Abstract
Screening patients for Cushing's syndrome can be done by measuring late-night salivary cortisol concentration. 1,2 This method is seen as accurate, sensitive and specific. 3 Moreover, it is non-invasive and there is no need for hospitalization. Accuracy, sensitivity and specificity are, of course, always dependent on the type of hormone assay that is used. An increased number of requests for salivary cortisol for research purposes prompted us to seek automation.
We modified an existing automated assay for serum cortisol (chemiluminescence immunoassay) and compared its performance with that of a radioimmunoassay (RIA) tailored to measure salivary cortisol.
From 49 patients, 83 samples were used for this comparison. Saliva was sampled at various time points with a collecting device (Salivettes with no preservative; Sarstedt, Etten-Leur, the Netherlands). There is a relatively high cortisone/cortisol ratio in saliva compared with serum. 4 While cross-reactivity of cortisone in a cortisol serum assay might not be very important, it is of major importance in salivary cortisol measurements. We therefore tested cross-reactivity with cortisone in the modified serum cortisol assay. Since no salivary assays have been described yet for the Architect i2000 (Abbott, IL, USA), we tested carry-over. Squamous cell carcinoma (SCC) antigen is a tumour marker, which can be measured in serum with the Architect i2000. As saliva is extremely rich in SCC, 5 carry-over of SCC from saliva into serum was tested for this purpose.
Salivary cortisol in the 83 samples was measured using two methods. The salivary cortisol RIA was used as instructed by the manufacturer (Spectria, Orion Diagnostica, Espoo, Finland). Sample volume was 150 μL. The serum cortisol assay was modified for measuring salivary cortisol. Modification meant a higher amount of sample (150 μL instead of 50 μL) and software adaptations for the calibration curve due to this change. This assay was run on the Architect i2000 (Abbott), which is a random access analyser. The carry-over experiment was performed five times on the Architect i2000 by measuring SCC in serum in duplicates before and after a duplicate salivary cortisol assay and comparing the SCC concentrations measured.
The intra- and inter-assay imprecisions (coefficient of variation) are shown in Table 1. The slope of the regression lines, determined using the Passing and Bablok method was different between low (<12 nmol/L) and high salivary cortisol values (1.00 vs. 0.85, respectively), with an intercept of 0.1 and 0.2 nmol/L, respectively. Correlation between the salivary cortisol RIA and the modified serum cortisol assay was high, below 12 nmol/L (r 2 = 0.920) as well as above 12 nmol/L (r 2 = 0.991). Bland-Altman bias plots are shown in Figure 1.
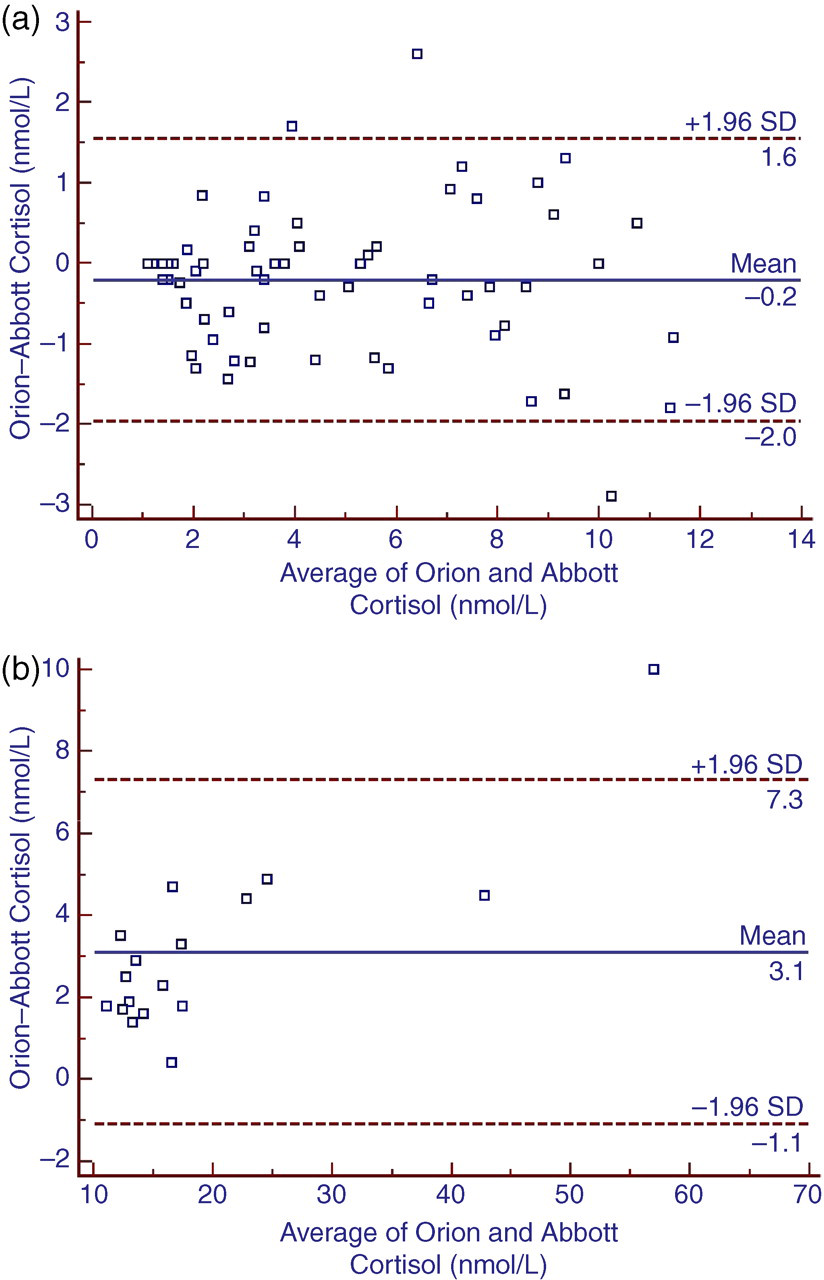
Bland-Altman bias plots of salivary cortisol determined by a salivary cortisol RIA (Orion) and a modified serum cortisol assay (Abbott); (a) <12 nmol/L (n = 65); (b) ≥12 nmol/L (n = 18)
Intra- and inter-assay variation
*CV estimated from results of duplicate measurements. CV, coefficient of variation; RIA, radioimmunoassay; CLIA, chemiluminescence immunoassay
Cross-reactivity with cortisone was <4% between 3 nmol/L and 20 nmol/L. No carry-over was observed in the SCC assay after assaying salivary samples for cortisol (<0.005% after a saliva sample with an SCC concentration of 1800 μg/L).
In conclusion, the modified serum cortisol assay gives comparable results with the salivary cortisol RIA and is comparable with, or better than, the salivary cortisol RIA with regard to reproducibility. Compared with other assays in the literature, 6–8 we find similar intra- and inter-assay imprecision. The modified serum cortisol assay has no significant cross-reactivity with cortisone. Moreover, there is no appreciable carry-over. We conclude that this assay can be used in clinical research. The assay might be of interest to the clinical laboratory as well, because it allows running salivary samples frequently on a random access analyser and thus satisfying the increased popularity of salivary cortisol measurements.
DECLARATIONS
