Abstract
Abstract
Background
This study aimed at evaluating the ability of cardiac troponin I (cTnI) measurement on the ACS:180 for prognostic risk stratification. Sequential admissions to the coronary care unit of a busy district general hospital were studied. All patients were followed up for a maximum of 4.3 years and cardiac events, either cardiac death, readmission with acute myocardial infarction or admission with acute unstable angina were documented.
Methods
Blood samples were taken on admission and at 4 and 12 h. A serum aliquot was taken and stored frozen at −70oC. The frozen aliquots were subsequently analysed for cTnI by chemiluminescent immunoassay using an ACS:180. Patients were categorized into those with or without ST segment elevation on the presenting electrocardiogram. The optimized decision threshold for mortality and event prediction was then determined by log-rank analysis and by construction of Kaplan-Meier survival plots.
Results
A total of 289 patients (196 men) median age 65.3 years, range 27.4–87.9 years were studied. Out of this, 139 had ST elevation myocardial infarction (STEMI) and 150 had suspected non-STEMI (NSTEMI). Full data were available from 278 patients. Admission cTnI did not predict any of the endpoints in the STEMI group. In patients admitted with suspected NSTEMI, admission and peak cTnI predicted increased risk of death or readmission with acute myocardial infarction. In addition, peak cTnI predicted increased risk of death.
Conclusion
A cTnI exceeding 0.16 μg/L on admission or during hospital stay predicted an increased cardiac event rate at four years in patients admitted with suspected NSTEMI.
Introduction
The measurement of the cardiac troponins, cardiac troponin T (cTnT) and cardiac troponin I (cTnI) are accepted as the ‘gold standard’ biochemical test for the diagnosis of myocardial infarction (MI) and management of patients with suspected acute coronary syndromes. 1,2 The methodology for cTnT estimation is effectively standardized as there is only a single manufacturer providing the methodology for this analyte. In contrast, cTnI is available from a large number of manufacturers and there is little agreement between methodologies despite efforts to standardize. This means that individual cTnI methods must be separately evaluated in order to demonstrate that they have an evidence base for routine clinical practice. In addition, although there is an extensive literature on the ability of both troponins to predict short-term outcomes and meta-analyses have been performed, 3,4 there have been few studies that have examined long-term prediction. There is currently one paper that has examined the ability of cTnT to predict outcome at more than three years follow-up 5 and none that have evaluated long-term outcome prediction for cTnI. A further criticism of the existing literature is a selection bias. Many of the large studies, especially those cited as evidence for current guidelines have been performed on clinical trial populations. Although these studies provide a very well-documented cohort, they have a very high prior probability of coronary artery disease. This means that there may be overstating of the diagnostic and prognostic abilities of the cardiac biomarker under investigation. It is therefore important to report studies, which represent current clinical practice and to ensure that long-term outcome data are available. This is especially true for cTnI determinations. A previous paper reported clinical evaluation and a six-month outcome study of the ACS:180 cTnI assay, comparing this cTnI assay with both conventional diagnostic criteria and using cTnT as a comparative reference methodology. 6 The study showed the two methods to be substantially identical for diagnosis and short-term outcome. The present paper reports the results of a four-year follow-up on the same patient cohort, but has only reported the cTnI data as there is already an extensive literature on cTnT, but there is a lack of data on cTnI.
Methods
Patient cohort and sample collection
The study has been reported in full previously. 6 Ethical permission for the study was obtained from the local ethics committee, and the study was performed in accordance with the Declaration of Helsinki. This was an unselected prospective observational study. All admissions to the coronary care unit (CCU) of a busy district general hospital with suspected acute coronary syndromes were enrolled in the study. There were no exclusions. Data were collected prospectively onto a proforma from the case-notes and missing data subsequently obtained by case-note review following discharge. All patients underwent standard management according to the protocols in use at the time. All patients had an admission and a follow-up electrocardiogram (ECG). If the ECG showed significant ST segment elevation, patients were thrombolysed. Patients without significant ST segment elevation were managed according to the recommendations of the European Society of Cardiology. 2 Blood samples were taken on admission, and at 4 and 12 h into vacutainers containing serum separator gel (Becton Dickinson, Oxford, UK), allowed to clot, centrifuged at 3000 rpm for 10 min and analysed using cTnT as the diagnostic cardiac biomarker. A serum aliquot was taken and stored frozen at −70oC. The frozen aliquots were subsequently analysed for cTnI by chemiluminescent immunoassay using an ACS:180 (Bayer Diagnostics, Newbury, UK). The assay imprecision was 3.0% at 2.3 μg/L, 2.9% at 8.48 μg/L and 3.9% at 15.2 μg/L and reported results in the range 0.01–200 μg/L with the manufacturer recommending a range of 0.2–200 μg/L.
Follow-up
All patients were followed up at four years from initial admission using the hospital medical records system and laboratory database. All readmissions were checked by casenote review. Further information was obtained by contact with the patients' general practitioners, where appropriate. Death, readmission with acute myocardial infarction (AMI), readmission with unstable anginal pain (UAP) or needs for urgent revascularization were recorded.
Statistical analysis
Patients were initially categorized into those with or without ST segment elevation on the initial ECG and the two populations analysed separately. Patients were then dichotomized into cTnI positive or negative using the measurements taken on admission and the peak value obtained during admission. This approach to sample timing has been used in all of the large studies cited in the literature as well as the only long-term follow-up study of cTnT, which was one of the reasons for its adoption in this study. 5,7–10 Patients were defined as cTnI positive if they had a value at or exceeding the decision cut-off using increments of 0.01 μmol/L starting from the detection limit of 0.1 μg/L. Log-rank statistics were then computed and Kaplan-Meier event curves constructed using the outcome measures being, death, death or readmission with AMI, death or readmission with AMI or UAP or the major adverse cardiac events (MACE), death or readmission with AMI or readmission with UAP or need for urgent revascularization at each discriminant level.
Results
A total of 289 patients (196 men, 93 women) were enrolled in the original study, median age 65.3 years, range 27.4–87.9 years. Median onset of symptoms was 6.08 h (range 0.25–793 h) and median time to worst pain 3.0 h (range 0.17–129.0 h). Risk factors are summarized in Table 1. Out of 289 patients, 139 had ST elevation myocardial infarction (STEMI) and 150 had suspected non-STEMI (NSTEMI) (Figure 1). There was no significant difference in time to first pain or time to worst pain between the STEMI and NSTEMI groups. Full data were available in 278/289 (96.2%) corresponding to 278/279 of the cases reported in the first publication. This consisted of 133/139 (95.7%) of the STEMI and 145/150 (96.7%) of the patients with suspected NSTEMI. This represents the difference between a clinical trial population and a realworld observational all comers study performed in the clinical environment. Overall cTnI values in the suspected NSTEMI group had a median of 0.14 μg/L (range <0.01–>200 μg/L) on presentation and 0.17 μg/L (range <0.01–>200 μg/L) with the peak skewed due to a significant number of undetectable values. Excluding these, the medians were 0.31 μg/L (95th centile range 0.01–120.31 μg/L) on admission and 1.26 μg/L (95th centile range 0.02–>200 μg/L) peak. The peak value occurred on admission in 34.5%, at 4 h in 26.2% and at 12 h from admission in 39.3%. In contrast, in patients with suspected STEMI, median cTnI was 3.18 μg/L (range <0.01–>200 μg/L) on presentation and 43.08 μg/L (range 0.02–> 200 μg/L) at peak with the peak value occurring on admission in 19.5%, at 4 h in 33.1% and at 12 h in 47.4%. There was no significant difference in time to first pain or time to worst pain between the STEMI and NSTEMI cohorts.
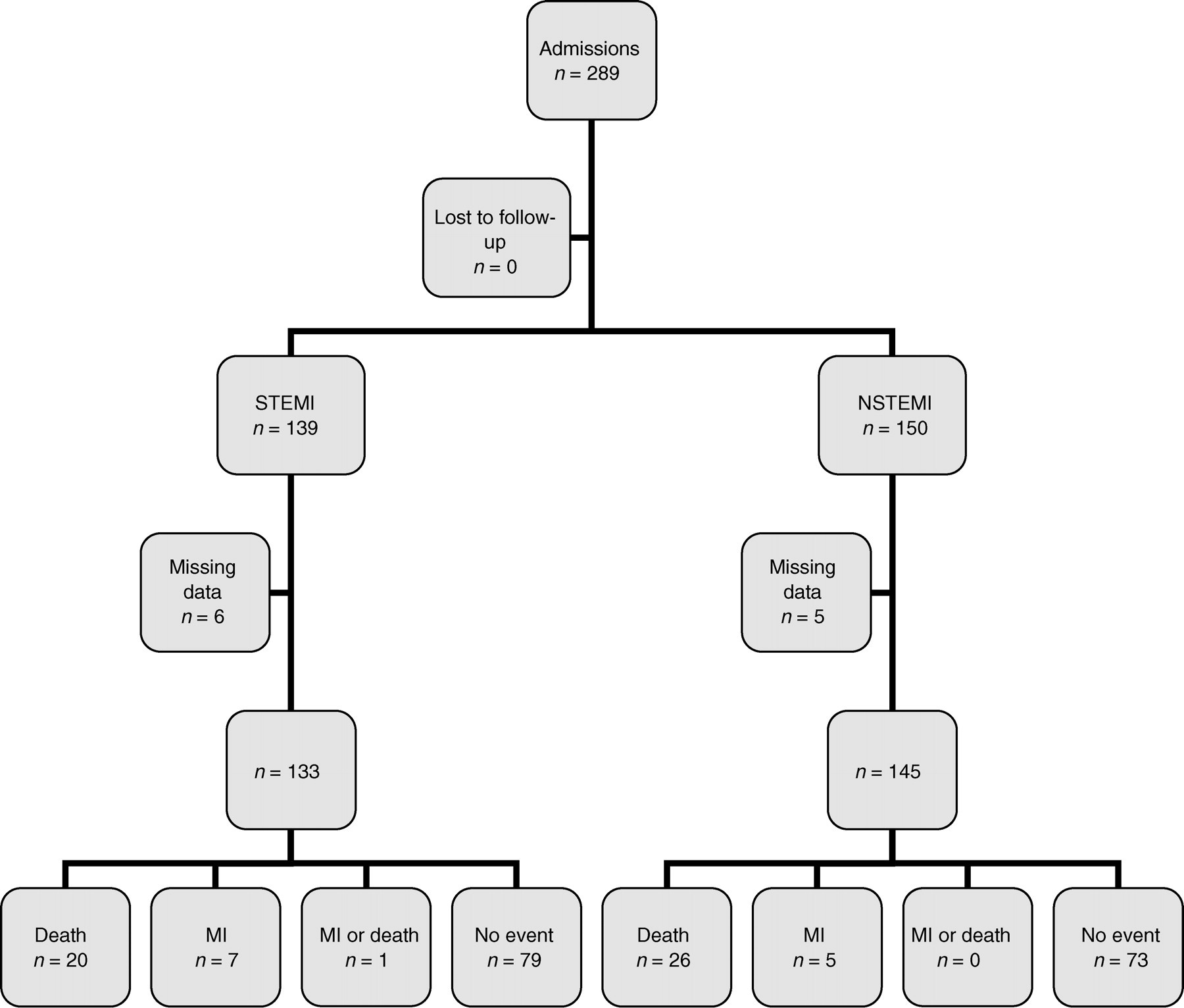
Enrolment of patients. MI, myocardial infarction; STEMI, ST elevation myocardial infarction; NSTEMI, non-STEMI
Risk factors (percentages shown in parentheses)
In patients admitted with STEMI, admission cTnI measurement did not predict any of the outcome measures studied either alone or in combination. In patients with suspected NSTEMI, both admission and peak cTnI predicted the outcome death or readmission with AMI. In addition, cTnI predicted the outcome of death. Neither admission nor peak cTnI predicted the combined endpoint of death or readmission with AMI or readmission with UAP, unstable angina or the combined endpoint of MACE. Representative Kaplan-Meier survival curves corresponding to a cTnI of 0.2 μg/L for admission and peak cTnI for the outcome death and MI are shown in Figures 2 and 3. The Kaplan-Meier curves for peak cTnI for the combined endpoint death and MI and death were essentially similar. The log-rank statistic for peak cTnI is 4.54, P = 0.0331 for the endpoint death and 5.55, P = 0.0185 for the combined endpoint death and MI (Figure 3). The curves show initial divergence at six months, consistent with the previous study on this population with the curves diverging with time, as would be expected with the known high event rate in patients with established ischaemic heart disease. In this study, death on follow-up included a large number of cardiac events, as the deaths were either sudden death or death on admission with suspected AMI. They therefore represent fatal AMI rather than hospital admission and discharge with a final diagnosis of AMI and are not counted separately as cardiac events. For this reason, it appears that there were more deaths than readmissions with MI with an unadjusted ratio of four cardiac deaths/AMI. In contrast, in a previous study of long-term follow-up of cTnT, there were an equivalent number of MIs and deaths on follow-up. 5 Interestingly, the survival curves in this study showed the same characteristics as in this study where the ability of cTnT measurement to predict death was only slightly superior to the ability to predict subsequent AMI. There is considerable discussion as to whether it is appropriate to report death, cardiac death, cardiac death and readmission with MI or combined endpoints in reporting outcome studies. The general consensus is that reporting death and death plus readmission with AMI as endpoints without attempting to define if this is due to a cardiac event is reasonable as it is very difficult to separate cardiac and non-cardiac death with certainty and death alone is the best single outcome measure. 11 In the current study, it is possible that patients with AMI were admitted elsewhere, but this is relatively unlikely as the population in the catchment areas is stable. A more likely explanation is the change in the underlying pattern of disease.
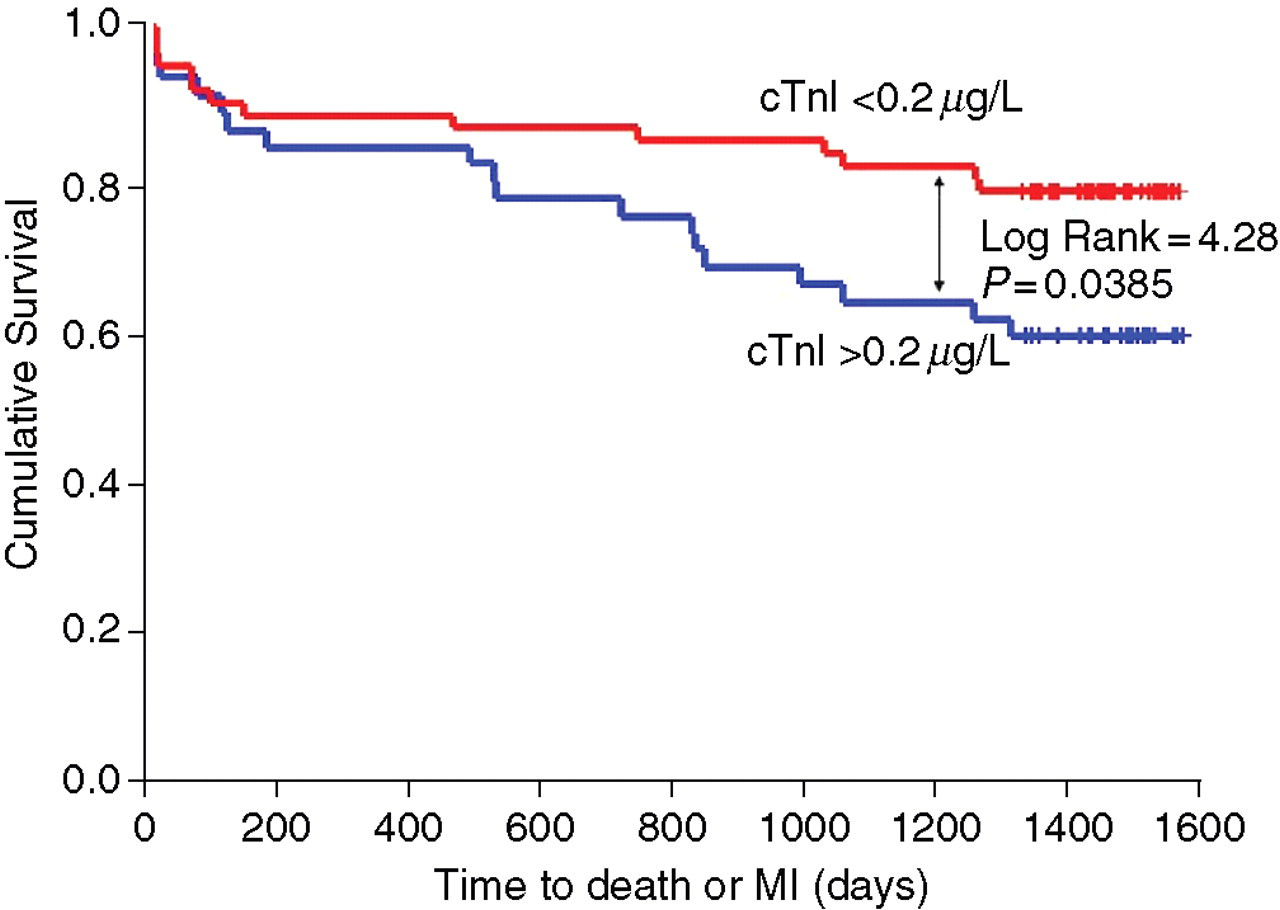
Kaplan-Meier plot of four-year event rate for the combined endpoint death or readmission with myocardial infarction (MI) for the admission sample
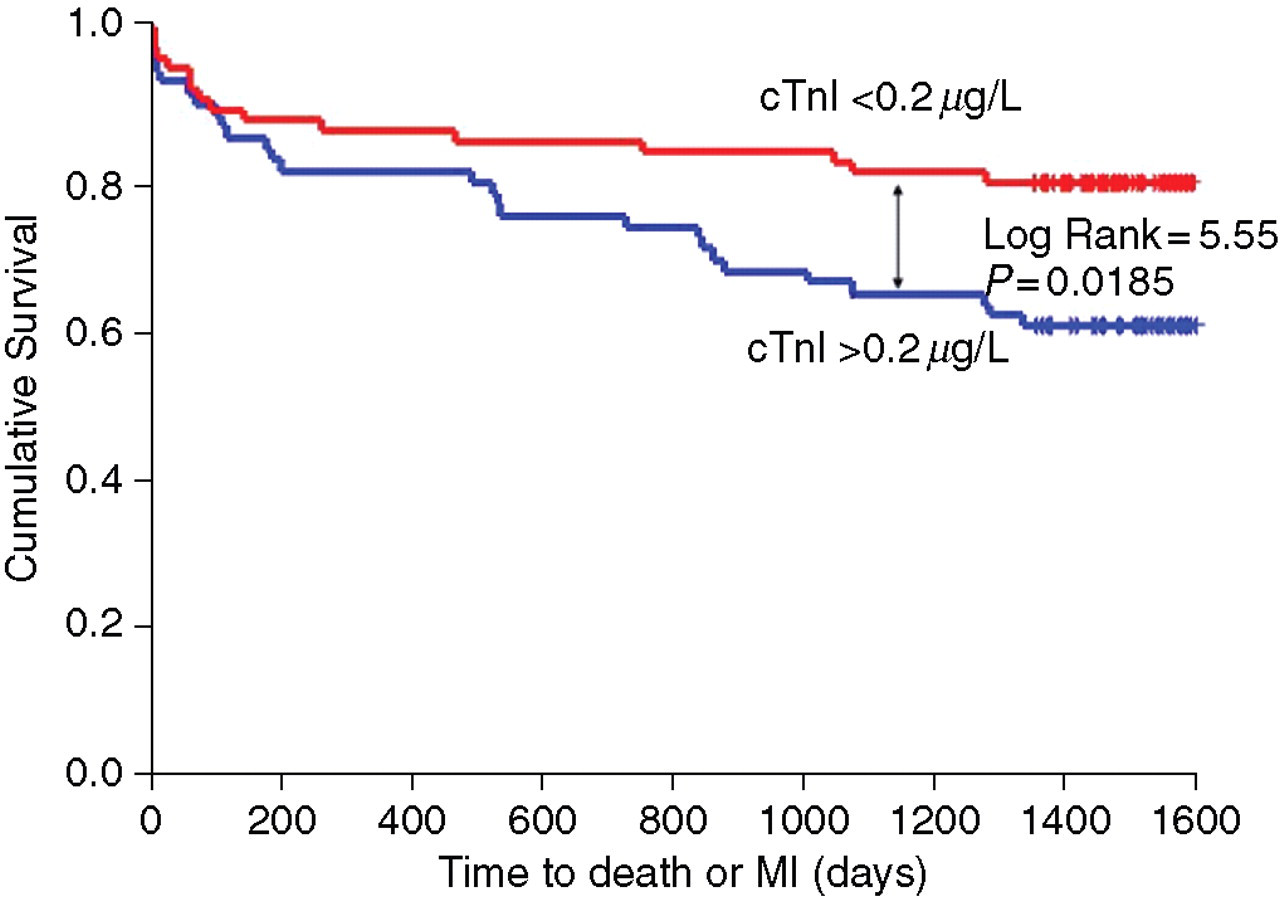
Kaplan-Meier plot of four-year event rate for the combined endpoint death or readmission with myocardial infarction (MI) for the peak cardiac troponin I (cTnI) value
Significant separation first occurred at a cTnI value of 0.16 μg/L. Across the range of cTnI values from 0.16 μg/L to 0.29 μg/L, no single cut-off value was significantly more efficient in discriminating between the endpoint death or MI (Table 2).
Log-rank values across cardiac troponin I (cTnI) cut points for the endpoint death or myocardial infarction
Discussion
The measurement of cTnI using the ACS:180 can be used for risk stratification in patients presenting without ST segment elevation on the ECG. This confirms and extends previous work, 12 but there are two important differences. First, the cohort studied was followed up for four years. There are no other studies reporting a four-year follow-up for cTnI currently reported in the literature. Secondly, the population represents admissions to a CCU rather than recruits to a clinical trial. Clinical trial populations are useful for hypothesis generation but suffer from significant problems with inclusion bias. The entry criteria for such trials exclude co-morbidities and actively select for coronary artery disease. This means that the prior probability of disease approaches 100%. In contrast, there were no exclusion criteria in this study. This means that the prior probability of disease was not the almost 100% observed in such trials and there is much less selection bias. The results are therefore more applicable to routine clinical practice. In contrast to previous studies with cTnT 7,13 and cTnI, 14 the admission measurement of cTnI did not allow risk discrimination. This may be a reflection of the population studied or of the sample size. Failure of peak cTnI to predict outcome in the STEMI cohort is unsurprising, as the patients received thrombolytic therapy, which will affect washout kinetics of the biomarker if reperfusion was successful. This is reflected in the fact that peak cTnI occurred before 12 h in an appreciable percentage of patients with STEMI implying variable degree of thrombolytic success. Patients were not routinely catheterized, so it is not possible to make comments on whether angiographic flow was adequately re-established. Clinical studies have shown that establishment of adequate reperfusion is the main determinant of survival in patients presenting with STEMI. 15
The results show optimal discrimination at a level below the 10% coefficient of variation (CV) point of the assay. Previous studies have suggested lowering the prognostic cut-off for cardiac troponins to values close to the detection limit of the assay with the recommendation that any degree of troponin elevation is of prognostic significance. 9 Although the 10% CV was originally proposed as the discriminant for risk stratification, there have been recent suggestions that to discriminate between a 10% CV and a 99th centile may be inappropriate. Two modelling studies have addressed this issue. One study considered the impact of imprecision on determination of the 99th centile and the impact this would have on clinical performance, concluding that the diagnostic impact would be minimal. 16 A second considered the effect of imprecision on receiver operating characteristic (ROC) determined cut-off points, with the same conclusion. 17 The ability of an assay to provide reliable diagnostic discrimination is determined by assay sensitivity as well as precision profile. This is borne out by this study where the optimal prognostic discrimination lies close to the detection limit of the assay and below the 10% CV point and shown little significant change across a range of cTnI values. It is interesting to speculate that as assay performance is improved, the 99th centile may progressively reduce to lower and lower values. As yet, a convincing normal distribution has not been demonstrated for any assay, with the majority of samples showing undetectable values. The need for very careful selection of reference populations has been demonstrated as apparently healthy individuals may have elevated troponin which carries prognostic significance. 18,19 Refinements of this cTnI assay, currently in development, can be expected to yield further improvements in diagnostic sensitivity and prognostic discrimination.
Although this study did not use a clinical trial population it does include a degree of selection bias as the patients were at high risk of ischaemic heart disease. It is therefore representative of a CCU population but not of a chest pain evaluation unit population. In conclusion, the ACS:180 cTnI assay is suitable for prognostic risk stratification in routine clinical use.
