Abstract
Abstract
Background
Plasma ammonia (PA) measurement is of key importance in the diagnosis and monitoring of some inherited metabolic disorders and to monitor subsequent treatment of hyperammonaemia.
Methods
Over a six-month period, patients' ammonia concentrations were measured in parallel, using an enzymatic–UV kit (Infinity Ammonia Liquid Stable Reagent, Thermo Electron Corporation, Australia) on an Olympus AU640 analyser (Olympus UK Ltd, Hertfordshire) and on our current dry chemistry system (Vitros 250, Ortho Clinical Diagnostic). Alanine amino transferase (ALT) was added to a human plasma sample to investigate its effect on the assessment of ammonia concentration.
Results
Both methods correlated well (InfinityTM kit = 1.12 × Vitros 250 + 39, R 2 = 0.95, n = 105). However, clinically important discrepancies ranging from 100 to 380 μmol/L were found in patients with acute liver failure. Ammonia concentration measured with the enzymatic Infinity™ kit increased by 137 μmol/L when ALT was added up to 17,000 IU/L.
Conclusions
ALT produces a positive interference in the enzymatic Infinity™ kit which may affect interpretation and influence further clinical action. However, this positive interference due to ALT does not fully explain the discrepant results observed between the two methods in patients with acute liver failure.
Introduction
Hyperammonaemia is a medical emergency because of the neurotoxic effect of ammonia on the central nervous system and requires urgent attention. The assessment of PA is used to support the diagnosis of some inherited metabolic disorders – primarily urea cycle defects and those involving the metabolism of organic acids – and to prompt further investigations, sometimes to monitor the severity of hepatic encephalopathy even though its correlation is variable and to monitor treatment of hyperammonaemia. 1 In our hospital, the principles of treatment range from reducing or stopping dietary protein, glucose infusion to limit catabolism and pharmacological treatment to favour the removal of excess nitrogen when PA concentration is between 150 and 350 μmol/L, to renal replacement therapy if ammonia is greater than 350 μmol/L or the patient does not respond rapidly to treatment. Therefore, the accurate measurement of PA concentration is of key importance for diagnosis of these disorders and for monitoring of treatment.
Several methods are available for the determination of PA concentration, based on the studies carried out in the late 1970s and 1980s. 2,3 The most frequently used methods are enzymatic, based on glutamate dehydrogenase (GLDH) that catalyzes the condensation of NH4 + and 2-oxoglutarate to form glutamate in the presence of NADH or NADPH. The concentration of ammonia is determined by monitoring the transformation of NAD(P)H to NAD(P)+ by a spectrophotometric technique at 340 nm.
Material and methods
An enzymatic–UV kit from Thermo Electron Corporation (Infinity™) was used on our AU640 analyser (Olympus UK Ltd, Hertfordshire) to compare the PA concentrations with our current method (Vitros 250, Ortho-Clinical Diagnostics, UK). A standard preparation of alanine amino transferase (ALT) (porcine heart) was purchased from Sigma Chemical Co (St Louis, MO, USA) and was used in further experiments.
As part of the evaluation for the introduction of a new kit, we carried out a comparison study between both methods. All PA requests were analysed in parallel with both methods over a period of six months. Venous blood was collected into lithium heparin tubes and was delivered to our laboratory, centrifuged and analysed within 30 min.
A blood sample from a volunteer was collected into lithium heparin, centrifuged and separated from cells within 15 min. Increasing amounts of ALT were added to aliquots of this plasma sample. No ethical approval was necessary to perform the comparison between methods and to carry out this experiment.
Results
The two methods correlated well (Figures 1a and b) when ALT was <50 IU/L (Infinity™ kit = 1.10 × Vitros 250 + 35, R 2 = 0.99, n = 33). We acknowledge that only two ammonia values over 200 μmol/L were included to calculate the correlation and it may not be completely representative. When ALT was over 50 IU/L the correlation (Figure 1c) showed greater scattering and the slope was 2.6 fold greater (Infinity™ kit = 2.86 × Vitros 250 + 35, R 2 = 0.52, n = 27). The absolute difference in PA concentration between the two methods appears to increase with increasing activities of ALT (Figure 1d).
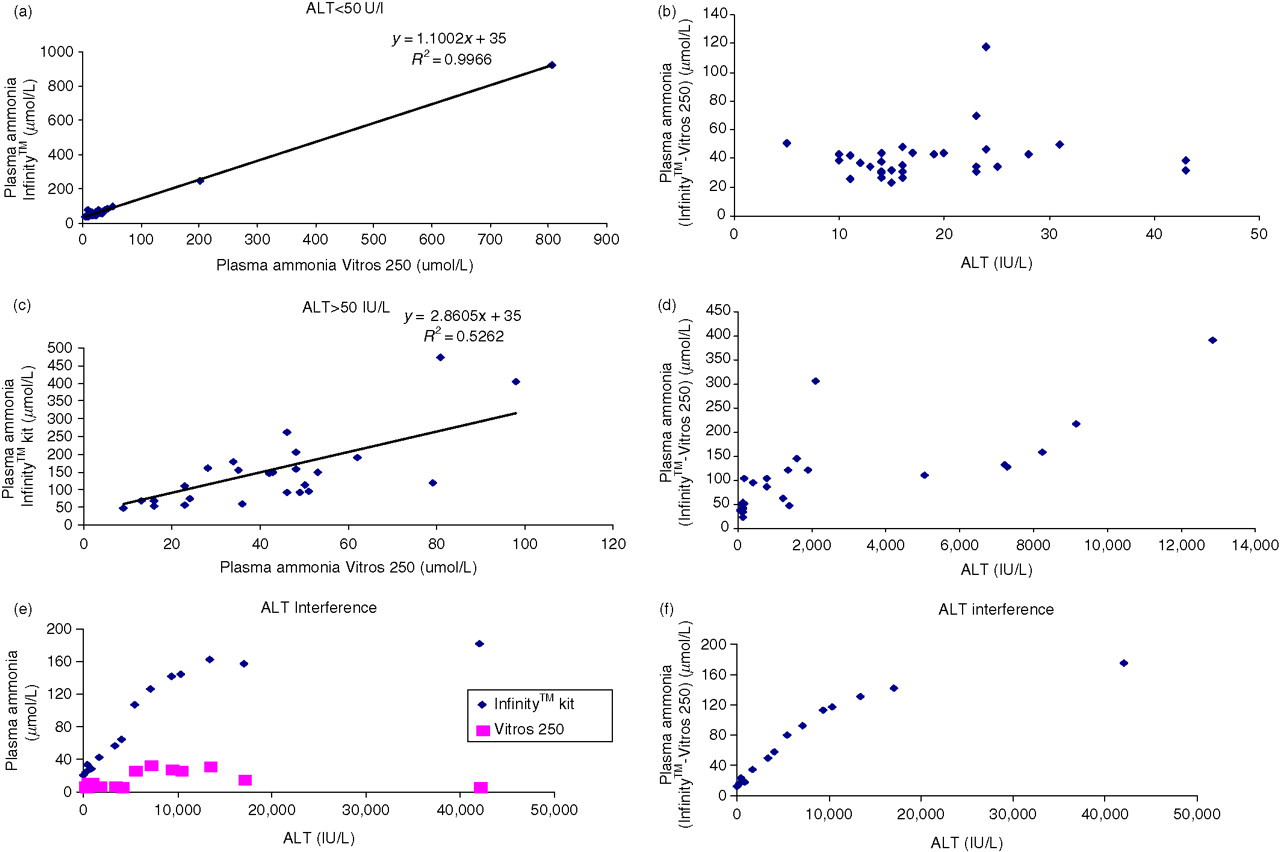
Plasma ammonia (PA) correlation and difference between the enzymatic – UV (InfintiyTM) kit and Vitros 250 method; (a) and (b) alanine amino transferase (ALT) <50 IU/L; (c) and (d) ALT > 50 IU/L; (e) PA concentration measured on both instruments when ALT varies from 14 IU/L to 42,000 IU/L; (f) PA difference between the enzymatic – UV (InfinityTM) assay and Vitros 250 method with increasing ALT activities
When a plasma sample was spiked with ALT up to 17,000 IU/L, PA measured by the enzymatic–UV kit increased from 21 μmol/L in the basal specimen to 158 μmol/L. This trend was not observed for the PA concentration measured on Vitros 250, and the net effect was an increase in the difference between the assays (Figures 1e and f).
Discussion
Overall the methods correlate well; however, clinically significant discrepancies in PA concentration were identified in patients with liver disease (Table 1).
Summary of plasma ammonia (PA) concentrations on patients with a variety of clinical conditions and moderate to high activities of alanine amino transferase (ALT)
PLAV, plasma ammonia measured on Vitros 250; PLAI, plasma ammonia measured with the enzymatic infinity™ kit. PA in patients 7 and 8 was monitored on several occasions
Infinity™ ammonia reagent is a one-step enzymatic assay – GLDH catalyzes the transformation of 2-oxoglutarate to glutamate in the presence of ammonia and NADH. The reagent contains lactate dehydrogenase (LDH) in excess to rapidly achieve equilibrium between pyruvate and lactate, in order that the endogenous pyruvate in the sample does not interfere with the assay. However, in the presence of significant ALT activity a second reaction plays an important role. ALT catalyzes the transformation of 2-oxoglutarate (in excess in Infinity™ reagent) and
Nevertheless, the interference due to ALT does not fully explain the difference observed in patients with acute liver failure (Table 1). In these patients, other dehydrogenases can be released into the systemic circulation and produce a transformation of NADH to NAD+, resulting in a spurious increase in PA concentration if the assay does not appropriately blank the sample.
Another issue is the contribution of ammonia to hepatic encephalopathy. The correlation between PA concentration and the severity of hepatic encephalopathy has been reported to be variable in patients with chronic and acute liver failure. 4,5 We speculate whether the disparity of results between different methods might contribute to the poor correlation found between PA concentration and clinical symptoms in these patients. In the Wales External Quality Assessment Scheme for ammonia, 47 laboratories in UK routinely use this GLDH enzymatic kit to measure PA concentration.
In conclusion, we encourage other users of ammonia methods to investigate whether their methods can be used to assess ammonia in patients with acute liver disease.
Footnotes
ACKNOWLEDGMENTS
We express our thanks to Wales External Quality Assessment Scheme (WEQAS) for providing data regarding the ammonia methods currently used in the UK.
