Abstract
Abstract
Background
Lysophosphatidic acid (LPA) plays important roles in a variety of biological responses, especially in the area of vascular biology, and the determination of its plasma concentration is believed to be important. Several mechanisms are known to be involved in the metabolism of LPA.
Methods
To identify factors that may determine the plasma concentrations of this important bioactive lipid, we examined its concentrations using an enzymatic cycling assay and related parameters in 146 healthy subjects.
Results
The LPA concentration was significantly higher in women (mean ± SD, 0.103 ± 0.032 μmol/L; n = 47) than in men (0.077 ± 0.026 μmol/L; n = 99). A multiple regression analysis showed a strong positive correlation between the plasma LPA concentration and serum lysophospholipase D (lysoPLD) activity, while the LPA concentration was correlated with the plasma lysophosphatidylcholine (LPC) concentration only in men. Other lipid-related parameters were only slightly correlated or were not correlated with the LPA concentration.
Conclusions
Our findings suggested that conversion from LPC by lysoPLD might be the major route for LPA production in plasma.
Introduction
The simple phospholipid lysophosphatidic acid (LPA), that is, 1-acyl-2-hydroxy-sn-glycerol-3-phosphate or 1-hydroxy-2-acyl-sn-glycerol-3-phosphate, is involved in a variety of (patho) physiological responses. 1–4 LPA binds to its specific G protein-coupled receptors and activates multiple signal transduction pathways, including those initiated by the small GTPases: Ras, Rho and Rac. LPA signalling has been implicated in such diverse processes as platelet aggregation, atherosclerosis, wound healing, vascular remodelling, tumour progression and brain development. 1–4 Recently, LPA has been revealed to be a platelet-activating component of mildly oxidized LDL and lipids isolated from human atherosclerotic plaques and LPA molecules present in the core of atherosclerotic plaques may trigger rapid platelet activation through the stimulation of LPA1 and LPA3 receptors. 5,6 These studies indicate the importance of LPA, and hence the regulation of plasma LPA concentrations, in vascular biology.
Unlike the mechanisms by which LPA elicits cellular responses, which are fairly well characterized, the regulation of plasma/serum LPA concentrations remains unclear. At present, the most important molecule for LPA production seems to be lysophospholipase D (lysoPLD), which forms LPA by hydrolysing lysophosphatidylcholine (LPC). 7–9 Recently, lysoPLD was purified from fetal bovine serum 7 and human plasma. 8 Interestingly and surprisingly, this lipase was found to be identical to autotaxin, which has been cloned as a tumour cell motility-stimulating factor and was originally isolated from melanoma cell supernatants. 10 Other factors also contribute to LPA formation in plasma/serum, including platelet activation. 4,9,11 Furthermore, vascular endothelial cells and blood cells express ecto-enzyme lipid phosphate phosphatases to degrade LPA, 12,13 thereby contributing to the regulation of the LPA concentration.
In this study, we measured the plasma LPA concentration in 146 healthy subjects as a step toward the introduction of LPA assays into laboratory medicine and to determine suitable reference values. Furthermore, to identify factors that may determine the plasma concentration of this important bioactive lipid, we examined the relationship between LPA concentrations and related parameters and identified factors that were correlated with the LPA concentration under physiological conditions.
Materials and methods
Samples
The samples used in this study were residual samples of those obtained for laboratory analyses (for medical checkups). Written informed consent was obtained for the use of the residual samples from all the participants. The study was approved by the Institutional Research Ethics Committee of the Faculty of Medicine, the University of Tokyo. To obtain the serum samples, whole blood specimens were directly collected into glass tubes and left to stand for 15 min at room temperature to allow the formation of blood clots; then, the serum was separated by centrifugation at 1500
Plasma lysophosphatidic acid and lysophosphatidylcholine measurements
For the plasma LPA and LPC measurements, suitable plasma samples were obtained from blood anticoagulated with 7.5 mmol/L EDTA plus 10% v/v of citrate- theophylline-adenosine-dipyridamole (BD, Franklin Lakes, NJ, USA), 14 as previously described. 15 While the plasma concentrations of LPA and LPC are easily altered, laboratory testing of these bioactive lipids for clinical purposes can be conducted reliably if the samples were prepared under stringent conditions. In vitro formation of LPA and LPC was negligible when whole blood samples were anticoagulated as described above, and all the procedures including the plasma preparation and preservation until measurement were performed at 4°C. 15
The concentration of plasma LPA was determined using an enzymatic cycling assay, as described previously. 16 Namely, LPA was hydrolysed with lysophospholipase to glycerol 3-phosphate, followed by enzymatic cycling using glycerol 3-phosphate oxidase and glycerol 3-phosphate dehydrogenase. The amplified concentrations of hydrogen peroxide, a product of the enzymatic cycling, were then measured colorimetrically. These LPA measurements were performed using an automatic analyser (Hitachi 7600; Hitachi High-Technologies, Tokyo, Japan).
The concentration of plasma LPC was determined using a specific enzymatic assay kit (AZWELL LPC Assay Kit, Alfresa Pharma Corporation, Tokyo, Japan), as described previously. 17 In brief, LPC was converted by lysophospholipase to glycerophosphorylcholine, from which choline was formed by glycerophosphorylcholine phosphodiesterase. Hydrogen peroxide was produced from choline by choline oxidase, and was quantified in the presence of peroxidase using an oxidative chromogenic reagent and 4-aminoantipyrine; the changes in absorbance were then measured. This LPC measurement was also performed using the Hitachi 7600 automatic analyser.
Serum lysophospholipase D activity assay
The serum lysoPLD activity was assessed based on the amount of choline released using LPC as the substrate, and confirmed to be stable; this assay is suitable for clinical laboratory testing, based on the results on within-run and between-run precision, interference, detection limit and linearity. 18
Plasma 5-hydroxytryptamine and β-thromboglobulin measurements
The in vivo platelet activation markers, plasma 5-hydroxytryptamine (5-HT) and β-thromboglobulin (β-TG), were measured as described previously. 19
Serum-oxidized LDL assay
Oxidized LDL, a biological marker of atherogenesis, was measured by a sandwich-type enzyme immunoassay for human oxidatively modified LDL using the mouse monoclonal antibody FOH1a/DLH3. 20 This antibody, which is specific for oxidized phosphatidylcholine, was used as the capture antibody and a horseradish peroxidase-labelled goat antihuman apolipoprotein-B (Apo B) IgG was used for detection. Copper-oxidized human LDL, prepared under controlled conditions, was used as a standard, and the results of the enzyme immunoassay were expressed in arbitrary units (U/mL).
Other measurements
Lecithin-cholesterol acyltransferase (LCAT) activity was measured using the Nescoat LCAT Kit-S (Alfresa Pharma Corporation); synthetic dipalmitoyl phosphatidylcholine was employed as the substrate, and the change in free cholesterol content was measured by a combined enzymatic method using cholesterol oxidase and peroxidase. 21 Ordinary serum chemical constituents were analysed using a Hitachi 7250 automated analyser (Hitachi High-Technologies). The complete blood count was measured using an SE9000 Automated Haematology Analyser (Sysmex, Kobe, Japan). The prothrombin time (PT), activated partial thromboplastin time and fibrinogen were measured using a CA-1500 Automated Coagulation Analyser (Sysmex).
Statistics
The SPSS Professional Statistics software program (SPSS Inc., Tokyo, Japan) was used for all the statistical analyses.
Results
Plasma lysophosphatidic acid measurements in healthy subjects
By employing an enzymatic cycling LPA assay 16 and a plasma preparation method suitable for LPA measurement, 15 we examined the plasma LPA concentrations in 146 (99 men and 47 women) healthy subjects. The distribution of the plasma LPA concentrations in men and women are shown in Figure 1a and b, respectively. The mean ± SD of the plasma LPA concentration among the 146 healthy subjects was 0.085 ± 0.031 μmol/L and the plasma LPA concentration was significantly higher in women (0.103 ± 0.032 μmol/L) than in men (0.077 ± 0.026 μmol/L) (Figure 1c). The reference values for the plasma LPA concentration, determined at 2.5–97.5 percentile interval, were 0.01–0.13 μmol/L and 0.04–0.17 μmol/L for men and women, respectively.
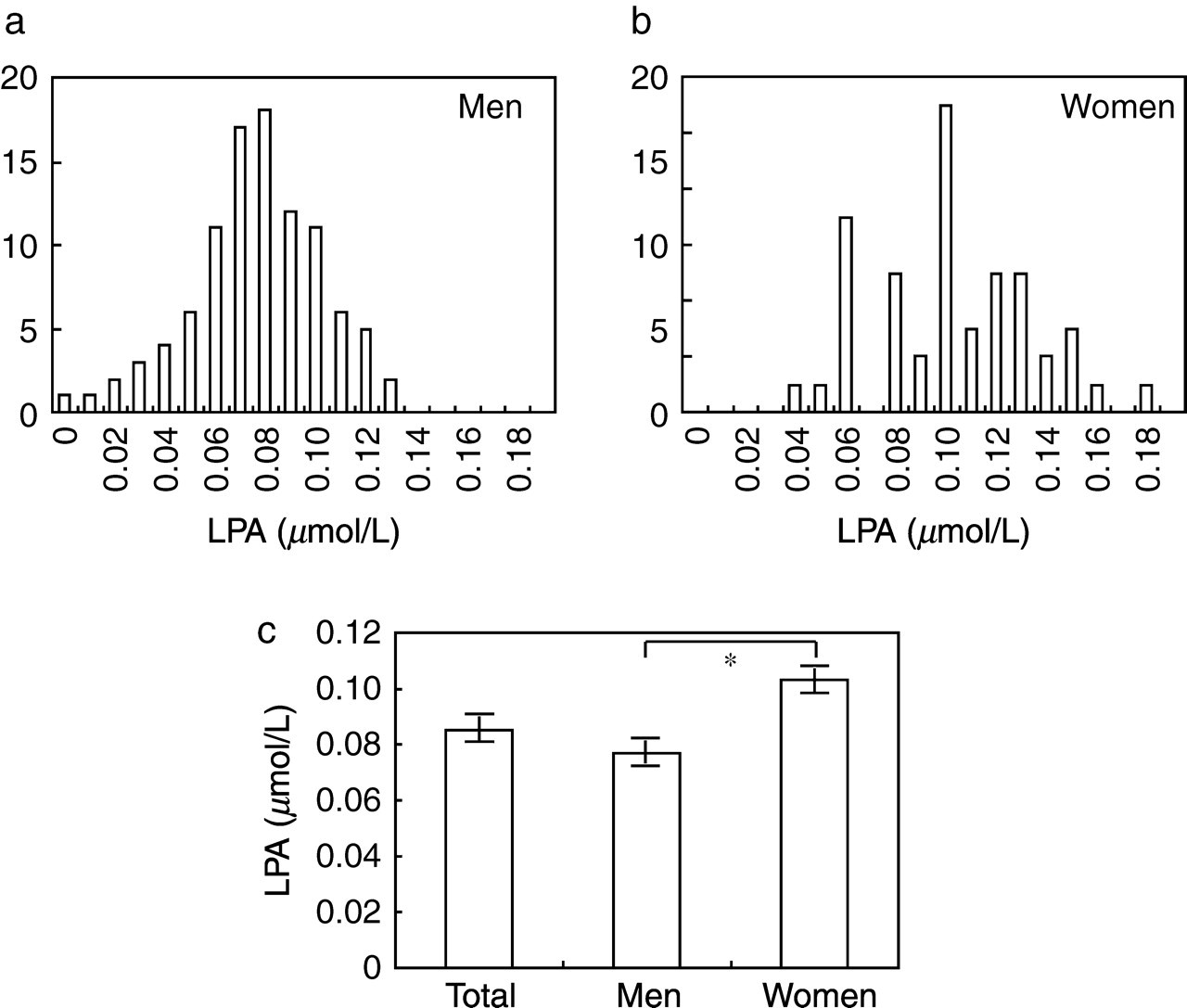
Measurement of plasma lysophosphatidic acid (LPA) concentrations in healthy subjects. The distributions of plasma LPA concentrations in 99 men (a) and 47 women (b) subjects are shown. In (c), the columns represent the mean ± SE of the plasma LPA concentrations of the total (men and women), men and women groups. The concentration was significantly higher in plasma samples obtained from women, as assessed by Mann-Whitney U test (P = 0.000003)
A weak but significant negative correlation between the plasma LPA concentration and age was observed in men (r = −0.270; Figure 2a), but not in women (Figure 2b).
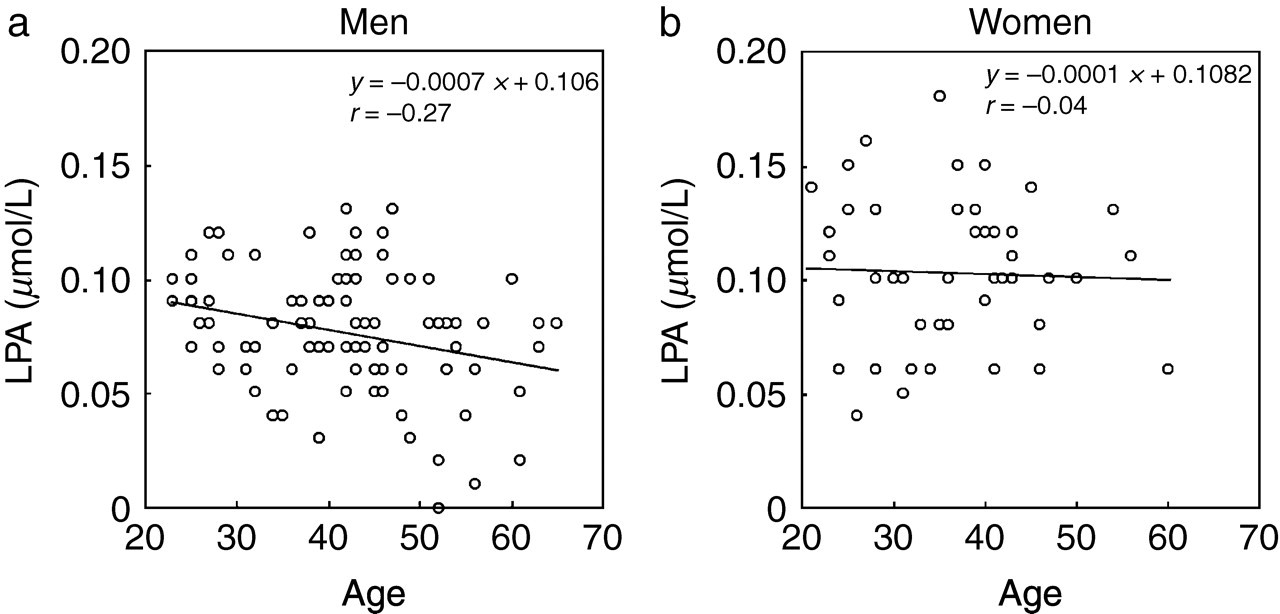
Correlation between plasma lysophosphatidic acid (LPA) concentration and age. Analyses for men (a) and women (b) are shown
Simple linear regression analyses between the plasma LPA concentration and other parameters
We then measured other parameters in these healthy subjects and searched for correlations with the plasma LPA concentrations. In a simple linear regression analysis, we found a strong positive correlation between the LPA concentration and serum lysoPLD activity: r = 0.439, 0.543 and 0.595 in men, women and total (men and women) groups, respectively (Figure 3a). On the other hand, a significant negative correlation was observed between the LPA concentration and haematocrit (r = −0.201, −0.350 and −0.456), fibrinogen (r = −0.256, −0.370 and −0.282) and LCAT (r = −0.274, −0.372 and −0.381) in men, women and total groups, respectively for r values. The LPA concentration was not significantly correlated with the platelet count or the LPC (Figure 3b), β-TG or oxidized LDL concentrations in men, women or total groups.
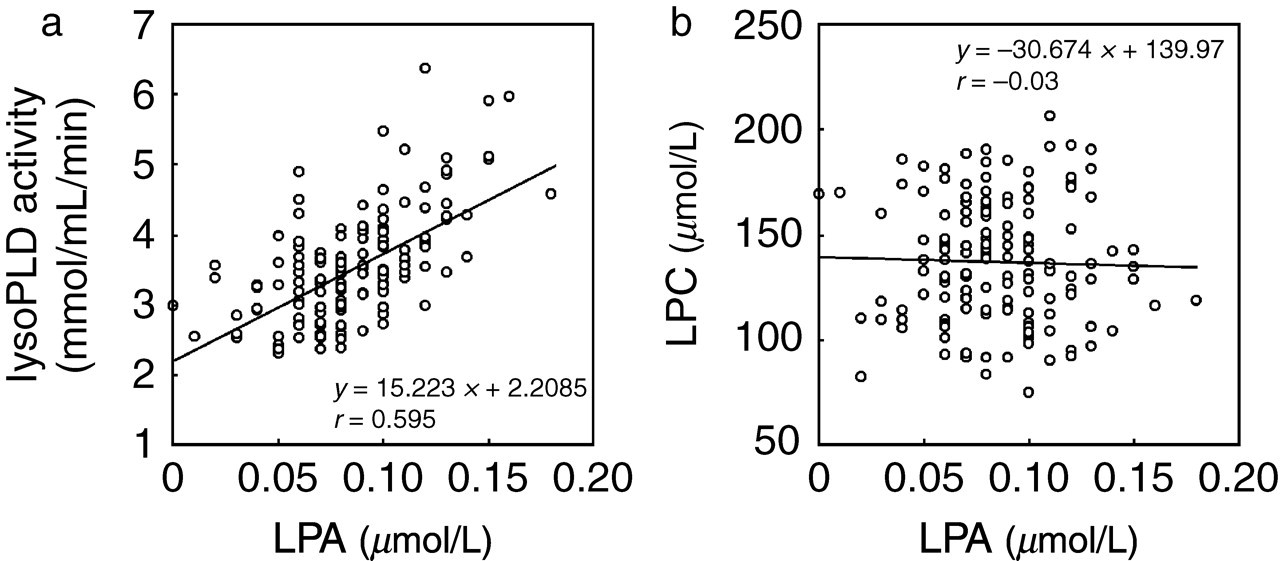
Correlation between plasma lysophosphatidic acid (LPA) concentration and the lysophospholipase D (lysoPLD) activity level (a) or the lysophosphatidylcholine (LPC) concentration (b)
Multiple regression analysis for plasma lysophosphatidic acid concentration
To identify factors independently associated with the plasma LPA concentration, a multiple regression analysis was performed (Table 1). Serum lysoPLD activity was confirmed to be strongly correlated with the LPA concentration in men, women and total groups. No other parameter was identified as an independent determinant of the LPA concentration, although haematocrit, lipoprotein (a) (Lp[a]), free cholesterol and LCAT were statistically correlated with the LPA concentration in the total group. In men, age, plasma LPC concentration, plasma 5-HT, total bilirubin (T-Bili) and free fatty acid (FFA) were significantly correlated with the plasma LPA concentration, while haemoglobin, Apo AII, free cholesterol and oxidized LDL were significantly correlated with the plasma LPA concentration in women.
Multiple regression analysis for plasma lysophosphatidic acid concentration
LysoPLD = lysophospholipase D; haematocrit = haematocrit; LCAT = Lecithin-cholesterol acyltransferase; Lp(a) = lipoprotein(a); Chol = cholesterol; LPC = lysophosphatidylcholine; 5-HT = 5-hydroxytryptamine; FFA = free fatty acid; PT-INR = internal normalized ratio of prothrombin time; β-TG = β-thromboglobulin; Apo CII = apolipoprotein CII; Hb = haemoglobin; Apo AII = apolipoprotein AII; LD = lactate dehydrogenase
Discussion
In this study, aimed at the introduction of the plasma LPA assay into laboratory medicine, we examined the plasma LPA concentration in 146 healthy subjects; the mean ± SD was 0.085 ± 0.031 μmol/L. The LPA concentration was significantly higher in women (0.103 ± 0.032 μmol/L) than in men (0.077 ± 0.026 μmol/L), which most likely reflects the higher lysoPLD activity in women than in men. 18 The reference values for the plasma LPA concentration, determined as the 2.5–97.5 percentile interval, were 0.01–0.13 μmol/L and 0.04–0.17 μmol/L for men and women, respectively. These observations are basically similar to those made in a previous report. 16 Although a weak but significant negative correlation between the plasma LPA concentration and age was observed only in men (but not in women), its significance remains to be solved. A multiple regression analysis showed a strong positive correlation between the plasma LPA concentration and the serum lysoPLD activity, which is again consistent with our recent finding that the plasma LPA concentration was strongly correlated with lysoPLD activity in patients with chronic hepatitis C, although the number of samples examined was rather small. 22
LPA, the smallest and structurally simplest phospholipid, is a lipid mediator with multiple biological activities that accounts for many of the biological properties of serum. LPA was long thought to be produced during serum formation, based on the fact that the LPA concentration is much higher in serum than in plasma, and to be released from activated platelets. 11 However, the amount of LPA released from isolated platelets, even after strong activation, reportedly accounts for only a small part of serum LPA, 23,24 although the examination of platelet-depleted rats showed that half of the LPA in serum is produced via a platelet-dependent pathway. 4,9 In fact, we confirmed that the plasma LPA concentration was hardly correlated with platelet-related parameters, such as the platelet count and the in vivo platelet activation markers plasma 5-HT and β-TG. 19
Recent research on the involvement of platelets, plasma and phospholipases in LPA synthesis has demonstrated multiple pathways for LPA production in serum and the involvement of several phospholipases, including phosphatidylserine-specific phospholipase A1, secreted phospholipase, A2-IIA, LCAT and lysoPLD. 4,9,24 However, it is now clear that serum lysoPLD, which is identical to the tumour cell motility-stimulating factor autotaxin, is the most important molecule for the synthesis of LPA. LysoPLD produces LPA from lysophospholipids (such as LPC, lysophosphatidylethanolamine and lysophosphatidylserine) released from activated platelets through the actions of phospholipases that are abundantly expressed in these anucleate cells, while lysoPLD produces LPA by hydrolysing LPC during plasma incubation. 4,7–9 Our present finding that the plasma LPA concentration is closely correlated with lysoPLD activity supports this idea. Furthermore, this finding is also consistent with the recent finding that in serum from heterozygous mice deficient in lysoPLD/autotaxin, both lysoPLD activity and the LPA concentration were about half of those in wild-type mice, suggesting that lysoPLD is responsible for the bulk of LPA production in serum. 25 In vivo, not only the production of LPA (mainly through lysoPLD), but also the degradation pathway through lipid phosphate phosphatases (i.e. ecto-enzymes that catalyse the dephosphorylation of a number of bioactive lipid mediators, including LPA) 12,13 might be involved. However, it seems unlikely that these LPA degradation enzymes are determinants of the plasma LPA concentration, based on our present result.
Oxidized LDL is known to play an important pathogenetic role in atherosclerosis, and its thrombotic complication – atherothrombosis. 20 In this context, LPA is formed during the mild oxidation of LDL and is the active compound in mildly oxidized LDL and minimally modified LDL, suggesting that LPA may be the primary platelet-activating lipid of atherosclerotic plaques. 5,6 However, a clear correlation between oxidized LDL and LPA was not observed, although a weak correlation was seen only in women.
Considering the various and important roles of LPA, a plasma LPA assay may be promising as a clinical laboratory test in the future. However, extreme care should be taken when preparing plasma samples for LPA measurement because the enzyme lysoPLD and its substrate LPC coexist in the plasma. For example, the concentration of LPA dramatically increased after incubation; the LPA concentration after 180 min of incubation was about 15 times higher than that in the control sample. 15,18 Unfortunately, this critical point was not considered when plasma LPA concentrations were measured in previous reports. Patients with ovarian cancer were reported to have significantly higher plasma LPA concentrations (mean, 8.6 μmol/L; range, 1.0–43.1 μmol/L). 26 However, in the same study, the values for the healthy control group (mean, 0.6 μmol/L; range, <0.1–6.3 μmol/L) were much higher than those obtained in the present study. Our present study, employing an automatic LPA measurement method and suitable plasma samples, may pave the way for the introduction of LPA assays into laboratory medicine.
Footnotes
Acknowledgements
This work was supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan, the Japan Science and Technology Corporation and the Japanese Society of Laboratory Medicine Fund for the Promotion of Scientific Research.
