Abstract
Background
Point-of-care-testing (POCT) of haemoglobin Alc (HbA1c) is popular due to its fast turnaround of results in the outpatient setting. The aim of this project was to evaluate the performance of a new HbA1c POCT analyser, the Bio-Rad in2it, and compare it with the Siemens DCA 2000, Bio-Rad Variant II and Roche Tina-quant HbA1c Gen 2 assay on the cobas c501.
Methods
Imprecision of the four methods were compared by computing total imprecision from within-run and between-run data. A total of 80 samples were also compared and analysed by Deming regression and Altman–Bland difference test.
Results
Study of total imprecision of the in2it at HBA1c levels of 6.0% and 10.4% produced a coefficient of variation (%CV) of 3.8% and 3.7%, respectively. These results were more favourable as compared with the DCA 2000 but did not match the low imprecision of the central laboratory methods, the Bio-Rad Variant II and the Roche cobas c501. Comparison between the in2it and the central laboratory analysers, Bio-Rad variant II and cobas c501, revealed positive bias of 12% and 10%, respectively, supported by corresponding Deming regression equation slopes of +1.18 and +1.14. Comparison between the DCA 2000 and the central laboratory analysers revealed a bias that became increasingly positive with rising HbA1c concentrations with Deming regression analysis also revealing proportional and constant differences.
Conclusions
The in2it is a suitable POCT analyser for HbA1c but its less than ideal precision performance and differences with the central laboratory analysers must be communicated to and noted by the users.
Introduction
The Singapore National Health Survey conducted in 2004 showed that 8.2% of adults had diabetes mellitus and a further 12% demonstrated impaired glucose tolerance. 1 The Diabetes Control and Complications Trial (DCCT) 2 and UK Prospective Diabetes Study (UKPDS) 3,4 showed that diabetic patients with poor control over the hyperglycaemia condition had increased risks of developing life-threatening complications such as ketoacidosis, retinopathy, renal failure, neuropathy and atherosclerosis. Long-term glycaemic control is vital to the reduction of diabetes-associated complications and mortality.
The level of glycated haemoglobin (GHb), primarily the haemoglobin A1c (HbA1c) fraction, has been shown by the DCCT and UKPDS to correlate with the risk of developing diabetes-related complications. Measurement of HbA1c provides the clinician with an indication of a patient's average glycaemic control over the past two to three months and can be used for the assessment of glycaemic control and treatment of diabetic patients.
John et al. 5 in his recent paper on HbA1c standardization reviewed the methodology available for HbA1c measurement since the 1970s. Methods that are based on charge differences of the haemoglobin fractions include cation-exchange high-performance liquid chromatography (HPLC) and electrophoresis. On the other hand, affinity HPLC and immunoassay are methods based on structural differences. There are a wide variety of kits and methodologies available today. Some of these methodologies, particularly affinity HPLC and immunoassay, are employed in point-of-care-testing (POCT) devices for the measurement of HbA1c. POCT of HbA1c using a simple and portable device provides quick results to the clinicians at the site of patient care. However, just as HbA1c analysis is moving into the arena of global standardization, there is a need to ensure that POCT measurement of HbA1c provides reliable results that are comparable to central laboratory analysis.
Our tertiary care hospital's clinical biochemistry laboratory was measuring HbA1c using affinity HPLC with the Bio-Rad Variant II (Bio-Rad Laboratories, Hercules, CA, USA) and recently changed to immunoassay on the Roche cobas c501 (Tina-quant HbA1c Gen. 2, Roche Diagnostics, Indianapolis, IN, USA). In addition, our hospital's diabetes centre utilizes immunoassay on a POCT device Siemens DCA 2000 (Siemens Medical Solutions Diagnostics, Dublin, Ireland, UK). Recently, a new POCT device, in2it, manufactured by Bio-Rad (Bio-Rad Laboratories, Hercules, CA, USA), has become available. Here, we describe the analytical performance of the new in2it and compare it with the DCA 2000 and central laboratory methods on the Bio-Rad Variant II and Roche cobas c501.
Materials and methods
POCT analysers for HbA1c analysis
The Bio-Rad in2it is a cartridge-based analyser that utilizes boronate affinity chromatography that separates the glycated fraction of haemoglobin from the non-glycated fraction. An algorithm is used to calculate a standardized HbA1c value from the GHb result. The Siemens DCA 2000 uses latex immunoagglutination inhibition technology that is specific for GHb molecules having the same first few amino-acid residues as the glycated N-terminal of the β-chain of HbA1c. The analysis time on the Bio-Rad in2it is 10 minutes whereas the DCA 2000 requires about six minutes to complete one test.
Both POCT methods are standardized to the International Federation of Clinical Chemistry (IFCC) reference system for HbA1c and aligned to the DCCT standards via the National Glycohaemoglobin Standardization Program (NGSP).
Central laboratory analysers for HbA1c analysis
The Bio-Rad Variant II utilizes boronate affinity HPLC that separates the GHb from the non-glycated fraction. Similar to the in2it, a standardized HbA1c value is calculated from the GHb result via an algorithm. The Roche cobas c501 uses the Tina-quant HbA1c Gen. 2 method, which is a latex-enhanced competitive turbidimetric immunoassay that measures all haemoglobin variants that are glycated at the β-chain N-terminal and that have antibody-recognizable regions identical to that of the HbA1c. This second generation of Tina-quant immunoassay does not require sample pretreatment to remove labile HbA1c and tetradecyltrimethylammonium is used to remove interference from leukocytes.
Both central laboratory methods are also standardized against the IFCC reference method and traceable to DCCT and NGSP.
Evaluation studies on the Bio-Rad in2it
Analytical imprecision of the Bio-Rad in2it assay was assessed by analysis of two levels of in2it A1c quality controls. Within-run (n = 21) and between-run (n = 21) studies were performed and total laboratory imprecision was calculated. Imprecision data previously established in-house for DCA 2000, Bio-Rad Variant II and Roche cobas c501, using quality control material from the respective manufacturers, are also presented to allow comparison.
Comparison studies
Our subjects were 80 diabetic patients who were followed-up at our diabetes centre. The study was approved by the Institutional Review Board of the Singapore General Hospital and all subjects gave informed consent for participation. Capillary blood was collected from finger prick for testing on both the DCA 2000 and the Bio-Rad in2it. Venous blood was collected in ethylenediaminetetraacetic acid tubes for analysis on the Bio-Rad Variant II and Roche cobas c501. All venous samples were stored at 2–4°C until analysis, which was not later than four days from the date of collection.
Statistical analysis
Statistical analysis was performed on the Microsoft Excel and Analyse-It Statistical Add-on for Excel (Analyze-It Software Ltd, Leeds, UK).
Results
Data from the imprecision study on the Bio-Rad in2it are tabulated in Table 1. Data previously established in-house for DCA 2000, Bio-Rad Variant II and Roche cobas c501, using quality material from respective manufacturers, are also presented to allow comparison.
Imprecision of HbA1c assays
CV, coefficient of variation
Data from the method comparison study was analysed using the Altman–Bland difference test and Deming regression analysis. Log-transformation of data was performed for the Altman–Bland plots to mitigate proportional relationships between the absolute differences and averages (Figure 1). Antilogs of resultant Altman–Bland mean differences and 95% limits of agreement are then tabulated together with the Deming regression statistics (Table 2).
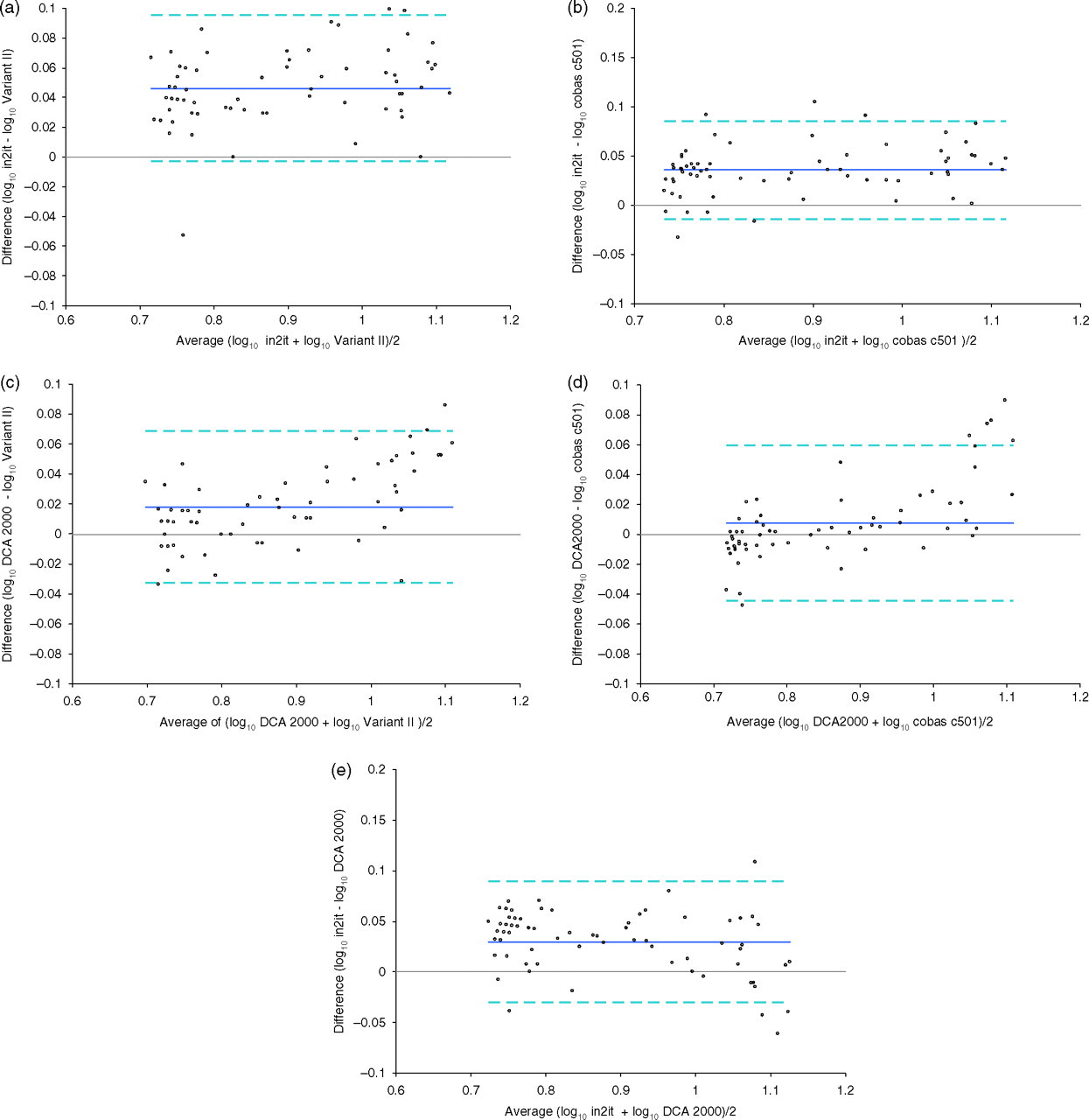
Altman–Bland Log–difference plots. (a) Agreement between in2it and Variant II. (b) Agreement between in2it and cobas c501. (c) Agreement between DCA 2000 and Variant II. (d) Agreement between DCA 2000 and cobas c501. (e) Agreement between in2it and DCA 2000
HbA1c method comparison: Deming regression analysis and Bland–Altman difference test
Discussion
It has been recommended that HbA1c assays should have a total intralaboratory imprecision (coefficient of variation, CV) of <5% (ideally <3%). 6,7 Our study revealed total imprecision of the new in2it to be satisfactory (CVtotal <5%) and more favourable as compared with the DCA 2000 (CVtotal 3.4–7.3%). Both POCT methods, however, could not match the low imprecision of the central laboratory methods, with both the Bio-Rad Variant II and Roche cobas c501 achieving CVtotal of 3% or less. It would have been ideal to concurrently study imprecision on all four analysers using pooled patient samples. The authors acknowledge the limitations of (1) comparing imprecision data from the in2it with historical imprecision data from the DCA 2000, Bio-Rad Variant II and Roche cobas c501 and (2) using manufacturer's quality control material instead of patient samples. Constraints of storage and stability of pooled whole blood samples prompted the latter strategy.
Comparison between the in2it and the central laboratory analysers, Bio-Rad variant II and cobas c501, revealed a positive bias of 12% and 10%, respectively. Deming regression analysis supported this observation with corresponding regression equation slopes of +1.18 and +1.14. Difference between the DCA 2000 and the central laboratory analysers was proportional to the level of HbA1c, with bias becoming increasingly positive with rising HbA1c concentrations. Furthermore, Deming regression analysis revealed proportional differences of 1.12% and constant differences of −1.18% HbA1c and −1.36% HbA1c, respectively. The new in2it also registered a 7% positive bias when compared with the DCA 2000. These differences are significant and indicate potential calibration differences between the POCT and central laboratory methods.
Interference from haemoglobin variants was not evaluated in our study. Literature 8–11 and manufacturer's data have shown that common haemoglobin variants (such as HbF, HbS, HbC and carbamylated-Hb) exert negligible interference on boronate affinity chromatographic analysis of glycated haemoglobin. With regards to immunoassays, both Siemens and Roche claim their assays on the DCA 2000 and Tina-quant are not affected by HbF of <10%. Literature also shows that the DCA 2000 and Roche Tina-quant immunoassays are not significantly affected by HbF of < 30% and suffer from negligible interference from other common haemoglobin variants such as carbamylated-Hb, HbAS and HbAC. 8,9,11,12 There are a myriad of uncommon haemoglobin variants and existing literature reveal that boronate affinity chromatographic and immunoassay methods of HbA1c analysis are minimally or unaffected by them. 8,9
The increasing interest and application of POCT devices arose from a desire to improve clinical services through a shorter turnaround time for laboratory tests. It has been recognized that POCT results should be comparable to results from analysers used in the central laboratory, especially if both categories of analysers are used within the same health-care organization. The Bio-Rad in2it is simple to use and can be easily operated by the nurse or doctor in the POCT setting. The Bio-Rad in2it should be able to compliment the busy central laboratory when used with a comprehensive quality assurance, quality control and documentation programme, coupled with thorough operator training. It is very important, however, that the less than ideal precision performance of this POCT device and the differences with the central laboratory analysers must be communicated to and noted by the users.
DECLARATIONS
