Abstract
This study was carried out to gain understanding about the sintering behaviour of highly crystallisable industrial waste derived silicate mixtures under direct heating and rapid cooling conditions. The materials used in this study were plasma vitrified air pollution control waste and rejected pharmaceutical borosilicate glass. Powder compacts sintered under direct heating conditions were highly porous; compacts with particle size < 38 μm reached a maximum density of 2.74 g cm− 3 at 850°C, whereas compacts with particles of size < 100 and < 250 μm reached maximum densities of 2.69 and 2.72 g cm− 3 at 875 and 900°C respectively. Further increase in sintering temperature resulted in a rapid decrease in density of the glass ceramics. Image analysis results were used to link the sudden drop in density to the increase in volume of microsized pores formed in the samples during sintering. In particular, compacts made from < 38 μm particles sintered at 950°C resulted in 65 vol.-% porosity with a pore size of ∼20 μm. Such materials can be used for sound and thermal insulation purposes.
Introduction
Municipalities incinerate waste materials in order to reduce waste volumes, recover energy and avoid excessive land filling. Municipal residues are incinerated at temperatures exceeding 850°C, resulting in fly ash (which may be treated in situ), air pollution control residues (APCr) 1 and bottom ash (also known as grate ash) as solid by products. It is estimated that the amount of waste treated in this way will increase to 20 Mt by 2020 (UK data).2,3 The composition of APCr varies according to the source, but it is rich in silica, calcia, alumina and other oxides 4 and can be modified by fluxing and melting using technologies such as the plasma vitrification process to form a stable solid. 5 Such vitreous materials can be completely or partially crystallised by controlling the process parameters to form glass ceramics.6,7 Glass ceramic materials can find structural or functional applications if their processing conditions, and therefore, microstructure and properties can be controlled. In general, waste derived glass ceramics made from waste residues are found to be chemically stable and exhibit improved mechanical and functional properties in comparison with the parent glass.6,7,8
Porous glass ceramics can find applications in sound insulation, thermal insulation and also as lightweight aggregates in concrete.
8
For example, Garcia
In the present study, a similar kind of PVW derived from APCr was mixed with borosilicate glass (BSG) residues from the pharmaceutical industry to develop a new family of porous glass ceramics. The influence of sintering parameters on density changes and on microsized porosity formation in the sintered body was investigated. The mechanisms responsible for the evolution of micron sized pores in the sintered bodies were also examined. An additional objective of this study was to investigate the reduction of processing time (and consequently total energy consumed) required to fabricate the final glass ceramic product based on optimisation of the processing conditions, which is discussed in detail elsewhere. 12
Materials and methods
Plasma vitrified waste and rejected pharmaceutical BSG (provided by Nuova OMPI, Italy) were used as raw materials. As received PVW (15 wt-%Al2O3–32.9 wt-%CaO–4.6 wt-%Fe2O3–1.4 wt-%MgO–40.9 wt-%SiO2–1.7 wt-%TiO2) 11 and BSG (5 wt-%Al2O3–10.5 wt-%B2O3–1.5 wt-%CaO–7 wt-%Na2O–75 wt-%SiO2) were crushed individually in a Retsch BB51 crushing device; the process was repeated ∼4 times with a decreasing gap between crushers. The fine powder obtained was graded into particle size fractions of < 250–100, 100–38 and < 38 μm using sieves. Of the total crushed powder, 15–20 wt-% was < 38 μm in particle size.
Plasma vitrified waste +10 wt-% BSG powder and 5 wt-% distilled water were mixed thoroughly in a mortar to obtain homogeneous mixtures. The wet powder mixture was pressed uniaxially in a hydraulic press to obtain green compacts of diameter 20 × 2 mm thickness; the compacts were dried overnight in an oven at 75°C. The composition was maintained constant by mixing similar particle sizes of PVW and BSG ( < 38 μm PVW to < 38 μm BSG, < 100 μm PVW to < 100 μm BSG, < 250 μm PVW to < 250 μm BSG). These samples are labelled as shown in Table 1.
Nomenclature of PVW+10 wt-% BSG samples

Sintering was performed on dry samples at temperatures in the range 800 to 1200°C under direct heating conditions (placing powder compacts into the furnace preheated at the sintering temperature) for 30 min followed by cooling in air to room temperature (RT). No visible cracks were found on the samples after cooling to RT. Density and water absorption (WA) measurements were performed by Archimedes’ method using the American Society for Testing and Materials standard procedure (C373-88). Samples were prepared for image analysis by grinding them to a 3 μm finish, by means of fine silicon carbide paper, followed by cleaning in distilled water using an ultrasonic cleaning bath. Optical microscopic images were analysed for pore size and pore volume using ImageJ open source software. Backscattered images from the prepared samples were taken using an ESEM Quanta 200 FEI NL scanning electron microscope (SEM).
The crystalline phases were studied by means of X-ray diffraction on powdered samples (Bruker AXS D8 Advance, operating with Cu
Results and discussion
Figure 1a shows the densities of powder compacts GC38, GC100 and GC250 sintered at temperatures between 800 and 1200°C. After sintering at 800°C, all powder compacts had similar low densities, indicating no significant viscous flow densification. However, above 850°C, all samples exhibited a drastic density change; sample GC38, as an example, rose from ∼1.9 g cm− 3 at 800°C to ∼2.7 g cm− 3 at 850°C.
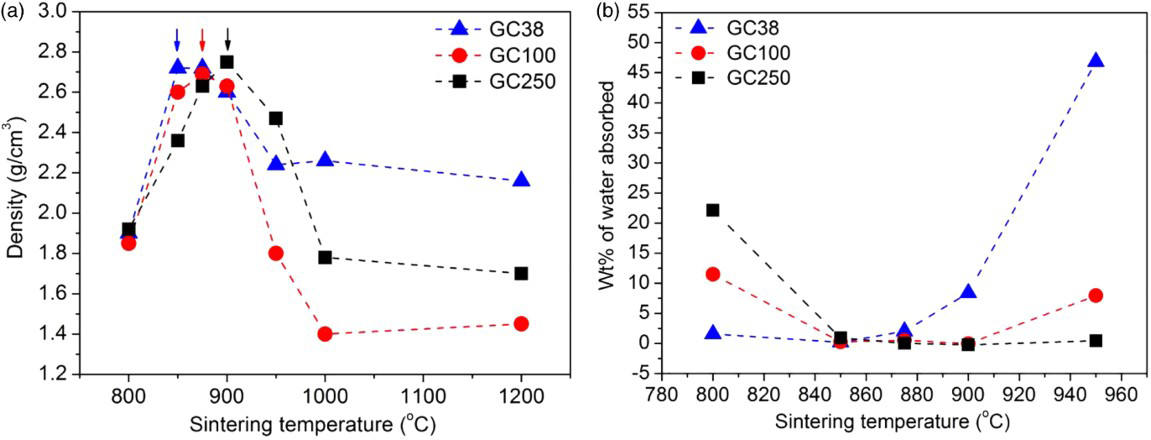
Comparison of influence of sintering temperature on
In good agreement with the classical theories of viscous flow sintering,
13
the densification was found to be more effective with fine particles ( < 38 μm) due to their higher specific surface energy; in fact, compacts made from coarser particles ( < 100 and < 250 μm) reached a density of ∼2.7 g cm− 3 by a further increase of sintering temperature up to 875 and 900°C. Alternatively, at a given temperature of 850oC, an increase in the annealing time to 45 and 60 min was necessary for GC100 and GC250 samples to reach maximum density of 2.7 g cm− 3, as demonstrated in Fig. 2. The time necessary to achieve similar densities can vary depending on the particle size distribution, extent of viscous flow at the given temperature and crystallisation ability of the material. In GC38 powder compacts, due to higher surface area, maximum densification occurred within 15 min of annealing time at 850oC, and the density remained constant up to 120 min. A similar effect was observed by Pascual
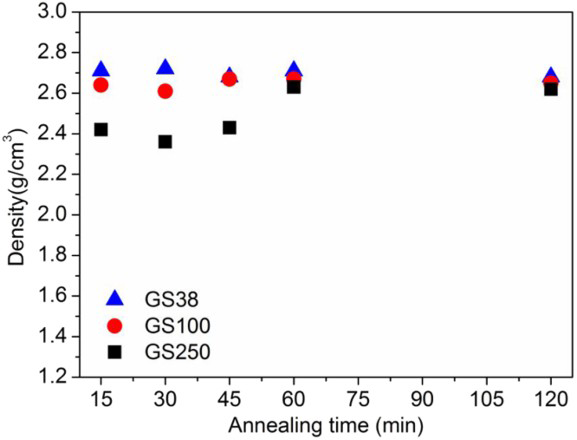
Density values of GC38, GC100 and GC250 samples sintered at 850°C for different holding times
The enhancement of densification, passing from 800 to 850–900°C, led to a remarkable decrease of WA, as illustrated by Fig. 1b. In particular, the densest samples had a WA < 1%.
Remarkably, as shown in Fig. 1a, treatments at temperatures above 900°C resulted in samples with a much lower density, especially from powders of intermediate size (GC100 samples). The decrease of density corresponded to a dramatic increase of WA (up to 50%), as illustrated in Fig. 1b. This phenomenon was attributed to the formation of a multitude of microsized pores, 15 particularly for GC38 samples, as illustrated in Fig. 3. In other words, while the densest sample exhibited only a system of isolated pores (Fig. 3a), the direct heating above 900°C resulted in the development of glass based foams (Fig. 3b) despite the absence of additives that could generate gasses by decomposition/oxidation reactions in the pyroplastic mass of softened glass particles undergoing sintering (glass based foams are well known to be developed by the addition of C, SiC or carbonates) 8 .

Optical microscopy images of GC38 compacts sintered at
Figure 4 reports quantitative data on pore volume and pore size obtained by analysing SEM images. Figure 4a shows that a pore volume of ∼5% was observed in sample GC38 at sintering temperatures of 850 and 875°C; this value rose to ∼65 vol.-% at 950°C. After sintering at 950°C, ∼50 vol.-% porosity was observed in sample GC100, but only 10 vol.-% porosity was measured in sample GC250. In Fig. 4b, the pore size is presented for samples processed at different temperatures. An average minimum pore size was recorded for the different particle size samples at their maximum densities (shown in Fig. 1a), and an increasing pore size was also observed outside this range. Thus, glass ceramics processed at 950°C are interesting for applications requiring highly porous materials but low pore size.
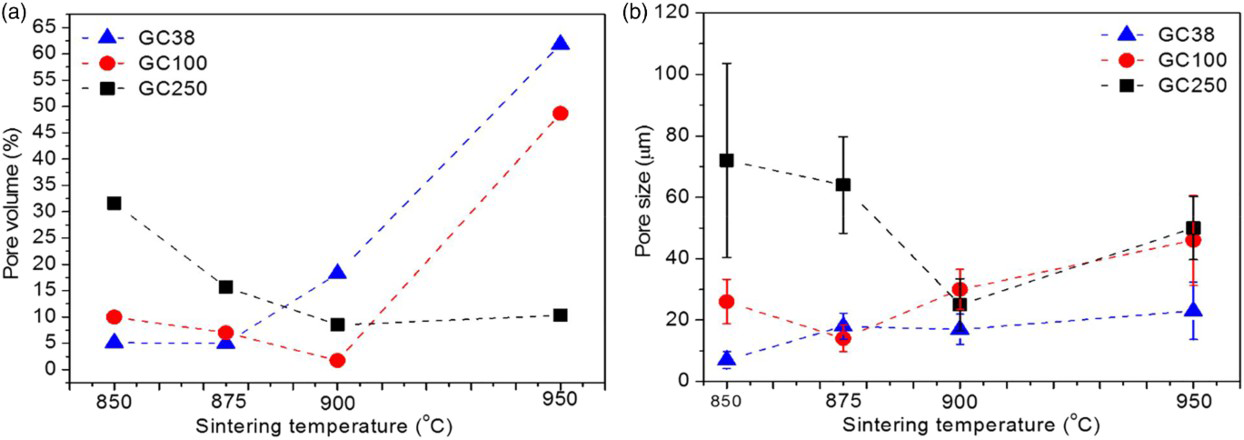
Influence of processing temperature on
The formation of micron sized pores presented in Figs. 3 and 4 was accompanied by other microstructural changes, inferred from X-ray diffraction analysis. Figure 5a shows that the densest samples, fired below 950°C, had limited crystallinity. The small peaks are consistent with the main diffraction lines of diopside, that is, Ca–Mg silicate (CaMgSi2O6, PDF#71-1496, peaks labelled with “A”), and gehlenite, a Ca alumino-silicate (Ca2Al2SiO7, PDF#74-1607, peaks labelled with “B”) belonging to the family of melilites. The sample from finest powders, GC38, appears the least crystalline, but this is attributable only to the above discussed promotion of viscous flow sintering: an enhanced densification removed many free glass surfaces, well known to catalyse the precipitation of silicates and alumina-silicates (by a surface nucleation mechanism). 16 This explanation is confirmed in Fig. 5b, illustrating a sensible increase of crystallisation degree with increasing sintering temperature; the almost flat background of the pattern for GC38 sintered at 950°C is an evidence of limited residual amorphous phase.
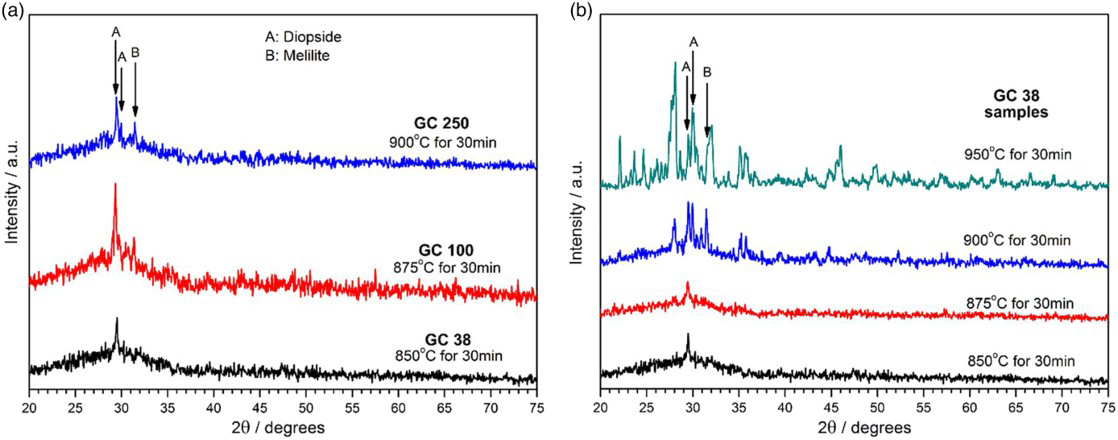
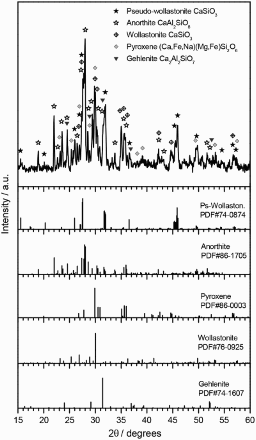
Detailed X-ray diffraction of GC38 sample sintered at 950°C
In GC38 sintered at 950°C, the two phases detected in Fig. 5a coexist with other more prominent crystalline phases, as shown by the upper pattern of Fig. 5b and as presented in more detail in Fig. 6b. The strongest peaks are attributed to pseudowollastonite (CaSiO3, PDF#74-0874) and anorthite (Ca2Al2Si2O8, PDF#86-1705). Minor peaks are associated with pyroxene ((Ca0.75Na0.249Fe0.018)(Mg0.937Fe0.063)Si2O6, PDF#86-0003), wollastonite (a further variant CaSiO3, actually known as “para-wollastonite”, PDF#76-0925) and gehlenite (as previously found). The effective presence of pure diopside is unlikely, considering that CaMgSi2O6 is only a variant of the complex series of solid solutions known as pyroxenes; the pyroxene phase reported in Fig. 6 could be seen as the result of the incorporation of iron (and sodium) ions in the diopside structure.
The correlation between crystallisation and porosity in glass ceramics has been the object of several investigations.17,18,19 It must be pointed out that the observed porosity evolution in the present samples could be due to the superposition of different effects, as outlined in the next paragraphs.
(i) Trapping of internal porosity – High heating rates generally imply a faster heating at the surface of samples. Such surface can form a non-porous viscous layer blocking the release of gases from the bulk of the material, creating (close) porosity in the interior of the sample. (ii) Crystallisation induced porosity – As discussed by Karamanov and Pelino,
17
, when a glass undergoes devitrification, the local reduction of volume associated with the formation of crystals creates voids. These voids are not compensated by the flow of the residual amorphous phase due to the increase of apparent viscosity (a glass embedding rigid, crystal inclusion is less prone to flow than the parent glass, without inclusions). The effect obviously depends on the composition, that is, on the specific volume changes of different phases: the same authors calculated a crystallisation induced pore volume of 4–4.5% for wollastonite and 10–12% for diopside (both phases effectively developed in our system). Karamanov and Pelino
17
and Bernardo (iii) Evolution of gasses from modification of the oxidation state – Glasses comprising iron oxide may present a “bloating” effect, due to oxygen release, in turn caused by the reduction of Fe3+ (ferric) into Fe2+ (ferrous) ions (the overall reaction is 2 Fe2O3 → 4 FeO + O2); this effect is the basis of recently developed highly porous glass ceramics8,19. The PVW glass contains more than 4.5 wt-% Fe2O311; ferrous ions were effectively found in the pyroxene phase, detected for the (highly porous) sample sintered at 950°C.
These three mechanisms could have individually or collectively been activated by the addition of BSG glass, leading to the formation of a porous glass ceramic. In fact, EDX mapping (not shown) of the backscattered SEM images in Fig. 7a and b revealed that pores concentrated in the amorphous zones corresponding to former BSG glass particles.
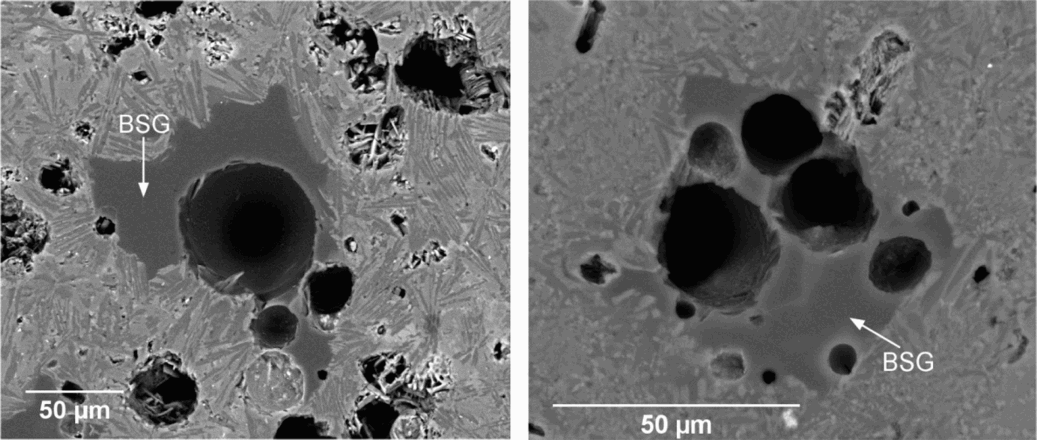
Backscattered SEM images of glass ceramic microstructures showing rounded pores in
The addition of BSG is thought to favour all the proposed mechanisms of pore formation, and the presence of alumina in the composition could have made BSG less sensitive to crystallisation 20 . The presence of BSG's low viscosity phase along with less viscous PVW could i) promote the sealing of internal porosity by enhanced viscous flow, ii) promote ionic interdiffusion and favour anisotropic crystal growth (the effect is well known for mullite 21 ; a confirmation of the effect in our system may be found in the fibrous crystals around the pores observed in Fig. 7a) and iii) favour the bloating, considering the key role of viscosity in the development of porosity.13,17,20
The exact balance between the different mechanisms responsible for pore formation should be clarified performing additional experiments, involving glasses of different composition. However, the present results are of relevance for the development of technical (porous) glass ceramics from this particular combination of wastes. For example, the possibility of sintering PVW+10 wt-% BSG within 30 min to form a porous, highly crystalline, glass ceramic is an attractive approach for developing materials for thermal insulation applications in the construction industry. Pore size and volume can be engineered by changing particle size and sintering temperature. This knowledge is relevant to industry to realistically consider waste derived glass ceramics for developing construction application oriented materials for thermal and sound insulation in modern housing.
Conclusions
Mixtures of PVW+10 wt-% BSG of different particle sizes were successfully sintered and crystallised to form glass ceramics by direct heating and normal cooling in air to RT. The glass ceramics made from particles of size < 38 μm showed densification within 15 min at 850°C. Increasing the sintering temperature led to an increase in the volume of micron sized porosity in all compacts. The balance between densification by viscous flow and crystallisation, the formation of voids induced by crystallisation and the reduction of iron oxide were suggested to be responsible for the drastic porosity increase in sintered compacts. Glass ceramics made from particles < 38 μm developed 65 vol.-% micron sized porosity with a pore size of ∼20 μm when sintered at 950°C. Potential industrial applications of the porous glass ceramics, for example, as building materials, should be facilitated by the cost effective fabrication process.
Footnotes
Acknowledgements
The authors would like to thank Dr. D. Deegan from Tetronics Ltd., for PVW and discussions during preparation of this paper. The support of the European Community's Seventh Framework Programme (FP7) through a Marie-Curie Research Training Network (“GlaCERCo” PITN-GA-2010-264526) is gratefully acknowledged.
