Abstract
Abstract
Background
Serum B-type natriuretic peptide (BNP) and the amino-terminal cleavage product of the prohormone (NT-proBNP) have been shown to be valuable parameters for the diagnosis of heart failure (HF) in the general population. Urinary BNP and NT-proBNP have also been suggested for diagnosis of HF. The present study investigated the diagnostic value of both serum and urinary NT-proBNP in selected groups of controls and patients diagnosed with HF.
Methods
Creatinine clearance and serum and urinary NT-proBNP were measured in 76 controls and in 47 patients diagnosed with HF (NYHA III and IV). Echocardiography was used to exclude cardiac dysfunction in the control population by the combined normality of left ventricular ejection fraction (LVEF), E/A ratio (echocardiographic early [E] and late, or atrial [A] phases of ventricular filling), deceleration time and LV mass index. All patients diagnosed with HF had LVEF <40%.
Results
NT-proBNP measurements in urine samples are subject to high variability. Receiver-operating characteristic area under the curve (AUC) for serum, urinary NT-proBNP and their products were 0.94, 0.72 and 0.93, respectively. Correction of urinary NT-proBNP for urinary creatinine content improved the AUC from 0.72 to 0.80. Negative predictive values for ruling out HF were 0.94, 0.67 and 0.89, respectively. Linear regression analysis revealed that creatinine clearance was more important in determining serum NT-proBNP concentrations than age.
Conclusions
Serum NT-proBNP is the best parameter to rule out HF. The product of the serum and urinary concentrations has equal value. Urinary NT-proBNP alone performs rather poorly. Renal function influences NT-proBNP concentrations more than age in this selected population.
Introduction
Heart failure (HF) is a major public health problem of increasing relevance. 1–3 It is important to realize that HF is the final stage of a variety of diseases affecting the heart. Hence, the population of HF patients is highly heterogeneous. Diagnosis of HF is based mainly on historical features (e.g. myocardial infarction), symptoms (e.g. dyspnoea), physical examination (e.g. third heart sound, leg oedema), chest radiography (e.g. interstitial oedema) and electrocardiography (e.g. atrial fibrillation). 4 Additional laboratory analyses can include B-type natriuretic peptide (BNP) and the N-terminal cleavage product (NT-proBNP), which have become well-established biochemical markers for diagnosis and prognosis of HF. 5–8
BNP (77–108 amino acids) and NT-proBNP (1–76 amino acids) are derived from proBNP (108 amino acids) by cleavage through the proteases furin 9 and corin. 10 proBNP itself is derived from pre-proBNP (134 amino acids) by cleavage of a signal peptide (26 amino acids). 11,12 Both peptides are released into the circulation from the cardiac ventricle in equimolar amounts. Release is induced by increased wall stretch, either mechanically or by volume overload. 13
NT-proBNP (8.5 kDa) is a low-molecular weight peptide that is filtered by the glomerulus. It is unclear whether it has any biological activity. In contrast, the smaller and biologically active BNP (3.5 kDa) acts as an antagonist of the renin-angiotensin-aldosterone system and protects the body from plasma overload by inducing diuresis, natriuresis and vasodilation. 12 In addition to renal clearance, BNP is also cleared from the circulation through binding to natriuretic peptide receptor C. This complex is then internalized and BNP is enzymatically degraded. The receptor is externalized again to the outside of the cell membrane. The third mechanism involves neutral endopeptidase 24.11, which also degrades circulating BNP. 14,15 Both mechanisms were shown to contribute equally to the clearance of BNP. 16 These two additional mechanisms might explain the longer half-life of NT-proBNP compared with BNP. 17
Recently, it was suggested to use the combined measurement of serum and urinary NT-proBNP in screening for left ventricular systolic dysfunction (LVSD) in the general population. 18,19 The aims of this study were to evaluate the Roche NT-proBNP immunoassay for measurement of urine samples and to investigate the diagnostic value of serum and urinary NT-proBNP in patients diagnosed with HF and in controls without cardiac abnormalities, as determined by stringent echocardiographic criteria.
Materials and methods
Study population
This study was approved by the Medical Ethical Committee of the University Hospital Maastricht. All patients had echocardiography, blood sampling and 24-h urine collection. The control group consisted of 76 patients who had no history of cardiac disease and were hospitalized for non-cardiac causes. Controls had normal echocardiography based on the following stringent and combined criteria: left ventricular ejection fraction (LVEF) >45%, E/A ratio (echocardiographic early [E] and late, or atrial [A] phases of ventricular filling) >1.0 for patients <55 years and >0.8 for patients ≥55 years, deceleration time <220 ms and left ventricular mass index (LVMI) ≤116 g/m2 in men and ≤104 g/m2 in women. In addition, 47 patients hospitalized for HF (NYHA III and IV) were recruited based on diminished LVEF (<45%) as determined by echocardiographic examination of the heart. A total of 121 serum and 89 urinary samples were analysed.
Laboratory analysis
Serum samples were analysed for NT-proBNP and creatinine. Creatinine clearance and a 24-h urine collection were used to estimate the glomerular filtration rate as a parameter of renal function.
NT-proBNP was measured on the Elecsys 2010 immunoassay system (Roche Diagnostics, Mannheim, Germany) with a sandwich immunoassay. The serum detection limit of the NT-proBNP immunoassay is 0.6 pmol/L. Between-day variation is 2.4% and 2.2% at concentrations of 51.2 pmol/L and 800 pmol/L, respectively (n = 60). The reference values for the identification and exclusion of acute coronary HF were shown to be age-dependent: 53 pmol/L (<50 years) and 106 pmol/L (≥50 years). 20,21 NT-proBNP concentrations are expressed in picomoles/litre (for conversion to picograms/millilitre they are multiplied by 8.457).
Serum and urinary creatinine concentrations were measured on the Synchron LX-20 system (Beckman Coulter, Fullerton, USA) with the Jaffe method using alkaline picrate. Between-day variation of serum creatinine is 2.2% and 0.93% at concentrations of 86.9 and 401 μmol/L, respectively (n = 93). The reference values are 71–110 μmol/L for men and 53–97 μmol/L for women.
Urinary NT-proBNP
In the studies by Ng et al., 18,19 urinary NT-proBNP was measured by an immunoluminometric assay (ILMA) with rabbit polyclonal antibodies. The Roche immunoassay used by Cortés et al. 22 utilizes two sheep polyclonal antibodies recognizing amino acid residues 10–21 and 39–50 of the molecule. 23 In our study, we also used the Roche immunoassay to measure NT-proBNP.
Recombinant human NT-proBNP (rhNT-proBNP; 1–76) was kindly supplied by Roche and was added to serum and urine samples, in which the NT-proBNP concentrations were below the detection limit of the immunoassay. Concentrations were measured in the range of 32–294 pmol/L. Stability of NT-proBNP in urine was tested by measuring NT-proBNP during a 48 h time span with aliquots of urine samples stored at different temperatures. The influence of urinary pH was tested by measuring urinary NT-proBNP before and after pH adjustment to a value of 7.4.
Echocardiography
One experienced cardiologist performed echocardiography for all subjects according to the standard clinical protocol used in our hospital. Data were collected on LVEF, LVSD, LV mass, LVMI and E/A ratio. The deceleration time was obtained from the electrocardiogram.
Statistical analysis
The Mann-Whitney U test was used for testing differences between groups for continuous variables and the χ 2 -test was used for categorical variables. The Spearman correlation coefficient (R s) was used as a measure of correlation. The receiver-operating characteristic (ROC) area under the curve (AUC) and the associated 95% confidence intervals were estimated. Optimal cut-off levels were calculated with combined highest sensitivity and specificity. Data were presented as medians with interquartile ranges and P <0.05 was considered statistically significant. The SPSS version 11.0.1 for Windows (SPSS, Chicago, IL, USA) statistical software package was used for statistical analyses.
Results
Assay feasibility
Feasibility of the NT-proBNP immunoassay for the determination of urinary NT-proBNP was checked by addition of rhNT-proBNP to urine samples of different subjects. Figure 1 shows the standard curves for NT-proBNP measurements in two representative serum and urine samples using the Roche immunoassay in the range of 32–293 pmol/L. In contrast to the 108% recovery found in serum samples, addition of rhNT-proBNP to urine resulted in lower recovery (16–55%). This was shown to be at least partly dependent on the urinary protein content of the sample (data not shown). The measurements were linear in the range tested.
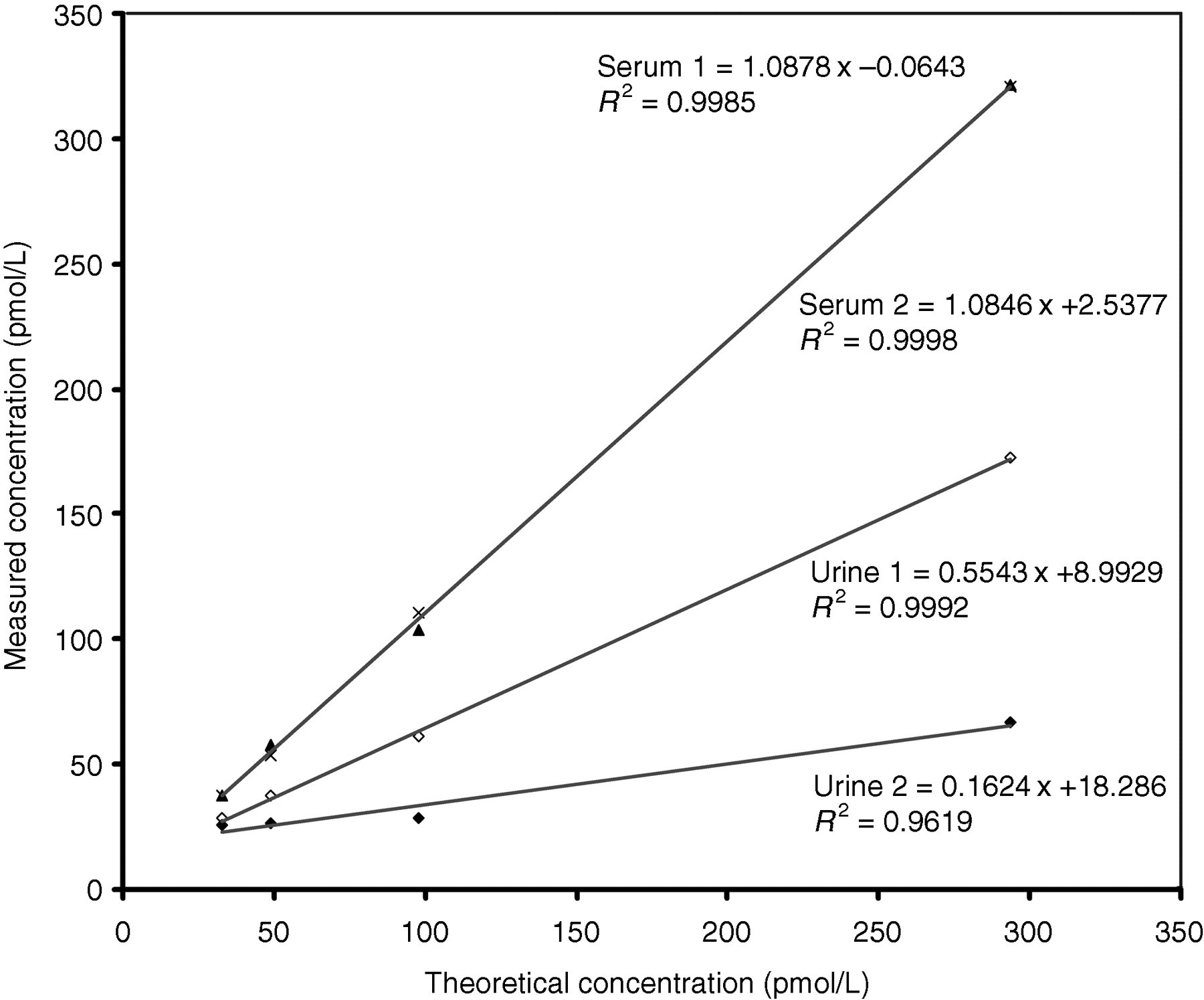
Standard curves for NT-proBNP measurement in serum and urine. rhNT-proBNP was spiked into two serum and two urine samples. The analytical response of the immunoassay was linear across the range of 32–294 pmol/L, independent of the matrix
Storage of serum and urine samples at room temperature for up to 48 h did not influence the concentration of NT-proBNP as measured by the Roche immunoassay. Urinary pH did not significantly influence NT-proBNP concentrations (after pH correction=0.8267×−2.5583; R 2 = 0.994).
Urinary NT-proBNP
Table 1 shows the characteristics of all subjects who participated in this study – 47 patients diagnosed with HF and 76 controls. HF patients and controls were not age-matched (P < 0.0001), and HF patients had lower creatinine clearance (P < 0.0001). As expected, HF patients had significantly higher serum and urinary NT-proBNP values (P < 0.0001).
Patient characteristics
Data are represented as median (interquartile range)
The Mann-Whitney test was used to calculate differences between controls and patients with HF. HF, heart failure; LVEF, left ventricular ejection fraction; LVMI, left ventricular mass index
*P < 0.0001; **P < 0.05
In Figure 2, the relation between serum and urinary NT-proBNP is shown. There was a statistically significant correlation between urinary and serum NT-proBNP concentrations (R s = 0.561, P < 0.01).
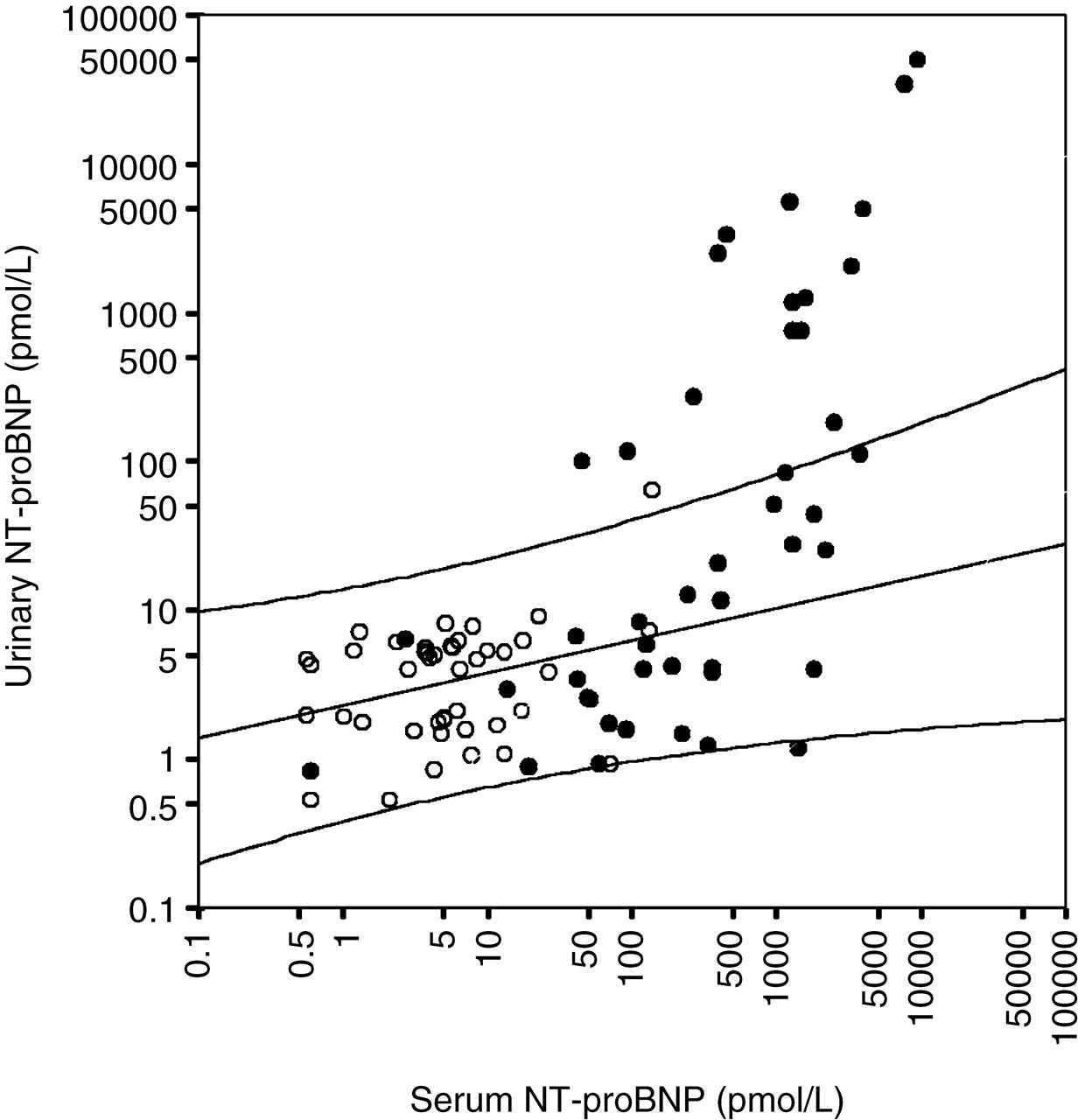
Relation of serum and urinary NT-proBNP. Open circles represent controls and closed circles represent heart failure patients. Spearman correlation and 95% confidence intervals were calculated with controls
Figure 3 shows the relation between serum and urinary NT-proBNP and age and creatinine clearance for both controls and patients diagnosed with HF. As calculated in the control population, serum NT-proBNP increased significantly with age (R s = 0.616, P < 0.01), as did urinary NT-proBNP; the latter, however, with a lower correlation coefficient (R s = 0.414, P < 0.01). Serum NT-proBNP showed an inverse correlation with creatinine clearance (R s = −0.777, P <0.01). Urinary NT-proBNP was also inversely correlated with creatinine clearance, but with a lower correlation coefficient (R s = −0.524, P <0.01).
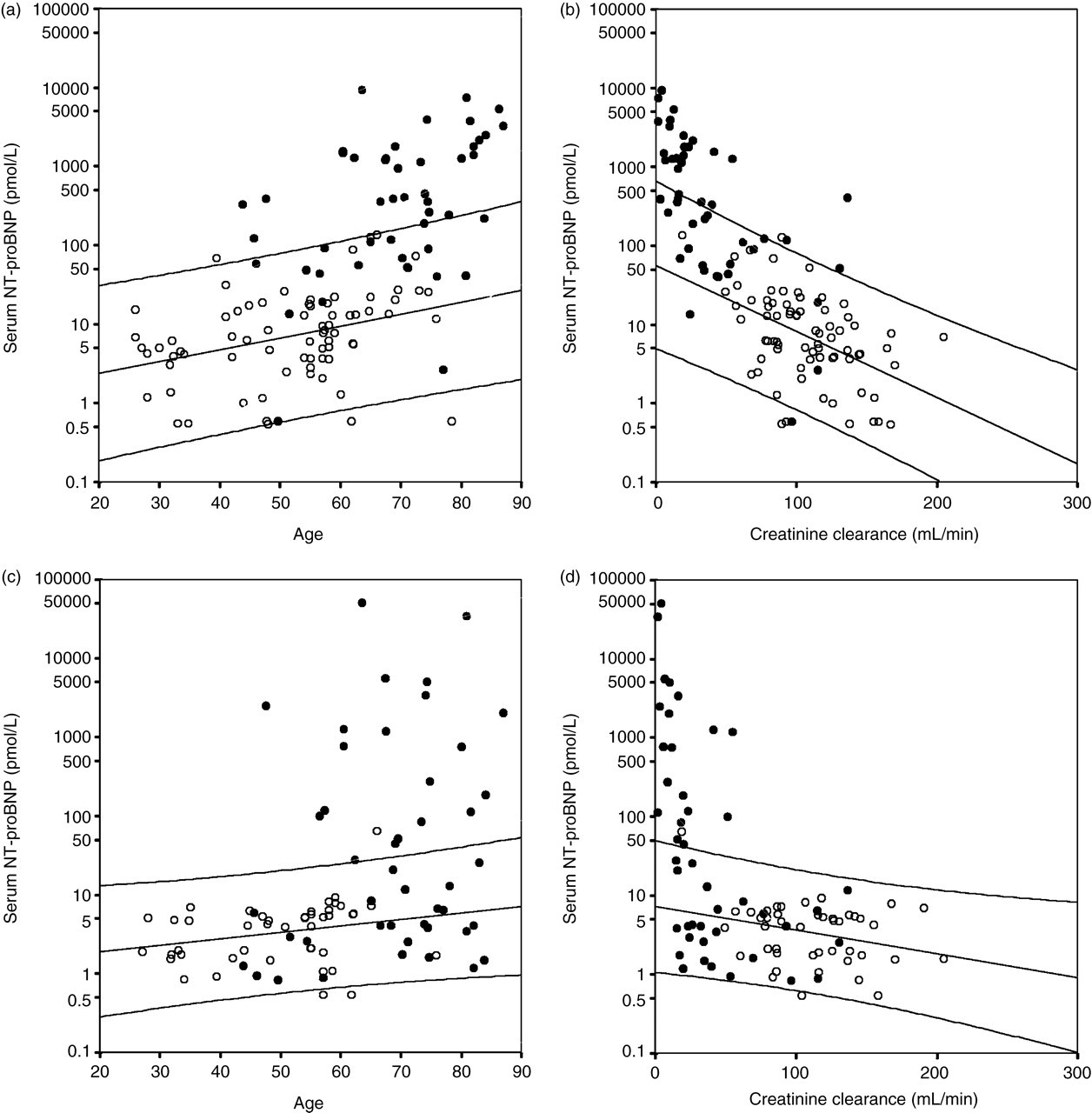
Serum and urinary NT-proBNP in relation to age and creatinine clearance. Open circles represent controls and closed circles represent heart failure patients. Spearman correlation and 95% confidence intervals were calculated with controls
Figure 4 shows the ROC curves for serum and urinary NT-proBNP, their product and the urinary NT-proBNP corrected for urinary creatinine for diagnosis of HF. Table 2 shows the ROC AUC analysis for serum and urinary NT-proBNP for diagnosis of HF with corresponding sensitivity, specificity and positive (PPV) and negative predictive values (NPV). Serum NT-proBNP had the highest AUC (0.940) and urinary NT-proBNP had the lowest (0.721). Correction for the urinary creatinine concentration increased the AUC to 0.804. The sensitivity of urinary NT-proBNP was only 53%, but the specificity was 98%. Urinary NT-proBNP had the highest PPV (96%) at a cut-off value of 10.5 pmol/L. Serum NT-proBNP had a sensitivity of 91%, a specificity of 92% and a PPV of 88% at a concentration of 33.5 pmol/L. The serum and urinary NT-proBNP product performed as well as, but was not superior to, serum NT-proBNP alone.
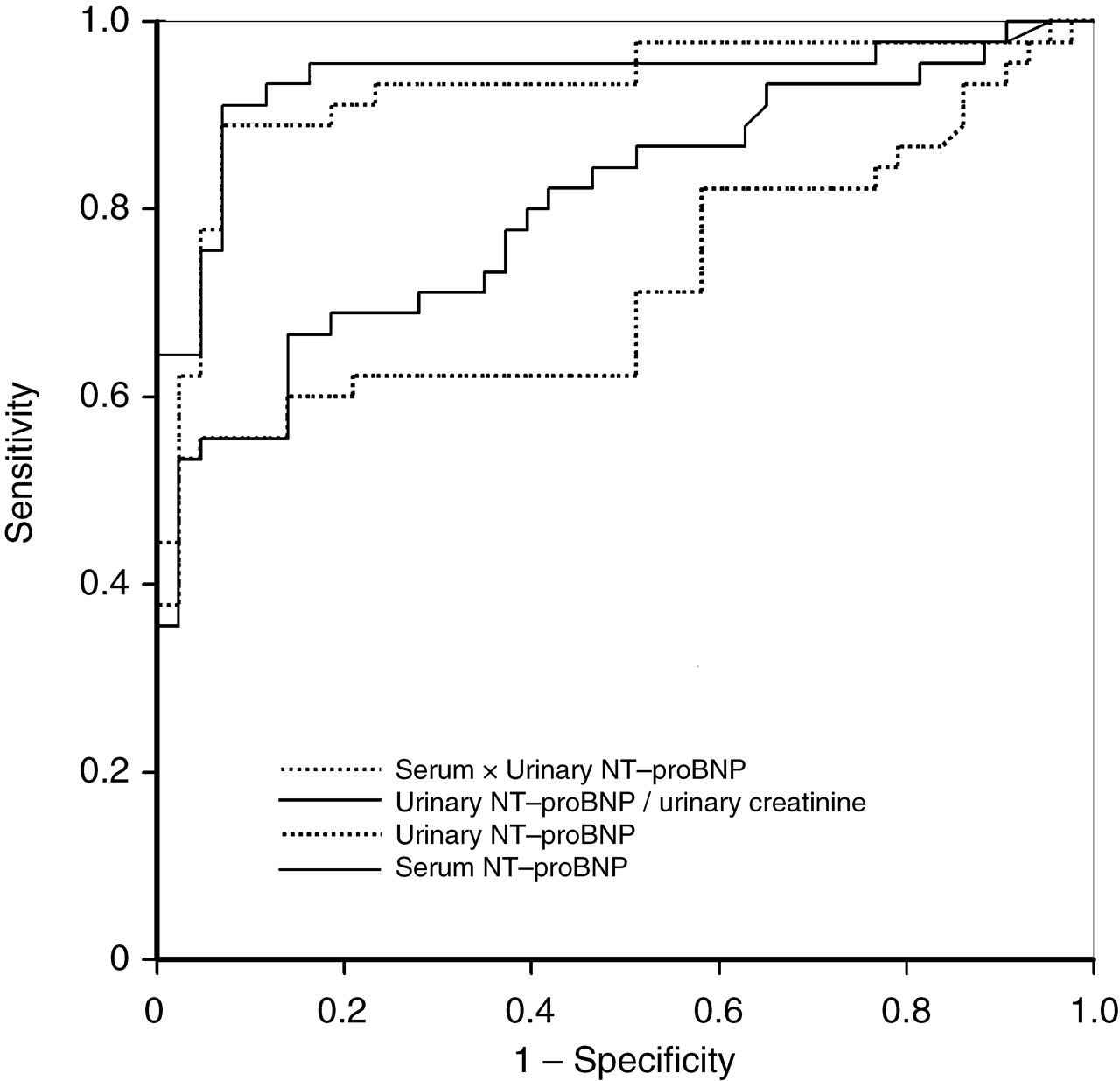
Receiver-operating characteristic area under the curve for the detection of heart failure for serum NT-proBNP, urinary NT-proBNP, the product of both and the urinary NT-proBNP corrected for urinary creatinine
Receiver-operating characteristic AUC analysis for ruling out heart failure
AUC, area under the curve; CI, confidence interval; PPV, positive predictive value; NPV, negative predictive value
Discussion
Circulating BNP and NT-proBNP have been widely accepted and are now used by clinicians as biomarkers for assessing cardiac functions. 5–8 Several papers reported the use of plasma or serum NT-proBNP measurements, but not urinary NT-proBNP measurements. Recent reports on the use of urinary NT-proBNP did not use the commercially available electrochemiluminescence immunoassay on the Elecsys 2010 platform that is marketed by Roche Diagnostics, 18,19 except for one. 23 The ILMA, originally evaluated with plasma samples, was used for the detection of urinary NT-proBNP. 22 Unfortunately, no details were provided on assay performance in urine samples. The first studies of this group reported that urinary NT-proBNP had diagnostic accuracy comparable with plasma NT-proBNP, and that urinary NT-proBNP may be suitable for diagnosis of HF. 18 In the second study, it was concluded that the most cost-effective strategy would be to apply these tests sequentially, using the urinary NT-proBNP test first to rule out LVSD and then subjecting the urine-positive cases to plasma testing. 19 Cortés et al. 23 reported good diagnostic value for urinary NT-proBNP for diagnosing HF with a ROC AUC value of 0.96 ± 0.02 (P <0.0001) compared with 0.98 ± 0.01 for serum NT-proBNP. The conclusion was that urinary NT-proBNP could be a simple non-invasive test for diagnosing HF.
In the present study, we evaluated the feasibility of the Roche NT-proBNP immunoassay for the determination of NT-proBNP in urine samples. First, we investigated the stability of NT-proBNP in urine samples and found that samples were stable for up to 48 h at room temperature. This is an important issue because patients have to collect 24-h urine samples and it may take longer than 48 h before these samples can be analysed or frozen in the laboratory. When we analysed rhNT-proBNP spiked in urine samples, the recoveries were rather low and varied substantially between 16 and 55%. In contrast, serum samples had a mean recovery of 108% as determined within the same concentration range. The results in urine were, however, linear in the range of 32–294 pmol/L. This might indicate an effect of the urine matrix resulting in lower recovery, which might at least partly explain the lower diagnostic value of urinary NT-proBNP when compared with serum NT-proBNP that was found. The analytical performance and diagnostic accuracy of this assay in serum samples have been evaluated elsewhere and these studies found excellent characteristics. 24,25
Urinary NT-proBNP increased with increasing concentrations of serum NT-proBNP. This is to be expected because NT-proBNP is filtered by the kidneys. From Figure 2 it can be argued that there seems to be a threshold value of serum NT-proBNP of approximately 100 pmol/L before it starts to appear in urine. This phenomenon can be explained by limited renal resorption with increasing serum concentrations. The increase in serum NT-proBNP with decreasing renal function can be explained by diminished renal filtration. Impaired renal function, however, implies a lower filtering capacity of the kidneys. This is in contrast to the increasing urinary NT-proBNP when renal function becomes impaired. Possibly, NT-proBNP is still small enough to be filtered, but this needs further investigation.
We then investigated the diagnostic value of serum and urinary NT-proBNP for diagnosis of HF. The results from this study are in line with earlier studies that showed the effectiveness of serum NT-proBNP for diagnosing HF in a variety of patient groups. 5,26–28 In a recent study among 20,536 patients that measured NT-proBNP according to the method described by Karl et al., 29 NT-proBNP concentrations were strongly predictive for HF and other major vascular events. 30 In contrast, the value of urinary NT-proBNP measurements for diagnosis of HF should be interpreted with care. Several factors influence the collection of proper urinary samples. Furthermore, the composition of urine varies greatly within and between individuals. These issues suggest that urinary NT-proBNP concentrations are less valuable than serum concentrations.
Conclusions
The results from this study are in line with earlier data showing that serum NT-proBNP is most effective in ruling out HF. However, with the Roche NT-proBNP immunoassay, the use of urinary NT-proBNP did not show the high values reported by others. 18,19,23 Using serum NT-proBNP is the most easy-to-use method with least variability. We recommend the use of serum and not urine for measurements of NT-proBNP for the diagnosis of HF.
