Abstract
After decades of debate on the mechanism for the formation of bainite, it is accepted that bainite grows via a displacive mechanism, i.e. as plate shaped transformation products exhibiting an invariant plane strain surface relief effect. But there is still much discussion on the diffusion or diffusionless nature of bainite. The purpose of this atom probe tomography study was to track the atom distributions during the bainite reaction in steels with different carbon and silicon contents. The steels were transformed over a wide range of temperatures (200–525°C) to elucidate the role of reaction rate and diffusion in the formation of bainite with and without cementite precipitation.
Introduction
Today, it is accepted that bainite grows via a displacive mechanism, 1 i.e. as lath shaped transformation products exhibiting an invariant plane strain surface relief effect. However, two different explanations about the growth nature of bainitic ferrite in steels are still discussed: the diffusionless hypothesis, which states that bainitic ferrite grows without any diffusion of carbon, and carbon supersaturation is subsequently relieved by partitioning to austenite, and/or through carbide precipitation; and the diffusional hypothesis, which states that bainitic ferrite growth is controlled by carbon diffusion, and not distinctly different in character from Widmanstätten ferrite, with carbide precipitation at the austenite/ferrite boundaries.
The criterion for distinguishing between the two bainite transformation theories is whether the newly formed bainitic ferrite has the paraequilibrium (PE) carbon content or if it is supersaturated in carbon. But it is a challenge to measure experimentally the carbon concentration of newly formed bainitic ferrite because the time taken for any carbon to diffuse into austenite can be extremely short.
Traditionally, the diffusionless nature of bainitic ferrite of bainitic ferrite growth has been confirmed indirectly in Si containing steels by determining the carbon content in the retained austenite at the end of the bainite reaction using X-ray diffraction (XRD) analysis and early atom probe analysis.2,3 Carbon concentrations of the residual austenite confirming the 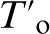 value,4,5 where austenite and ferrite of the same chemical composition have the same free energy, indicate that bainite initially forms having a full supersaturation of carbon. The partitioning of carbon into the residual austenite occurs immediately after formation.
value,4,5 where austenite and ferrite of the same chemical composition have the same free energy, indicate that bainite initially forms having a full supersaturation of carbon. The partitioning of carbon into the residual austenite occurs immediately after formation.
The purpose of this atom probe tomography (APT) study was to track the atom distributions during the bainite reaction to elucidate the role of carbon diffusion in the formation of bainite. The results show atomic experimental evidence on the 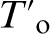 limit, the carbon supersaturation of bainitic ferrite and secondary processes, such as plastic accommodation and carbide precipitation, which are not essential to the mechanism of formation of bainitic ferrite, except where any precipitation of carbon trapped at dislocations influences the reaction rate by removing carbon from the supersaturated ferrite.
limit, the carbon supersaturation of bainitic ferrite and secondary processes, such as plastic accommodation and carbide precipitation, which are not essential to the mechanism of formation of bainitic ferrite, except where any precipitation of carbon trapped at dislocations influences the reaction rate by removing carbon from the supersaturated ferrite.
Approaching 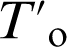 curve during bainite formation
curve during bainite formation
High carbon high silicon bainitic steels are known to exhibit extremely sluggish transformation to bainite at low temperatures,
6
which can be advantageous for monitoring carbon distribution during bainitic ferrite growth. The evolution of the carbon content in bainitic ferrite and austenite, as determined from XRD analysis and APT, during transformation at 200°C in Fe–4·3C–2·8Si–1·8Mn–1·3Cr (at-) steel, is shown in Fig. 1. The XRD data in Fig. 1a clearly indicate that, as the formation of bainitic ferrite progresses, the austenite is gradually enriched in carbon from the overall carbon content to that given by the 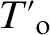 value and well before the PE phase boundary value (
value and well before the PE phase boundary value (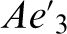 ).XRD results in Fig. 1a are an indirect manifestation of the diffusionless nature of bainite transformation so that the original bainitic ferrite retains much of the carbon content of the parent austenite.
).XRD results in Fig. 1a are an indirect manifestation of the diffusionless nature of bainite transformation so that the original bainitic ferrite retains much of the carbon content of the parent austenite.
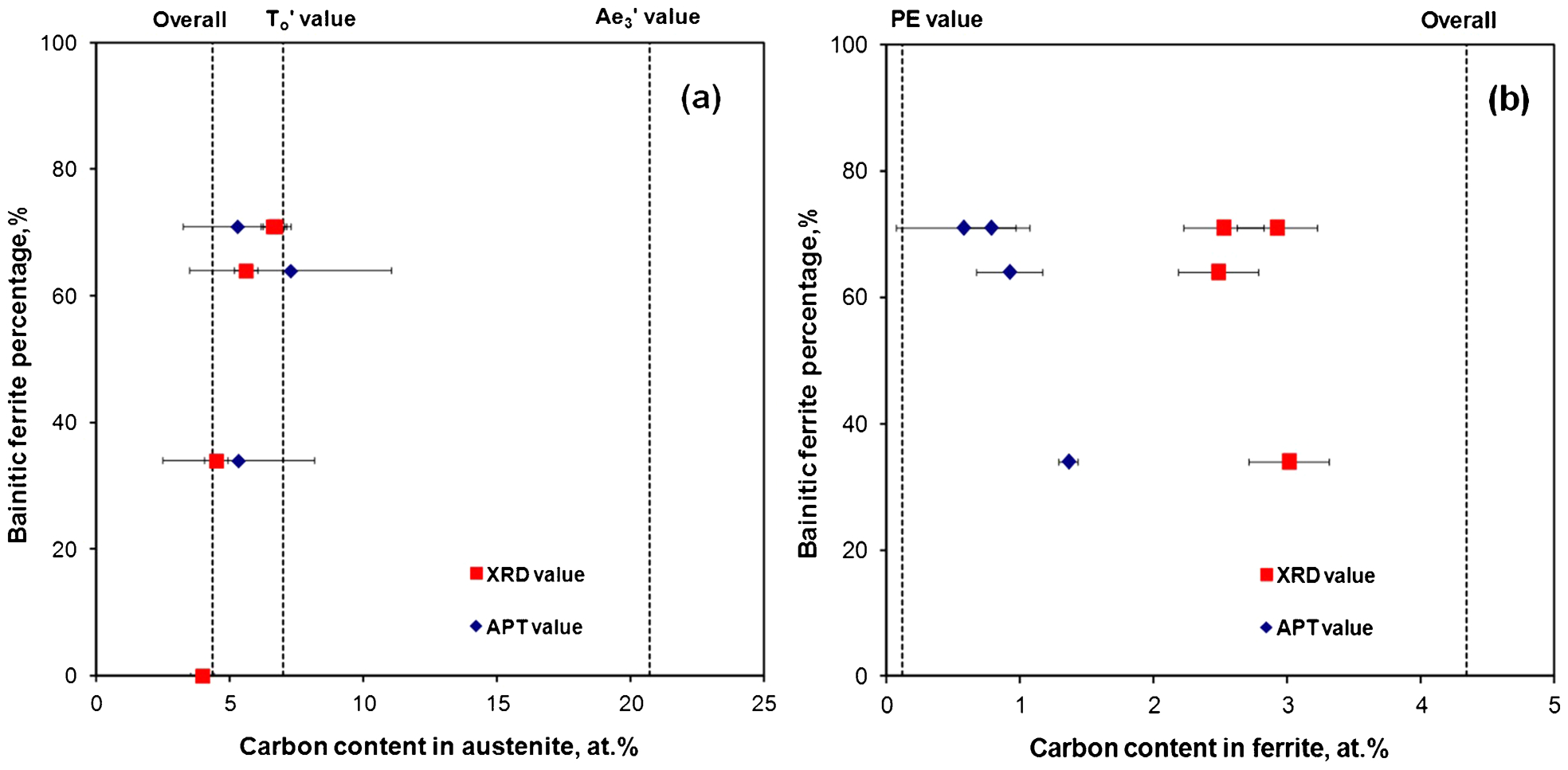
Carbon contents in a austenite and b bainitic ferrite determined by XRD analysis and APT in Fe–4·3C–2·8Si–1·8Mn–1·3Cr (at.-) steel as bainite transformation progress at 200°C
The APT values are estimated from proximity histograms across the interface between austenite and ferrite regions with a random solid solution. An example of three-dimensional carbon atom map showing austenite and ferrite regions is illustrated in Fig. 2.
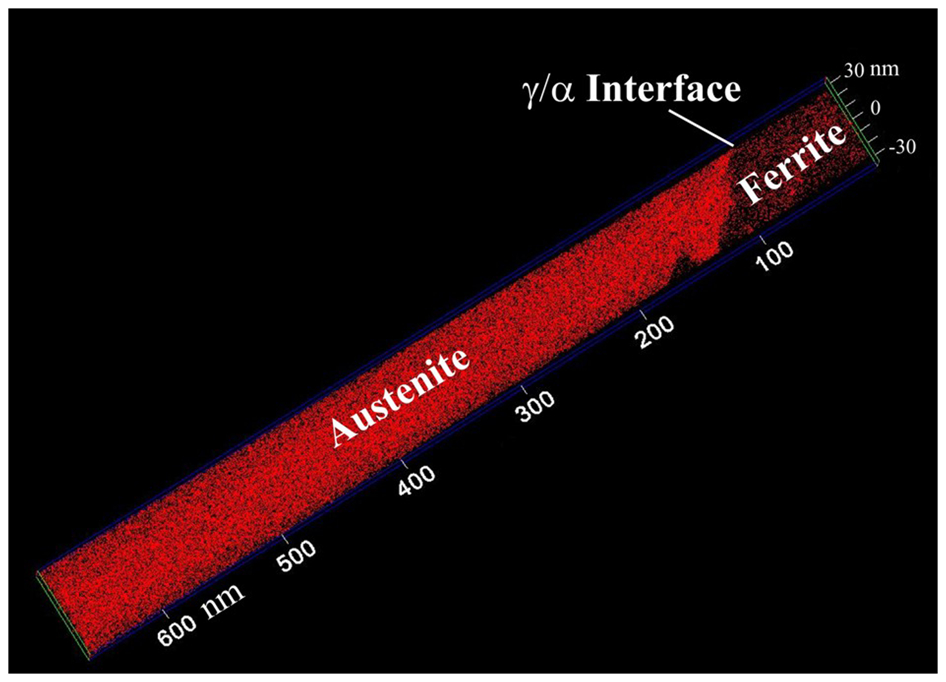
Carbon atom map showing ferrite/austenite interface: α is bainitic ferrite and γ is austenite
The trend of the APT values as a function of time in Fig. 1a may not be so obvious. This is a consequence of variations in the carbon distributions in different regions of austenite.
7
The austenite films entrapped between neighbouring subunits of bainitic ferrite have a higher carbon content than the blocks of residual austenite located between the sheaves of bainite. Moreover, nanosized austenite films can accumulate higher amounts of carbon during bainite formation.
8
Thus, Fig. 3 indicates the thickness in nanometres of the APT analysed austenite regions. Nanoscale austenite films with thicknesses in the range of 5–20 nm show APT values ranging from 9 to 14 at-, higher than the corresponding XRD and 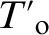 (7·0 at-) values and less than the
(7·0 at-) values and less than the 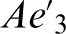 value (20·7 at-). Conversely, submicron austenite regions exhibit APT estimated carbon contents ranging from 4·77 to 6 at-, which is lower than the corresponding XRD and
value (20·7 at-). Conversely, submicron austenite regions exhibit APT estimated carbon contents ranging from 4·77 to 6 at-, which is lower than the corresponding XRD and 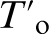 (7·0 at-) values.
(7·0 at-) values.
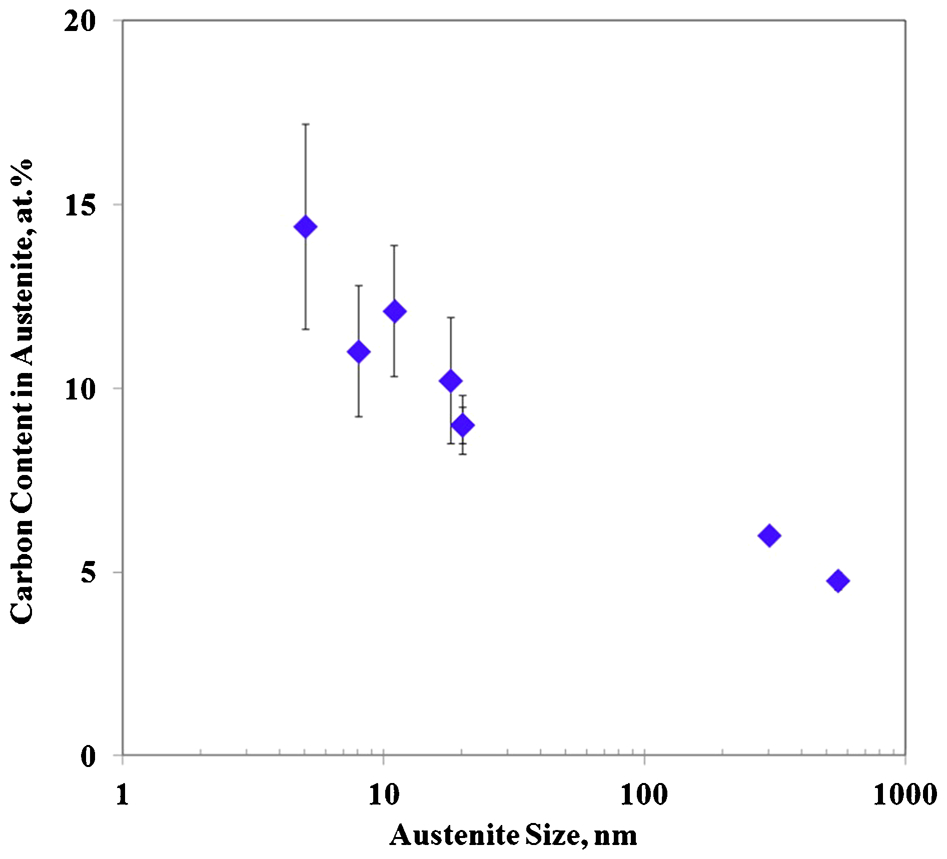
Carbon content in austenite measured by APT as function of thickness in nanometres of analysed austenite regions in Fe–4·3C–2·8Si–1·8Mn–1·3Cr (at-) steel isothermally transformed at 200°C
Carbon supersaturation of bainitic ferrite in bainite
The evolution of the carbon content in bainitic ferrite as bainite transformation progresses at 200°C in the same high carbon high silicon steel determined by XRD analysis and APT is shown in Fig. 1b. The APT estimates are from selected volumes of ferrite that did not contain any carbon enriched regions, such as dislocations and boundaries, whereas the higher XRD estimates are average estimates of larger volumes of bainitic ferrite that include such carbon enriched regions. APT values prove the presence of a high level of carbon in bainitic ferrite, which was well above that expected from PE with austenite (PE: 0·12 at-), and provide clear evidence of carbon supersaturation in bainitic ferrite at the early stage of transformation, confirming that the bainite transformation is essentially diffusionless in nature.
However, the lower reaction rate of bainite in comparison to martensite has been traditionally used to support the idea of a diffusional growth mechanism, as it seems natural to believe that trapping of carbon in the growing ferrite requires faster kinetics than those observed experimentally. In this sense, the carbon content of the bainitic ferrite was analysed by APT in Fe–XC–YSi–1·5Mn–0·5Cr (at-) steels with different carbon contents and silicon contents (1·3C–0·5Si, 1·3C–2·8Si and 4·3C–2·8Si in at-) that transforms over a wide range of temperatures (200–525°C) to elucidate the role of reaction rate and diffusion in the formation of bainite with and without cementite precipitation.
Examples of transmission electron microscopy images for the different morphologies of bainite identified in the studied steels are shown elsewhere. 9 The difference in carbide distribution, intralath and interlath respectively, allows identifying both upper and lower bainite morphologies in the studied steels, as summarised in Table 1. In addition, overall transformation kinetics data reported elsewhere10,11 of these bainitic steels are presented in Fig. 4a in terms of the start and finish reaction times. As expected, the bainite reaction is greatly retarded at lower transformation temperatures as carbon content is increased.
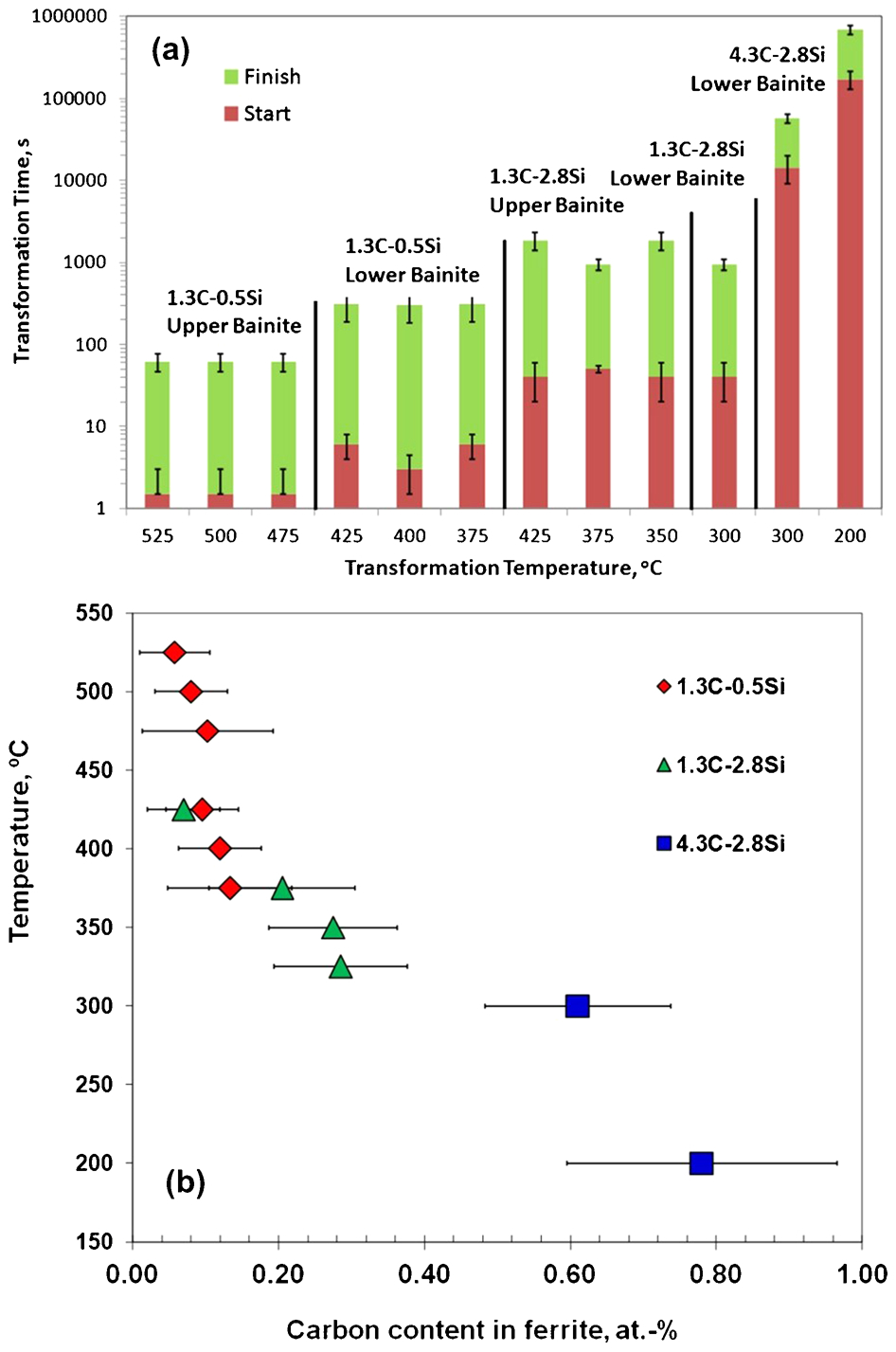
a start and finish bainite reaction times as function of transformation temperature and b carbon contents in bainitic ferrite determined by APT after bainite reaction at different temperatures in steels with approximated composition of Fe–XC–YSi–1·5Mn–0·5Cr (at-)
Carbide distribution and morphology observed after bainite transformation in steels with different carbon and silicon contents
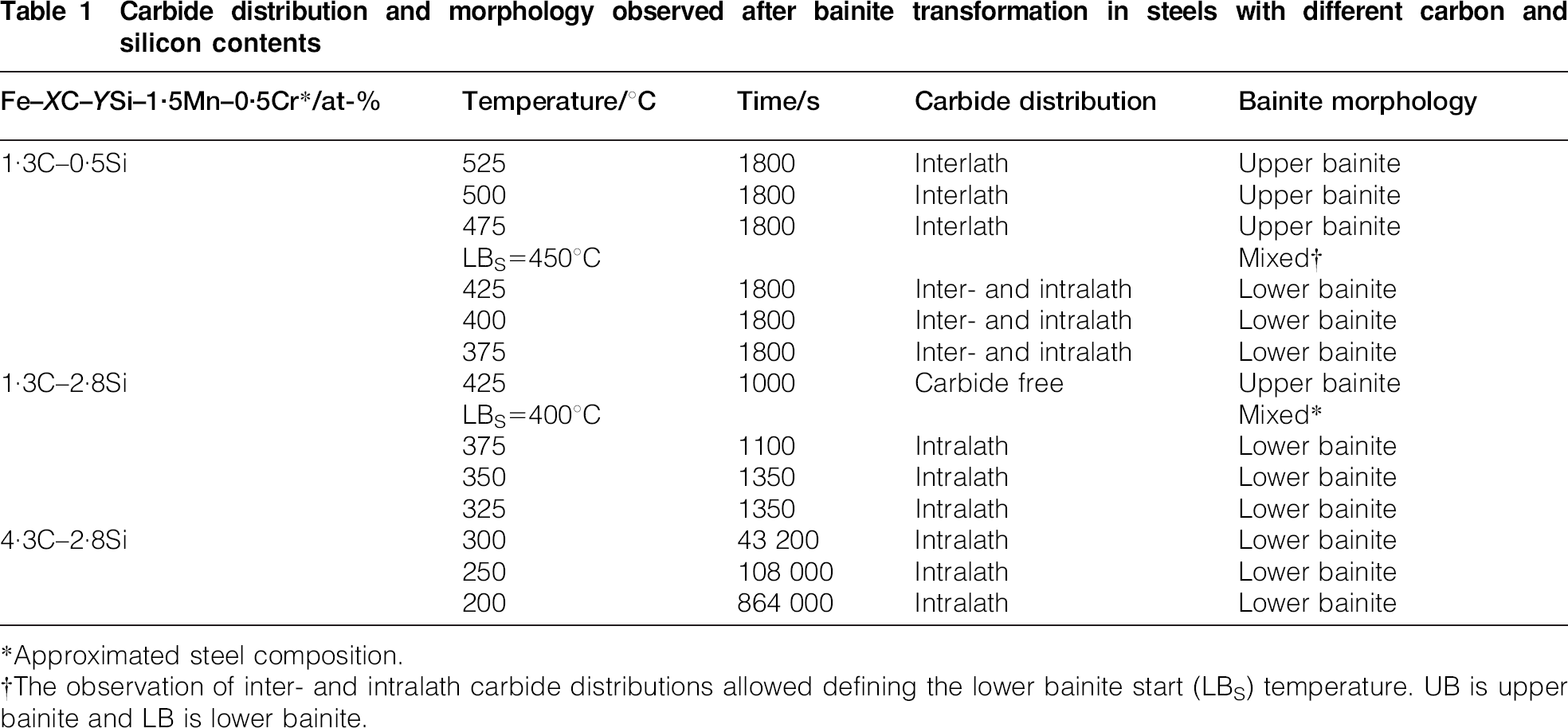
*Approximated steel composition.
†The observation of inter- and intralath carbide distributions allowed defining the lower bainite start (LBS) temperature. UB is upper bainite and LB is lower bainite.
The carbon content of the bainitic ferrite, as analysed by APT in the three steels after completion of transformation as a function of temperature, is shown in Fig. 4b. The results suggest that bainitic ferrite forms as a supersaturated solution of carbon, in spite of the fact that slow reaction rates had been determined at these temperatures. The explanation is that a sheaf of bainite consists of several subunits and the experimental reaction rate applies to the whole bainite constituent, whereas it is proposed that each subunit grows with a velocity high enough to trap the carbon of the parent austenite.12,13
What is also remarkable is that there is no essential difference in the observed results between steels that transform to upper or lower bainite. The examination of experimental data in Fig. 4b did not show any abrupt change of the carbon content in bainitic ferrite that could indicate a difference in bainite growth mechanism between high and low temperatures. This observation lends strong support for the opinion that the bainitic ferrite grows supersaturated with carbon independent of the transformation temperature and the overall reaction rate. As the transformation temperature is increased, carbon diffusion is enhanced, providing an opportunity for the decarburisation of the supersaturated ferrite soon after the growth event. The excess carbon may then partition into the residual austenite or precipitate in the ferrite in the form of carbides. Therefore, the observed carbon content in solid solution in bainitic ferrite comes from the competition between the rate at which carbon is partitioned from supersaturated ferrite into austenite and the rate with which carbides can precipitate from ferrite.
Effect of plastic accommodation defects on carbon partitioning into austenite
The shape deformation associated with a displacive transformation of austenite in steel can be described as an invariant plane strain with a relatively large shear component. Christian 14 demonstrated that when the shape strain is elastically accommodated, the aspect ratio of the plate adjusts so that the strain energy is equal to the driving force. In ideal circumstances, where the transformation interface remains sliding throughout, and where there is no friction opposing the motion of the interface, thermoelastic equilibrium occurs. 15
The thermoelastic equilibrium has been widely demonstrated for martensite, 16 but it has not been straightforward for bainite. One reason for this is that the bainite transformation occurs at higher temperatures than those of martensite, where the austenite is mechanically weaker. The shape deformation therefore causes plastic deformation, and the resulting debris from dislocations eventually blocks the transformation interface. 17
Carbon trapping at dislocations in the vicinity of the interface has been repetitively revealed in high carbon high silicon bainitic steels transformed at low temperature.18–20 The 6 at-C isoconcentration surface shown in Fig. 5 outlines the carbon enriched region around a dislocation and two austenite plates. It is evident from the proximity histogram across the dislocation/matrix interface in Fig. 5 that dislocations only trap the carbon atoms, as originally suggested by Kalish and Cohen. 21
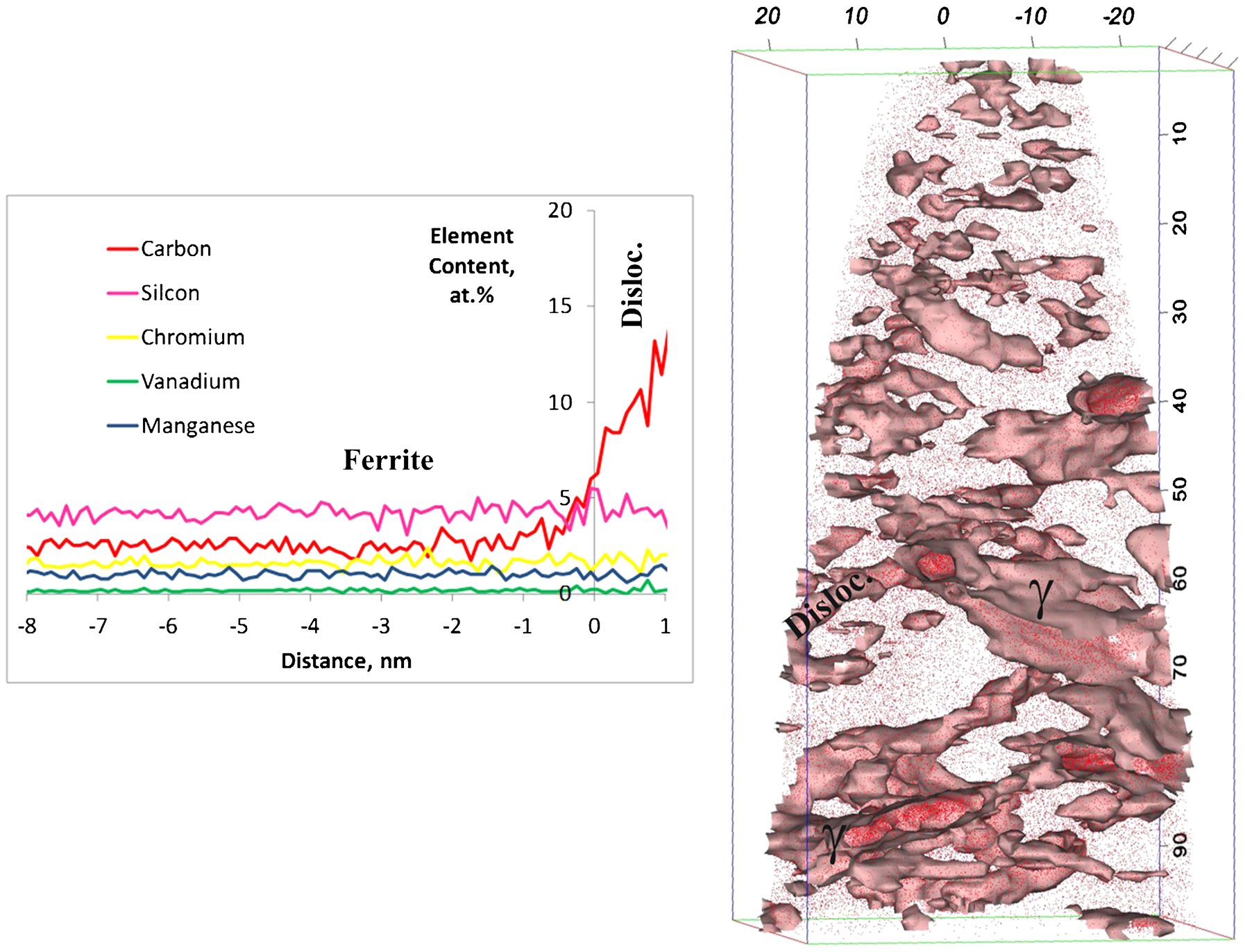
Carbon isoconcentration surfaces at 6 at-C and proximity histograms across dislocation in ferrite in vicinity of two austenite plates: γ is austenite
The average carbon levels of different Cottrell atmospheres detected by APT in the studied steels are listed in Table 2. The range (6–16 at-) for the amount of carbon trapped at dislocations is remarkable and has no apparent correlation with the overall carbon content and the transformation temperature. Pereloma et al. 22 found by APT that the extent of solute segregation to a dislocation depends on its position relative to other defects. In addition, Cochardt et al. 23 pointed out that the atmosphere around a screw dislocation can bind more than twice the number of carbon atoms associated with an edge atmosphere. Therefore, some significant variation in the carbon level at different dislocations or different segments of dislocations would be expected.
Carbon trapped at dislocations determined by APT after bainite transformation in steels with different carbon and silicon contents
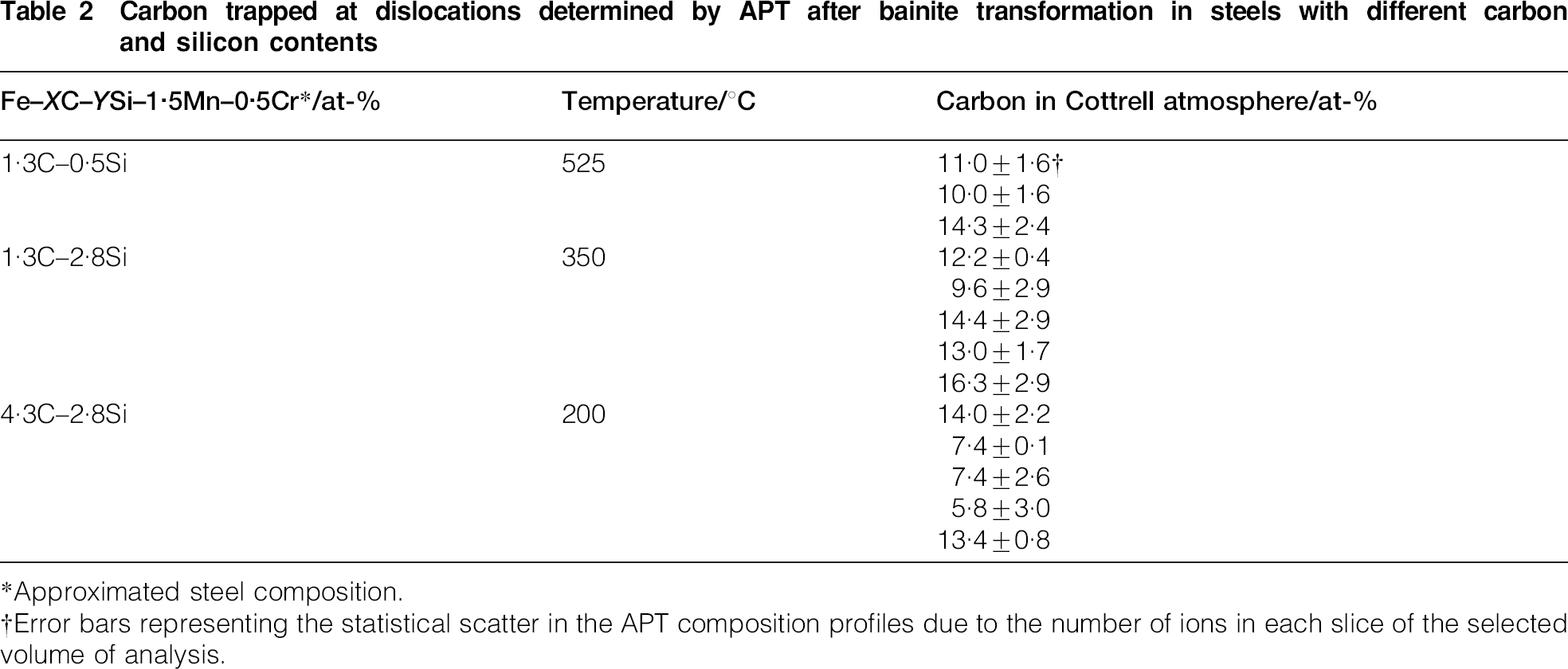
*Approximated steel composition.
†Error bars representing the statistical scatter in the APT composition profiles due to the number of ions in each slice of the selected volume of analysis.
It is believed that solute segregation on dislocations in the vicinity of the interface plays an important role in carbon redistribution during bainite transformation. Dislocations in austenite are inherited in the bainite after transformation and segregation at dislocations is expected to bind and, hence, prevent or hinder the carbon atoms from diffusing out of the ferrite lattice. This explains the small extent of carbon enrichment detected in the residual austenite as determined from XRD analysis and APT, during transformation at 200°C in Fe–4·3C–2·8Si–1·8Mn–1·3Cr (at-) steel (Fig. 1a). Moreover, the increase in the amount of carbon in bainitic ferrite as the transformation temperature decreased (Fig. 4b) is consistent with the fact that, the lower the reaction temperature, the higher the dislocation density of bainitic ferrite. 24
Cluster and carbides formation from supersaturated ferrite
There are many observations that reveal that lower bainitic carbides nucleate and grow within supersaturated ferrite in a process identical to the tempering of martensite. 25 The precipitation of intralath carbides during lower bainite reaction is a secondary process, not essential to the mechanism of formation of bainitic ferrite, except where any precipitation influences the reaction rate by removing carbon from the supersaturated ferrite. However, as discussed below, there are other complications, which can influence any changes in the sequence of precipitation during bainite formation.
The average carbon contents of different carbides formed during lower bainite reaction at different transformation temperatures in the studied steels were estimated from APT data with the use of proximity histograms across carbide/ferrite interfaces; the results are shown in Fig. 6. Examples of a carbon atom map showing carbon segregation across carbide particles are reported elsewhere. 26 Based on the carbon content, different types of carbides detected were as follows:
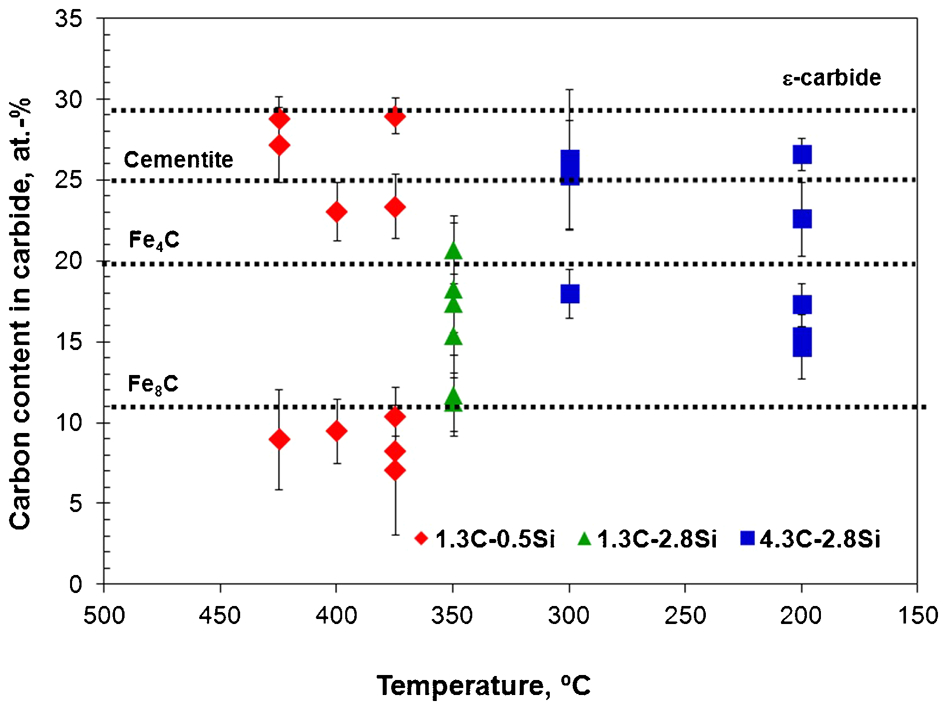
Average carbon contents estimated from APT data of different particles formed during lower bainite reaction at different temperatures in steels with approximated composition of Fe–XC–YSi–1·5Mn–0·5Cr (at-)
preferential agglomeration of carbon atoms in the bainitic ferrite matrix with a carbon content of 11–16 at-C and without evidence of substitutional solute partitioning
particles with a carbon content of 17–26 at-, which might correspond to cementite or Fe4C carbon clusters with ∼20 at-C. Cementite particles ideally contain a carbon content of 25 at-. However, an apparent low carbon concentration of cementite has also been reported in earlier atom probe studies. 27 All the particles detected with this level of carbon have also shown clear evidence of substitutional solute partitioning across the interface. This additional information helps to discriminate between cementite and carbon clusters
particles with a higher carbon content ranging from 27 to 30 at-C were identified as ϵ-carbides.
Matas and Hehemann 28 first suggested that the initial carbide formed in lower bainite is ϵ-carbide, which is subsequently replaced by cementite. In the present work, both ϵ-carbide and cementite precipitated inside bainitic ferrite in the 1·3C–0·5Si (at-) steel (Fig. 6). The rate at which the ϵ-carbide converts to cementite increases with temperature, depends on the steel composition. In fact, a high Si concentration retards the reaction, as is observed at all the temperature tested in the 1·3C–2·8Si (at-), and 4·3C–2·8Si (at-) steels in Fig. 6. Bhadeshia and Edmonds 29 also failed to detect ϵ-carbide in high silicon, medium carbon steels, even during the early stages of the lower bainite transformation.
The detection of cementite instead of ϵ-carbide in lower bainite was rationalised in terms of carbon trapping at dislocations. 30 The detection of cementite as the transformation temperature decreased in Fig. 6 is consistent with the fact that the lower the reaction temperature, the higher the dislocation density in the bainitic ferrite. 24 Carbon segregation at dislocations in the vicinity of the austenite/ferrite interface is other possible reaction that competes for carbon during carbide precipitation.
Carbon atom clusters, i.e. particles with a carbon content of 11–16 at-C and without evidence of substitutional solute partitioning, represented in Fig. 6 were also detected in the three steels at all the tested temperatures. The nature of the carbon atom clustering processes seems to be associated with a redistribution of solute to lattice defects 31 and they might signify the onset of or precursors to carbide precipitation during the bainite reaction. 32
Conclusion
Bainite grows by displacive transformation; the growth is accompanied by a shape deformation which is an invariant plane strain with a large shear component. The transformation is diffusionless, but carbon atoms partition into the residual austenite (or precipitate as carbides), shortly after growth is arrested. The precipitation of carbides is therefore a secondary event.
Footnotes
Acknowledgements
Research was supported by ORNL's Shared Research Equipment User Facility, which is sponsored by the Office of Basic Energy Sciences, Scientific User Facilities Division, US Department of Energy. The authors also gratefully acknowledge the support of the Spanish Ministry of Science and Innovation for funding this research under the contract MAT2010-15330 respectively.
