Abstract
Background:
Because excessive reduction in activities after back injury may impair recovery, it is important to understand and address the factors contributing to the variability in motor responses to pain. The current dominant theory is the “fear-avoidance model”, in which the some patients' heightened fears of further injury cause them to avoid movement. We propose that in addition to psychological factors, neurochemical variants in the circuits controlling movement and their modification by pain may contribute to this variability. A systematic search of the motor research literature and genetic databases yielded a prioritized list of polymorphic motor control candidate genes. We demonstrate an analytic method that we applied to 14 of these genes in 290 patients with acute sciatica, whose reduction in movement was estimated by items from the Roland-Morris Disability Questionnaire.
Results:
We genotyped a total of 121 single nucleotide polymorphisms (SNPs) in 14 of these genes, which code for the dopamine D2 receptor, GTP cyclohydrolase I, glycine receptor α1 subunit, GABA-A receptor α2 subunit, GABA-A receptor β1 subunit, α-adrenergic 1C, 2A, and 2C receptors, serotonin 1A and 2A receptors, cannabinoid CB-1 receptor, M1 muscarinic receptor, and the tyrosine hydroxylase, and tachykinin precursor-1 molecules. No SNP showed a significant association with the movement score after a Bonferroni correction for the 14 genes tested. Haplotype analysis of one of the blocks in the GABA-A receptor β1 subunit showed that a haplotype of 11% frequency was associated with less limitation of movement at a nominal significance level value (p = 0.0025) almost strong enough to correct for testing 22 haplotype blocks.
Conclusion:
If confirmed, the current results may suggest that a common haplotype in the GABA-A β1 subunit acts like an “endogenous muscle relaxant” in an individual with subacute sciatica. Similar methods might be applied a larger set of genes in animal models and human laboratory and clinical studies to understand the causes and prevention of pain-related reduction in movement.
Background
Pain-evoked limitation of activities is one of the most costly morbidities of illness [1]. Several studies have replicated the finding that bed rest or a decrease in activity may impair recovery from an acute back pain episode [2–5]. Clinical practice guidelines recommend early activity in the management of acute low back pain [6,7].
If decreased activity in the presence of back pain retards recovery, it may be important to understand why some patients decrease their activity more than others after a similar injury. Most of the relevant work to date has focused on psychological causes of reductions in motor activity in pain patients [8,9] paying relatively little attention to factors intrinsic to the motor system or direct connections with afferent pain systems. For example, Lethem et al [8] proposed the fear-avoidance model to explain a possible mechanism of inactivity and disability in patients with chronic musculoskeletal pain. According to this hypothesis, if an individual misinterprets back pain as a signal of reinjury or continued tissue damage, his fear of injury may cause him to systematically avoid movements that typically increase pain [10]. The resulting inactivity may lead to deconditioning, contractures, and disability.
Psychological variables almost certainly explain some of the variation in the motor response to injury, but we propose that one should also examine the contribution of inter-individual differences in the wiring and neurochemistry of the motor system itself. Lund et al. [11] have proposed a plausible link from pain inputs to motor circuits, the “pain-adaptation model.” In this model, pain afferent activity decreases activity of the muscle groups that move a joint in the pain-provoking direction and increases the activity of muscle groups that antagonize such movement. These motor adjustments reduce movement velocity and limit excursions, thereby protecting against pain.
One way to examine potential differences in motor control processes is to test for polymorphisms in genes encoding molecules that regulate motor systems. We and others have already been studying the association between short lists of candidate genetic polymorphisms and the severity of acute and chronic pain [12–14] and pain-related mood change [15]. Arrays of 500,000 or more single nucleotide polymorphisms are commercially available, making possible whole genome association studies in large cohorts of pain patients. It occurred to us that in cohorts where one had a good measure of motor function, one could use the same whole genome scan data to search for genes that predisposed to a greater decrease in activity, given similar injury and pain level. Many current spine pain studies will include enough data to make this possible, because a decade ago, leading spine researchers [16] agreed that studies should routinely incorporate either the Roland-Morris [17] or Oswestry Disability questionnaires [18], which question the patient about many motor functions.
We propose that there may be genetic polymorphisms that directly affect the motor system to cause a maladaptive level of guarding and hypoactivity. In preparation for our studies to test this hypothesis, we have examined the literature on motor systems to suggest some candidate genes. We previously proposed a method for prioritizing genetic polymorphisms for clinical studies in which pain intensity is the primary endpoint [19]. We have adapted this into a method to prioritize polymorphisms that may contribute to reduction in movement in the presence of pain, and will describe this method and our resulting list of plausible candidate genes.
Results
High priority motor control candidate genes
The prioritization process generated the candidate gene list shown in Table 1, plus additional genes, not shown, that scored lower than those in Table 1. Our literature search showed that there was virtually no work specifically on the neurochemistry of pain-related motor changes in mammalian models. Therefore, the ratings for “relation to motor control” just reflect the degree of certainty that the molecule is present at key sites in motor control systems, from basal ganglia down to the neuromuscular junction.
High-priority candidate polymorphisms that may affect motor control.
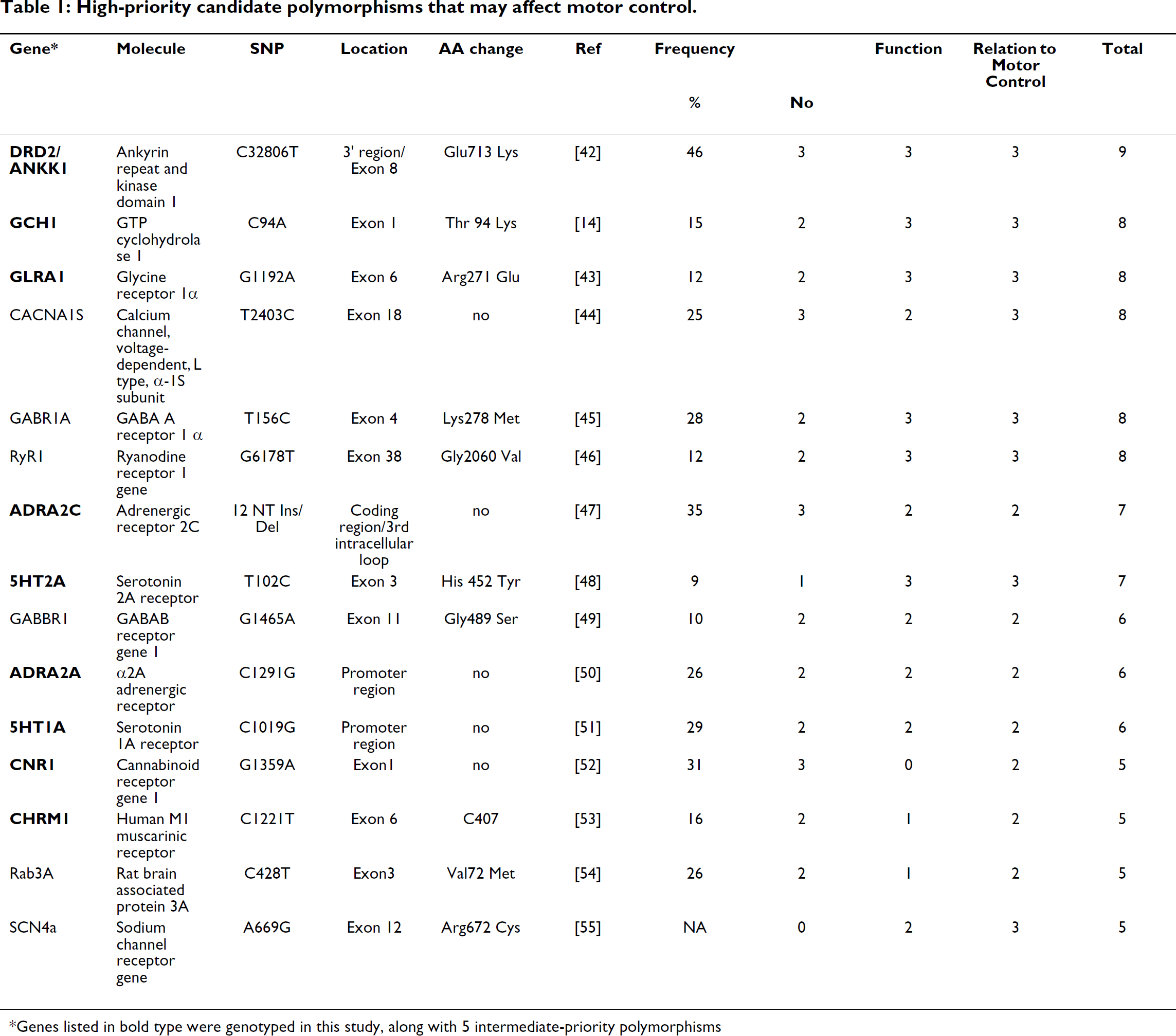
Genes listed in bold type were genotyped in this study, along with 5 intermediate-priority polymorphisms
Non-genetic factors contributing to movement reduction
Table 2 shows the Roland-Morris Disability Questionnaire items that we included in the motor limitation subscale. Figure 1 shows that patients with higher SF-36 bodily pain magnitude ratings reported greater movement limitation. Pain rating contributed 31% of the variance in movement impairment in patients who chose nonoperative treatment, and 16% of the variance in those who chose surgery. Table 3 shows other non-genetic contributors to variation in movement impairment. For this and subsequent statistical analyses, we did not include pain after observing that a two-factor, gene × pain analysis of covariance for movement impairment yielded many more significant genetic associations than would have occurred by chance, even for lists of genes that were not on our motor candidate list. Our only explanation for this was that because the Roland-Morris movement questions include wording referring to both pain and movement, this might produce unanticipated biases of analyses that include both pain and movement. Therefore we simplified the analysis by recognizing that all patients presented to the study with leg and back pain – an entry criterion – and dropping pain magnitude from the analysis. This step corrected the number of SNP associations to the number expected by chance.

Impairment of movements involved in everyday activities on the day of study entry (Roland Morris Disability Questionnaire subscale, Table 2) vs. magnitude of “bodily pain” (SF-36) in the month prior to study entry. The patients are Maine Lumbar Spine Study sciatica cohort patients who subsequently chose nonsurgical (left) or surgical treatment (right). Some y axis values have been slightly altered to let the reader estimate the number of patients with overlapping data points. Not surprisingly, patients with more pain report limitation of more activities.
Roland-Morris Disability Questionnaire.

Note: In the Maine Lumbar Spine Study, patients were asked to answer yes or no to each question to describe their condition “today,” at study entry after referral to a specialist for acute or subacute sciatica. The total score is the number of items answered “yes.” We selected the items in bold type as a “movement impairment” subscale.
Variables that contribute to baseline Roland-Morris motor limitation subscale score.

Note: In the Maine Lumbar Spine Study, patients were asked to answer yes or no to each question to describe their condition “today,” at study entry after referral to a specialist for acute or subacute sciatica. The total score is the number of items answered “yes.” We selected the items in bold type as a “movement impairment” subscale.
Table 3 shows that greater reduction in movement was associated with lower scores for the SF-36 Vitality subscale (R2 = 29%); the choice of surgical treatment (R2 = 16%); the patient receiving workers' compensation (R2 = 1.5%), the presence of neurological deficits on exam (R2 = 4%), and lower scores, indicating greater impairment, on the SF-36 Emotional Role subscale (R2 = 12%). A combination of all of the variables in the model explained 44% of the variance in the movement reduction subscale.
Analysis of 14 movement candidate genes and movement reduction
Resources permitted us to genotype a set of SNPs from 14 of these genes: 9 of the top priority 15 genes in Table 1, and 5 lower priority genes that were included in the Laboratory of Neurogenetics' array of 130 polymorphic genes of interest to a consensus of neuropsychiatric and addictions researchers. No SNPs in the fourteen genes we tested – the dopamine D2 receptor, GTP cyclohydrolase I, glycine receptor α1 subunit, GABA-A receptor α2 subunit, GABA-A receptor β1 subunit, α-adrenergic 1C, 2A, and 2C receptors, serotonin 1A and 2A receptors, cannabinoid CB-1 receptor, M1 muscarinic receptor, and the tyrosine hydroxylase, and tachykinin precursor-1 molecules – showed a significant association with the movement score after a Bonferroni correction for the multiple candidate genes tested (Additional file 1). The two SNPs in the α-2C adrenergic receptor that were nominally significant in the additive model (without correction for multiple tests, Table 4) made small contributions to the variance in movement scores – approximately 0.6% and 0.9% (partial R2). Neither of these SNPs is known to cause a functional change in molecular function. Haplotype analysis of this gene using all four ADRA2C SNPs genotyped in this study showed a nominally significant association (p = 0.02) with movement scores which loses significance when one corrects for the 22 haplotype blocks examined.
Association of four ADRA2C markers with movement scores in sciatica patients
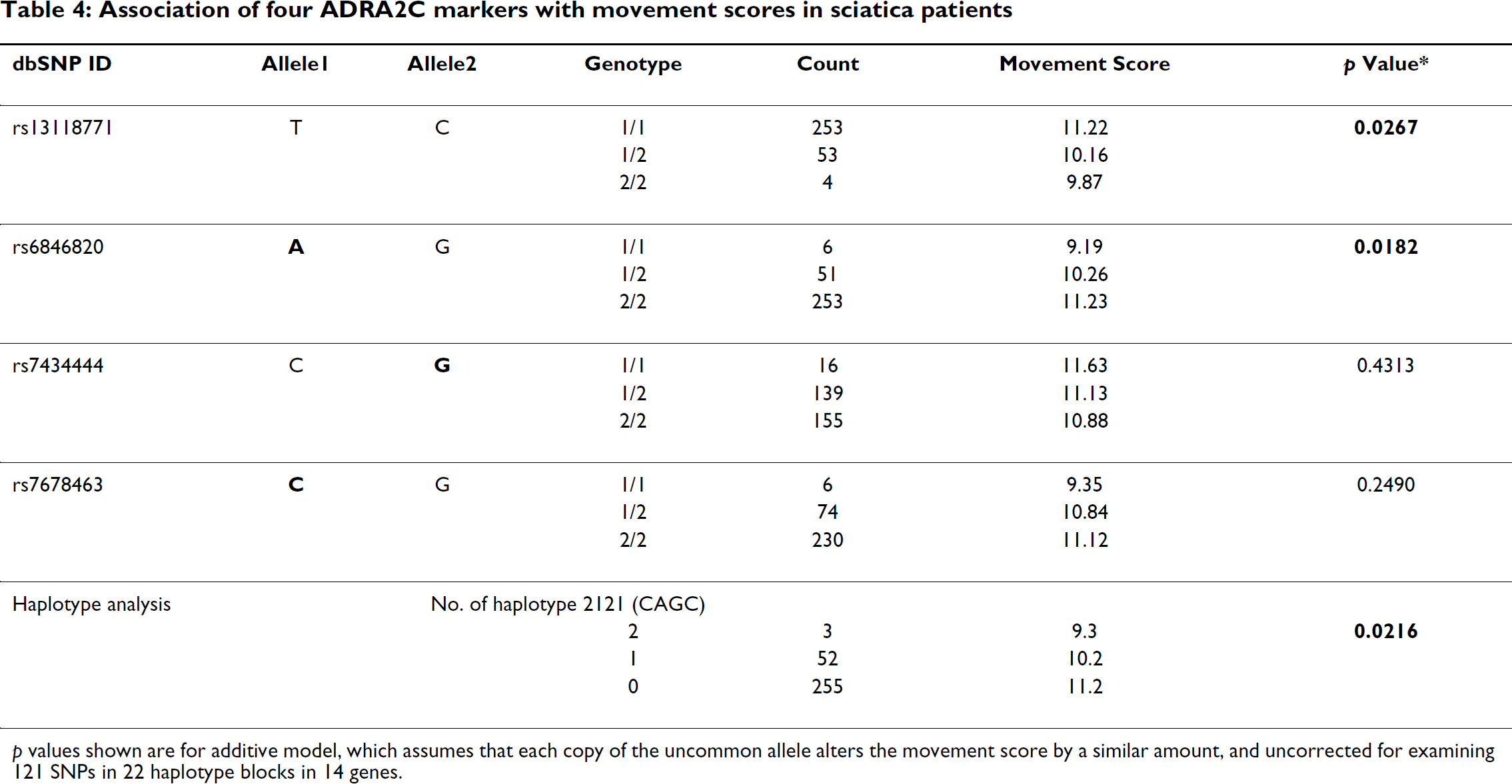
p values shown are for additive model, which assumes that each copy of the uncommon allele alters the movement score by a similar amount, and uncorrected for examining 121 SNPs in 22 haplotype blocks in 14 genes.
Haplotype analysis of one of the blocks in the GABA-A receptor β1 subunit showed that a haplotype of 11% frequency was associated with less limitation of movement at a significance level (p = 0.0025; Table 5) almost strong enough to correct for testing 22 haplotype blocks.
Association of six GABRB1 markers with movement scores in sciatica patients

p values shown are for additive model, which assumes that each copy of the uncommon allele alters the movement score by a similar amount, and uncorrected for examining 121 SNPs in 22 haplotype blocks in 14 genes.
Power analysis indicated that our study had sufficient power (85%) to detect effect of a single SNP that contributed 5% (partial R2 = 0.05) of the movement variance after correcting for Bonferonni error using α = 0.00357 (=0.05 divided by the number of genes tested, 14).
Discussion
Our literature review showed that the genes for many molecules associated with motor control circuits have common polymorphisms. Such polymorphisms might conceivably contribute to excessive guarding shown by some patients with pain.
The present study of 14 of these candidate genes revealed an association of a GABA-A β1 subunit haplotype with the amount of motor limitation (Table 5). Two copies of the haplotype reduced movement limitation scores almost 30% compared with no copies. The nominal p value of 0.0025 is small enough to be interesting even if one corrects for the 22 haplotype blocks examined, yielding an overall p value = 0.055. If confirmed by subsequent studies, we would hypothesize that this variant of the gene serves as an “endogenous muscle relaxant”, with greater GABAergic motor inhibitory activity that reduces spasm and permits more activities.
We cannot speculate about the specific biochemical mechanism by which this haplotype might affect motor function. The finding that the haplotype analysis was much more sensitive than single SNP analyses suggests that a movement-facilitating SNP that we did not directly test may be present on the haplotype we identified. Compared to other GABA receptors, the GABA-A receptor β1 subunit has been little studied [20], and there is no commonly used specific antagonist, agonist, or knockout mouse. The published literature states that benzodiazepine muscle relaxant effects are primarily mediated through GABA-A α2, α3, and α5 receptor subunits [21]. However, interest in the β1 subunit is growing because of possible associations with autism [22] and bipolar disorder [23], so the gene may soon be better understood.
The nominally significant associations of two ADRA2C SNPs and a haplotype with motor limitation scores are not persuasive because of the modest size of the effect and the many genes tested. However, this gene is known to have common polymorphisms with strong effects on cardiovascular regulation and mood, such as a deletion affecting about 40% of chromosomes in African Americans [24,25], not tested for in the current study of a Caucasian population. Alpha-2 adrenergic receptors mediate inhibition of motor tone at the spinal level [26], so this gene should be revisited in future studies.
Because of several limitations in our study methods, we consider these findings tentative. The first limitation is that the Roland-Morris scale items that we used are retrospective and subjective assessments of the patient's ability to carry out various movements of daily life. Moreover, we chose a subset of these items to represent changes in movement without any formal validation studies. Objective measures of motor function would have been more convincing, but these were not available for this cohort.
Secondly, we tested only 14 genes, chosen by an anatomical analysis the emphasized the final two or three neurons in the motor control pathway. Molecules influencing more rostral levels of control might also be candidates. For example, Kleim et al. [27] have reported a BDNF polymorphism to be associated with modified plasticity in the human motor cortex. A more thorough search could be done using a whole genome association array, but this would require a sample size in the thousands [23] to provide adequate power.
Thirdly, we would have been interested in searching for an interaction between gene effect and pain level on movement, similar to the gene, pain, and depression analysis we have done on the same cohort [15]. For example, some polymorphic alleles might produce a major reduction in movement only in the presence of severe pain. However, this analysis was not appropriate in this case because the scale items mention both pain and various movements.
Finally, long-term decrease in movement is more directly relevant to the economic burdens of pain than the acute effects that we examined above. The analytical approach demonstrated above might also be used to search for effects of genes on a chronic decrease in movement. We chose to examine acute pain and movement data as an initial demonstration of this method for several reasons: (1) Almost all of our 290 patients who gave a DNA sample had pain at the baseline measurement, while a minority had persistent pain at one year or later time points. Sample size is crucial in screening multiple candidate genes [19]. (2) A shorter time window in which patients are studied under similar methods and treated by the same clinicians may be more sensitive to genetic effects than a longer period during which many types of environmental variability overshadow gene effects. (3) The processes involved in chronic pain and movement limitation are probably more complex than acute pain, including deconditioning, muscle atrophy and contractures, chronic psychiatric morbidity, and occupational factors.
Although physicians commonly prescribe “muscle relaxants” such as cyclobenzaprine, carisoprodol, and methocarbamol for acute pain, the database supporting their effects on muscle and clinical usefulness is limited [28]. Development of new treatments will require expansion of animal research on the neurochemical mechanisms of pain-related guarding. We propose that, as has been demonstrated in genetic studies of pain [14], genomic screens in humans may be useful in to prioritizing targets identified in animal studies.
Conclusion
We have illustrated a method to use routinely measured variables in musculoskeletal pain studies to screen for molecules that may be associated with the excessive pain-related decrease in movement that increases risk of chronic disability. After correction for multiple comparisons, we found a modestly significant association between a haplotype in the GABA-β1 receptor subunit with motor limitation scores. Replication of this finding and the use of such methods with larger samples or direct measures of movement [29,30] might open up a new facet of the relationship between pain and movement.
Methods
Prioritization of “motor control” polymorphisms
Although muscle stiffness and reduction of movement in pain patients is widely discussed in the literature [31,32], there is no evidence regarding its molecular basis. Hence we reviewed the motor control research from animal and human studies to compile a list of molecules which appear to be involved in normal motor control and in diseases where muscle tone is altered and movement restricted and muscle stiffness.
We searched the literature for reports of the transmitters, receptors, and signaling molecules mediating control of limb and trunk movements, tone, and posture. We searched PubMed for review articles on motor control mechanisms and for citations of neurotransmitters, receptors, or signaling molecules relevant to motor cortex, brainstem, basal ganglia, cerebellum, locus coeruleus, corticospinal tract, ventral horn, interneuron, Renshaw cells, and neuromuscular junction. We prioritized polymorphisms in the genes for these molecules similar to the criteria proposed by Belfer et al. [19]. Three scores of 0–3 were assigned to each polymorphism according to (a) the strength of evidence supporting involvement of the gene in motor control; (b) population frequency of the polymorphism; and (c) strength of evidence that the polymorphism is associated with an altered clinical or molecular phenotype.
A). Strength of evidence supporting the involvement of gene in motor control and muscle tone regulation
We assigned one point if a single paper reported anatomical, physiological, or pharmacological evidence plausibly associating the molecule with motor control, two points for multiple reports, and three points for multiple reports that show alterations of that molecule alter muscle tone in animals or in patients.
B). Population frequency of polymorphism
Given equal effects on function, more common polymorphisms more efficient to study because there are sufficient numbers of patients with one or two copies of the uncommon allele. We assigned zero points if the population frequency of the variant is less than 3%, one point for 4–9%, two points for 10–29% and three points for 30–50%.
C). Functional effects of polymorphism
We made a PubMed and Panther database search and assigned one point if the variant changed an amino acid, two points for one report that the variant changes the amount of mRNA or protein expression or function or is associated with a clinical phenotype, and three points for independent replication of any of these types of evidence.
Patients
Participants were members of the sciatica group in the Maine Lumbar Spine Study (MLSS [33]). The MLSS was a prospective cohort study conducted by approximately half of Maine's orthopedists and neurosurgeons who actively treated spine disease. Patients were enrolled between 1990 and 1992, and surgical discectomy or non-surgical treatment was chosen based upon clinician judgment and patient preference. Patients completed questionnaires at a baseline assessment, and then at 3, 6, and 12 month follow-up, and then annually through year 10. After completion of the 10 year study, the NIDCR and MLSS investigators developed a collaboration to add a genetic component to the study, under a protocol approved by the NIDCR Institutional Review Board. Patients were invited to contribute a DNA sample. Two hundred ninety of the original 507 enrollees provided DNA and had usable baseline data.
Pain measure
The primary measure of pain for this study was the Bodily Pain intensity item on the Short-Form-36 (SF-36) quality of life instrument [34] at baseline. Patients responded to the question “How much bodily pain have you had during the past 4 weeks?” by choosing from “very severe,” “severe,” “moderate,” “mild,” “very mild,” and “none.”
Selection of “movement impairment subscale” from Roland-Morris
Table 2 lists the 23 items in The Roland-Morris Disability Questionnaire. We selected the 16 items that refer to a pain-related reduction in movement to comprise a “movement impairment subscale.” Each “yes” answer contributes one point to the score.
Genotyping methods
DNA samples
Genomic DNA was extracted from lymphoblastoid cell lines using a salting out protocol [35]. Additional DNA samples were obtained from saliva samples collected using Oragene self-collection kits and the DNA extracted according to the manufacturer's recommendation.
Selection of genetic markers
One hundred twenty-one SNPs in the 14 genes studied were selected based on the haplotypes reconstructed by Haploview [36] or SNPHAP [37] with HapMap project genotype data. A SNP selection pipeline based on a double classification tree search algorithm [38] was used to capture the haplotype complexities and the tag SNPs (seleceted with Haploview for haplotype blocks).
Genotyping
We genotyped the DNAs using Illumina GoldenGate chemistry on Sentrix Universal-96 Arrays. The Illumina array used interrogates 1536 SNP simultaneously, using a custom primer assay design (GS-0007064-OPA). 500 ng of DNA per well was genotyped using standard Illumina protocols. Arrays were imaged using an Illumina BeadStation 500 GX and data analyzed using GenCall 6.2.0.4 and GTSreports v5.1.2.0 software (Illumina). Ten percent of DNA samples were run in duplicate in order to obtain an estimate of genotyping reproducibility. The overall error rate was <0.005. Average genotyping completion rate was >0.98. Hardy-Weinberg equilibrium for each marker was tested with Chi-square tests using the R package “genetics” [39] or exact tests [40]. No deviation from the expected Hardy-Weinberg equilibrium values were observed for any of the values analyzed.
Inference of haplotypes
Haplotype phases – i.e., how the directly measured SNP alleles were distributed into two chromosomes in each patient – were inferred by the expectation – maximization (EM) algorithm (SAS/Genetics, Cary, North Carolina, USA).
Statistical analysis
Multiple linear regression was applied to examine the association between movement and each SNP, adjusting for eight other variables: age, sex, SF-36 Vitality, SF-36 Emotional Role, nonsurgical vs. surgical treatment, workman's compensation, marital status, and neurological exam deficits. An additive genetic model was tested by recoding the three SNP genotypes as 0 for the homozygote of the common allele, 1 for the heterozygote, and 2 for homozygote of the uncommon allele. In the haplotype analysis, first haplotype phases were inferred using expectation-maximization (EM) algorithm, where the probability was assigned to each pair of haplotypes which each subject possessed. Then, stepwise regression [41] was applied to test the association between movement score and haplotypes; the linear regression model included all haplotypes with frequency greater than 1% and the eight covariates.
List of abbreviations used
GABA: Gamma-aminobutyric acid
GTP: Guanosine triphosphate
NIDCR: National Institute of Dental and Craniofacial Research
Competing interests
Drs. Max, Belfer, Wu, Kingman, Goldman, and Atlas are listed as coinventors on a patent application for the use of a diagnostic test for GCH1 polymorphisms to predict the level of chronic pain.
Authors' contributions
BKM carried out the review of motor neurochemistry, prioritized the polymorphisms, and cowrote the manuscript. MBM conceived of the project and analysis methods and cowrote the manuscript. TW did the statistical analyses, with advice of AK. CAH, IB, and CK carried out the genotyping. QY, CAH, and DG designed the genotyping array. DG provided senior scientific guidance on bioinformatics and genotyping issues. LGC provided advice on motor system physiology and clinical phenotype. SJA and RBK designed and carried out the original clinical study and advised on interpretation of the current analyses. All authors read and approved the final manuscript.
Additional material
Additional file 1
Association of SNPs in motor control candidate genes with movement scores in sciatica patients. Each SNP tested in the study is identified, with the number of subjects and mean movement limitation score for each genotype, and significance levels, assuming an additive model for allele effects.
Click here for file [http://www.biomedcentral.com/content/supplementary/1744-8069-3-20-S1.doc]
Footnotes
Acknowledgements
Supported by NIDCR Intramural Grant DE00366 (MBM) and NIAAA Intramural Grant AA000303 (DG). The Maine Lumbar Spine Study was supported by grants from the Agency for Healthcare Research and Quality (HS-06344, HS-08194, and HS-09804). SJA is supported in part by funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. We thank Yen A. Wu, MPH for preparing the clinical dataset. The funding bodies had no input into the design or publication of the current study.
