Abstract
Background
In the laboratory evaluation of suspected paracetamol poisoning, a non-invasive sample type that avoids venepuncture would be an attractive alternative to plasma, particularly in the paediatric setting. Salivary paracetamol measurement has not previously been evaluated in the published medical literature in the setting of deliberate self-poisoning (DSP).
Methods
In-house validation experiments (recovery, stability and lower limit-of-detection) were performed on pooled saliva samples using a Roche Acetaminophen assay on a Roche/Hitachi 917 analyser. A clinical study of comparison of paired saliva and plasma samples was also conducted involving adult patients presenting with DSP of paracetamol, the results of which were published previously.
Results
The validation experiments using pooled saliva samples showed: (i) mean recovery (paracetamol concentration 37.5–525 mg/L) 100.01% (±0.02 SD); (ii) precision of repeated assay over 24-h period CV <4%; (iii) lower limit-of-detection 0.9 mg/L. The clinical study of 21 patients with mean plasma paracetamol concentration of 48 mg/L (range 0–130) and mean saliva concentration 62 mg/L (range 0–183) showed good correlation between saliva and plasma concentrations (r 2 = 0.91).
Conclusions
The Roche Hitachi 917 Acetaminophen assay is suitable for the measurement of paracetamol in saliva. Further studies comparing plasma and saliva samples from patients with DSP and plasma paracetamol concentrations in the hepatotoxic range are warranted. If salivary samples are suitable for risk-stratifying patients with paracetamol DSP, venepuncture may be avoided, which would be attractive, especially in the paediatric setting.
Introduction
Timed plasma paracetamol concentration is used in paracetamol poisoning to predict severe hepatotoxicity and guide antidote (N-acetylcysteine) therapy. 1,2 A non-invasive sample type that avoids venepuncture, such as saliva, would be an attractive alternative to plasma, particularly in the paediatric setting.
Previous studies in the setting of therapeutic use of paracetamol have shown a close correlation between plasma and saliva paracetamol concentrations. 3–6 Saliva paracetamol concentration in the setting of paracetamol deliberate self-poisoning (DSP), however, has not been evaluated in the published medical literature (MEDLINE and EMBASE English language articles to January 2008). One small unpublished series of 13 patients presenting with paracetamol overdose to a hospital in Dunedin, New Zealand, found saliva and plasma paracetamol concentrations had a close correlation (r 2 = 0.987). 7
The Roche Acetaminophen automated colorimetric assay has not been evaluated using saliva samples in the published literature. One manual colorimetric paracetamol assay however, has previously been studied with saliva samples. 4
The aims of this study are to evaluate the Roche Acetaminophen assay using saliva samples and compare plasma and saliva paracetamol concentrations in patients with paracetamol DSP.
Materials and methods
Reagents and equipments
Paracetamol was measured using the Roche Acetaminophen assay on a Roche/Hitachi 917 analyser. The assay uses a colorimetric method that involves enzymic hydrolysis of paracetamol (by arylacrlamidase) and reaction with colour reagents (o-cresol and a periodate catalyst) to produce an indophenol. 8 Routine calibration and maintenance were performed according to the manufacturer's specifications. Assay time for paracetamol is 20 min.
Sample preparation
The in-house validation experiments used pooled saliva samples obtained from five laboratory volunteers not receiving paracetamol. The clinical comparison study used saliva samples (approximately 2 mL) collected in a sterile container. All saliva samples were centrifuged (4000 RCF for 5 min) prior to assay, and the supernatant separated for analysis. Sample preparation is important, as there is usually impurities and froth in the saliva sample collected, and the final yield of supernatant is much less than the collection volume. A minimum volume of 25 μL of sample is needed for the analysis, and together with dead volume, 100 μL final volume is required.
Methods
In-house validation experiments performed included recovery, stability and lower limit-of-detection. In the recovery experiment varying volumes of a paracetamol standard aqueous solution (600 mg/L) were dried to a powder in a vortex evaporator. Two millilitres of pooled saliva samples were then added and mixed by vortexing and then centrifuged for 5 min at 4000 RCF to remove the impurities. The supernatant was then aspirated and assayed. The highest paracetamol concentration that could be studied was 525 mg/L.
The stability experiment re-assayed samples from the recovery experiment in a random order, 12 times over 24 h. 9 The samples were re-centrifuged after 9 h and remained on the analyser for the duration of the experiment. Two changes of assay reagent kit were accompanied with routine recalibration. The lower limit-of-detection experiment re-assayed the negative standard saliva sample 20 times and was determined as mean + 3 SD. 9
A clinical comparison study was undertaken thereafter which has been published in detail elsewhere. 10 Briefly, paired saliva and heparin plasma samples were collected at least 4 hours post paracetamol ingestion from all eligible and consenting adult patients presenting with paracetamol DSP to the Emergency Department at Royal Perth Hospital, a large teaching hospital, during the study period (Aug 2006–Jan 2007). The study protocol was approved by the Royal Perth Hospital Ethics Committee (approval number 2006/126).
Statistics
Results are given as mean (and standard deviation) unless otherwise specified. Statistical analysis (including r2 value and linear regression) was performed using Microsoft Office Professional Edition 2003 (Excel).
No external funding source was used.
Results
In-house validation experiments
The average recovery of paracetamol in the concentration range 37.5–525 mg/L was 100.01% giving a proportional error of −0.01% (Table 1, Figure 1). The precision of all samples over 24 h was CV <4% (mean CV 1.7%, SD 0.70). The mean of the zero standard pooled sample was −0.3 mg/L (SD 0.4 mg/L). The lower limit-of-detection was calculated as 0.9 mg/L.
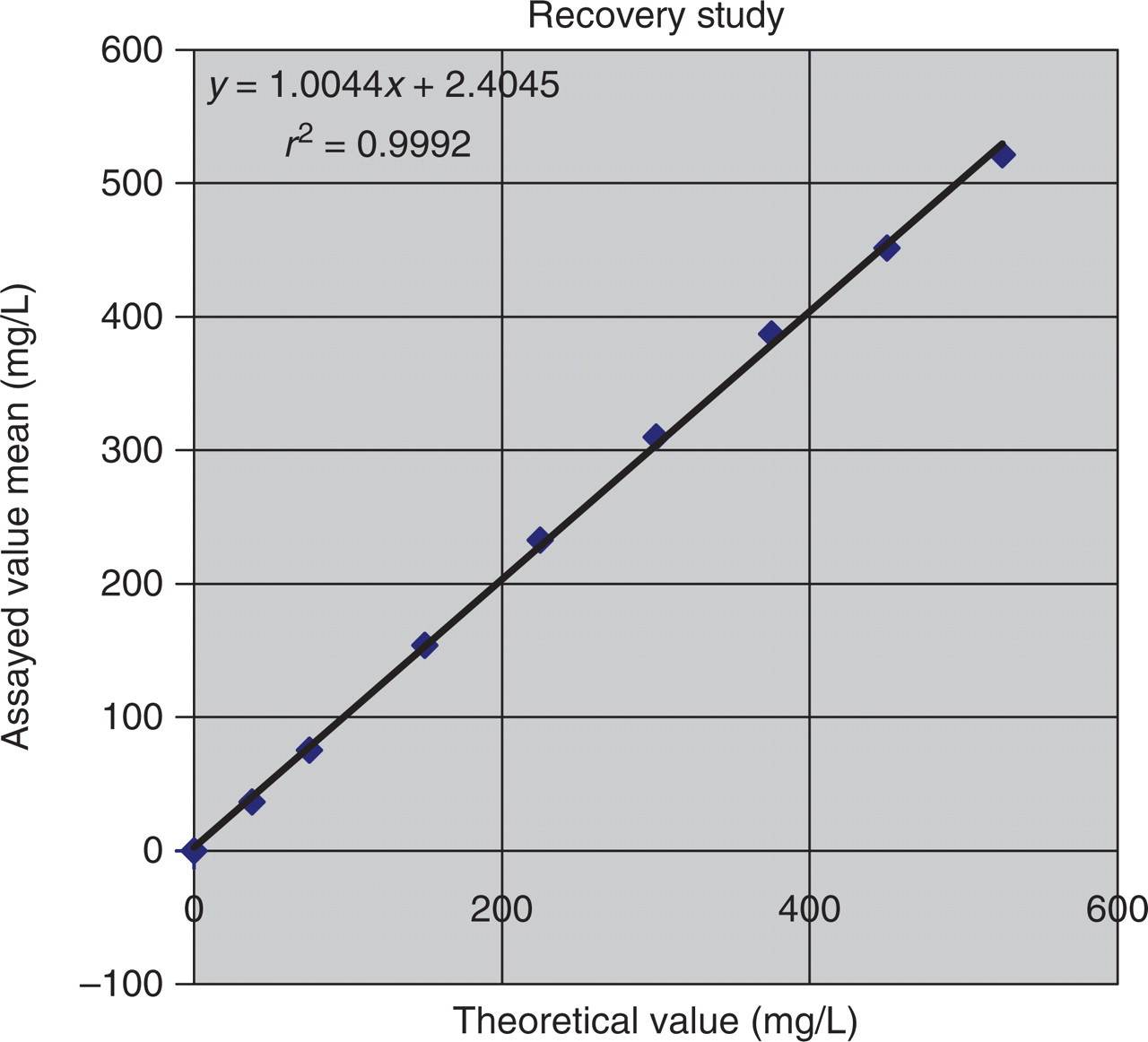
Comparison plot of salivary paracetamol recovery experiment results. Results of nine pooled saliva samples augmented with paracetamol standard and assayed in duplicates
Salivary paracetamol recovery experiment results
Results of nine pooled saliva samples augmented with paracetamol standard and assayed in duplicates. Paracetamol concentration in milligrams/litre. SD, standard deviation
Clinical comparison study
The mean plasma paracetamol concentration was 48 mg/L (range 0–130), mean saliva concentration 62 mg/L (range 0–183) and the correlation between the 21 paired plasma and saliva samples was r 2 = 0.91. 10
Discussion
Preliminary in-house validation of the Roche Acetaminophen assay using saliva samples found performance of saliva in recovery, lower limit-of-detection and stability experiments was similar to that of pooled serum samples (from manufacturer's published data). 8 The unpublished New Zealand study evaluating salivary paracetamol in the setting of DSP used the Abbott TDx Acetaminophen assay, which used a fluorescence polarization immunoassay method. The study also used parafilm (for salivary stimulation) in some participants and samples were frozen prior to assay. Correlation between saliva and plasma paracetamol concentrations in the 13 participants was high (r 2 = 0.987 (p < 0.05)). One manual colorimetric assay with method similarities to the Roche paracetamol assay was previously used in a comparison study of 20 healthy adult volunteers administered therapeutic doses of paracetamol. 4 Correlation between saliva and serum paracetamol concentrations in patients in that study was r = 0.67. However, samples were collected at 30 and 60 min postingestion; correlation within the first hour postingestion has been shown to be reduced. 3,6 All samples collected in our clinical study of paracetamol DSP were at least 4 h postingestion and showed a close correlation (r 2 = 0.91) between saliva and plasma paracetamol concentrations. Limitations of the clinical study included small sample size (n = 21) and plasma paracetamol concentrations that were below concentrations associated with severe liver toxicity in the majority of participants (20 of 21 patients).
In conclusion, the Roche Hitachi 917 paracetamol assay is suitable for the measurement of paracetamol in saliva. The paracetamol assay in this study, which is rapid and automated, would be suitable for this purpose. However, plasma paracetamol concentrations in the clinical study that showed close correlation between plasma and saliva concentrations were predominantly below that associated with severe hepatotoxicity warranting further studies that include samples with higher plasma paracetamol concentrations. If salivary samples are adequate for risk-stratifying patients with paracetamol DSP or toxicity, venepuncture may be avoided. This would be attractive, especially in the paediatric setting.
