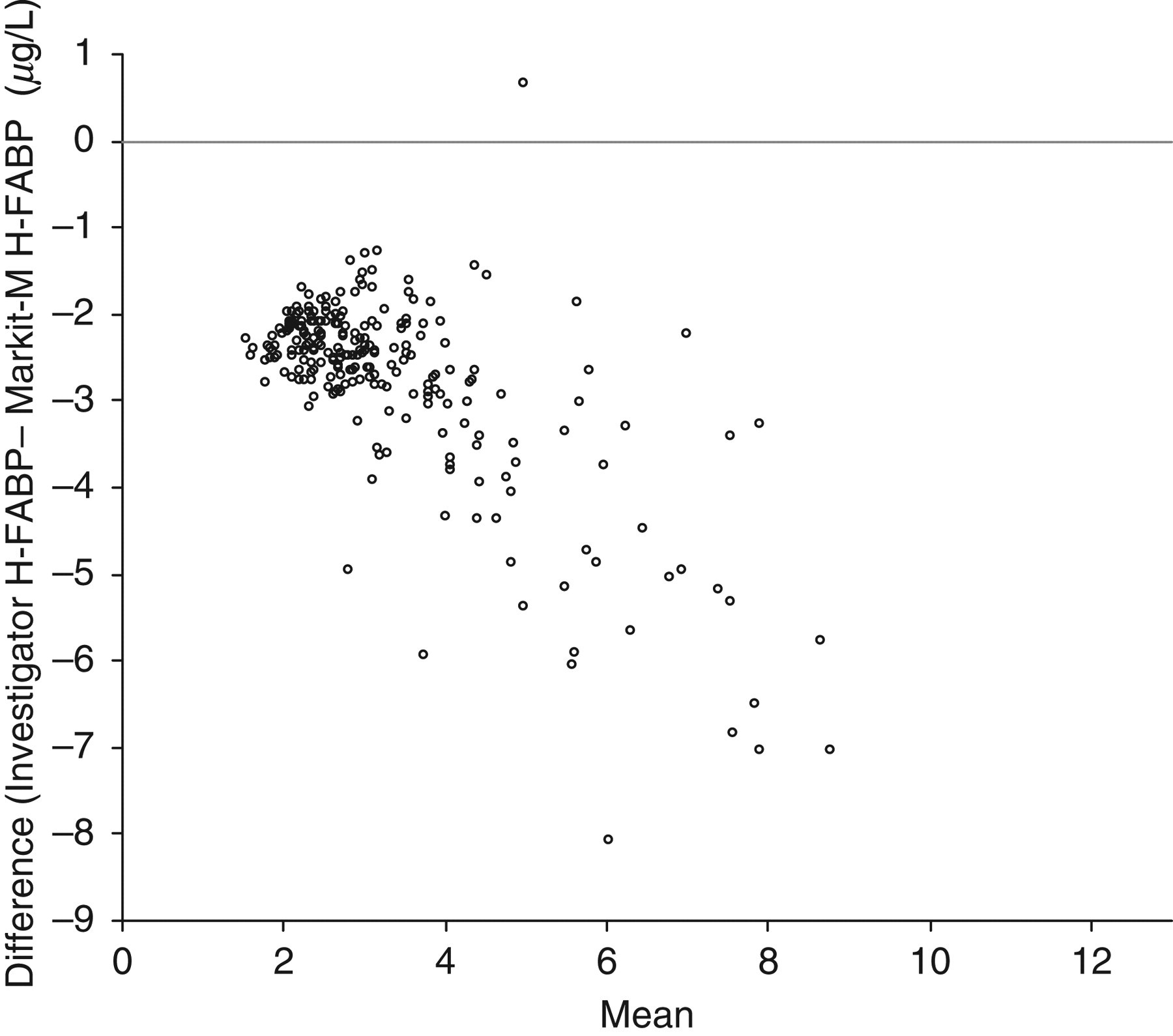
Bland–Altman plot comparing Evidence Investigator and MARKIT-M assays for H-FABP for all subjects (both genders and all degrees of renal function)
The 99th centile values in subjects with eGFR >60 mL/min for the Evidence Investigator H-FABP were 5.3 and 5.8 μg/L and for the MARKIT-M H-FABP were 8.3 and 9.1 μg/L in female and male subjects, respectively. Gender comparison showed a tendency to higher values in male subjects but this did not achieve significant difference with either assay (Mann-Whitney U test, P > 0.2) (see Figure 2). There is an increase in H-FABP with age in subjects with normal renal function for both assays (Rs = 0.48, P < 0.0001, n = 174).
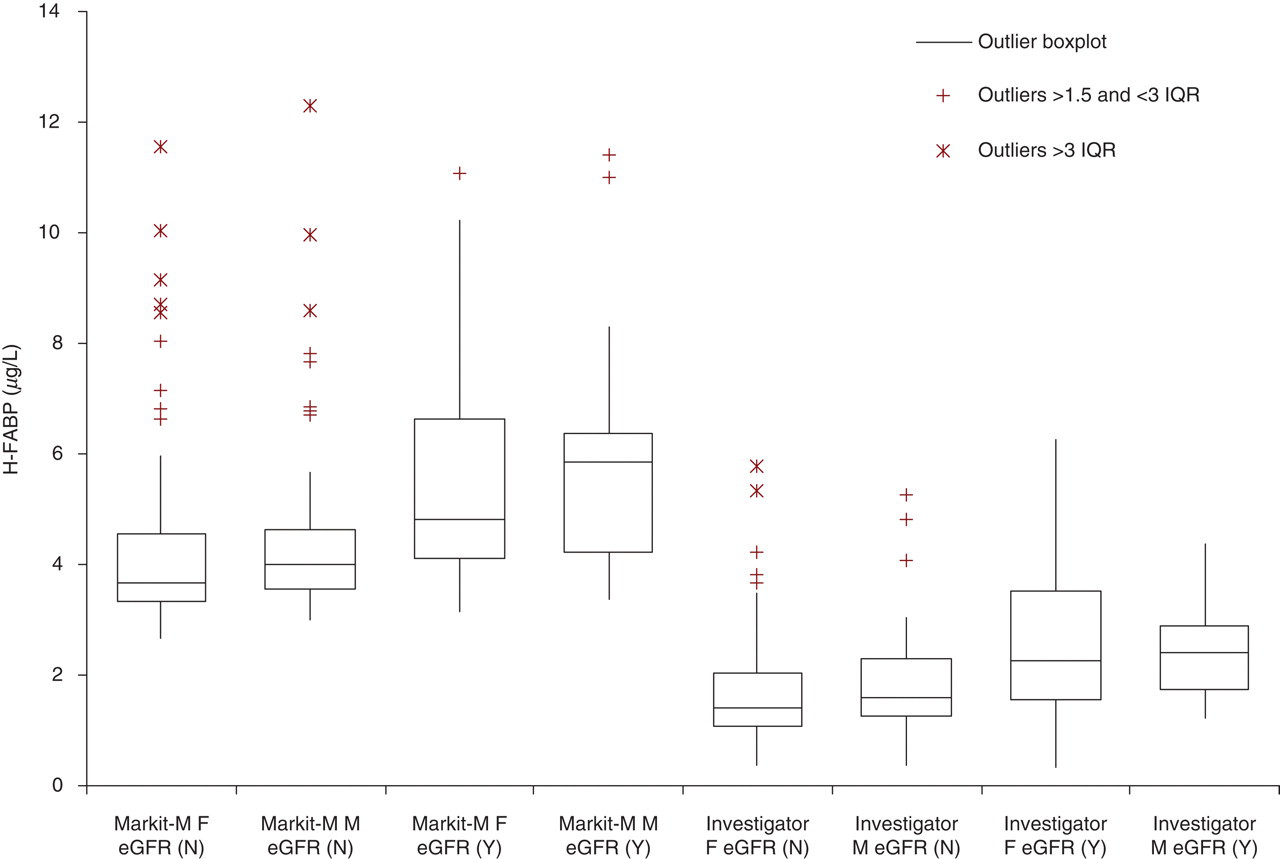
H-FABP distributions subdivided by assay method, gender and renal function. Box and whisker plots (median, interquartile range [IQR] outliers as described in the inset). Methods: Investigator or Markit-M; M or F (male or female). Estimated glomerular filtration rate (eGFR) N ≥ 60 mL/min, Y ≤ 60 mL/min
Comparison of samples from subjects (n = 68) with impaired renal function (eGFR <60 mL/min) showed higher values than from subjects with normal renal function. The difference between the median values was 0.71 μg/L (Mann-Whitney U test P < 0.0001) with the Evidence Investigator assay and 1.09 μg/L (Mann-Whitney U test P < 0.0001) with the MARKIT-M assay.
Both assay systems were used to measure recombinant H-FABP and showed a linear relationship. The Evidence Investigator H-FABP was equivalent to [0.27 × rH-FABP + 1.30] and the MARKIT-M H-FABP was equivalent to [0.70 × rH-FABP − 5.03]. Cross measurement of the Evidence calibrators on the MARKIT-M gave values of 168 ± 45% (mean ± SD) of expected values and MARKIT-M on Evidence gave values of 67 ± 13%.
Discussion
We have previously demonstrated that H-FABP is an independent prognostic marker for survival after an ACS. The purpose of this study was to define the cut-off values that should be used to guide the identification of individuals at increased risk after ACS. Serum H-FABP has been recognized since 1988 as a marker of acute myocardial infarction, but has never achieved acceptance either because there was insufficient outcome data to support its use or because the analytical performance was inadequate. 5 Therefore, as there is no international standard, we have evaluated two commercially available assays with good laboratory performance.
This is the first study to report on the 99th centile values of H-FABP in a sizeable Caucasian population spanning all age groups (including those aged above 70 yr) and after excluding those with renal dysfunction. Our data have defined the 99th centile value for both assays and noted the linear relationship between the two assays. The constant bias and comparison with recombinant H-FABP 7 suggests that the difference between assays is due to a calibration difference rather than a difference in antibody specificity. Should H-FABP become more widely used in clinical practice as the clinical data suggest, then an international standard will need to be prepared.
The 99th centile values found in this report are consistent with the values reported for the Evidence Investigator assay by Di Serio et al., 8 who reported a mean value of 3.3 μg/L and a range of 0.6–11.1 μg/L. Our previous report suggested that the 99th centile value using the MARKIT-M assay was 5.8 μg/L. We felt it was necessary to revise this as the data were collected on a relatively young cohort of subjects with a mean age of 45 yr in whom neither renal function nor troponin status was studied (unpublished data). Moreover, they represented a healthy group of blood donors rather than a population who were already being seen in a clinical setting. Finally, they would not have been entirely comparable with patients complaining of chest pain who in our experience have median age >72 yr. 2
Our data have also confirmed that H-FABP increases with both increasing age 9,10 and renal function 11 but does not show the clear gender difference noted by Pelsers et al. 9 or Niizeki et al. 10
We have defined the 99th centile value for H-FABP in a population of primary and secondary care outpatients. It is of note that H-FABP has tighter precision at the decision-making value than troponin offering the potential for improved risk stratification in patients with ACS. We have confirmed that H-FABP increases with renal dysfunction and age, but have not confirmed the difference between genders in the UK population that has been reported previously in subjects from the Netherlands and Japan. Finally, we expose the need for an international standard for the harmonization of H-FABP assays.
DECLARATIONS
