Abstract
Background
The diagnosis and treatment of intrahepatic cholestasis of pregnancy (ICP) has important implications on fetal health. The biochemical parameter commonly used in the diagnosis of ICP is the determination of the concentration of total serum bile acids (TSBA). However, bile acid profile, especially lithocholic acid (LCA) analysis is a more sensitive and specific biomarker for differential diagnosis of this pathology and also could be an alternative to evaluate the efficiency of ursodeoxycholic acid (UDCA) for ICP treatment.
Methods
Serum bile acid (SBA) profiles including LCA determination, were studied in 28 ICP patients using a capillary electrophoresis method. The effects of UDCA treatment on bile acid profile, were analysed in 23 out of 28 ICP patients and the two samples obtained before and 15 days after treatment were compared. Two samples taken as controls were also obtained from each of five patients without therapy.
Results
A dramatic decrease in LCA concentrations and maintenance of TSBA concentrations were found in all patients after UDCA therapy, whereas SBA profiles together with LCA values did not change in patients without therapy.
Conclusion
We propose LCA as an alternative biomarker and a more sensitive parameter than TSBA to evaluate the effectiveness of UDCA treatment, at least in ICP patients from Argentina.
Introduction
Intrahepatic cholestasis of pregnancy (ICP) also called obstetric cholestasis in some parts of the world is a reversible form of cholestasis that develops in the third trimester of pregnancy and disappears spontaneously after parturition.
Although obstetric cholestasis causes extreme discomfort in the mother, the most important consequences are that it carries risks for the fetus such as premature birth, fetal distress, low weight for gestational age and intrauterine death. 1,2 The most efficient medical management that can improve maternal condition and may prevent the perinatal complications for ICP without adverse effects is the administration of ursodeoxycholic acid (UDCA) during pregnancy. Thus, as soon as obstetric cholestasis is diagnosed, the mother should be considered a high-risk patient and UDCA treatment coupled with close maternal–fetal control are indicated. 3,4 The most common biochemical parameter used in the diagnosis of ICP and follow-up of these patients after UDCA treatment, is the determination of total serum bile acids (TSBA). 5 However, recently, we have demonstrated that individual serum bile acids (SBA), especially lithocholic acid (LCA) measurements, provide more information than TSBA determination in the differential diagnosis of ICP from asymptomatic hypercholanaemia of pregnancy (AHP). 6 AHP was identified as a subgroup of pregnant woman with high concetrations of TSBA showing no pruritus and normal liver function test. 7 In our work, elevated concentrations of TSBA and clear differences between AHP and ICP in the SBA profile were observed. We found that the concentrations of conjugated dihydroxy derivatives were high but without elevations of cholic acid (CA) and LCA levels. 6
LCA, a monohydroxy bile acid, produced from the 7α-dehydroxylation of chenodeoxycholic acid (CDCA), is more toxic than the other bile acids because of its hydrophobic properties. 8,9 Since the toxicity of LCA has been described in several studies 10–15 and some of them have reported that UDCA could be converted to LCA, 16 certain apprehension to treat patients with UDCA during pregnancy is observed. Over the last few years, the increasing interest in the study of bile acid profiles in different diseases has led to improvements in the analytical methodology to determine SBA paterns. One of the latest technological advances capable to simultaneously analyse compounds with different hydrophobicity is capillary electrophoresis (CE). Relevant features of performance such as simplicity, very high resolution in a short time of analysis and low cost of operation, makes CE an alternative methodology in the analysis of SBA profiles including LCA determination. 17,18 Taking into account LCA toxicity and scarce information available about its concentrations in serum after UDCA treatment, we aimed to establish the effect of UDCA therapy on the complete SBA profile, especially on LCA levels, and also to estimate if LCA could be a better marker than TSBA to evaluate the effectiveness of UDCA treatment, using CE as a convenient methodology for these two purposes.
Materials and methods
Settings and study design
A longitudinal study was performed in normal pregnant women and patients with ICP at the ‘J.M. Penna’, Hospital from the Government of the City of Buenos Aires, associated with the University of Buenos Aires, Argentina.
This study was performed according to the principles of the Declaration of Helsinki and was approved by the Institutional Review Board and the Bioethical Commitee of our Institution. Written consent was obtained in every case.
Patients
During the study period, 20 healthy pregnant women and 28 ICP patients in the third trimester of pregnancy were studied.
Diagnosis of ICP was based on the presence of pruritus with elevation of TSBA higher than 11 μmol/L and at least one of the two aminotransferases like alanine-aminotransferase (ALT) or aspartate-aminotransferase (AST) higher than the upper normal limits (40 UI/L and 31 UI/L, respectively), during the second half of an otherwise uneventful pregnancy all of them with normalization of cholestasis after delivery, and the absence of infection by hepatitis viruses (HAV, HBV and HCV), autoimmune diseases, moderate (≤2 drinks/day) to severe alcohol intake, HIV infection, skin diseases or biliary obstruction.
Pruritus was arbitrarily measured in an ordinary scale: grade 1 intermittent, nocturnal, and slight; grade 2, continuous diurnal and nocturnal, from slight to moderate; grade 3 severe and grade 4 severe but also accompanied with insomnia or itching lesions. Only one case with TSBA below the cut-off level was included because the patient suffered from pruritus grade 4 together with personal history of ICP. Complete medical, obstetrical and perinatal data were recorded. Weight, APGAR score 19 and gestational age evaluated by the Capurrós method 20 were registered in newborns. Small-for-gestational age was defined as birth weight for gestational age lesser than the specific 10th percentile cut-off of a published Argentinean fetal growth reference. 21 Weight less than 2500 g at birth was considered as low birth weight, and gestational age less than 37 weeks was considered as preterm. 21 Signs of fetal distress, such as meconium-stained amniotic fluid, clinical or Doppler ultrasonographic signs of hypoxaemia, low birth weight, preterm, small-for-gestational age and APGAR score at the first minute less than 7 were considered as newborn complications.
Twenty-three patients with ICP were treated with UDCA (900 mg/day) until delivery and two samples were obtained from each patient: one at the moment of the diagnosis (before treatment) and the second, 15 days after the begining of the treatment.
Two samples taken within 15-day periods, were also obtained from five ICP patients who did not give their consent to receive UDCA therapy. Additional clinical data of the patients studied are shown in Table 1.
Clinical and perinatal characteristics in normal pregnant women and patients with intrahepatic cholestasis of pregnancy (ICP)
Results are expressed as means ± SEM
*P < 0.01
†Fetal distress, low birth weight, small for gestational age and APGAR score <7 at first minute
Methods
Liver function tests
Serum samples were obtained after a fasting period of 8 h, and aliquots were frozen at −20°C until bile acid determinations were performed. Activities of ALT, AST, alkaline phosphatase (ALP), gamma-glutamyltranspeptidase and concentrations of total and conjugated bilirubin were carried out by routine automated techniques.
TSBAs and SBA profile
Total SBA, CA, CDCA, deoxycholic acid (DCA), LCA and UDCA, in their free, glycine and taurine derivative forms were assessed by CE. A detailed description of the analytical method performed in this study has been described by Tripodi et al. 18 Briefly, simultaneous determinations of 15 SBA in serum samples were performed using an off-line C18 solid-phase extraction procedure for sample clean-up and concentration. This step was followed by the complete separation of the bile acids using cyclodextrine-modified micellar electrokinetic chromatography with UV detection in less than 12 min. The limit of detection of this method were: 0.80–1.1 μmol/L for free bile acids forms, 0.19–0.27 μmol/L for glycine-derivative forms and 0.30–0.40 μmol/L for taurine-derivative forms. The imprecision of the method expressed as relative standard deviation (RSD) was 7%. The same determinations were carried out in serum samples taken from patients and controls.
Statistical analysis
Results were expressed as mean ± SEM. Shapiro-Wilkś W test of normality was performed. Differences between groups were analysed by Student's t-test. Levels of significance were established at P < 0.05.
Results
Women with ICP had significant concentrations of total and direct bilirubin, aminotransferases and ALP higher than normal pregnant women. Only six out of 28 ICP patients (21.4%) had gammaglutamyl transpeptidase levels slightly above the upper reference range of 32 UI/L (Table 2). Evaluating the normality of the data using a Shapiro-Wilkś W test, we have observed that bile acids were not normally distributed (P < 0.05).
Biochemical characteristics in normal pregnant women and patients with intrahepatic cholestasis of pregnancy (ICP) before and after ursodeoxycholic acid (UDCA) treatment
Results are expressed as means ± SEM. For total bilirubin, the reference range is up to 18.8 μmol/L; for direct bilirubin the reference range is up to 5.1 μmol/L. The reference ranges for AST, ALT and ALP are 12–31, 12–40 and 90–240 UI/L, respectively
*Represents significances of the data from ICP patients before UDCA treatment with respect to normal pregnant women. P < 0.03
†Represents significances of the data from ICP patients after UDCA treatment respect to ICP patients before UDCA treatment. P < 0.03
‡Represents significances of the data from ICP patients after UDCA treatment respect to ICP patients before UDCA treatment. P < 0.02
Effects of UDCA administration: clinical, perinatal and liver function tests
The effects of UDCA administration were studied in 23 patients out of 28 ICP women. All ICP-treated patients presented severe pruritus (grade 3 or 4) but it disappeared or was markedly reduced during UDCA treatment, except one case where no improvement of this symptom was observed. Five ICP patients without UDCA treatment presented slight to moderate pruritus (grade 1 or 2) remaining unchanged during 15-day treatment. They also showed a slight rise in the concentrations of the biochemical markers of cholestasis. Personal or familiar history of ICP was found in seven out of 28 patients. Labour at term was induced in five patients because of ICP, caesarean was performed in five and preterm delivery was observed in two patients. No neonatal death was observed during the study. Additional data are shown in Table 1. Table 2 shows a significant reduction in all liver functions tests after UDCA treatment.
Effects of UDCA administration on SBAs
Women with ICP, before UDCA treatment, have significantly higher levels of TSBA than clinically healthy pregnants (51.7 ± 7.2 vs. 7.3 ± 0.6 μmol/L, P < 0.001). After UDCA administration, we have observed a dramatic decrease in LCA (P < 0.002, Figure 1a, Figure 2, Table 3), whereas TSBA levels remained elevated in all ICP-treated patients (Figure 1b, Table 3) probably due to the therapy that produces an increment in UDCA concentrations from 4.5 μmol/L to 27.2 μmol/L (P < 0.002) in which 38.2% of the last value corresponds to UDCA in the free form and the other 61.8% to the conjugated form. We have also observed a reduction in DCA values after UDCA treatment (P < 0.05, Figure 1c, Figure 2) with no significant differences in CA and CDCA (Figure 2). Table 3 shows the raw data, both before and after, for all the 28 patients individually for TSBA and LCA.
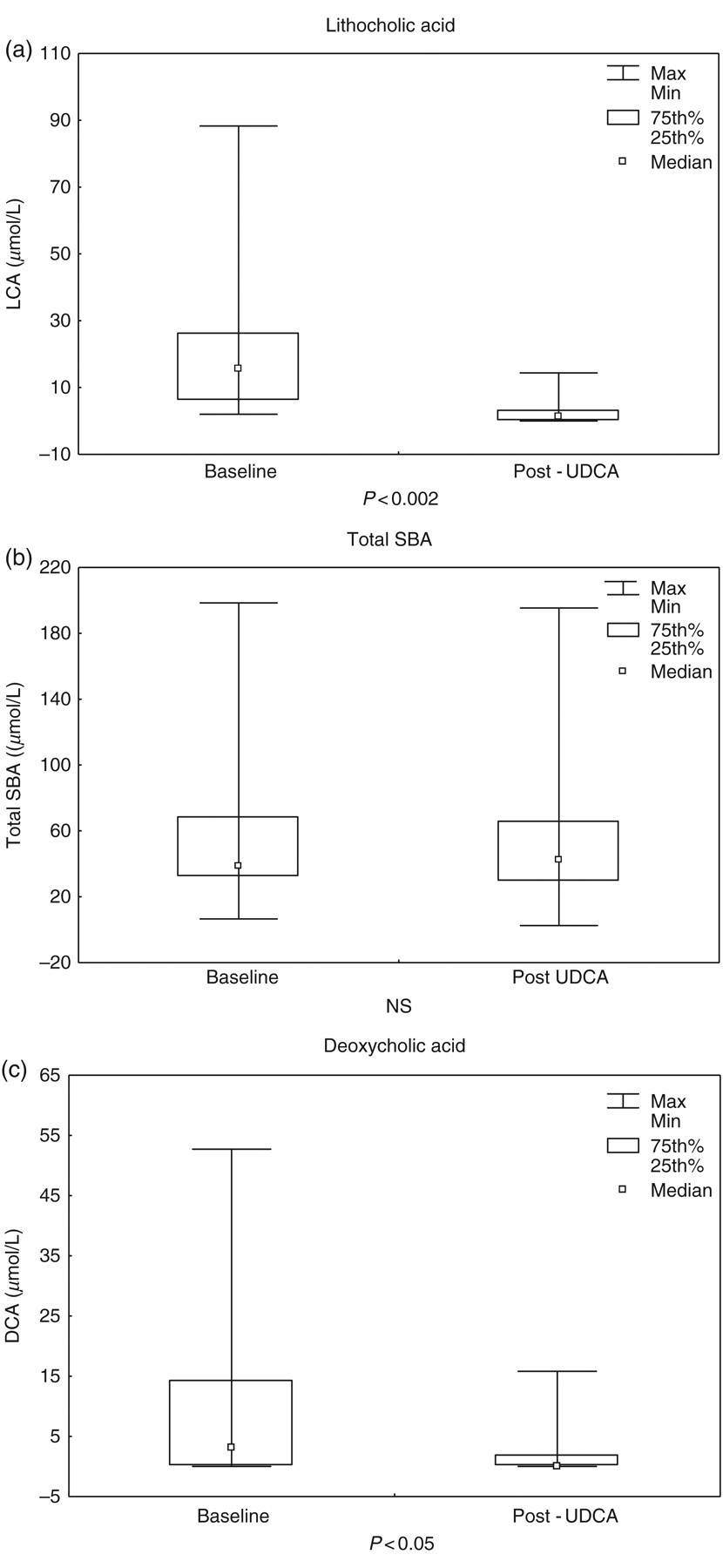
The effect of ursodeoxycholic acid therapy in lithocholic acid (a), total serum bile acids (b) and deoxycholic acid (c) levels in 23 intrahepatic cholestasis of pregnancy patients
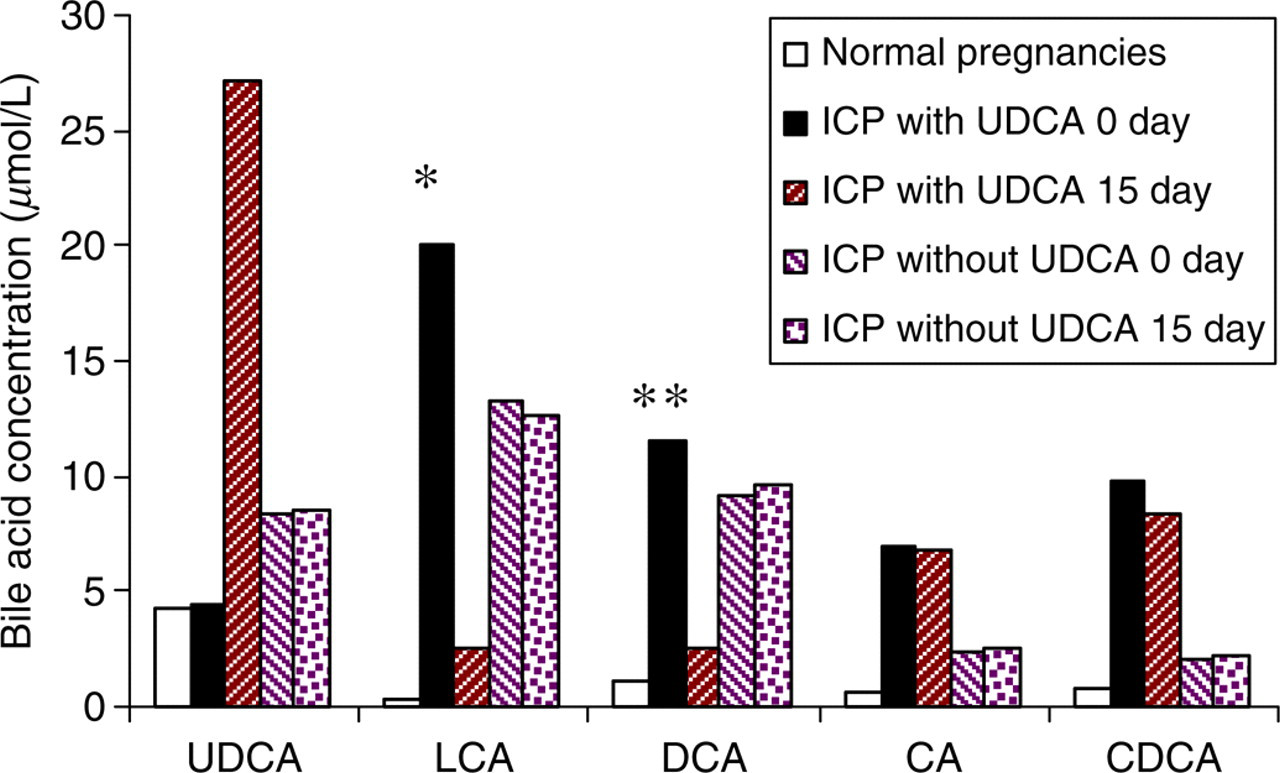
Comparison of serum bile acid profiles (mean ± SEM) in normal pregnancies (n = 20) and intrahepatic cholestasis of pregnancy patients with (n = 23) and without (n = 5) ursodeoxycholic acid (UDCA) treatment. Bile acids are expressed as the sum of their free, glycine and taurine forms. *P < 0.001 (difference in lithocholic acid from day 0 to day 15 with UDCA treatment), **P < 0.03 (difference in deoxycholic acid from day 0 to day 15 with UDCA treatment)
Comparison of individual total serum bile acids (TSBA) and lithocholic acid (LCA) concentrations (μmol/L) in serum from patients with intrahepatic cholestasis of pregnacy (ICP) with and without ursodeoxycholic acid (UDCA) treatment
+Denotes with UDCA treatment; −denotes without treatment; ND, not detectable
The whole group of UDCA-treated patients studied had high baseline LCA concentrations showing a signficant decrease after UDCA therapy in all patients (100%), whereas DCA decrease was observed in 73.9% of treated patients (17 out of 23) and TSBA concentrations are highly overlapped before and after treatment.
As mentioned before, we have observed that, except in one case, ICP-treated patients had a dramatic change in their pruritus scores. This change in symptom highly correlated with the fall in LCA levels after UDCA treatment (correlation coefficient: 0.996).
Figure 3 shows the electropherogram of one patient before (Figure 3a) and after (3b) UDCA administration.
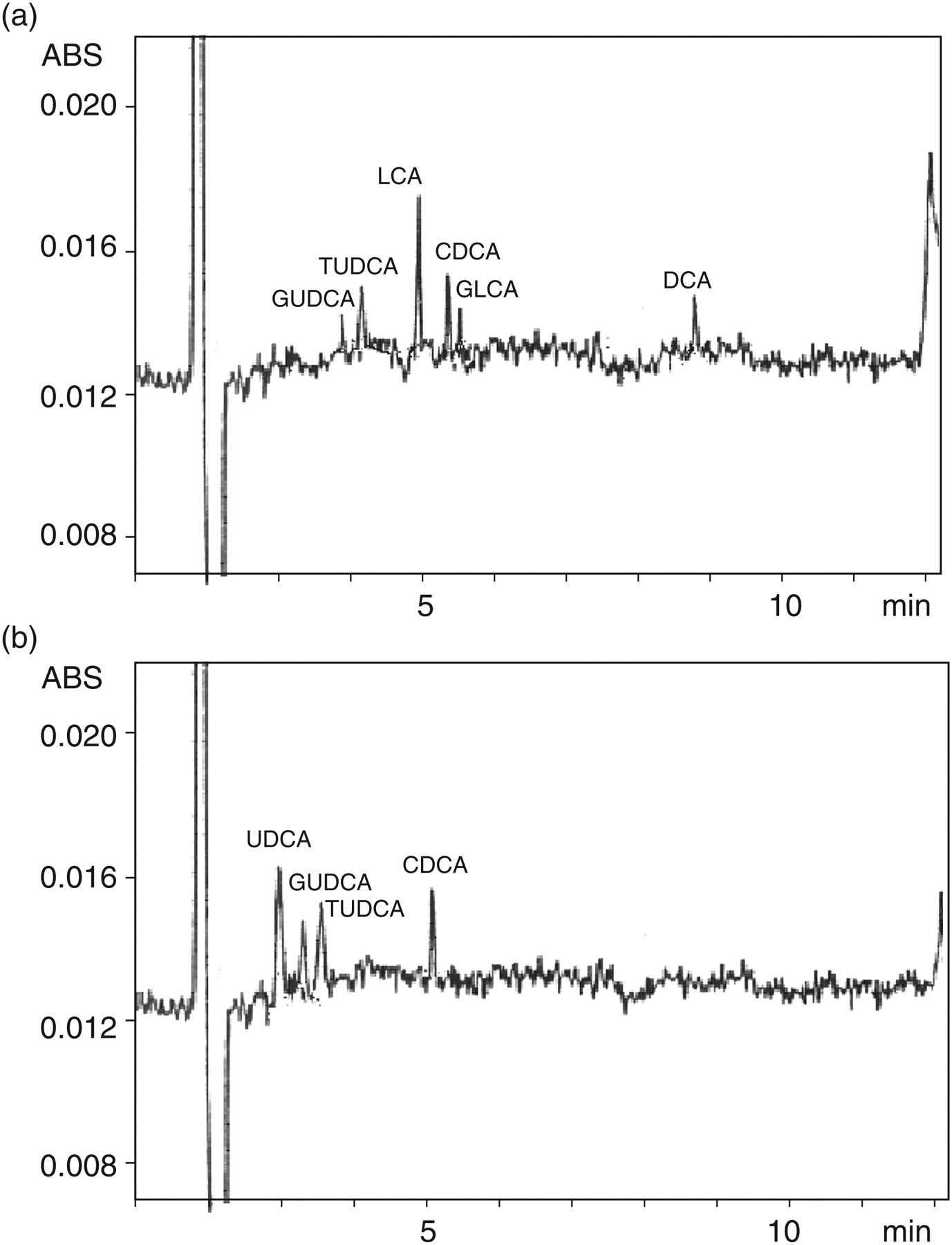
Electropherogram of a patient with pregnancy cholestasis showing serum bile acid profiles. (a) Before ursodeoxycholic acid (UDCA) treatment (total serum bile acid [TSBA] 23.2 μmol/L, glycoursodeoxycholic acid [GUDCA] 1.8 μmol/L, tauroursodeoxycholic acid [TUDCA] 2.2 μmol/L, lithocholic acid [LCA] 7.3 μmol/L, chenodeoxycholic acid [CDCA] 5.1 μmol/L, glycolithocholic acid [GLCA] 2.4 μmol/L, deoxycholic acid [DCA] 4.4 μmol/L). (b) After UDCA treatment (TSBA 21.2 μmol/L, UDCA 8.1 μmol/L, GUDCA 3.0 μmol/L, TUDCA 4.2 μmol/L, CDCA 5.9 μmol/L).
After analysing SBA profiles from ICP patients not treated with UDCA, we found a rise in total and individual bile acids with high LCA values and we could not observe a significant difference in their concentrations and their profiles during the period of study (Figure 2).
Conclusions
In this study we describe modifications in the complete SBA profile determined by CE, between the groups of patients with ICP-treated and non-treated with UDCA. Although the mechanisms of UDCA protection still remain unclear, it is known that UDCA administration is at present the most efficient therapy in ICP condition. UDCA therapy may be associated with its ability to reduce the hydrophobicity of the bile acid pool. UDCA is hepatoprotective where it tends to increase bile flow by acting as a choleretic agent 22 and it displaces the more hydrophobic and toxic bile acids from hepatic membranes in cholestatic conditions. 23,24 As it is known that LCA is the most hydrophobic bile acid, 25 its decrease is possibly the more significant one. However, some authors have reported the probability that UDCA could be converted to LCA 16 increasing its concentrations during UDCA treatment, and eventually also fetal morbidity. However, Brites et al. 24 have determined some bile acids by thin layer chromatography (TLC) and they have found that LCA concentrations remained constant during UDCA administration.
In our work, we have observed a relief of pruritus together with a dramatic decrease in LCA concentrations in all patients following UDCA therapy. Disagreement between the results observed by Brites and ourselves could be attributed to differences in the populations studied or in the methodologies applied in each case. Nevertheless, LCA concentrations did not increase in both the studies.
A reduction in DCA concentrations was also observed though LCA and DCA did not reach the normal pregnancy values during the study.
At present TSBA determination is used to diagnose ICP condition and also to monitor UDCA treatment. In a previous work, we have reported that TSBA concentrations overlapped with those of healthy pregnant women with hypercholanaemia, making it difficult to obtain a differential diagnosis solely on the basis of TSBA measurement. In the present study, we have also found that TSBA concentrations overlapped before and after UDCA therapy (Figure 1b). Instead, LCA concentrations were high in all ICP patients but decreased only in the group of treated patients. Thus, the mentioned results indicated that LCA is a more sensitive marker than TSBA to evaluate the therapeutic monitoring of ICP.
We have also observed that a decrease in the concentrations obtained in individual SBA did not have any time effect, since SBA profiles in ICP patients without therapy did not change during 15 days. This result shows that the decrease in LCA and DCA concentrations may be due only to UDCA treatment. We are aware of the fact that the number of cases in the group of ICP patients without UDCA therapy (n = 5) is small for comparison but it is now difficult to find patients who are not treated with UDCA. Nevertheless, SBA profile is similar to those obtained with the other ICP patients showing increments of total SBA, LCA, DCA and free bile acid levels compared with the group with normal pregnancy and thus it confirmed ICP diagnosis.
Previous reports, 3,24 have shown a decrease in CA and CDCA after UDCA treatment, but we could not find a significant difference in their concentrations within the group of patients studied during and after 15-day treatment.
However, in agreement with the available literature, 5,24,26,27 we found that UDCA treatment improves pruritus and hepatic biochemical tests without adverse effects.
With regard to the concentration of transaminases, we have found a relatively low concentration of AST and ALT indicating that, probably, damage of liver cells was not significant enough. With accordance to the bibliography, 28 ALP was found to be high in ICP patients, mean while gammaglutamyl transpeptidase was found to be slightly elevated only in six out of the 28 patients. As ALP concentrations are of poor diagnostic value in ICP and gammaglutamyl transpeptidase concentrations are only highly elevated in cases of MDR3 gene mutation, 28 they were not taken into account as a criteria for selection of patients in this study.
In conclusion, a dramatic decrease in LCA concentrations is observed in all patients during UDCA therapy, whereas TSBA concentrations are maintained, showing that LCA determination helps to evaluate the evolution of UDCA therapy and it is more sensitive than TSBA. Our conclusions are based on results of the pattern of SBAs performed using a CE methodology that can assure the simultaneous separation of 15 bile acids in their free and tauro- and glyco-congugated forms determined with high precision and accuracy and in a short time of analysis. Moreover, taking into account the usefulness of bile acid determination as a tool for diagnosis or therapeutic monitoring, it is important to emphasize that using the CE method, a complete SBA profile including LCA could be obtained in less than 1 h including the off-line sample clean-up and bile acid CE quantitation, so that the clinician could get these assay values back no longer than one day.
In brief, from the results obtained by a CE method and clinical history of ICP patients, we propose LCA as an alternative biomarker and a more sensitive parameter to evaluate the effectiveness of UDCA treatment in these patients.
At present, further investigations are needed to estimate if LCA determination can be used with the purpose of enhancing the prognosis of perinatal complications in ICP pregnant women.
Footnotes
Acknowledgements
We thank the Obstetrics and Gynecology Division Staff and the Laboratory Staff of J.M. Penna Hospital for their continuous cooperation and Dr Jorge Muse for assistance. This study was supported, in part, by the National Council of Scientific and Technological Research (CONICET) and by the University of Buenos Aires (UBACyT B128).
