Abstract
With appropriate electropulsing parameters, the microstructure of steels can be manipulated towards a state that possesses favourable physical, mechanical and chemical properties. This work demonstrates the application of designed electropulsing to pearlitic steel, transformation induced plasticity steel, austenite stainless steel and clean steel in order to generate novel microstructures that are difficult to achieve by other conventional thermomechanical processes. The principles for the design of electropulsing processing have been discussed. The method is particularly suitable for fabrication of the advanced multiphase and multicomponent alloys.
Introduction
When the electric potential differences are imposed on a metallic material, the free electrons in metal drift from low to high electrical potentials and are in the mean time scattered by the atomic cores. The scattering causes unsymmetrical distribution of electrons around a dislocation, which leads to a net electromagnetic force. The force promotes dislocation migration and atom hopping. This kinetic effect of electropulse on metals is called electromigration.1,2 A thermodynamic calculation reveals that the electropulse promotes a structural evolution in materials towards the state with lower electrical resistance. 3 The latter implies less scattering from atomic cores to the drifting free electrons. 4 There are three considerations to arrange the microstructure of a material so as to minimise its electrical resistance. Microscopically, atoms can be arranged to a structure possessing minimum electrical resistance. For example, a crystal structure with long range order has less electrical resistance than that of the amorphous structure with only short range order. The experimental observations of electropulse promoted alloy solidification5,6 and electropulse enhanced metal glass crystallisation7,8 are within this theoretical frame. Mesoscopically, the orientation of crystals in metals can be arranged toward a certain direction so that the electrical resistance along the electrical current direction is at minimum. For example, a face centred cubic crystal with lower electrical resistance at <100> than that at <111> can be arranged so that current flows along <100> direction. The observed electropulse induced texture formation is within this consideration. 9 The recent reported electric current induced epitaxial growth of Cu–Sn crystals has enhanced this scientific understanding. 10 The fundamental reason behind this is the anisotropic electrical properties of the crystalline metals. Macroscopically, the phases and components in the materials can be arranged to a configuration so that the total electrical resistance of the material achieves minimum. For example, the conductive phases can be configured to a state with maximum percolation effect. Due to the configuration dependent electrical property of a material, electropulsing can, inversely, impose an effect on the configuration of phases and components. This is most appealing because the conventional thermomechanical process is not able to handle this type of requests. For a multiphase and multicomponent material, e.g. steel, equilibrium calculation by means of chemical free energy minimisation tells the amount and chemical constitution of equilibrium phases. The configuration of the phases in the condition of that the total interface energy in the materials remains constant does not affect the equilibrium. However, this affects the electrical resistance of the materials and hence the electropulse associated system free energy. Manipulation of the configuration of phases and components enables to achieve optimum physical, mechanical and chemical properties of the alloys. On the other hand, manipulation of an object on its motion and location in a bulk material is an attractive topic due to its importance in biomedical applications. 11
Steels are multiphase and multicomponent alloys with a couple of solid–solid phase transitions. The phase transformations are sensitive to the mobility of chemical constitutions. The electrical properties between the different phases in steels are different. Electropulsing can, therefore, affect the configurations of phases and components in steels. The task of the present work was to use the electropulsing to synthesis novel microstructures. The overall aim of the research was to validate the capability of using electropulsing to tailor the microstructure of steels.
Experiments and results
Four types of steel samples are received from various steelmaking companies. Their names and chemical constitutions are listed in Table 1. Those steels are to be electropulsed at specific thermomechanical states with different electropulsing parameters in order to achieve the designed microstructures and properties.
Chemical composition of steel sample

The pearlitic steel wires have been cold drawn to 80 reduction before electropulsing. The microstructure shows typical elongated cementite and ferrite plates in alternating arrangement, as presented in Fig. 1a. There are two reasons for preparing the sample suing cold work:

a before electropulsing; b after electropulsing
the kinetic consideration of the effect of electropulsing on metals reveals that the electromigration is mainly due to the unsymmetrical distribution of the free electrons around the dislocations. The cold work increases the dislocation density and hence will be beneficial to the electropulsing effect
the thermodynamic consideration of the effect of electropulsing indicates that the electropulsing intends to reduce the system electrical resistance. The cold work may increase the electrical conductivity of the materials so that the electropulse associated thermodynamic driving can be enhanced. It should be emphasised that the relation between materials electrical resistance and strain is not monotonic. Careful design of the strain is required. The preparation for the sample enables the microstructure transformation taking place in minimum time duration.
An electropulse with peak current density 9×109 A m–2 and pulse duration 150 μs has been implemented on the cold worked pearlitic steel sample at ambient temperature. The pulse is in a damped sinusoidal waveform. The scanning electron microscope characterises the microstructure of the electropulsed steel, as illustrated in Fig. 1b. The cementite plates are turned into nanoscale particles throughout the electropulsed area. The structural evolution of pearlitic steel using similar electropulsing parameters has been reported in details previously.12,13 The experimental measurement shows that the electrical resistance is dropped into 76 of its original value before electropulsing. The strength of the electropulsed steel is slightly higher than that before electropulse. Considering that the residual stress in cold work has been removed by electropulsing, the actual change of the mechanical properties of the electropulsed steel is significant. Further cold work could be implemented to the electropulsed steel to achieve its optimum strength.
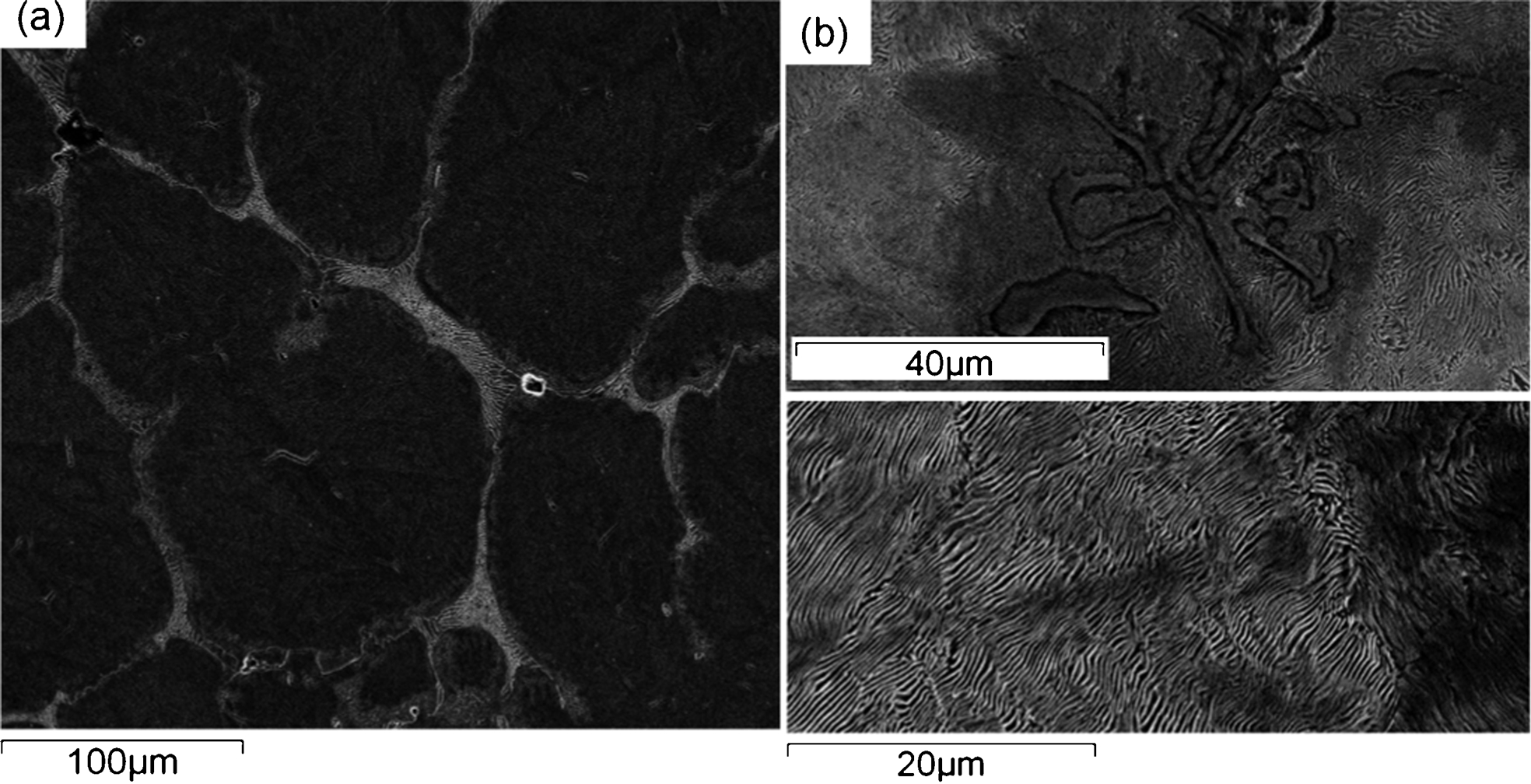
Dislocations exist in steels even without any cold working. The low dislocation density in steels, however, affects the structural evolution rate. The phase transition after hot rolling of steels causes residual stress. This can be used as part of thermodynamic driving force. The hot rolled transformation induced plasticity (TRIP) steel sample has been subjected to electropulsing treatment at ambient temperature. The microstructure before electropulsing is demonstrated in Fig. 2a. It is statistically anisotropic and without noticeable preferred orientation for any phases inside the steel. After the 30 000 electropulses of current density 2×107 A m–2, pulse width 20 μs and pulse frequency 1 Hz, the microstructure changes into that demonstrated in Fig. 2b. The stripes are in line with the current flow direction. Such patters have not been noticed after 20 and 100 pulses. It shows the accumulative effect of electropulsing on microstructure evolution. Electropulsing rearranged the microstructure of steel.

a before electropulsing; b after electropulsing
To prove that the observed phenomenon cannot be explained fully by electromigration mechanism, 316L stainless steel has been testified using the electropulsing treatment. The steel forms secondary phases at an annealing temperature <1273 K. The formation of the secondary phases is detrimental to the stainless steel because it causes heterogeneous distribution of Cr and Mo. Electropulsing treatment, if its effects on alloys are mainly to increases the mobility of microscale species, will accelerate the formation of the secondary phase. In this group of experiments, two samples are annealed simultaneously at 1208 K for 1 h. One sample was connected to electropulsing but another without. The electropulsing parameters are: current density 6×107 A m–2, pulse width 20 μs and pulse frequency 1 Hz. The images obtained by the scanning electron microscope are presented in Fig. 3; Fig. 3a is from the sample without electropulsing and Fig. 3b is from the sample with electropulsing treatment. The electropulsing demoted the formation of the secondary phase particles. Obviously, the formation of small crystal particles causes the appearance of interface that enhances the scattering to free electrons. The thermodynamic effect of electropulsing on metals prevents the secondary phase formation at high annealing temperature, which is positive to the corrosion resistance of the stainless steel.

a without electropulsing during annealing; b with electropulsing during annealing
In principle, as introduced earlier in this work, the configuration of phases can affect the thermodynamic state of electric current carrying materials. This can be used to process the clean steel. The received clean steel samples are molten and then solidified. In one case electropulsing did not allow to pass through the melt but in another case electropulsing was introduced in the process. Other environments are kept as similar as possible. The electropulsing parameters for this treatment are: current density 1·2×105 A m–2, pulse width 20 μs, pulse frequency 1 Hz and total treatment time 20 min. The optical microscope indicates that the ingot without electropulsing treatment contains randomly distributed MnS particle in matrix but the ingot after electropulsing treatment has MnS particles segregated to the surface, as demonstrated in Fig. 4. The MnS inclusions are rarely seen in the central part of the ingot. MnS particles possess much higher electrical resistance than that of the steel matrix and are rearranged to the surface of the ingot by the passing electropulses. A detail report of this experimental procedure can be found elsewhere. 14 The aim of the present work was to integrate all experiments to figure out the mechanism of electropulsing treatment.
Distribution of MnS inclusion in steel a without electropulsing and b with electropulsing treatment; upper graph is from surface area and lower graph is from middle of ingot
Discussion
Consider a material consisting of two phases: one has much high electrical resistivity than the other. The total electrical resistance of the material cannot be calculated simply by the rule of mixture for two phase composites. The geometric morphology of each phase and the spatial configuration of both phases affect the total electrical property of the material significantly. This can be demonstrated schematically in Fig. 5. The dark phase in Fig. 5 is supposed to have electrical resistivity ρd. The electrical resistivity of the white matrix in Fig. 5 is ρw, where ρd>ρw. The volume fractions of both phases are the same in Fig. 5a–d. In Fig. 5a, the total electrical resistance of the material is approximated as Ra = ρdL/S, where L is the length of the sample and S is its cross-section area. This gives a value close to the electrical resistance of the dark phase. There is no electrical percolation formed. In Fig. 5b, the total electrical resistance can be estimated by Rb = ρwL/Sgap, where Sgap is the area of gap between the fragmented particles, as indicated in Fig. 5b. Sgap in Fig. 5c is much bigger and in Fig. 5d is the largest among the listed four cases. Therefore, one has Ra>Rb>Rc>Rd. According to the thermodynamic prediction of electropulse promoted microstructural transformation, the structure will evolve from that illustrated in Fig. 5a to c and eventually towards Fig. 5d.
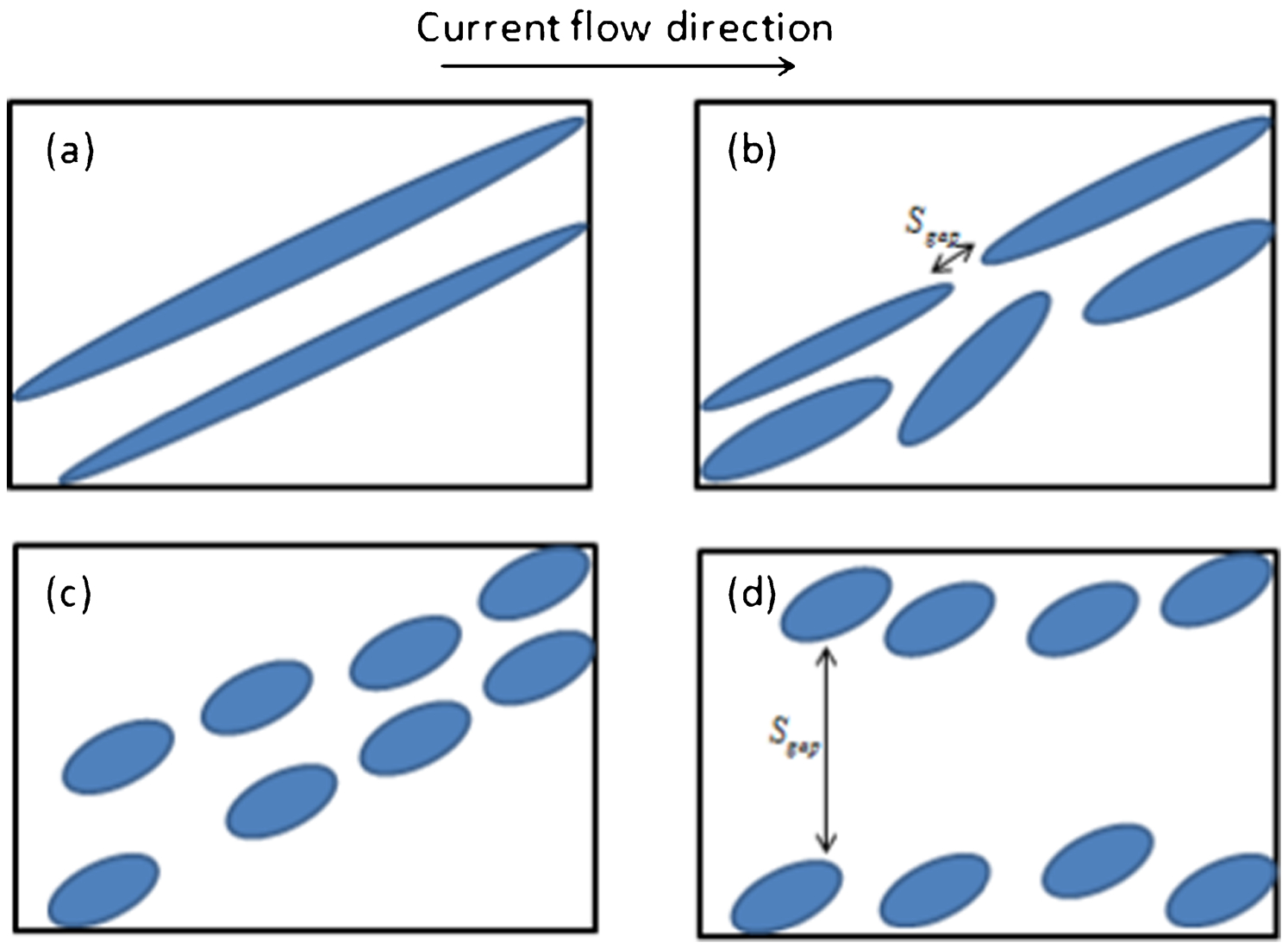
Schematic diagram of two-phase composites with different phase configurations
The structural evolution of pearlitic steel demonstrated in Fig. 1 can be immediately understood by the above explanations. Cementite phase has much large electrical resistivity than that of ferrite phase due to the high carbon and low iron composition in cementite phase. The lamellar interconnected cementite phase makes the electrical resistance of the pearlitic steel large. Fragmentation of the lamellar structure and formation of the disconnected spherical cementite particle allow the formation of more percolation routines along the ferrite phase. Therefore, the cementite plates try to break into small pieces to reduce the electric current associated system free energy. The cold work provides sufficient high stored energy to enable this structural transformation and the high dislocation density enables the high electromigration enhanced carbon diffusivity to accomplish the kinetic requirement. The observed structural transformation hence occurred at ambient temperature. The nanostructured steel is fabricated. The experimental measured electrical resistance reduction proves the theoretical analysis. The observation of that Fig. 1b is similar to Fig. 5c rather than to Fig. 5d is due to the solid state phase transition and short duration. In Fig. 4, the electropulsing treatment was in liquid state and was lasted in 20 min, a structure similar to Fig. 5d is observed. The cleanness of the steel can be improved if the surface layers containing the inclusions are removed.
If the dark phase demonstrated in Fig. 5a can be aligned along the electric current flow direction, the total materials electrical resistance is low. This is the case observed in Fig. 2 for TRIP steel. The pearlitic laminar rearranged in a structure parallel to the electric current flow direction. This helps the ferrite phase to form percolation channel with minimum flow path so that to reduce the total electrical resistance. The difference between Figs. 1 and 2 is due to the electropulsing parameters. A close examination of Fig. 2 reveals many fragmented cementite particles within the pearlitic phase, particular when the orientation of cementite is not parallel to the flow direction.
Figure 3 reveals a complete different case. The segregations of the Cr and Mo are reduced under electropulsing treatment. This effect, however, needs to be analysed carefully. The free energy change due to the electropulsing is Δwe>0. The chemical free energy difference in the secondary phase formation is Δwc<0. The change of total interface energy in the secondary phase formation is Δwi>0. The electropulsing treatment will certainly prevent the formation of secondary phase when Δwe+Δwc +Δwi≥0. In the case when Δwe+Δwc+Δwi<0, the effect of electromigration may steel accelerate the formation secondary phases due to the electropulsing enhanced solute diffusivity. A quantitative calculation is able to demonstrate this. The calculation requires the composition dependent local electrical resistivity, and will be presented in a separate work.
Summary
A series of experiments have been carried out to investigate the effect of electropulse on phase and component reconfigurations in the multiphase and multicomponent steels. The electropulsing enhanced-mobility and electropulsing-induced structural evolution have been used to produce novel microstructures. The nanostructured pearlitic steel possesses high electrical conductivity and high strength. The electropulsing prohibited secondary phase formation can help to improve the high temperature corrosion resistance of stainless steel. The observed stripe patterns in TRIP steel indicate the possibility to control the metallographic texture in materials processing. The electropulsing induced inclusion segregation at surface reveals a new way to improve the cleanliness of the steel.
Footnotes
Acknowledgements
The authors are grateful to TATA, Royal Academy of Engineering, EPSRC, DSTL and POSCO for financial support of this research.
