Abstract
Background
Tumour markers are frequently used in clinical practice and the reason for ordering varies considerably and often seems to be inappropriate. We carried out a survey of Italian laboratories on their current pattern of use.
Methods
Forty-four laboratories located in health-care institutions with inpatient beds were surveyed about the organizational, clinical and methodological aspects of tumour markers ordering.
Results
Thirty-one laboratories (70%) filled in and returned the questionnaire. Overall, 977,786 tumour marker tests were scrutinized. The pattern of tumour marker use did not seem to be influenced by the institutional setting, by availability of oncology facilities or by adoption of clinical guidelines. In addition, the information flow from clinicians to the laboratory and vice versa was poor and informal.
Conclusions
Monitoring tumour marker pattern use can provide valuable information for health-care decision makers, highlighting potential inadequacies in laboratory services but also identifying problems in other areas of health-care delivery that could benefit from educational programmes.
Introduction
Tumour markers are widely used in laboratory practice. Testing for tumour markers should only be performed if it is beneficial in terms of improving the patient's health and if there is a reduced overall cost of care. 1 However, available data show that both the pattern and the reason for tumour marker ordering vary considerably. 2–5 By the end of the 1980s, our general laboratory became more involved in monitoring the tumour markers' use in clinical practice. 6 In 1987, we verified whether the carcinoembryonic antigen (CEA) tests were properly requested in order to identify any bias of cost due to inadequate use. 7 Two thousand and six hundreds seventy-one CEA orders were evaluated, of which 43% appeared to be inadequate. A similar scenario was also seen for other tumour markers (CA125, AFP and PSA) in a survey performed in a small sample of Italian general practitioners (from Barbato PC et al., La Società Italiana di Medicina Generale. Vol. 3 2003). Performing unnecessary tumour marker tests may expose patients to potential risks such as hazardous treatments or may induce fear and anxiety in patients such as for a false-positive result, and finally it may represent a useless cost for the health-care system. Monitoring the pattern of tumour marker use is a tool for identifying potential inadequacies in all stages of the laboratory practice, from the test request (pre–preanalytical phase), to the collection and specimens' handling (preanalytical phase), to sample assay (analytical phase) and to the test results' report and interpretation (postanalytical phase).
Last but not at least, the role of the clinical laboratory in supporting clinical trials in oncology must be considered as these are an important source of clinical requests for tumour marker measurement. Thus, several variables related to organizational, clinical and methodological aspects of tumour marker requests should be evaluated to determine whether and which of these areas are problematic and which are most likely to benefit from educational programmes.
In order to evaluate the current use of tumour markers in Italy, we surveyed a sample of health-care institutions.
Methods
A questionnaire developed by the members of Working Group on Tumour Markers of Italian Society of Clinical Chemistry and Laboratory Medicine (SIBioC) was discussed and further refined by the Clinical Guidelines Group of the European Group on Tumour Markers (EGTM). The questionnaire consists of five sections. The first concerns general aspects of the health-care institutions surveyed (i.e. type of institution, type of oncology facilities, whether accredited or certified and if so by whom) and also includes questions to assess how well laboratory and clinical staff communicate. Sections 2–4 contain questions concerning the use of the markers in clinical practice (i.e. number of markers currently requested in blood and in other fluids, assay methods and cut-off points used, clinical guidelines). Section 5 focuses on the role of laboratory in clinical trials in oncology. For each area, the questionnaire included multiple-choice questions, where the responders could select more than one choice, as well as opportunities for responders to provide free text comments. A copy of the questionnaire was provided as an appendix.
The survey was addressed only to the laboratories located within health-care institutions that have inpatient beds and that regularly perform tumour marker measurements. The target area was Veneto, the second most populous region of North Italy, with 4,800,000 inhabitants. Public health-care institutions were selected using a list available on the website of Regione Veneto, while private health-care institutions were selected from a list provided by the Italian Association of Private Health Institutions, which collates information for all private institutions covered by the Italian health-care system insurance. The questionnaire was sent both by e-mail and post to the Directors and/or the Heads of Departments of Laboratory Medicine of all the selected institutions in December 2006. All responses were received by the end of June 2007. Non-responders were reminded at six-monthly intervals, first by e-mail as first line and subsequently if necessary by telephone until at the end of 2007.
Results
Sample size
The surveyed sample covered 44 laboratories, representing a total of 14,701 inpatient beds (74% of all those available in the Veneto Region in 2005), i.e. 28 public laboratories (representing 13,729 inpatient beds, i.e. 81% of all available within the region in the public sector) and 16 private laboratories (972 inpatient beds, i.e. 32% of all available in private institutions in the region). Overall, 31 laboratories (70%) filled in and returned the questionnaire, 25 (89%) in public and six (38%) in private institutions.
Three laboratories in public institutions did not perform tumour marker determinations and were excluded from further analysis.
Overall, the 28 laboratories surveyed performed 977,786 tumour marker tests from January to December 2005. The proportion of tumour marker requests performed, as a percentage of the total number of laboratory tests, was 1.5% (0.82–2.26) for public health institutions compared with 1.83% (1.64–1.85) for private institutions. Similarly, the percentage of tumour marker requests was 1.48% (0.82–2.14) for institutions with at least one oncology department versus 2.11% (1.59–2.26) for institutions in which no oncology facilities were available.
General information
Overall, 15/28 (68%) of surveyed institutions had at least one oncology department (14/22 [64%] public and only 1/6 [17%] private institutions), while 9/28 (32%) did not have any type of oncology facilities (4/22 [18%] public and 5/6 (83%) private institutions). Four public institutions (14%) did not report complete data on this topic. Medical oncology units were located in 14/18 (78%) and radiotherapy services in 8/18 (44%) of public institutions.
Only 14/28 (50%) surveyed laboratories were formally accredited and/or certified by an external body. In 5/28 (18%), the accreditation was in progress whereas 9/28 (32%) institutions were neither certified nor accredited.
How did laboratory and clinicians communicate?
When asked whether laboratory and clinicians did communicate, 25/28 (89%) of laboratories stated that no clinical information was regularly provided with tumour marker requests. Twenty-three/twenty-eight laboratories (82%) declared that the communication between laboratory and clinicians was on demand and only 14% of laboratories organized educational programmes such as clinical audits. Three laboratories did not report details on this question. Laboratory staff most frequently contact clinicians to request clinical information for particular cases (21 of 28 laboratories [75%]) whereas clinicians most frequently contact the laboratory to request interpretation of tumour marker results (24 of 28 hospitals [86%]) (Figures 1a and b).
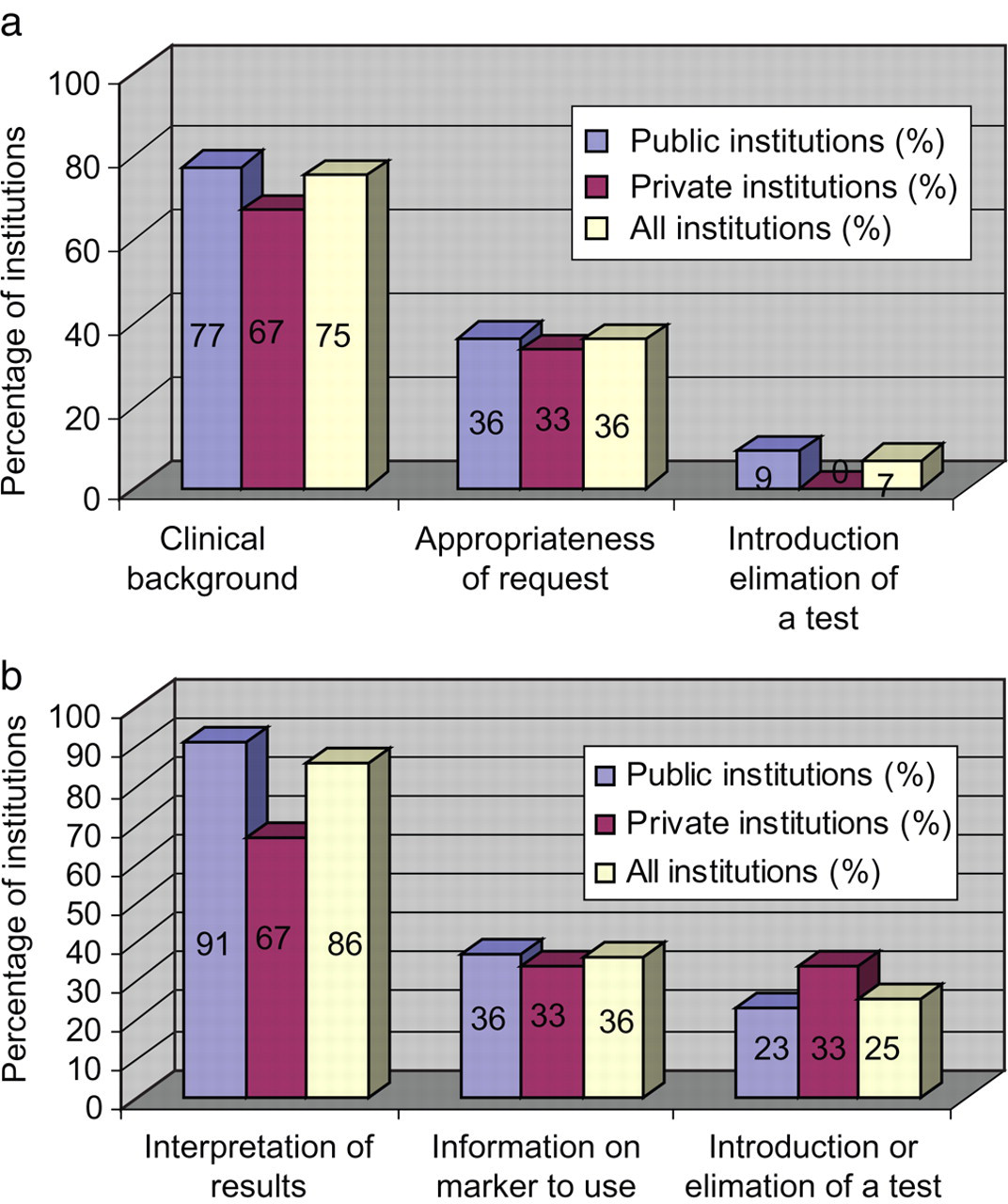
(a) Frequently asked questions: from laboratory to clinicians. Units in vertical axis is the percentage of institutions. (b) Frequently asked questions: from clinicians to laboratory. Units in vertical axis is the percentage of institutions
Which and how many tests are requested? (case mix and cut-off)
Overall, the percentage of requests was 34.5% for tPSA, 18.3% for CEA, 10.6% for CA19.9, 9.9% for AFP, 7.6% for CA15.3, 7.1% for fPSA and 6.6% for CA125. Other tumour markers accounted for less than 6% of the total requests, with individual marker percentages ≤1%.
Requesting patterns were similar in public and private laboratories (Table 1) and in institutions with or without oncologic facilities (Table 2), although in the public institutions a wider range of tumour markers were performed.
Tumour markers performed in health public, private and all institutions
Numbers are expressed as percentage of total tests performed in 2005
ND, not done
Tumour markers perfomed in institutions with or without oncology departments
Numbers are expressed as percentage of total tests performed in 2005
ND, not done
The ADVIA Centaur (Bayer Diagnostics, Tarrytown, NY, USA), Modular Analytics and Elecsys (Roche Diagnostics, Indianapolis, IN, USA) and ARCHITECT (Abbott Diagnostics, Abbott Park, IL, USA) were the most frequently used platforms for tumour marker measurements, representing 93% of all methods and assays used. The most commonly used cut-off point was that provided by the manufacturer's kit insert (66%), followed by values used in the scientific literature (25%). The cut-off point was calculated using healthy or benign groups as controls only in 8% of the cases. Among the private institutions, 95% of laboratories used data from the kit insert (only 61% of public institutions) and the remaining 5% obtained data from the scientific literature (29% of public institutions). Only 10% of laboratories from public institutions calculated the cut-off points from healthy controls.
Different cut-off values were sometimes quoted even by laboratories apparently using the same criteria to choose the cut-off point. For example, upper limits of the reference range quoted by laboratories using the same ARCHITECT (Abbott Diagnostics, Abbott Park, IL) assay for CEA and stating that they use manufacturer's instructions ranged from 3.5 to 10 ng/mL (Table 3).
Upper normal reference ranges used for tumour markers frequently requested
*Values marked with * are expressed in U/mL
§Test systems reported in the table are: ARCHITECT (Abbott Diagnostics, Abbott Park, IL, USA), ADVIA Centaur (Bayer Diagnostics, Tarrytown, NY, USA), IMMULITE 2000 (Diagnostic Products, Los Angeles, CA, USA), and Modular Analytics (Roche Diagnostics, Indianapolis, IN, USA), LIAISON (DiaSorin, Saluggia, Italy)
Overall, six laboratories (5 public, 1 private) analysed tumour markers in other biological fluids, including BTA™ in urine, CEA, AFP and/or NSE in pleural fluid and AFP, CA 125 and/or CA 19.9 in peritoneal effusions. However, these applications constitute fewer than 1% of the total number of tumour marker measurements performed by respondent laboratories.
Does the declared use of clinical guidelines influence how the markers are requested?
Fourteen/twenty-eight (50%) laboratories stated that their clinicians did not refer to clinical guidelines for tumour marker ordering (9/22 [41%] public and 5/6 [83%] private institutions), 5/28 (18%) declared that clinicians followed recommendations in their clinical practice (23% public institutions), 5/28 (18%) of responders did not know what practice their clinicians followed (23% public institutions) and 4/28 (14% private institutions) did not reply to this question (3/22 [14%] public and 1/6 [17%] private institutions).
Overall, 5/28 (18%) of the laboratories declared that their clinicians followed recommendations in tumour marker requesting; tumour markers were firstly ordered in clinical scenarios such as for the disease course monitoring and for the response to therapy, with the exception of PSA for prostate cancer, CA 125 for ovarian cancer, CEA for colon cancer and HCG for testis cancer, that were also requested for diagnosis and/or screening of high-risk patients.
The use of clinical guidelines did not appear to influence tumour marker workload and test mix (Table 4). We also obtained the same results when we evaluated the possible impact of accreditation certification on tumour marker requesting (data not shown).
Tumour markers performed in institutions adhered to clinical guidelines and not
Numbers are expressed as percentage of total tests performed in 2005
ND, not done
Are markers requested for research purposes?
Only one laboratory belonged to a public institution declared as being aware of contributing to a clinical trial.
Discussion
We carried out a survey to evaluate the current use of tumour markers in routine laboratory practice in one of the most large and representative regions of North Italy.
The pattern of tumour marker requesting was not influenced by the type of hospital or by the availability of oncology facilities. Requesting patterns (both test mix and workload) are similar in public and private institutions, which is of some concern because only 17% of private institutions have oncology facilities, which are available in 64% of public institutions. This may in part reflect routine ordering of tumour markers by non-oncology medical departments and general practitioners. In fact, in Italy, oncology patients may be cured by diverse clinical specialists, including surgeons, gynaecologists and urologists, especially in minor health-care institutions, whereas in major hospitals, the care of these patients is specifically managed in oncology or radiotherapy departments, which would guarantee a higher proficiency level in provided care.
This could explain why the most popular test ordered (34% of total requested) is PSA, a marker whose measurement is almost routine in many general practices. 8
Similar observations have been reported by other authors, 2–4 who attributed the variation in the number and reasons for tumour marker requests to the specialization of the requesting physicians. Clinicians not usually involved in cancer patient care (e.g. family practitioners and medical/surgical clinicians) tend to order tumour markers more frequently, particularly for diagnostic purposes, while oncologists order proportionately lower numbers in a more appropriate manner.
These data were confirmed in the present study on comparing the number of tumour marker requests (expressed as a percentage of the total number of laboratory tests) made in institutions with at least one oncology department with those made in institutions without an oncology department even if the available data are based on a small sample of institutions.
Somewhat surprisingly, awareness of clinical guidelines does not appear to have influenced the number of tumour marker requests. In fact, a similar pattern of tumour markers use was observed whether or not respondents stated that clinical guidelines were followed or not.
In a previous survey conducted in the same geographic area to verify how tumour markers were used in the follow-up of breast and colorectal cancer, we found that a larger number of tumour markers was used on a routine basis than those recommended by clinical guidelines. 5 In particular, only one of the 29 surveyed institutions used CA15.3 alone for the postoperative follow-up of patients with primary breast cancer, while eight used two markers and the other 20 routinely used three or more markers. A similar situation was observed for colorectal cancer (only 3 hospitals used CEA alone, 15 used two markers and 11 used 3 markers).
The relatively restrictive nature of the recommendations provided by clinical guidelines 9 would be expected to lead to decreased requests in those hospitals following clinical guidelines but this was not observed. This may reflect the clinical awareness of ongoing uncertainty relating to the clinical effectiveness of tumour markers, i.e. the impact of their measurement on the clinical outcome. In fact, as reported in the clinical guidelines update on tumour markers in breast cancer of the American Society of Clinical Oncology Committee, 10 very few randomized-controlled trials that can support the efficacy or otherwise of tumour marker measurements have been conducted. Many previous studies are methodologically inadequate, commonly recognized shortcomings including their retrospective rather than prospective nature, patient groups that are too small and failure to provide adequate descriptions of how patients were treated or how data analysis was conducted in different treatment subgroups. The apparent lack of effect of the guidelines may reflect failure to achieve the ‘local ownership,’ which has been identified as being critical to their success, as reported by Sturgeon. 11 Over-requesting may be the result of inexperience, test ordering protocols that are difficult to change, lack of knowledge about the most appropriate use of tests, fear of a ‘diagnostic error’, pressure to adhere to perceived standards of care and/or the need to meet patients' demands. A prime example of the above is represented by PSA in the prostate cancer screening. In fact, even if the clinical benefit of prostate cancer screening with PSA was not definitely demonstrated, routine use of PSA for diagnostic purposes in asymptomatic men is a common practice. 12
Naturally, the present study has some limitations. Firstly, our projections were based on a small sample of private laboratories (only 38% of private hospitals surveyed) that may not have been representative of this setting. For this reason, our results are preliminary and should be confirmed in a more large size sample. Secondly, our study was performed to evaluate how tumour markers are routinely used in a laboratory practice in a regional-scale area so that it may not be possible to generalize to other settings.
Thirdly, in our survey, there was no direct attempt to determine whether the tests were ordered appropriately and the information about adoption of clinical guidelines was obtained in an indirect way because the questionnaire was sent to the director of the central laboratory of each hospital and not ideally to the requesting clinician. This approach is very questionable for several reasons, among which the laboratory could not know whether and how their clinicians use tumour markers. In fact, in this survey, 5/28 (18%) of surveyed laboratories stated they did not know whether clinical guidelines were in place or not, and 4/28 (14%) did not answer this question, confirming that communication between the clinician and the laboratory staff, as we have previously highlighted, 5 continues to be a great challenge. Furthermore, another reason could be that the application of guidelines by the clinician could vary depending on the tumour marker or the type of tumour. Thus, our estimates about the number of institutions that actually followed clinical guidelines could be biased.
In our 1998 survey, 5 about 50% of the respondent laboratories received no clinical information relating to tumour marker requests, a situation that appears to have worsened, with 89% respondents to the present survey stating that no clinical information is routinely provided. This may in part reflect increased automation and increased demand for high-throughput and rapid turn-around times in clinical laboratories.
It is important to note that approximately 80% of the surveyed laboratories do not communicate with clinicians in a systematic manner, but instead rely on informal contacts. The analysis of the most frequently asked questions from clinicians to the laboratory and vice versa suggests that necessary information could be provided fairly simply, by re-engineering both the order form (in printed and electronic formats) so as to force provision of basic clinical information (i.e. reason of requesting) and the result reporting form, so as to incorporate additional information, e.g. the assay method, variability data (i.e. total, analytical and biological error) and possible caveats relating to the particular result (e.g. major causes of false-positive results such as urinary tract infections for PSA).
From the results of the present survey, we can draw the following conclusions concerning the examined geographic area:
Bidirectional communication between clinicians and laboratory is poor and informal; Tumour marker requesting patterns are remarkably similar in different institutions in the region, both in terms of the percentage of total laboratory tests and in terms of the tumour markers measured; Variables believed to impact the quality of health care in oncology, such as availability of oncology facilities, adherence to guidelines or size of the institution, do not appear to affect how tumour markers are requested; Variables believed to impact on the quality of laboratory medicine, such as certification or accreditation at present, do not seem to impact how tumour markers are requested; The awareness of laboratory of the test requested for clinical trials in oncology seems anecdotal; We believe that surveys on the state of art in tumour marker requests should be periodically performed in order to monitor the impact of educational programmes or of the clinical guidelines in order to provide professional feedback to decision makers in health policies.
DECLARATIONS
This study is based on an idea of the Focus Group on Clinical Guidelines of the European Group on Tumour Markers (EGTM): Prof. MJ Duffy, Dr C Haglund, Prof R Klapdor, Dr R Molina, Dr P Stiebe, and Dr C Sturgeon, who realized a similar experience in a European scenario.
Finally, special thanks are due to Catharine M. Sturgeon, whose comments were gratefully received.
