Abstract
The aetiology of haemolytic disease is diverse and the diagnosis often relies on laboratory testing. We describe a case of intravascular haemolysis, which illustrates that significant intravascular haemolysis can occur in the absence of any abnormal haematological findings. Despite gross haemoglobinuria at presentation, the haemoglobin and reticulocyte counts were both within reference limits and a normal blood film was observed. Subsequently, acute tubular necrosis occurred secondary to haemolysis, and acute renal failure was evident by day 2. However, the haemoglobin decreased slowly reaching a nadir of 75 g/L on day 6 (reference interval 130–180 g/L). A diagnosis of paroxysmal cold haemoglobinuria secondary to mycoplasma infection was subsequently made. Biochemical analysis was useful in this case to confirm that the gross pigmentation in the samples received could be attributable to intravascular haemolysis.
Introduction
Intravascular haemolysis can be caused by hereditary, acquired and iatrogenic conditions. Haemoglobinuria is one of the most prominent clinical signs of excessive intravascular haemolysis 1 and diagnosis is often made on the basis of haematological investigations. Decreased haemoglobin concentration and increased numbers of reticulocytes are key features. Biochemical findings include elevated activities of lactate dehydrogenase (LDH), aspartate aminotransferase (AST) and an unconjugated hyperbilirubinaemia. In this report, we demonstrate that significant intravascular haemolysis can occur in a patient with apparently normal haematological findings.
Case report
A 23-year-old man presented to the accident and emergency department with a three-day history of headache, multiple joint pains, rigours, vomiting and a mild cough. The past medical history included aortic coarctation aged 12 days, which was corrected surgically, but nil else of note. A regular follow-up demonstrated no cardiovascular sequelae. On examination, he was pyrexial with a temperature of 37.9°C and with a blood pressure of 137/66. Physical examination was unremarkable, but significant red discolouration of the urine was noted.
The results of initial haematological investigations were unremarkable: haemoglobin 147 g/L (reference interval: 130–180 g/L), platelet count 159 × 109/L (reference interval 150–400 × 109/L) and reticulocytes 0.080 × 1012/L (reference interval 0.020–0.100 × 1012/L).
Both serum and urine samples were grossly pigmented (Figure 1) and results of serum biochemistry investigations were consistent with intravascular haemolysis. Activities of AST and LDH were significantly elevated at 165 U/L (reference interval 0–35 U/L) and 1049 U/L (reference interval 0–248 U/L), respectively. Alkaline phosphatase activity was normal at 54 U/L (reference interval 30–120 U/L) and alanine aminotransferase (ALT) was 84 U/L (reference range 0–45). Haptoglobin was only slightly depleted at 0.41 g/L (reference interval 0.45–2.05 g/L), C-reactive protein (CRP) elevated at 113 mg/L (reference interval <5 mg/L) and serum bilirubin was 62 μmol/L (reference interval 1–25 μmol/L), with an unconjugated fraction of 51 μmol/L. The analysis of the urine sample on admission showed no red blood cells (<1/cmm). The Medi-Test urine combi-stick test was positive for haemoglobin, and this was confirmed by the spectrophotometric analysis, which showed the presence of an oxyhaemoglobin peak at 410–418 nm. Of note, creatinine was elevated at 134 μmol/L at presentation (reference interval 74–110 μmol/L).

Grossly pigmented urine (left) and serum (right)
A blood film was requested which showed no abnormality; however, the biochemistry results were consistent with haemolysis and haemoglobinuria. Repeat blood tests confirmed the initial findings. Acute renal failure was diagnosed, the patient rapidly became anuric, was started on temporary dialysis and a renal biopsy was performed on day 2. This showed the presence of damaged erythrocytes and cast material predominantly within medullary tubules, indicative of acute tubular necrosis caused by haemolysis. Despite such substantial haemolysis, the depletion of haemoglobin was slow (Figure 2) and reached a nadir of 75 g/L on day 6; at which point haemodialysis was performed. At this time, serum albumin concentration had fallen from 39 g/L on admission to 26 g/L. A reticulocyte count of 0.062 × 1012/L was measured on day 5. The patient also showed symptoms of a chest infection, with a chest X-ray revealing shadowing in both his left and right lung fields. This was treated with intravenous antibiotics.
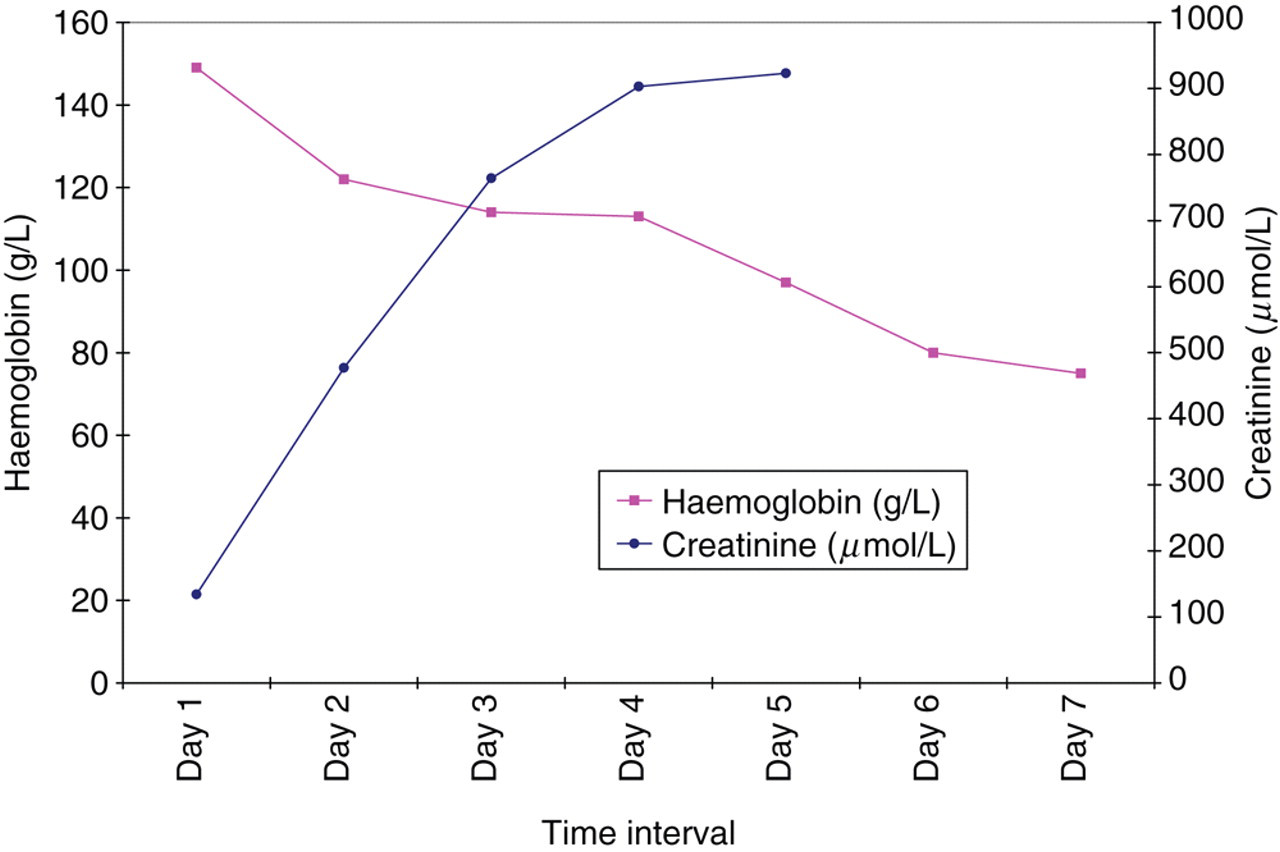
The slow depletion of haemoglobin and acute renal failure secondary to paroxysmal cold haemoglobinuria
Paroxysmal cold haemoglobinuria (PCH) was diagnosed using the Donath-Landsteiner test, and was subsequently found to be secondary to mycoplasma pneumonia. Mycoplasma antibody was not detected at presentation, but a sample taken one month later gave a positive agglutination at a titre of >1280. These results are consistent with recent mycoplasma infection as titres do not peak until 4–6 weeks postinfection.
Discussion
In a typical case of haemolytic anaemia, the haematological findings are diagnostic at presentation and the key features of anaemia in the presence of increased reticulocytes and undetectable haptoglobin are observed. 1
In our case, the haematological findings at presentation were unremarkable. PCH is a rare type of autoimmune haemolytic anaemia and typically presents in young children. The estimated incidence is 0.4 cases per year per 100,000 people. 2 Exposure to temperatures below 20°C in the peripheral circulation allows the binding of the Donath-Landsteiner IgG antibody to glycosphingolipids present on erythrocytes. The antibody has biphasic action: it binds to the P-antigen at lower temperatures and efficiently fixes complement at 37°C when the red cells are returned to the central circulation. Complement-induced erythrocyte lysis then takes place. PCH often occurs postinfection and this patient was diagnosed with mycoplasma pneumonia.
The slow decline in haemoglobin (Figure 2) was surprising, given the significant haemoglobinuria and biochemical findings. The acute anaemia in some published paediatric cases of PCH has been severe enough to cause sudden death. 2,3 Wynn et al. 3 reported six cases of PCH presenting in childhood and prominent red cell agglutination was evident in the blood film in all six cases, which contrasts the normal blood film reported in our case. Heddle et al. 4 reviewed 42 cases of PCH and found that the admission haemoglobin ranged from 25 to 125 g/L with a mean value of 68 g/L. The degree of haemolysis varies among patients because of antibody titre and affinity, but in our case there was a surprising delay in the deterioration of the haematological parameters. There was also an absent reticulocyte response. However, PCH is often associated initially by a relative or absolute reticulocytopenia, and this may represent coexistent viral inhibition of erythropoesis. 3,4 Haptoglobin concentration was also near normal at presentation. Haptoglobin is consumed during intravascular haemolysis, as it covalently binds free haemoglobin forming haptoglobin–haemoglobin complexes, which are cleared by the reticuloendothelial system. However, haptoglobin is a positive acute-phase protein and concentrations increase in inflammation. It is therefore of interest that the CRP was elevated on admission at 113 mg/L (reference interval <5 mg/L), indicating inflammation; therefore, the haptoglobin concentration may have been higher than expected due to the acute-phase response.
This case demonstrates that in patients with PCH, markers of red cell breakdown, such as increased reticulocyte count and blood film appearance, may initially be normal. These findings appear to take place even in the presence of clinical and biochemical evidence of haemolysis, with free haemoglobin in blood and urine. In this instance, low levels of haptoglobin and raised levels of enzymes such as LDH appear to be more sensitive indicators of haemolysis.
