Abstract
Background
To improve current alpha-fetoprotein (AFP) assays, which are expensive and time-consuming, a specific AFP reagent has been developed for practical use in our newly developed high-speed, highly sensitive pulse immunoassay (PIA) system, in which a latex immunoagglutination reaction is carried out under a high-frequency pulse voltage, leading to an enhanced immunological reaction.
Methods
We evaluated the assay performance (reproducibility, sensitivity, dilution linearity, interference) of the newly developed automated AFP PIA compared with the current AFP assay.
Results
Using pooled serum samples, the within-run reproducibility resulted in a correlation variation of 3.6–4.7%. The AFP assay detection limit was determined to be 2.5 μg/L. Linear sequential dilution was found up to nearly 700 μg/L. Even up to an AFP concentration of 1.0 g/L, the prozone phenomenon was not observed. Free and conjugated bilirubin, haemolytic haemoglobin, chyle and rheumatoid factor did not show any test interference. Using AFP-positive serum samples from 114 patients, the correlation between our PIA and a chemiluminescence immunoassay resulted in an excellent correlation coefficient of 0.994.
Conclusions
The performance of AFP reagents in the PIA device shows that the system has excellent speed and equal sensitivity and specificity compared with the most highly sensitive conventional method. Our PIA system thus appears ready for use in the clinical diagnosis setting.
Introduction
The latex agglutination turbidity method has a well-known application as a simple immunological analytical method. An antibody (or antigen) is bound to small latex particles (diameter 0.05–0.5 μm) and when an antigen (or antibody) in a sample reacts, the latex particles agglutinate. This agglutination causes a change in turbidity in the solution, and the related change in absorbance is a quantitative measure of the concentration in the sample. Latex agglutination is believed to occur by Brownian motion of the antigen and antibody. Because this diffusion is a comparatively slow process, a reaction time on the order of hours is needed. However, because clinical diagnostics require a high throughput, practical latex agglutination assays should be carried out on the order of minutes.
In general, latex agglutination assays show a lower sensitivity when compared with enzyme-linked immunosorbent assays (ELISA) and chemiluminescence immunoassays (CLIA), and it is generally insufficient for detecting infectious diseases and tumour markers. However, the latex agglutination method has the advantage of being a simple and cost-effective method. On the other hand, ELISA and CLIA assays are more expensive due to cost of enzymes, conjugation procedures and stability requirements, as well as costs related to shortening the assay time.
Thus, Tamiya and Karube 1 invented the pulse immunoassay (PIA) method, a novel methodology in latex agglutination, to equal the sensitivity of the most highly sensitive conventional method. The PIA method 2–5 is a technology to enhance, in a very high-speed and high-sensitivity mode, an immunological agglutination reaction. Briefly, a reagent consisting of 1–3 μm latex particles coated with an antibody (or antigen) is mixed with a sample (presumably containing an antigen or antibody of interest), and this suspension is placed between two electrodes. A pulse voltage is applied over the electrodes for several tens of seconds, and latex particle agglutination is enhanced. Using this method, the amount of antigen or antibody of interest in the sample can be measured. 6 Applying a pulse voltage to a latex suspension results in strands of particles, called pearl chains, due to dielectric polarization of the latex particles and subsequent electrostatic attraction. When the voltage is switched off, the pearl chains immediately return to single particles in the case of a negative sample. However, when an immunological reaction has occurred in the case of a positive sample, when the voltage is switched off, the pearl chains remain. Repeating this pulse mechanism, increases the probability of antibody-antigen contact thus enhancing the immunological latex agglutination reaction.
In our previous study of instrument basics, we reported that the PIA method resulted in assay sensitivity on the same order as ELISA and CLIA assays. 5 The latex agglutination reaction, when pulse voltage is applied, results in an increased reaction speed of the immunological agglutination reaction and higher sensitivity. This has been verified using samples spiked with standard reference material of myoglobin 4 and AFP 5 in the buffer.
In this study, using the AFP reagents with our newly developed PIA instrument, we compared the assay speed, sensitivity and specificity with conventional methods and proved its clinical use.
Materials and methods
Materials
Buffers
Glycine-buffered saline (GBS) consisted of 50 mmol/L of glycine, 150 mmol/L NaCl and 0.9 g/L of NaN3, adjusted to pH 8.2 with NaOH.
Standard
Human alpha-1-fetoprotein standard (code no. X0900) was obtained from Dako A/S (Glostrup, Denmark).
Antibody
Polyclonal rabbit immunoglobulin against human alpha-1-fetoprotein (code no. A0008) was obtained from Dako A/S.
Clinical samples
Serum samples were collected from Department of Clinical Laboratory of Saitama Medical Center of Jichi Medical University and stored frozen at −80°C before assaying. The AFP specimens of ambulatory patients and inpatients of the Saitama Medical Center were used following approval from the ethics committee of Jichi Medical University. The AFP concentration in samples from the centre were determined by CLIA using the LUMIPULSE® AFP-N′ (Fujirebio Co., Tokyo, Japan).
Reagents
Bovine serum albumin (BSA) was obtained from Sigma Chemical Co (MO, USA). Polystyrene latex (particle diameter 1.5 μm, 10 g/L) was obtained from Sekisui Chemical Co. Ltd (Osaka, Japan).
Methods
Purification of antibody
Rabbit IgG fraction against human alpha-1-fetoprotein antibody was purified using a DEAE-cellulose column and dialysis (17.5 mmol/L phosphate buffer).
Preparation of IgG-bound latex
One volume of 10 g/L latex particles in GBS was added to one volume of purified IgG fraction of anti-human alpha-1-fetoprotein antibody. The suspension was gently stirred at 37°C for 2 h. The latex-IgG suspension was twice-centrifuged and washed with 10 g/L BSA-GBS buffer. The latex-IgG reagent was subsequently diluted to 5 g/L and preserved at 4°C until use.
Standards
We diluted human alpha-1-fetoprotein standard with physiological salt solution containing 10 g/L BSA to obtain standards at concentrations of 2.5, 5.0, 10, 20, 40, 80, 160, 320 and 640 µg/L. The value of AFP is based on the standard reference material (WHO International Standard of AFP) of NIBSC (National Institute for Biological Standards and Control).
Apparatus
The PIA instrument is built of three modules (Figure 1): a dispensing unit, a microscopic unit and a power unit to apply the pulse voltage. The instrument is able to carry out three assays simultaneously. It is possible to dispense a solution ranging from 4.0 μL to 30 μL using a disposable tip of 30 μL. Furthermore, this dispensing unit is able to mix a solution by repeating aspiration and dispensing. The voltage of the pulse power supply can be preset from 0 Vp-p to 50 Vp-p. The frequency, producing a rectangular wave output, ranges from 1 kHz to 10 MHz. The microscopic unit contains an objective lens (×20), microscope tube (tube length 120 mm, incident-light LED illumination), extended lens (×1.5) and charge-coupled device (CCD) camera (1.3 million pixels).
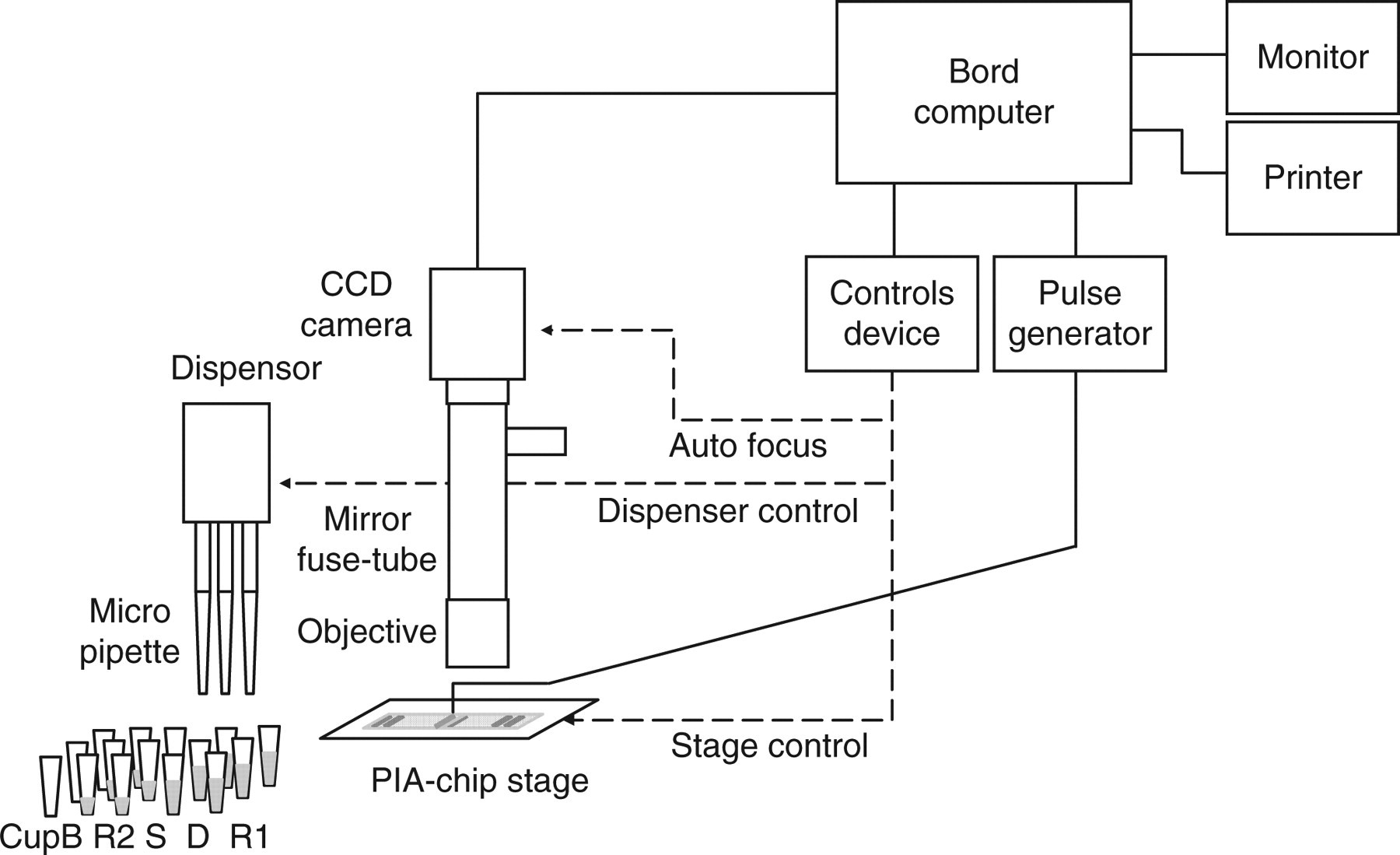
Schematic diagram of the pulse immunoassay system. R1: buffer solution; D: dilution solution; S: sample; R2: latex reagent. Cup B: Sample is mixed with the reagents R1 and R2 and this cup serves for the first reaction. The holder of this cup (incubator) can be set at 37–70°C
Microreactor
A Pyrex glass plate containing three microchannels was designed as a microreactor for the PIA (and called a PIA-chip). Microchannels were etched to a depth of 10 μm, width of 0.5 mm and length of 5 mm; therefore, the reaction chamber volume in each microchannel was 0.025 μL.
Assay procedure
By following steps 1–8 (below) with the prototype of our PIA instrument, optimal running conditions were developed for automated measurements. Assay reproducibility, detection limit, dilution linearity and other parameters were determined using this protocol.
Serum dilution solution: 10 g/L BSA-saline (36 µL) was added to serum (4 μL) and mixed by repeated aspiration and dispensing; Reagent R1: 4 μL of the diluted serum solution was added to GBS (20 μL) and mixed by repeated aspiration and dispensing; Reaction solution: Reagent R1 (4 μL) was added to cup B, and after 15 s, 4 μL of Reagent R2 (AFP-coated latex) was added and mixed by repeated aspiration and dispensing; After 15 s, 1.5 μL of the reaction solution was dispensed on the PIA-chip, thereby filling one microchannel on the chip through capillary force. Up to three microchannels can be filled at the same time; A pulse voltage (frequency, 400 kHz; voltage, ±11 V; rectangular wave) was applied during 90 s over the electrodes forming the capillary on the PIA-chip; After 40 s, images were taken using the microscope and CCD camera and uploaded onto the PC (10 images per microchannel); Particle distribution analysis was carried out from data of these images; the agglutination ratio (the number of two or more agglutinated particles divided by the total number of particles) was calculated and the sample concentration determined from the standard curve; Finally, the measurement results were provided as agglutination ratio and sample concentration.
Statistical analysis
Within-run and between-run were determined with a nested analysis of variance. Linear regression analysis (least-squares method) was used for correlation studies. Statistical analysis was performed with Excel 2003 (Microsoft Corp., USA) software.
Results
The calibration curve of AFP (Figure 2A) is shown at concentrations ranging from 0 to 640 μg/L.
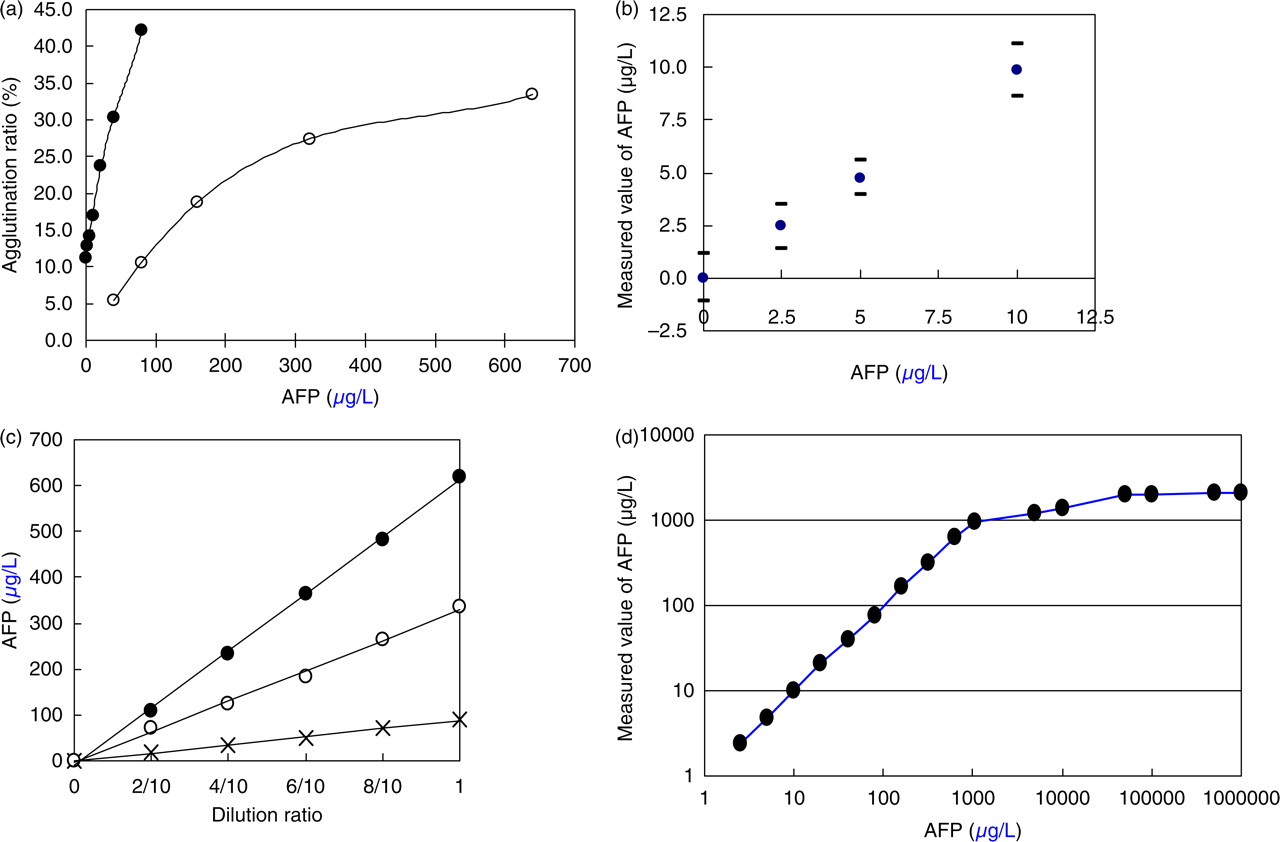
Analytical characteristics of pulse immunoassay of alpha-fetoprotein (AFP). (a) Calibration curve for pulse immunoassay of AFP. The agglutination ratio is expressed as the number of two (•) or three or more (○) agglutinated particles divided by the total number of particles. (b) Detection limit. The mean value ± 3SD (n = 6) determined is plotted against AFP concentrations 0, 2.5, 5.0, and 10 μg/L. (c) Linearity. The dilution ratio (up to 2/10) is plotted against measured AFP values of three serum sample pools (L, M, H). (d) Prozone phenomenon. Dilutions from the high AFP concentration 1.0 g/L are measured and plotted against the theoretical value.
Reproducibility
Using pooled serum samples of three concentrations (L, M, H), the within-run reproducibility (20×) resulted in a coefficient of variation (CV) of 3.6–4.7%, indicative of excellent reproducibility. Moreover, when these pooled serum samples (preserved at −30°C) were freshly thawed to room temperature and measured that same day for a consecutive 20 days, the between-run reproducibility showed a CV ranging from 4.0% to 5.9%, again indicative of excellent reproducibility (Table 1).
Precision of the pulse immunoassay for the determination of alpha-fetoprotein
SD, standard deviation; CV, coefficient of variation
Detection limit
The AFP calibrator (10 μg/L) was diluted with the dilution solution to 5.0 μg/L and 2.5 μg/L. Each concentration (including 0 μg/L, dilution solution) was measured six times and the average value and standard deviation (SD) were determined. In Figure 3B, the average value and SD interval (+3SD and −3SD) are given. From these experiments, the detection limit was determined to be 2.5 μg/L (Figure 2B).
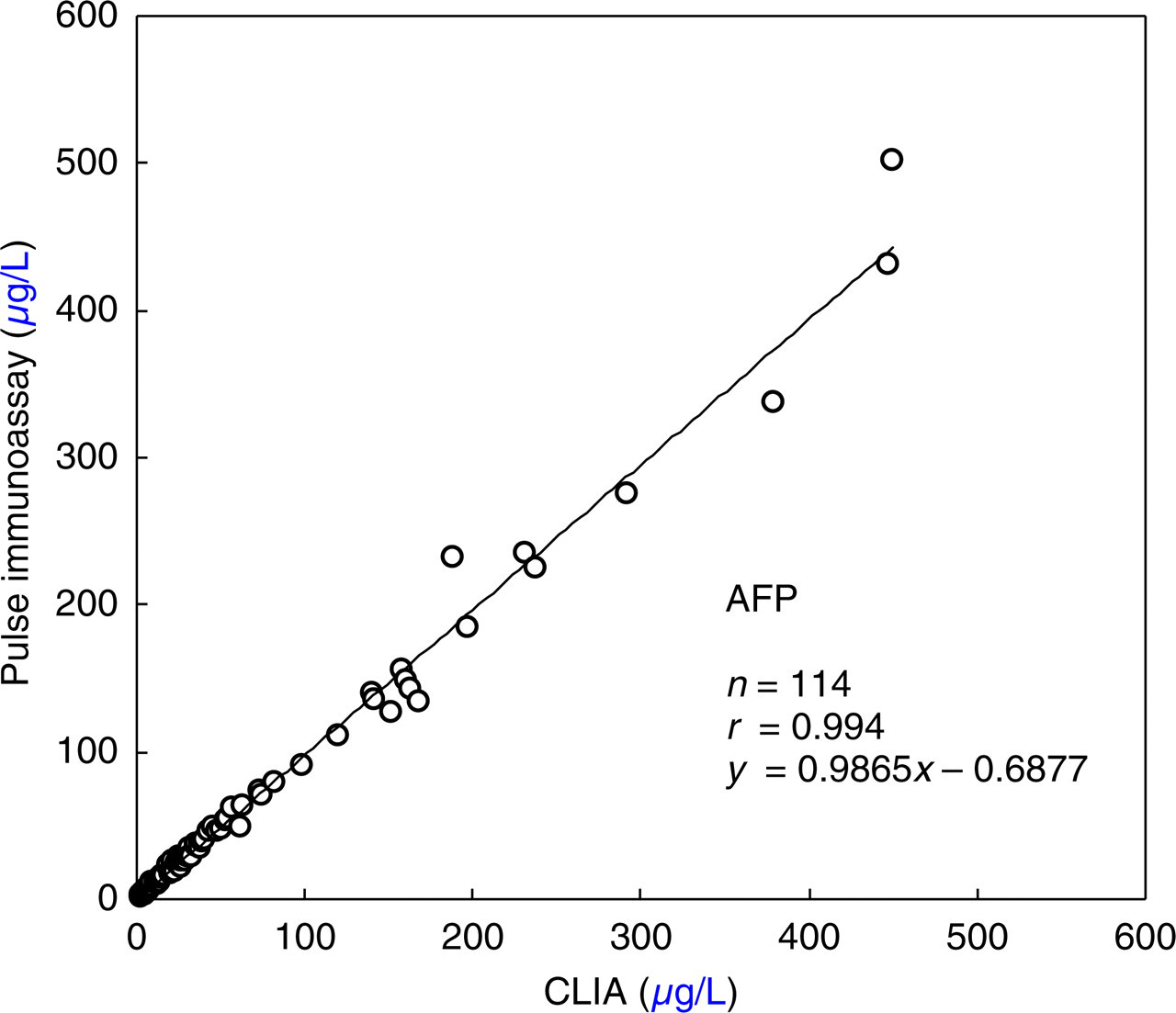
Correlation between pulse immunoassay and the chemiluminescence immunoassay (CLIA) method for the determination of alpha-fetoprotein in 114 serum samples. Pulse immunoassay = 0.99(CLIA) − 0.69, r = 0.994
Dilution linearity
For each of the three pooled serum samples, sequential dilution (five times) with the dilution solution was carried out. All three showed a good linearity, passing through the origin of the graph (Figure 2C). Linear regression analysis gave the relationships of: (L) y = 90.9 × −1.51, r = 0.998; (M) y = 331.4 × −2.64, r = 0.998; (H) y = 622.2 × −9.36, r = 0.999.
Prozone phenomenon
The high AFP calibrator (1.0 g/L) was diluted with the dilution solution, as shown in Figure 2D. As a result that the samples of 0.1–1.0 g/L of the concentration of AFP were measured, the agglutination ratio became a plateau near 42.5% (AFP approximately 2000 μg/L). Even up to an AFP concentration of 1.0 g/L, the prozone phenomenon was not observed.
Interference influence
To evaluate the effect of potentially interfering substances, in separate experiments, we spiked five different substances into five-fold-diluted pooled serum. Free and conjugated bilirubin, haemolytic haemoglobin, chyle (Interference Check A Plus, Sysmex Co, Kobe, Japan), and rheumatoid factor (Interference Check RF Plus, Sysmex Co.) were evaluated. From the concentrations studied, we did not find any interference influence (Table 2).
Evaluation of potentially interfering substances
Correlation with CLIA
Using positive serum samples from 114 patients, the correlation between our PIA and CLIA resulted in an excellent correlation coefficient of 0.994 (Figure 3).
Discussion
Using our newly developed PIA system (PIS), we applied AFP reagents and found excellent basic data results for the reproducibility, detection limit, dilution linearity, prozone phenomenon and interference influence. A very good correlation between our PIA system method and the conventional CLIA method was found.
In our method, the immunological reactivity was increased by mixing a sample with conjugated latex (diameter, 1–2 μm) and incubating it at 56°C for a short time (15 s) to enhance the Brownian motion between the AFP antigen and the antibody-coated latex particles. It is believed that the assay sensitivity is higher when there are no free AFP antigen molecules in the reaction solution. We achieved this condition by applying a pulse voltage over the reaction solution; due to dielectric polarization of the latex particles, particles are attracted in an electrostatic manner, and the antigen–antibody reaction causes particle agglutination in so-called pearl chains. 4,5
In conventional basic research, a latex diameter of 2–3 µm is used to easily form and disperse pearl chains. Even though smaller particles show a higher diffusion in solution, our novel system using latex particles with diameter of 1–2 μm showed an enhanced immunological reaction and pearl chain formation. Specifically, the total reaction time in our extremely high-speed method is 160 s, i.e. incubation time was 30 s (non-specific adsorption reaction time, 15 s; first reaction enhancement time, 15 s) and the pulse immunological reaction assay time was 130 s (pulse voltage application time, 90 s; re-dispersion time, 40 s). In comparison, the CLIA-based LUMIPULSE® AFP-N has a reaction time of about 25 min.
However, when enhancing the immunoreactions, unwanted non-specific reactions may interfere concomitantly. Rheumatic factor in a patient sample is one factor that may cause a non-specific reaction. 7,8 To avoid non-specific reactions, various options are available, such as coupling of F(ab′)2 fragments to latex or preprocessing of the patient sample by absorbing or otherwise inactivating the interference factor. 9–11 In the most widely used latex agglutination methods, non-specific agglutination reactions are suppressed by mixing the patient sample with a specific buffer solution (with incubation of about 5 min at 37°C), and subsequently mixing this sample solution with the latex reagents. To overcome non-specific reactions in our method, a high-temperature 56°C shock for only 15 s was used, because high speed is an essential feature.
In conventional Brownian motion-based latex agglutination methods, one of the problems is the so-called prozone effect, which occurs as a result of excess antigen in the sample. 12 Although prozone effect has been seen to be rare with AFP concentrations of up to 1.0 g/L, various options to avoid the prozone phenomenon are utilized. For example, the mixing of small and larger latex particles, 13 the combining of polyclonal and monoclonal antibodies 14 or using latex particles with a different reactivity are positive factors, while difficulty in controlling lot-to-lot variation and increasing costs are negative factors. It is believed that in conventional latex agglutination methods, the prozone phenomenon occurs faster than theoretically predicted (from the numerical balance between antigen and antibody molecules) due to the progressive formation of a three-dimensional network at high concentrations. Conversely, our method does not show the prozone effect, even with high AFP concentrations, as can be seen in Figure 2D. It is thought that in the PIA agglutination method, the numerical balance between antigen and antibody is favourable to avoid the prozone effect because the latex particles are forced to form pearl chains due to electrostatics.
In conclusion, our PIS is a high-speed, highly sensitive method. Moreover, the chip reaction chamber has a volume of 0.025 μL, and the chip will increase efficiency by simultaneously detecting several items from a single drop of blood. We expect an inexpensive disposable chip to become available in the near future with the potential to halve the cost of a pulse immunoassay compared to conventional methods. The clinical significance of this technology in infectious disease control is its speed, as various viral antigens or their antibodies can be detected very quickly. In addition, the small sample volume facilitates the diagnostic testing of patients from whom blood is difficult to collect, such as infants and older adults. Finally, other applications such as allergy chips that quickly detect various allergens are possible.
Footnotes
Acknowledgements
The fully automated measurement instrument for pulse immunoassay used in this study was supported by a commercialization subsidy (20050309-01) from the Organization for Small and Medium Enterprises and Regional Innovation, Japan (SMRJ). We thank K. Kubo (FSI) for assisting the development of particle size distribution software.
