Abstract
Background
Critical result reporting is a requirement for accreditation by accreditation bodies worldwide. Accurate, prompt communication of results to the clinician by the laboratory is of extreme importance. Repeating of the critical result by the recipient has been used as a means to improve the accuracy of notification. Our objective was to assess the accuracy of notification of critical chemical pathology laboratory results telephoned out to clinicians/clinical areas. We hypothesize that read-back of telephoned critical laboratory results by the recipient may improve the accuracy of the notification.
Methods
This was a prospective study, where all critical results telephoned by chemical pathologists and registrars at Tygerberg Hospital were monitored for one month. The recipient was required to repeat the result (patient name, folder number and test results). Any error, as well as the designation of the recipient was logged.
Results
Of 472 outgoing telephone calls, 51 errors were detected (error rate 10.8%). Most errors were made when recording the folder number (64.7%), with incorrect patient name being the lowest (5.9%). Calls to the clinicians had the highest error rate (20%), most of them being the omission of recording folder numbers.
Conclusion
Our audit highlights the potential errors during the post-analytical phase of laboratory testing. The importance of critical result reporting is still poorly recognized in South Africa. Implementation of a uniform accredited practice for communication of critical results can reduce error and improve patient safety.
Introduction
Laboratory data forms an integral part of patient care and is therefore an important source of medical error affecting patient safety. Errors during the analytical phase have been emphasized, with very little attention being paid to errors during the pre- and postanalytical phases of testing. Lundberg 1 originally highlighted critical result reporting and defined a critical value as a result suggestive of imminent danger to the patient unless appropriate therapy is promptly initiated.
Critical result reporting is one of the requirements for accreditation, as implemented by laboratory accrediting agencies such as the Joint Commission on Accreditation of Healthcare Organizations (JCAHO) and the College of American Pathologists (CAP) in the United States. 2 In South Africa too, critical result reporting is a requirement for accreditation by the South African National Accreditation System (SANAS), 3 thus necessitating pathology departments to have a written protocol dealing with the verbal transmission of results to clinicians. This procedure should detail why, how and when to telephone results, and who should receive results as set out in the new Clinical Pathology Accreditation (CPA) standards guidelines. 4 Similar recommendations for the out-of-hours reporting of markedly abnormal laboratory results have been published by the Royal College of Pathologists, including the identification of an abnormal result, the communication thereof, the importance of a completely filled-in request form to permit out-of-hours communication between the laboratory and the care-provider. 5 Most laboratories have parameters and definitions for critical values. Accurate and prompt communication to the clinician is very important, with the implementation of repeating the critical results by the recipient used to improve accuracy. The objective of this audit was to assess the accuracy of notification of critical laboratory results telephoned to clinicians/clinical areas. We hypothesized that reading-back of the telephoned critical laboratory result by the recipient may improve the accuracy of notification.
Methods
Setting
The Chemical Pathology Laboratory at Tygerberg Hospital (Cape Town, South Africa) participated in this study. Tygerberg Hospital is an academic, tertiary hospital in the Western Cape province of South Africa. Ethical approval was obtained from the University of Stellenbosch and patient confidentiality was maintained. The study was conducted during April 2008. All critical results communicated during this period were logged by the chemical pathologists and registrars in the department. The laboratory has an established list of critical values and results (see Appendix). When a test has a pre-established critical value or result, immediate notification to a care-giver must be made. The recipient of the result was asked to repeat the message (patient name, test result, patient folder number) and any error made by the recipient was recorded. The professional capacity of the recipient was logged as well.
Statistical analysis
Excel data sheets were used for statistical calculations.
Results
Of 472 outgoing telephone calls from the laboratory, 51 errors were detected (error rate 10.8%). The major categories of error were incorrect name of patient, incorrect test result and incorrect folder number. Most of the reported errors were made when recording the folder number (64.7%), with incorrect name of patient being the lowest (5.9%) (Figure 1). When the errors with folder numbers were omitted, the error rate decreased to 3.8%. Results telephoned to clinicians had the highest error rate (20%), most of them being the omission of recording folder numbers or incorrect folder numbers (Table 1). The lowest error rate (3.7%) was noted with reporting results to staff nurses (most results were received by ward sisters).
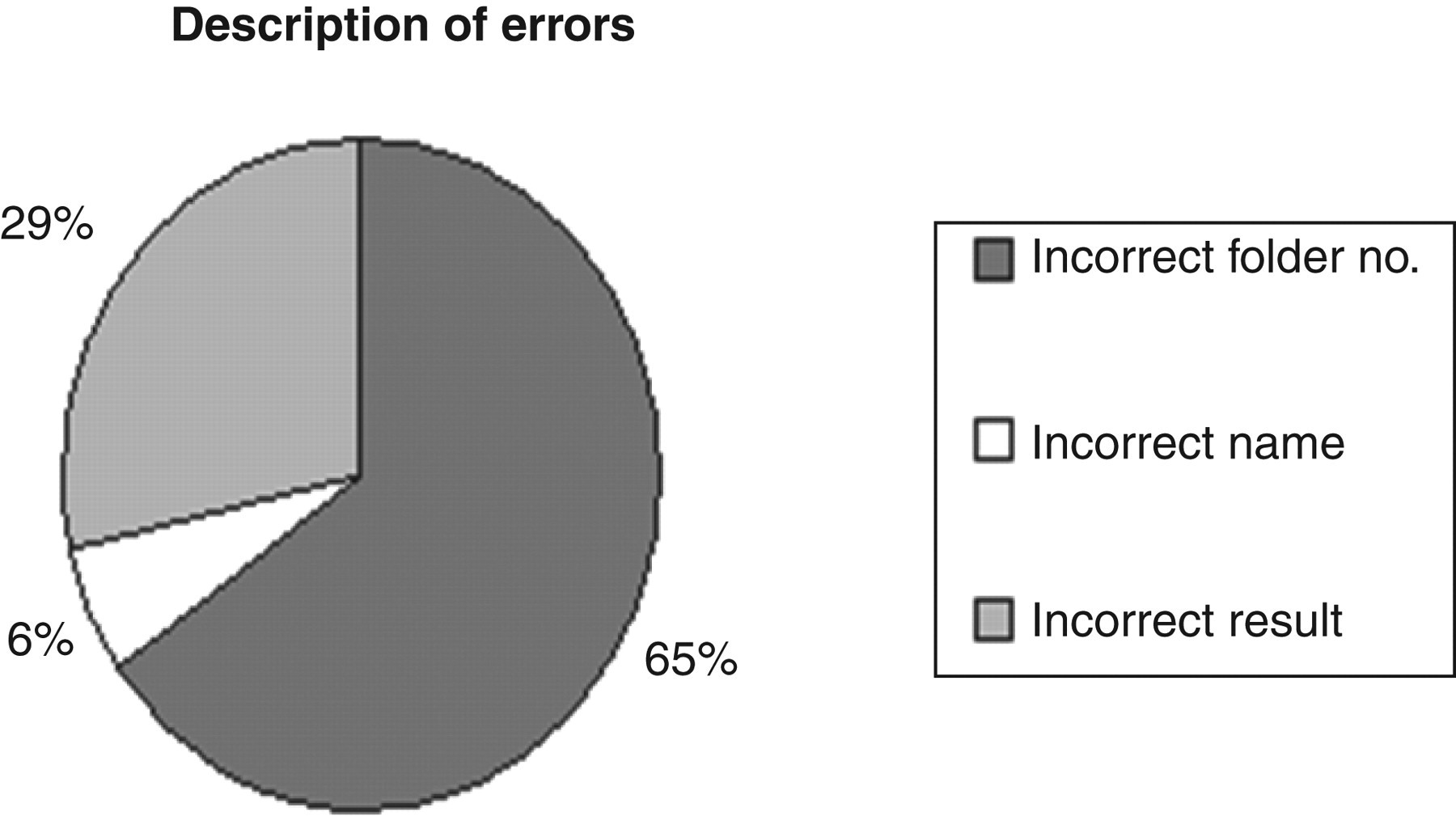
Description of errors
Distribution of telephone calls and errors of critical results by recipient
Discussion
Laboratory data are important to patient care and estimated to affect 70% of medical diagnoses. 6 Many laboratory errors occur outside the analytical phase, and these include errors associated with pre- and postanalytical events. 6 Until recently, there has been little interest in errors occurring during the postanalytical phase. This article highlights the importance of error reduction by read-back of critical results by the recipient and shows that errors during the communication of critical results occur at a low but constant rate. Using the five stages of conducting an audit, as set out in the National Institute for Clinical Excellence (NICE) guidelines, 7 this audit is only at stage three and provides preliminary results on the error rate, as the full audit cycle (stages four and five) have not yet been completed. According to the set guidelines, an audit is only complete after examining implementation of improvements based on results found using the preliminary data.
Two patient identifications (name and folder number) were used to communicate results, resulting in an error rate of 10.8%. With omission of the folder number, the error rate reduced to 3.8% in keeping with previous studies. 8 Regarding medical staff, physicians received only 12.7% of the calls, but had the highest rate of errors, highlighting the need of the read-back of results by this group.
There appears to be a general lack of enthusiasm among clinical staff receiving critical results, despite their potentially serious impact on patient care and consequent medicolegal implications.
Our local accreditation body, SANAS, requires laboratories to have a local policy on the reporting of critical results, but no laboratory in South Africa has yet to acknowledge the JCAHO recommendation requiring the person contacted by telephone to read-back the critical value in order to minimize error. 9 Development of national policies applied in accordance to uniform or internationally accredited standards – such as those of the CPA, JCAHO and CAP may lead to improved communication that is timely, accurate, complete, unambiguous and understood by the recipient; with consequent benefits of a reduced error rate and improved patient safety.
Footnotes
Acknowledgement
We thank Dr R Wicomb for proofreading this manuscript.
APPENDIX
Pre-established critical values for our laboratory
| Analyte | Severely low | Severely high |
|---|---|---|
| Sodium (mmol/L) | ≤120 | ≥155 |
| Potasium (mmol/L) – haemolysed | ≤3.5 | ≥6.0 |
| Potasium | ≤3.0 | ≥6.0 |
| Glucose (mmol/L) | ≤3.0 | ≥20 |
| Calcium (mmol/L) | ≤1.6 | ≥2.8 |
| Magnesium (mmol/L) | ≤0.5 | |
| Phosphate (mmol/L) | ≤0.55 | |
| Lithium (mmol/L) | ≥1.5 | |
| Pseudocholinesterase (kU/L) | ≤3.0 | |
| Bilirubin (μmol/L) (neonates) | ≥250 | |
| Osmolality – serum (mOsm/kg) | ≤250 | ≥310 |
| pH | <7.2 | |
| Triglyccrides (mmol/L) | >1.5 | |
| CSF protein (mmol/L) | >3.5 | |
| CSF glucose (mmol/L) | <1 | |
| Sudden severe change in renal function |
