Abstract
Laboratories evaluated whether an interference was causing a false-positive PSA for the Immulite 2000 immunoassay after a time course of increasing prostate-specific antigen (PSA) in a post-prostatectomy patient led to salvage therapy, which had no effect on the elevated PSA. Serial dilutions of PSA for the patient sample (6.1 ng/mL; post-prostatectomy reference range: <0.1 ng/mL [undetectable]) were linear (r > 0.99). However, the PSA measurement was reduced to 0.1 ng/mL after pretreatment of the sample with heterophilic antibody blocking reagent. PSA was undetectable (<0.1 ng/mL) when measured using two alternative immunoassays. These results were consistent with the presence of heterophilic antibody interference for the Immulite 2000 assay. In this case, heterophilic antibody interference with PSA measurement must have originated during the period of post-prostatectomy monitoring, and the apparent progressive increases in PSA may have been due solely to the progressive increase of this heterophilic antibody assay interference. In the absence of clinical correlation, positive PSA monitoring results should always be assessed for heterophilic antibody interference for at least one time point.
Introduction
Heterophilic and human anti-animal antibodies are well known as a potential cause of interference in immunoassays. 1–9 In previously reported cases involving measurement of prostate-specific antigen (PSA), discoveries of the assay interference have been made either immediately after a first measurement 10 or immediately after prostatectomy. 11–14 Here we report a case of falsely elevated PSA due to heterophilic antibodies that developed in a patient nearly 3 yr after radical prostatectomy for prostate adenocarcinoma. The possibility of an interference was questioned only after aggressive therapeutic intervention showed no effect on PSA even though the time course of apparently progressively increasing PSA was typical for recurrence of prostatic adenocarcinoma.
Case report
A 51-year-old patient was initially found to have an elevated PSA (15.9 ng/mL) on screening. He was subsequently diagnosed with Gleason 7 (3 + 4) adenocarcinoma of the prostate. Radical prostatectomy in August 2004 revealed stage pT2c,N0,M0. The Gleason score 15,16 refers to the histological pattern of adenocarcinoma, with scores ranging from 2 to 10. Lower scores correspond to more well-differentiated tumours and higher scores correspond to more poorly differentiated tumours. Tumour staging using the TNM system 17 refers to the tumour size/extent (T), lymph node metastases (N) and distant metastases (M). A T2c,N0,M0 tumour involves the bilateral prostate and does not have any regional lymph node or distant metastases. A higher Gleason score and tumour stage is associated with a worse prognosis.
Postoperatively, serial PSA measurements for the patient were undetectable (<0.1 ng/mL; post-prostatectomy reference range: <0.1 ng/mL) for nearly 3 yr. However, in June 2007 his PSA rose to 1.4 ng/mL and continued to rise over the next 3 months to as high as 4.1 ng/mL. Radiological evaluation showed no evidence of metastatic or locally recurrent disease. The patient was treated with hormonal therapy, systemic chemotherapy and pelvic radiation. During therapy, the PSA measurements declined briefly on two occasions but still remained high (3.1–7.9 ng/mL) (Figure 1). The laboratory was asked to investigate the possibility of a heterophilic antibody interference in February 2008 when PSA continued to rise even after aggressive therapy was completed. All PSA measurements to that time had been performed using the Immulite 2000 immunoassay (Siemens Healthcare Diagnostics, Deerfield, IL, USA).
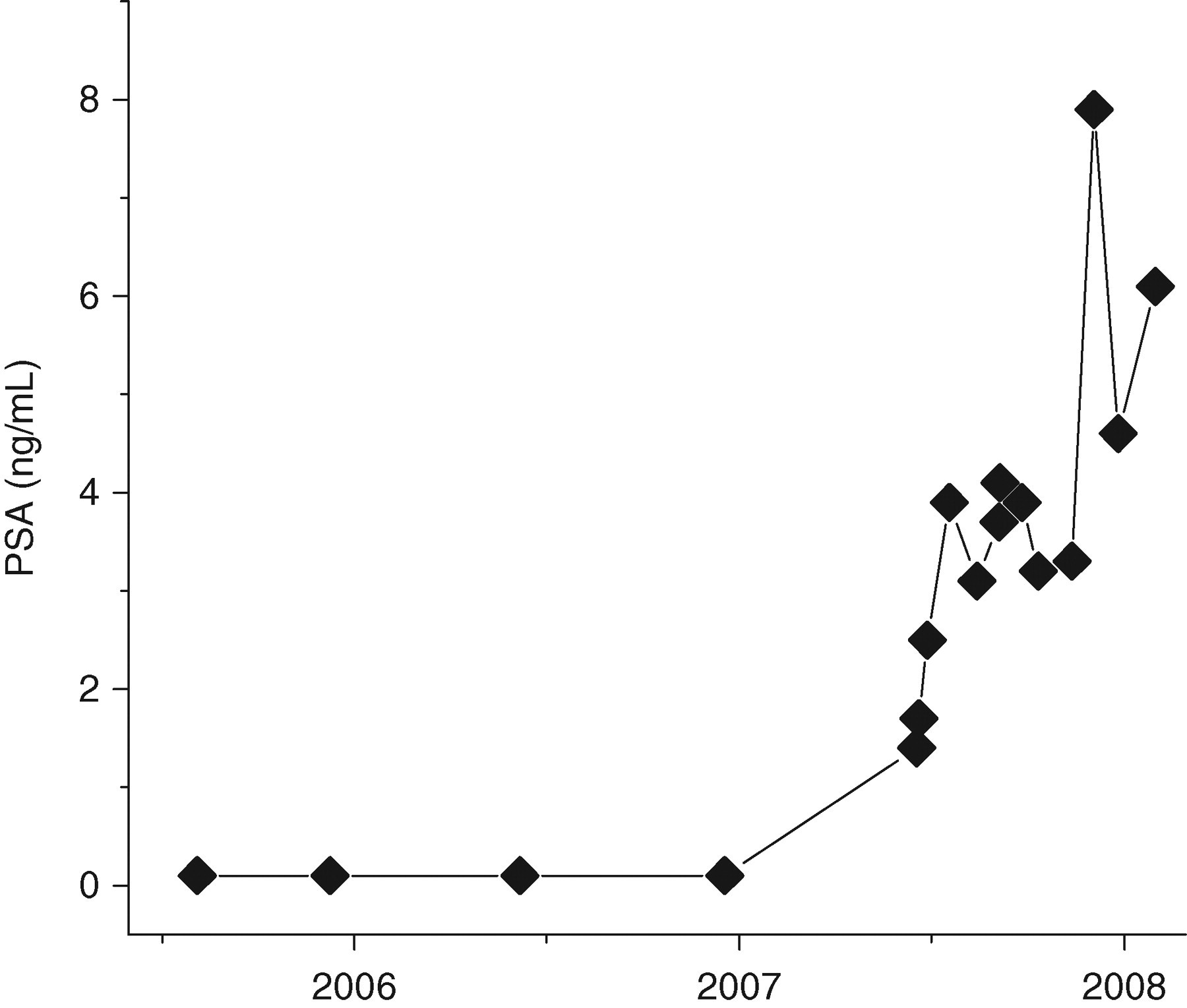
Serial post-prostatectomy prostate-specific antigen measurements for patient
Serial dilutions (2/3, 1/3) of the sample were prepared manually using a diluent consisting of bovine serum albumin (6 g/dL) in phosphate-buffered saline (pH 7.4). (This buffer is used routinely in our laboratory for manual dilutions for immunoassays, and may differ from the Immulite 2000's online diluent primarily in its lack of any preservative; such manual dilution experiments are always accompanied by dilution measurements for a control sample.) PSA was measured on the diluted samples using the Immulite 2000 immunoassay and assessed for linearity. The dilution samples revealed essentially linear PSA results, although there was a non-zero intercept; dilutions on a control patient with similar PSA concentration (patient diagnosis: prostatitis, with no histological evidence of prostate cancer) were also linear but with an approximately zero intercept (Figure 2).
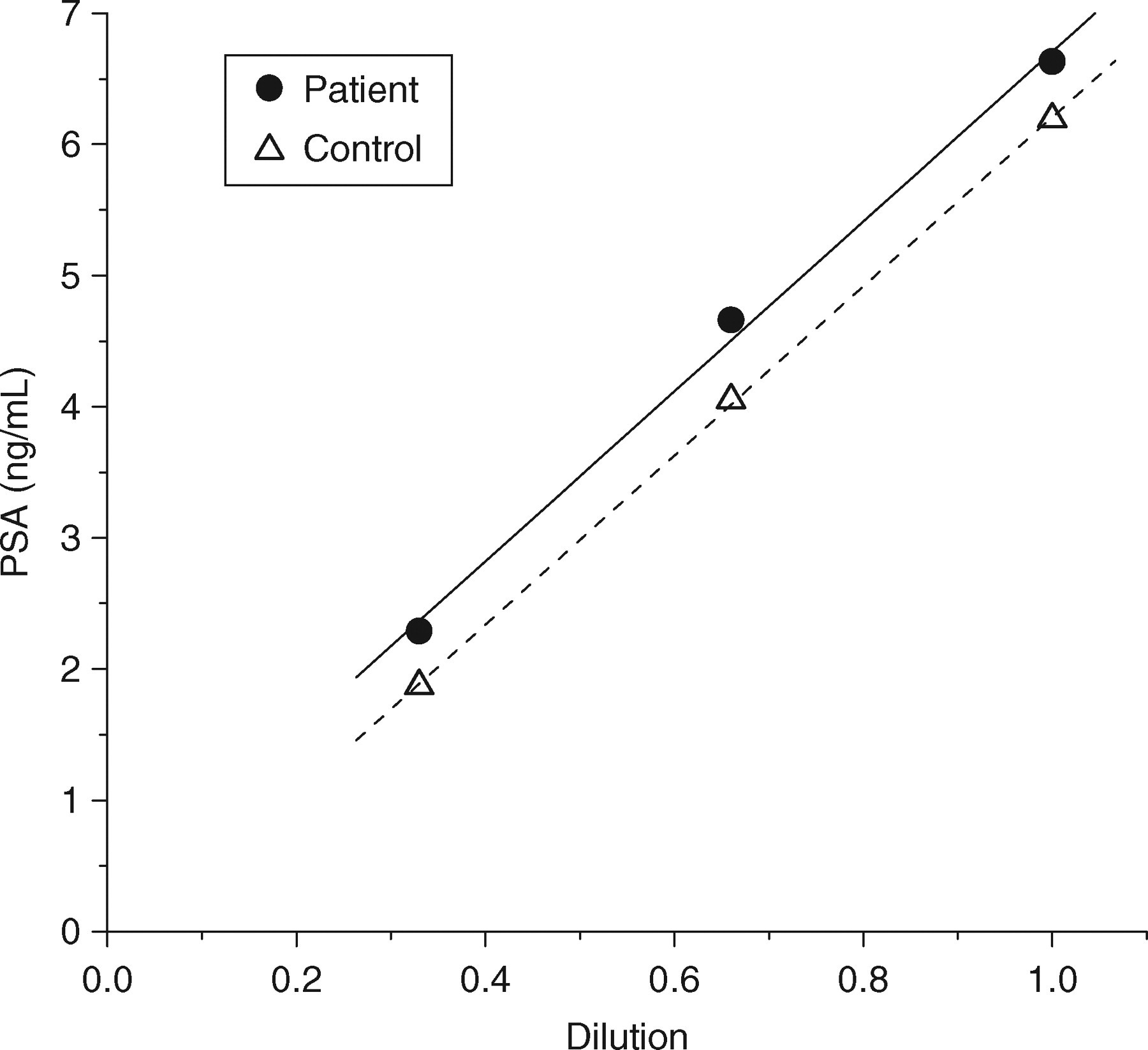
Results of sample dilutions for prostate-specific antigen measurements. Correlation coefficients (r) for both dilution series were >0.99
Pretreatment of the patient's sample with heterophilic antibody-blocking reagent (Scantibodies Laboratory, Inc., Santee, CA, USA) decreased the apparent PSA from 6.6 ng/mL (untreated) to 0.1 ng/mL (treated) as measured using the Immulite 2000 assay. The control patient measured 6.2 ng/mL (untreated) and 5.9 ng/mL (treated). Additional aliquots of the patient sample were sent to referral laboratories for PSA measurements conducted by two different alternative immunoassays (Hybritech, Beckman-Coulter Inc., Fullerton, CA, USA; Roche Modular E170, Basel, Switzerland). PSA measurements using the alternative PSA assays each returned results of <0.1 ng/mL (Table 1).
Prostate-specific antigen assay formats, antibodies (Ab) and patient results
*Post-prostatectomy reference range
In order to evaluate the possibility of an interfering monoclonal paraprotein, serum protein electrophoresis was also performed on the patient's sample (Hydrasys, Sebia Inc., Norcross, GA, USA). The patient's serum protein profile was normal in all aspects, with no elevation in gamma globulins or any apparent abnormality or monoclonal paraprotein present (data not shown). In addition, measurements for the patient's sample in a series of other assays on the Immulite 2000 immunoassay analyser produced results only within reference ranges (Table 2), i.e. there were no unexpected results that would suggest an antibody interference in these other assays.
Additional Immulite 2000 assay formats, antibodies (Ab) and patient results
hCG, human chorionic gonadotrophin; TSH, thyroid-stimulating hormone
*Reference range
During the interval of evaluation and treatment following detection of apparently elevated PSA, the patient was at various times treated with the following drugs: aspirin (anti-inflammatory), bicalutamide (nonsteroidal antiandrogen), dexamethasone and prednisone (anti-inflammatory corticosteroids), docetaxel (antineoplastic), famciclovir (antiviral), gentamicin (aminoglycoside antibiotic), goserelin (parenteral synthetic agonist analogue of gonadotrophin-releasing hormone), loratadine (non-sedating H1-blocker), lorazepam (benzodiazepine anxiolytic), ondansetron (antiemetic), pseudoephedrine (sympathomimetic anticongestant) and prochlorperazine (antiemetic). In two imaging studies, the patient was also administered Prostascint, a 111Indium-labelled IgG1 conjugate reactive with the glycoprotein prostate-specific membrane antigen (PMSA). None of these common drugs or treatments has been reported to be cross-reactive with PSA immunoassays, or to have induced an immune response interfering with PSA assays.
Discussion
Results from measurements of PSA for the patient sample following heterophilic antibody-blocking reagent pretreatment and from two alternate PSA immunoassays indicated positive interference for the patient's current PSA-positive Immulite 2000 immunoassay results. It is clear that the PSA assay interference had arisen in this patient since the time that post-prostatectomy monitoring had begun. It is possible, although not definite, that all of the apparently elevated PSA measurements made for this patient during the post-prostatectomy monitoring period were solely the result of a new onset and progressively increasing interference due to a heterophilic antibody. The development of increasing interference may have mimicked the results that would be typical for recurrence of prostate cancer. To our knowledge there is no previous report of such a temporal pattern of development of interference.
False-positive immunoassay results can cause additional laboratory and radiological studies, and in some patients, overtreatment for presumed recurrence/residual disease. 18 In previous reports of heterophilic antibody interference with PSA measurements, clinical suspicion for a possible interference was raised quickly when elevated PSA was unexpected following prostatectomy 11–14 or screening. 10 In the case reported here, PSA was undetectable for nearly 3 yr following prostatectomy. Detection of an apparent rise in PSA was of course the very basis for monitoring, and such a rise was therefore not regarded with suspicion and was not initially questioned.
Dilution experiments are recommended as a first-pass investigation of potential heterophilic antibody interference. 5 However, such experiments have poor negative predictive value. 19 In this case, the dilutions gave linear results, and could be interpreted only as giving no evidence of an interference rather than as evidence of no interference. In contrast to the dilution experiment, the heterophilic-blocking reagent experiment was essentially definitive with respect to concluding that a heterophilic antibody interference was present. It should be noted, however, that a negative result for such an experiment would also not rule out an interference, as it is difficult if not impossible to have appropriate controls for such an experiment. Both dilution and blocking reagent experiments can be performed easily and immediately onsite, and are therefore useful to quickly suggest heterophilic antibody interference. It is advisable, however, to make at least one additional alternative assay measurement irrespective of dilution or blocking reagent results.
The aetiology of heterophilic antibodies is not fully understood. 2,20–23 Whereas assay interfering antibodies are generally polyclonal, 7 reactive monoclonal paraproteins are also a potential cause of immunoassay interference. 24 Serum protein electrophoresis may be able to identify abnormalities in gamma globulins in such cases, and this reasoning was the basis for examination of this patient's serum by this method. The results of this assessment were negative in this case, however. The time course of rising apparent PSA concentrations was not incompatible with that of a secondary antibody response as, for instance, the time course of antibody production in response to infectious disease. 25
The exact causes for irregularities in the time course of PSA measurements in this case are uncertain. The decline in apparent PSA results from the most elevated result due to heterophilic antibody interference seen in Figure 1, may be caused by corticosteroid suppression of antibody production, when the patient was treated with dexamethasone prior to chemotherapy. In a study by Nixon et al., 26 biological variation of PSA is reported to have a geometric mean of the coefficient of variation of 7.3% (across a range of PSA concentrations, with the 95th percentile of coefficient of variation = 19.2%); for an analytical interassay coefficient of variation of 5%, the mean critical difference between PSA measurements was estimated to be 20.5%. The Immulite 2000 PSA immunoassay has an interassay coefficient of variation of approximately 6%. Thus, individual consecutive pairs of PSA measurements in the time course shown in Figure 1 would not meet all criteria for reasonable certainty of a difference between them; nonetheless, the overall pattern of increase was certainly compatible with the interpretation of a recurrence of disease.
Among the different manufacturer's PSA assays on which the patient's sample was assayed, the Immulite 2000 PSA assay is distinct in its use of a goat polyclonal antibody. Among the additional Immulite 2000 assays for which the patient's samples were assayed, the PSA assay is distinct both in its use of a polyclonal solid-phase antibody, and of a single-step incubation rather than a 2-step incubation that includes a wash step after binding to the solid-phase antibody and before introduction of the enyzme-linked antibody. Despite the overlap of antibody types among the assays listed in Tables 1 and 2, the restriction of interference for this patient to only the Immulite PSA assay was apparently due to the exact format and antibodies for that assay. Although one-step sandwich immunoassays are more likely to be subject to antibody interferences than are other formats, 5 immunoassays of any format are potentially subject to interference by heterophilic antibodies. 5,7
In summary, this case report indicates that PSA measurements exhibiting a transition from undetectable to detectable PSA in post-prostatectomy patients could be due to heterophilic antibody interference. This is particularly true in situations where there is no clinical evidence of disease spread or recurrence. The laboratory should highlight this possibility to clinicians. Whereas dilution and antibody-blocking experiments may be expedient in suggesting a heterophilic antibody interference, an assessment for interference should ultimately be made using an alternative immunoassay.
