Abstract
This review considers the compositions, the main process routes, microstructure and structural properties of microalloyed steels. The background and brief history are followed by sections dealing with aspects of precipitation, which control grain size and dispersion strengthening in ferrite–pearlite steels, the approaches to modelling thermomechanical processing and the influence of multiple additions of transition metals on properties. High strength acicular ferrite/bainite steels used for linepipe are included and lead to super bainite steels. Around 12% of the world strip production is processed by the thin slab direct charging route, which is considered in some detail. The weldability of microalloyed steels now embraces joining using friction stir welding, which is discussed. Over the years, many approaches have been developed to predict the structural properties of these steels. They comprise several quantifiable microstructural features including possible atom clusters, relatively recently identified through atom probe tomography. A comprehensive collection of references is provided.
Introduction
This review follows two previous reviews by the present author on ‘microalloyed steels’ (MA steels). 1,2 Since the second of these was published in 1992, an excellent book by Gladman 3 and several reviews closely related to MA steels have appeared. 4–7 In addition, reviews on specific elements in MA steels have been published. These include the influence of additions in MA steels of niobium by DeArdo, 8 an overview of titanium MA steels by Pickering 9 and reviews of vanadium by Langneborg et al. 10,11 and Baker 12 and of zirconium by Baker, 13 together with the effects of aluminium nitride in steel by Wilson and Gladman. 14 In the main, these publications deal with MA steels having a ferrite–pearlite (FP) microstructure.
What, therefore, is new and justifies yet another review? In the intervening years, there has been a continual drive to produce high strength structural steels at a lower cost; therefore, MA steels have progressed in several directions. A better understanding of the role of both the microalloying additions and the deformation processes, usually hot rolling, has been sought. Thermomechanical processing has been refined through a better understanding of the relationships between processing parameters, especially those involving controlled rolling, and microstructure and properties. Direct charged thin slab processing of MA steels is an example of a developing route (by 2007, 12% of the World's strip), which, for some products, is more economic and has environmental advantages over conventional processes. The extension of high strength MA steels into non-FP microstructures having yield strengths σy>500 MPa for such applications as pipeline and automobiles is well advanced. However, the application of friction stir welding (FSW) to these steels, which reduces residual stresses associated with the fusion welding processes, is at much earlier stage of commercial exploitation. At a fundamental level, the strength contributions from nanoprecipitates is being investigated, which, in some cases, may change the way estimates of contributions to strength based on microstructure are summated. The modelling of hot working processes involving MA steels is work still in progress. All these areas will be included in this review. However, with ∼130 000 hits on Google for ‘microalloyed steels. the work reviewed is inevitably selective. It is based on well cited papers that the author has been aware of for some time, and recent papers that make a novel contribution to the field from the many research groups worldwide that I follow.
Background
The estimated world production of steels of all classes for 2014 is ∼1.65 billion tonnes, of which 50% is accounted for from China. 15 Of this total, an ∼12%, equal to some 200 million tonnes, a substantial component, is made up of MA steels. 16
Compared to mild steel, with a lower yield strength σy of 150–200 MPa, current MA steels have σy values in the range of 350–800 MPa, with the potential to exceed 1000 MPa. 17
Microalloyed (MA) or high strength low alloy (HSLA) steels before the 1980s, contained typically, 0.07–0.12% carbon, up to 2% manganese and small additions of niobium, vanadium and titanium (all usually max. 0.1%) in various combinations. 18 (The steel compositions are given in wt-%, throughout the paper, unless stated otherwise.) Other elements that might be present include molybdenum, zirconium, boron, aluminium, nitrogen and rare earth metals.
Vervynct et al. 7 compiled a useful table of alloying elements normally present in MA steels, which, with the addition of zirconium and boron, is reproduced in Table 1. The microalloying elements are used to refine the grain microstructure and/or facilitate dispersion strengthening through precipitation. They are normally regarded as having a low hardenability effect. 19
Alloying elements frequently used in microalloyed steels
Controlled additions of sulphur, and occasionally tellurium, are also added to improve the machinability. 19 The original aim was to develop high strength and toughness in FP steels in the as rolled condition. Owing to their superior mechanical properties, they allowed a more efficient design, with improved performance, even under difficult environmental conditions. Furthermore, they permit reductions in component weight and manufacturing cost. For more information on the physical metallurgy of the elements of importance in MA steels, attention is drawn to the book by Gladman. 3
To avoid any confusion, the present writer prefers to avoid the use of the term ‘high strength low alloy steels, HSLA steels’ and use ‘microalloyed steels’ to describe those with microadditions of niobium, titanium, vanadium and zirconium, either singly or in combination, forming carbides, nitrides or carbonitrides, with a face centred cubic structure. Low alloy steels, a much earlier defined class of steels than MA steels, are generally regarded as containing < 3.5 wt-% total of alloying elements and included Cr (0.5–2.5%), Mo ≤ 3% and V ≈ 1%. These steels were developed as creep resistant steels, but had an upper temperature limitation < 400°C, with applications, as discussed by Oakes and Barroclough, 20 in the earlier British aero gas turbines, and by Robertson, 21 in coal power energy producing plants. The Cr Mo grades were normalised, while the Cr Mo V types were used in the normalised and tempered condition, to develop the alloy carbides in a bainitic microstructure, which conferred their particular properties. While the earlier MA steels sought to avoid transformation to acicular ferrite (AF) and bainite, modern MA steels, built upon a sounder understanding of the processing routes and the development of microstructure than was available before the 1980s, embrace these phases. 17
In MA steels, FeNb consumption has grown threefold during the last 25 years, and the most part of this growth has been noticeable over the past 10 years. Figure 1 shows the trend in ferro-niobium production. Furthermore, the development of MA steels was far greater and far quicker for flat products than for long products. 22
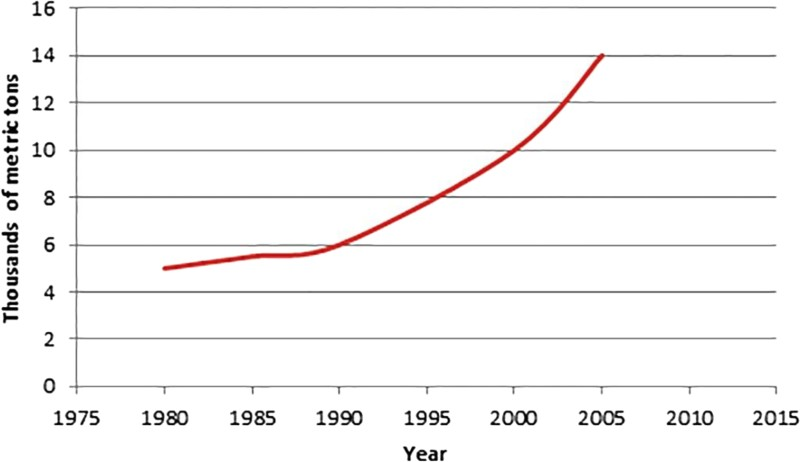
Ferro-niobium (FeNb) consumption in West Europe (EU15) (CBMM-NPC data) 15
The material is produced preferably by a thermomechanical rolling process, also known as controlled rolling, possibly with accelerated cooling, which maximises grain refinement as a basis for improved mechanical properties. Before thermomechanical processing, the steel was heated into the austenite temperature range for all of the precipitates to be taken into solution; after forming, the material must be quickly cooled to 600 to 540°C. 18 Medium carbon directly quenched MA steels, avoiding FP microstructures, can also be forged. 23
Microalloyed steels lie, in terms of performance and cost, between carbon steel, or mild steels, and low alloy steels mentioned above. Until around 1980, low alloy steels were designed to have a yield strength between 500 and 750 MPa without heat treatment. The weldability is at least equal to that of mild steel and can be improved by reducing carbon content while maintaining strength. Fatigue life and wear resistance are superior to similar heat treated steels. The disadvantages are that ductility and toughness are not as good as quenched and tempered (Q&T) steels. 18
Cold worked MA steels do not require as much cold working to achieve the same strength as other carbon steels; this also leads to greater ductility. Hot worked MA steels can be used from the air cooled state. If controlled cooling is used, the material can produce mechanical properties similar to Q&T steels. Their machinability is better than Q&T steels because of their more uniform hardness and their FP microstructure. 18 Because FP MA steels are not Q&T, they are not susceptible to quench cracking, nor do they need to be straightened or stress relieved. However, because of this, they are through hardened and do not have a softer and tougher core like Q&Tsteels. 18
Current UK standards that contain specifications for MA steel grades are BS EN 10025-3:2004 and BS EN 10025-4:2004 (UK/EU). Industry specific grades include C38, C38 modified (includes tellurium), C42, C42 modified (includes tellurium), 49MnVS6, Vanard series and Hypress series (for narrow strip products). 18 Table 2 gives the mechanical properties for various grades of pipeline steels discussed throughout this paper.
Mechanical properties for pipeline steels
History of MA steels
The main development of these steels has taken place over the past 50 years and was initially concentrated on niobium additions. The term microalloying, as applied to steels, is generally accepted as emanating from the paper by Beiser 24 published in1959, which reported the results of small additions of niobium to commercial heats of a carbon steel. However, it has not been recognised that microalloying, as such, first occurred some 35 years earlier, when small additions of zirconium were added to plain carbon steels, and the effects reported by Field 25,26 and by Beckett. 27 Like many other scientific and engineering innovations, military conflict was also the driving force in the case of the development of zirconium steels. During the period of a few years immediately preceding the entry of the USA into the First World War in 1918, the US War Industries Board decided upon an intensive experimental programme with the aim of possible large scale production of zirconium steels suitable for light armour. However, this appeared to be discontinued when the conflict ended, and interest moved to low alloy Q&T steels, normally containing chromium, molybdenum and vanadium additions of 0.5–3%.
Much of the early work on the niobium MA steels, which re-emerged in the late 1950s and early 1960s, was concentrated in the USA and UK. In the UK, the University of Sheffield, British Iron and Steel Association based in Sheffield and Swinden Laboratories of the United Steel Corporation based nearby in Rotherham all made significant contributions. The history of this development has been well documented in an excellent review by Morrison, 5 who, 28 together with Woodhead, 29 played a major part in understanding the role of niobium carbide in contributing to dispersion strengthening and grain refining of ferrite, leading to greater strength than found in mild steels. His initial paragraph, which is quoted here, sets the scene: ‘In 1958, it was announced in the Journal of Metals that the Great Lakes Steel Corporation, a division of the National Steel Corporation of the USA, had entered the market with its GLX-W series of niobium-treated steels, the first steel company in the world to do so. What made this development so special was the very small, relatively low cost addition of niobium used, 0.005 to 0.03%, and the relatively large resultant strengthening effect, combined with good toughness. Also, the niobium was added to an ordinary semi-killed C–Mn steel (mild steel or mild carbon steel) and changed its strength level from a low yield strength of around 300 MPa to a high yield strength of up to 415 MPa for the GLX-60-W grade, equivalent to a conventional alloy steel.’
History has shown that some of the major advances owe their success to the chance simultaneous appearance of several, apparently disparate, facets. This was certainly the case with MA steels. The marketing by the Great Lakes Steel Corporation in the USA occurred in the same decade as the series of publications by Hall, 30 Petch 31 and Cottrell, 32 which provided the first real understanding of the factors that control the strength and, to some extent, the toughness of crystalline material.
The Hall–Petch equation conveniently allows σy, the lower yield stress (or often, in practice, the 0.2% proof stress), to be related to the ferrite grain size d
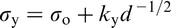
Over the same period, the first high resolution (0.8 nm) transmission electron microscope (TEM), the Siemens Elmiskop I, became available, and in the UK, precipitation in Q&T low alloy steels was one of the first areas studied using this new equipment. Electron micrographs had appeared in a number of papers emanating from the USA before this time. These were mainly concerned with steels but, apart from utilising the higher resolution then available compared with the optical microscope, little attempt was made to extend the interpretation; this was due mainly to the studies being based on surface replicas. Following the pioneering work of Heidenreich 33 in 1949, Hirsch et al. 34 at Cambridge successfully prepared thin foils of a number of alloys, including steels. The theories of kinetic and dynamic diffraction contrast were developed over a period of years, which allowed the details of features observed in foil TEM specimens, such as grain boundaries, dislocations and precipitates, to be interpreted. 35 This is well described by Hirsch. 36
Over the past two decades, the electron back scattering diffraction technique, originally devised for the TEM, has been widely used with an SEM to obtain information on details of grains and subgrains and particularly their boundaries, 37 through the availability of versatile software. This has provided opportunities to quantify what were regarded formerly as difficult phases, such as bainite and martensite/austenite phase, which are important constituents in some modern high strength/high toughness MA steels.
A fourth factor was the developments associated with steelmaking and hot rolling, the latter to be the main route for producing MA steels. New stricter procedures were needed to ensure that niobium steels, in particular, achieved their potential properties. These involved a knowledge of the solubility limits of niobium carbide and nitride, 3,8 to ensure that the precipitates that formed during casting were taken into solution before rolling. This often involved running soaking pits at higher temperatures than normal practice. Furthermore, it soon became apparent that the number of rolling passes and their temperature needed to be controlled and hence the birth of ‘controlled rolling’. 38–40 Within a short time, this led to the introduction of computer control in many other aspects of steelmaking, and later, the application of the results of academic computer modelling to aid the whole complex process of achieving a small, < 10 μm, homogeneously distributed ferrite grains. 41–43
Figure 2 shows schematically how the microstructure and properties of plate steels changed over time with advances in alloy design and processing. 7,44
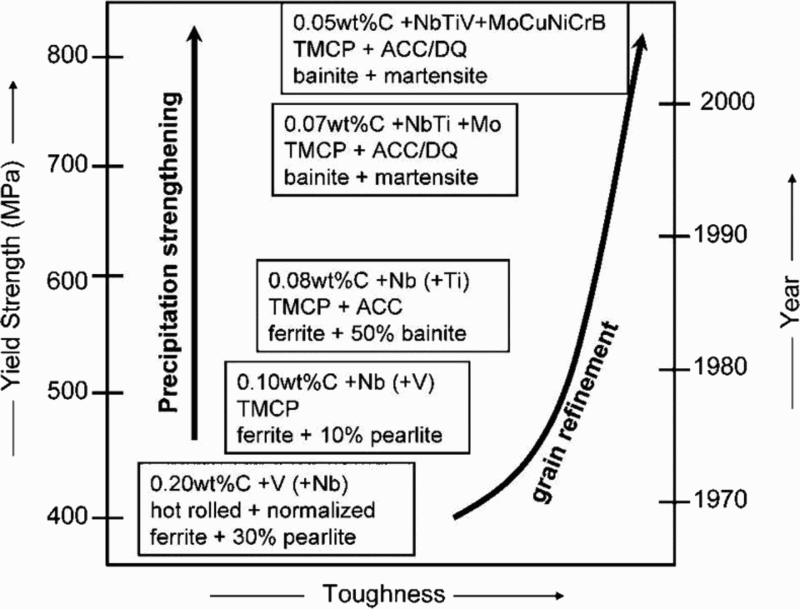
It is obvious from Fig. 2 that the accelerated cooling after rolling was largely responsible for the very high strengths attainable, practically independent of composition. With suitable cooling practices, σy levels >690 MPa (X100) can be achieved in low C steels containing < 2 wt-%Mn and with carbon equivalent and weld cracking parameter near 0.5 and 0.2 respectively. 6
Chemical compositions and precipitation in MA steels
DeArdo et al. 6 posed the question ‘what is the role of the microalloying element (MAE) in obtaining strength levels in these steels?’ It is answered by considering the early steels (pre-1980), where air cooling of plate and high coiling temperatures of strip were used. As noted above, these were the FP steels with strengths up to ∼420 MPa (X60) for gauges up to 18 mm. The most obvious contributor to strength was grain refinement, as was clearly shown by quantitative optical microscopy. There is no doubt that the MAE was responsible for this contribution through its effect on austenite conditioning. Other contributions included solid solution strengthening by the Mn, Si and others, including the MAE, when retained in solution. Equations have been published quantifying these effects. 3 The other contribution to strength, claimed by researchers studying these early steels, was dispersion hardening by transition metal carbides and nitrides. 2,3
The precipitation of carbides and nitrides occur at three different stages during the processing of MA steels. 2,3 Type 1 precipitates are formed during the liquid phase and during or after solidification, on the liquid/solid interface and in delta ferrite. These precipitates are very stable, and while they are normally too large to influence recrystallisation of austenite, the smallest may effectively retard coarsening in austenite during reheating or during a welding cycle. 45
Type 11 particles are precipitated in austenite after solution treatment and during hot deformation, such as controlled rolling, as the temperature is decreasing. 46 The precipitates are strain induced and can retard the recrystallisation of austenite. Grain refinement of MA steels is mainly due to this group of particles. 47–49
Finally, type 111 particles are formed during or after the austenite to ferrite phase transformation, nucleating on the austenite/ferrite interface and in ferrite. 50 Dispersion strengthening in ferrite normally occurs through these changes, and a fine precipitate dispersion is usually observed.
The carbides and nitrides of the transition metals, which precipitate in MA steels, are B1 NaCl (Fm3m) type compounds. Several precipitate nucleation processes have been recognised in MA steels. These include homogeneous precipitation, resulting in coherent precipitates with strain fields, leading to semicoherent and incoherent precipitates, interphase precipitation, heterogeneous precipitation on grain boundaries and dislocations, where the latter is often referred to as strain induced precipitation (SIP).
A pre-precipitation grouping of atoms, nanoprecipitates, known as Guinier–Preston (GP) zones in non-ferrous alloys and also described as ‘clusters’ in MA steels, has received increased attention over the past decade due to the development of atom probe tomography (APT). Several APT studies on niobium MA steels have been reported. 51–56 These nanoprecipitates are assuming importance, due to the claim that they provide a significant contribution to the yield strength. 57 Following a 1250°C homogenisation, water quenching and tempering at 600°C in a salt bath for 300 s, in a steel containing controlled atomic concentrations of 500 ppm Nb and 250 ppm C and N, monolayer GP zones were detected by Danoix et al. 53 These were platelets comprised of niobium and nitrogen atoms, ∼4 × 4 nm in size, lying on {100}α planes. The amount of iron and carbon in the GP zone was ∼ nil. They 53 also claimed that the homogeneous nucleation mechanism for the nitrides, which nucleated heterogeneously on dislocations, was completely different from that of carbides. However, different results were obtained by Breen et al., 55 who used APT to study a steel containing 0.03C–0.007N–0.084Nb, finish rolled at 879°C and coiled at 567°C. Samples were aged to investigate the Nb (C,N) precipitation in ferrite. Table 3 compares the nanoprecipitate composition following three aging treatments. It can be seen that the longer aging results in more C and N atoms being trapped within the nanoprecipitate, which had a similar composition to the nominal composition of the steel. Even after aging for 40 min at 700°C, the nanoprecipitate size of ∼2.6 nm length × 2.8 nm width, was still considered to be less than the critical size for coherence loss with the ferrite matrix. As most MA steels are used in the as rolled condition, it is relevant to examine how hot deformation, rather than aging, influences the development of nanoprecipitates.
Bulk atomic compositions were calculated on IVAS using background correction and decomposition of molecular ions.
Pereloma et al. 51,56 used APT to compare niobium clustering in a Nb–Ti MA steel, deformed above and below the non-recrystallisation temperature T nr, which represents the start of the inhibition of complete static recrystallisation during cooling between rolling passes. Using a Gleeble simulator, they studied a steel containing 0.081C–0.064Nb–0.021Ti–0.003V–0.017N. After austenitising at 1250°C and roughing at 1100°C, samples were cooled either to 1075 or 825°C, deformed at a strain rate of 5 s− 1 to a strain of 0.75, before cooling. Similar distributions of >70 nm TiN and (Ti, Nb)(C,N) were found in all specimens. A temperature of 1075°C was too high for Nb–C clusters to form, and therefore, SIP of NbC did not occur. However, after 825°C deformation, a relatively high number density of Nb–C clusters occurred, as did SIP of NbC. This implies that the lower deformation temperature of 825°C resulted in a higher dislocation density, leading to an increase in the number of nucleation sites for Nb–C clusters. These clusters might be considered as precursors to SIP via heterogeneous nucleation of NbC. The above work was extended by Kostryzhev et al., 54 to include aspects of the strengthening of the steel through the possibility of clusters being cut, which is discussed below. Coherent precipitates are cut by dislocations, leading to a different strength mechanism than incoherent precipitates. This distinction becomes very important when estimating yield strength from microstructure.
Few observations of coherent precipitates in MA steels have been verified. This is in part, due to the very small size at which they lose coherence. Coherence strain fields associated with transition metal carbides and nitrides have very rarely been reported for precipitates in MA steels, vanadium carbonitride 2 being an exception (Fig. 3. Figure 4 shows that the calculated misfit of vanadium carbide and nitride in austenite is much less than for niobium carbide and nitride. 58,59 Estimates of the limiting size of some coherent carbides and nitrides if they precipitated in ferrite are given in Table 4, which shows the size when coherence begins to be lost is significantly smaller for NbC than VC. The estimated V–C cluster diameter is in the range of 3–13 nm, which is larger than that given for NbC in Table 4, based on misfit alone.
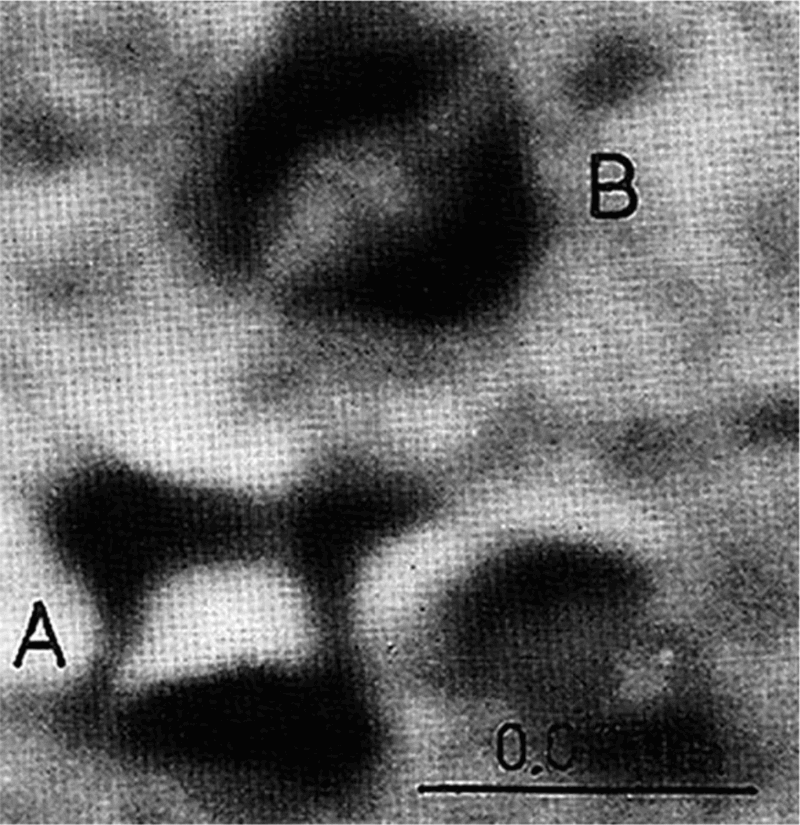
Image (TEM) of coherence strain fields associated with vanadium carbide particles (A: double arrowhead strain field contrast of disc particle; B: double lobe strain field contrast of spherical particle) 2
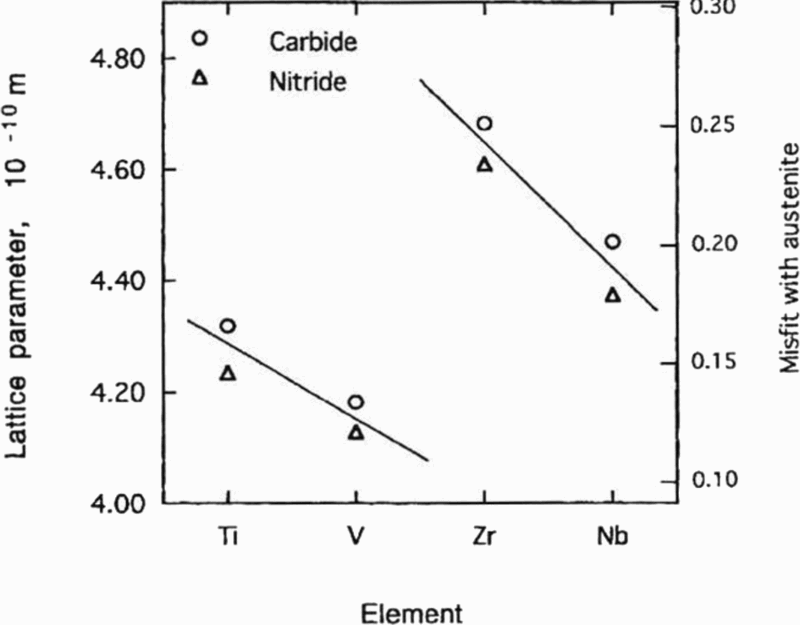
Variation of lattice parameters and misfit with austenite of some transition metal carbides and nitrides found in microalloyed steels 58
Estimated limiting sizes for coherence of platelet carbides and nitrides found in ferrite in microalloyed steels*
ε1 and ε2 are the calculated strains parallel and perpendicular to the major axis of the disc shaped particle of diameter x 1 and thickness x 2.
The data in Table 4 are based on the cube on edge orientation relationship first established by Baker and Nutting (BNOR) for tempered steels
60
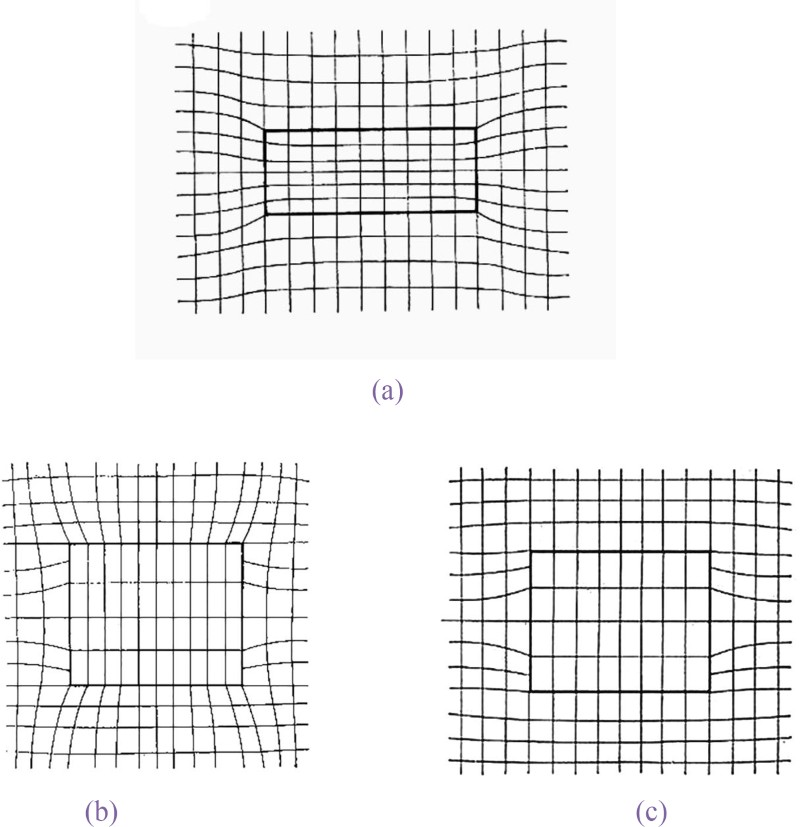
a cross-section of disc shaped coherent particle with small negative mismatch, b cross-section of disc shaped coherent particle with small positive mismatch and c cross-section of disc shaped with positive misfit normal to plane of disc 35
Incoherent disc precipitates of VN are seen in Fig. 6, where strain fields are no longer present. The fine NbC particles in Fig. 7, present on a replica, would provide dispersion strengthening.

Incoherent vanadium nitride disc precipitates mainly in section 1

TEM micrograph of carbon replica which has extracted ˜2 nm particles114
Interphase precipitation occurs during the transformation of austenite to ferrite and is found, with the possible exception of ZrC and ZrN, for all the transition metal carbides and nitrides,
50
as well as silver
61
and copper
62
precipitates in steels. Dunne
62
pointed out that the many studies by Honeycombe and co-workers,
50
usually with highly alloyed ternary or quaternary laboratory steels, and often in the isothermally treated condition, producing high volume fractions of precipitates, were undertaken to elucidate the fundamental mechanisms of interphase precipitation. These are summarised in terms of the morphologies that have been established: interphase precipitation (planar) interphase precipitation (curved) continuous fibre/lath growth precipitation from supersaturated ferrite.
Planar interphase precipitation is typified by parallel sheets of densely populated sheets, related to ferrite by the BNOR, 60 and appear to have a regular spacing (Fig. 8). Continuous fibres comprise parallel laths, akin to a very fine pearlite. Details of the mechanisms have been reported in many publications, including Refs. 1–5, 11, 50, 61 and 62.

Interphase precipitation in vanadium microalloy steel 1
While interphase precipitation is well established in isothermally transformed steels, it appears to be less important in commercially processed Nb and Ti–Nb as rolled MA steels. For these steels, this leaves heterogeneously nucleated precipitation associated with grain boundaries, leading to grain refining, and precipitation associated with dislocations (SIP) as the main classes of precipitates. However, compared to the other microalloying elements, vanadium has a much greater solubility in austenite and therefore remains in solution to a much greater extent during processing in the austenite range. Vanadium carbonitride interphase precipitates that form above 700°C are recognised as making an important contribution to strengthening, which becomes general V(C,N) precipitation below 700°C. The fibrous morphology of V(C,N) is sparse and is never a dominant microstructure. 11
Vervynct et al. 7 considered the principal function of alloying elements in FP microalloy steels to be ferrite strengthening by grain refinement, dispersion strengthening and solid solution strengthening, all implied by Petch. 31 Solid solution strengthening is closely related to the alloy content, while grain refinement and dispersion strengthening depend on the complex interaction of alloy composition and thermomechanical processing. For example, the strengthening effect of niobium, the most widely studied element in MA steels, occurs mainly by three microstructural mechanisms: ferrite grain refinement due to austenite grain boundary pinning, retardation of recrystallisation and dispersion strengthening. 6 Each of these mechanisms will be considered more fully below. In addition, alloying elements are selected to influence the reduction in the temperature at which austenite transforms to ferrite and pearlite during cooling. In this way, a fine grained product is produced, which is a major source of strengthening and toughness.
DeArdo et al. 6 consider that ‘the year 1980 represents a bench mark in the strength of microalloyed (MA) steels. From the early days of the 1960s to approximately 1980, the steels being microalloyed were low hardenability steels with FP microstructures and σy ≤ 420 MPa. These steels were used to develop the principles and interrelationships of microalloying, controlled rolling and air cooling. They were characterised by relatively higher carbon contents and moderate manganese levels, and exhibited FP microstructures after air cooling. Around 1980, both the line-pipe and the automotive industries desired strengths >420 MPa, that could be readily supplied with fine grained FP steels. Clearly, higher strength microstructures were required. The obvious choices were the lower temperature transformation products: matrices comprised of non-polygonal ferrite, AF, the bainites and martensite, either as monoliths or as mixtures. To achieve these microstructures, the combination of higher hardenability and high cooling rates was required. Furthermore, much additional research was needed to reach the required goals consistently and with uniform results. From the processing side, the solution to this dilemma was using water cooling after hot rolling. This was accomplished in the mid-1980s for plate processing by interrupted accelerated cooling (IAC) 63,64 and interrupted direct quenching (IDQ) 65 in plate mills, Fig. 9. Runout table water spray cooling to the coiling temperature in hot strip mills had been in practice since the 1960s, but not as a microstructural control tool for increasing strength. This was because of the higher carbon contents of the steels of that era. The benefits of faster rates of cooling and lower coiling temperatures were exploited for achieving higher strengths later with steels of lower carbon contents.’
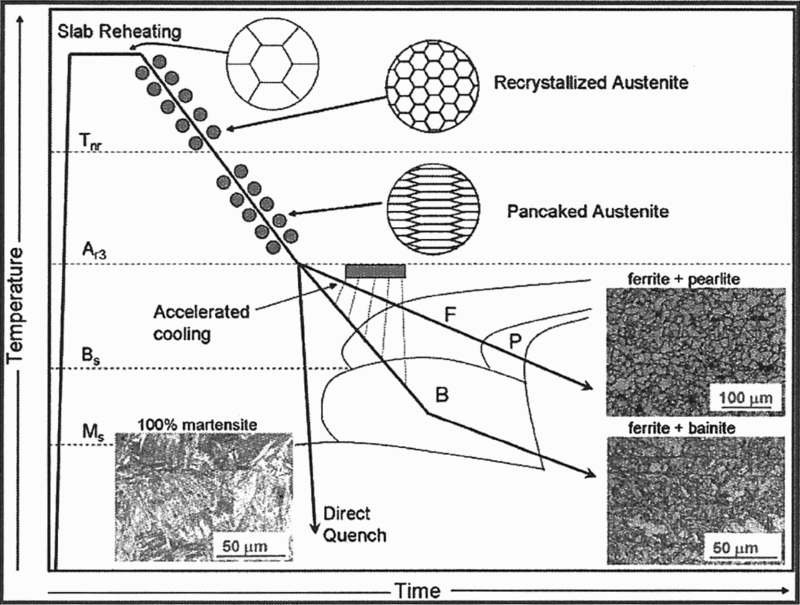
Schematic diagram of thermomechanically controlled processing (TMCP) and microstructures that result from this process 7
Controlled rolling and controlled cooling
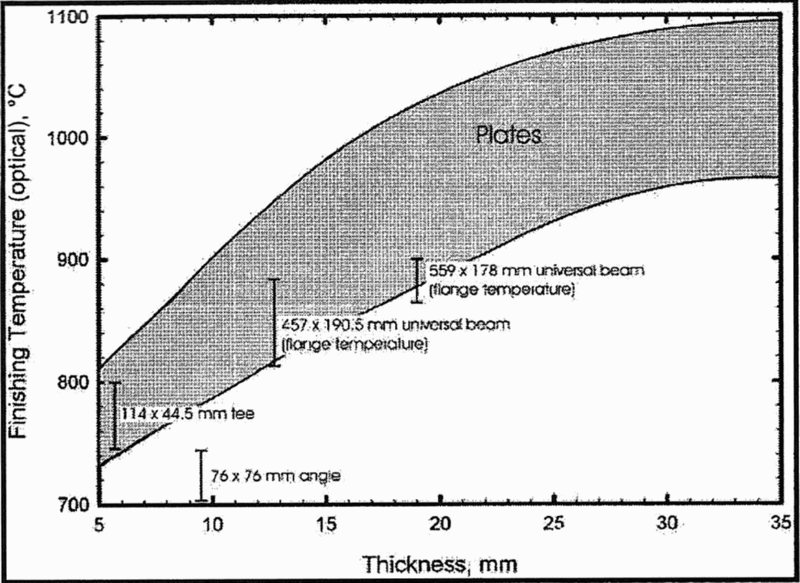
The importance of developing a small grain size in terms of increases in both strength and toughness is evident from the initial work of Hall 30 and Petch 31 mentioned above. As stated by Llewellyn, 66 ‘the traditional route (before the 1970s) to a fine grain size in FP structural steels has been to incorporate grain refining elements, such as aluminium, and then to normalise the materials from about 920°C after rolling.’ He also comments on the fact that ‘when normalizing was carried out on a niobium treated steel to improve the impact properties, the strength advantage was forfeited. There was therefore a need for an alternative route to a fine grain size in structural steel plate which would overcome both the cost and strength penalties associated with traditional normalizing. As described by Morrison 5 , ‘the key to obtaining a fine grain size in a low niobium steel (∼0.02 wt-%) is the low finishing rolling temperature (FRT), which occurs naturally in thin plates and cross-country mills.’ Mackenzie 67,68 collected data from 68 plates, finished rolled from 11 to 38 mm, using 14–36 passes, which took between 3 and 6 min in the mill, with finishing temperatures between 800 and 1100°C. He found that finishing temperatures below ∼900°C gave acceptable notch ductility, equating to as rolled plates ≤ 13 mm in thickness. Figure 10 shows that much greater thicknesses could be tolerated in sections due to their lower rolling rate. As far back as 1958, Vanderbeck 69 reported that ‘European steel producers were adopting lower than normal finishing temperatures during rolling, in order to refine the microstructure and improve properties’. The idea of using a rolling schedule to produce a fine grain size may have originated from so called cross-country mills, a versatile ‘jobbing’ finishing mill, often used for producing sections or bar in a steel works, where the workpiece usually passed only once through a set of rolls, which were spread over a large area, with the final set some distance from the starting point. Thus, the rolling master was always dealing with a loss of heat, and often, the steel was black on reaching the finishing stand. However, at Round Oak Steelworks in Staffordshire, it was noted that, when the steel was black at the finish of rolling, the strength and toughness properties were always superior to when the steel finished red. It was on this mill that the writer undertook some of his first controlled rolling trials in 1963.
‘Controlled rolling or rolling over a deliberately lowered temperature range compared with conventional hot rolling, is now a widely accepted technique for the production of microalloyed steels.’ This statement by Kozasu 70,71 is as true today as when it was first made in 1968, when the aim of controlled rolling was to produce a steel with a fine polygonal ferrite grain size. Today, this is extended to include acicular and bainitic microstructures. 17
In his review of controlled rolling, Tamura 40 states that ‘the fundamental difference between conventionally hot-rolled and controlled-rolled steels lies in the fact that the nucleation of ferrite occurs exclusively at austenite grain boundaries in the former, while it occurs in the grain interior, as well in the latter, leading to a more refined grain structure.’
Conventional controlled rolling is an example of a thermomechanical controlled process (TMCP) which manages the temperature and deformation during hot rolling, to control the austenite microstructure at the start of transformation. Here, it is essential that recrystallisation of austenite is avoided. The original approach was to introduce a high density of nucleation sites into austenite grains for nucleation of ferrite grains, by hot rolling in the austenite phase field.
After transformation to ferrite and subsequent recrystallisation, as first described by Hanemann and Lücke, 72 this leads to a refined microstructure. Ferrite grain refinement is due to two mechanisms: fine recrystallised austenite grains formed by hot rolling at intermediate temperatures and austenite deformation below the recrystallisation temperature, which enhances the nucleation of ferrite. 70
The first step in the controlled rolling process is to control the austenite grain size during the soaking stage. This is set by the temperature, which is necessary to take into solution the microalloying particles, which have formed during cooling following solidification during casting. As is well established, the austenite grain size is related exponentially to the soaking temperature. Therefore, a balance exists between the temperature necessary to dissolve the particles, the resultant austenite grain size and the economics of high soaking pit temperatures. 7,70 The importance of the correct soaking temperature for a given steel is well illustrated by Lagneborg et al. 8 for Ti–V–N steels. Reducing the soaking temperature from 1250 to 1100°C reduced σy by ∼40 MPa and the ductile–brittle transition temperature by 15°C, due to a reduction in the dissolved amount of titanium and vanadium.
Kozasu et al. 70 divided the controlled rolling process into three ranges, associated with changes in the austenite and ferrite grain structures.
First is the deformation in the austenite recrystallisation temperature range. Deformation above 1000°C normally develops coarse recrystallised austenite grains, which transform to a relatively coarse ferrite and upper bainite. The size of the austenite grain size obtained by recrystallisation decreases with increasing strain, which is introduced by the rolling reduction, and eventually reaches a limiting value. 73
Second is the deformation in the unrecrystallised range. Deformation in the intermediate temperature range from 1000 to 900°C refines austenite by repeated recrystallisation, leading to fine grained ferrite. The austenite grains are elongated (pancaked) and deformation structures result. 74 A build-up of strain is often associated with the formation of twins or deformation bands, which increase the number of potential sites for ferrite nucleation, and this, as mentioned above, is regarded as one of the most important aspects of controlled rolling. 40,75
Third is the deformation in the (γ+α) two-phase region. Deformation below the recrystallisation temperature produces ‘warm worked’ austenite, which leads to a finer ferrite grained microstructure. The third stage deformation has a much larger influence on the final mechanical properties than the first two stages. Rolling to just above the Ar 3 temperature can result in equiaxed ferrite grains and a substructure, produced by deformation of the recently formed grains. The transition temperature was shown to decrease in a linear manner with an increase in total reduction in ranges 2 and 3. 76
Kozasu et al. 70 do not consider rolling below Ar 1, or the changes in microstructure in terms of the role of dislocations. This has been addressed more recently Vervynckt et al. 7
An important concept is the non-recrystallisation temperature T nr, which represents the start of the inhibition of complete static recrystallisation during cooling between rolling passes. The most common method of determining T nr consists of simulating successive rolling passes and representing the mean flow stress (MFS) versus the inverse of the absolute temperature graphically for each of the simulated passes, 7 as seen in Fig. 11. Here, T nr appears as a change in slope of the MFS curve. Vervynckt et al. 7,75 proposed extending the controlled rolling process to four regions, as defined in Fig. 11. Data were obtained for an MA steel containing 20 ppm Ti and 18 ppm N, based on a simulation of a 23 plate rolling pass schedule, with fixed interpass times of 20 s and a cooling rate of 1°C s− 1. In Fig. 11a , the flow stress increases with decreasing temperature in region 1, while the curves change their shape entering region 11. As strain increases and temperature decreases in region 111, the flow stress decreases, before increasing again in region 1V. These are better interpreted in Fig. 11b , where the MFS is plotted against the inverse absolute temperature.
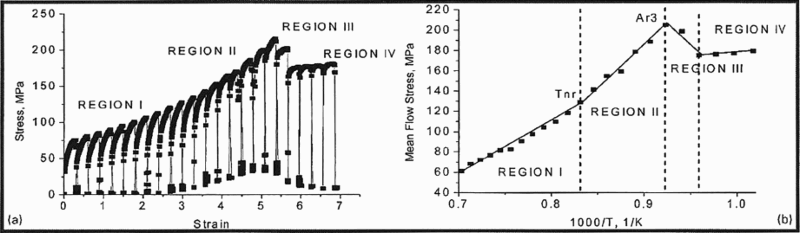
Region I corresponds to deformation at high temperature. Austenite recrystallises completely between passes, and there is no accumulation of dislocations. The increase in flow stress is solely due to the decrease in temperature.
In region II, the change in slope indicates that dislocations are being accumulated. The flow stress increases more rapidly because of the inhibition of recrystallisation between passes.
Region III is characterised by a significant decrease in MFS and corresponds to the start of the γ → α transformation. Here, the intercritical two-phase rolling takes place.
Region IV corresponds to warm rolling in ferrite.
The intersection of the straight regression lines fitted to regions I and II defines T nr, that fitted to regions II and III defines Ar 3 and that fitted to regions III and IV defines Ar 1. These are only for the conditions used here, i.e. fixed interpass times of 20 s and a cooling rate of 1°C s− 1.
Hot rolling takes place on a falling temperature gradient, and the influence of the recrystallised state of austenite on the transformation behaviour and continuous cooling transformation (CCT) curves of MA steels was first investigated by Smith and Siebert. 77 They showed that the ferrite start line Ar 3 was shifted to shorter times when austenite was in the unrecrystallised state. Mixed ferrite grain sizes were found when large strains were introduced in a temperature range in which only a fraction of the austenite grains recrystallised. The regions where unrecrystallised grains transformed, i.e. < T nr, usually lead to ferrite of a different grain size from the ferrite originating from the recrystallised austenite in adjacent regions.
The state, or condition of the austenite before transformation, is therefore one of the major factors that determines the ferrite grain size. In addition to the grain size of austenite, the potential ferrite nucleation sites must be taken into account in any relationship with the ultimate ferrite grain size. Priestner and de los Rios 78 introduced the term S v, which is the grain boundary surface area per unit volume of austenite, also described as the ‘effective grain size’, to account for the elongated grains. S v is now often used to include boundary and intragranular nucleation sites. 71 Austenite, which has a large value of S v, would, by definition, have a large capacity for nucleation of ferrite, and as has been pointed out by De Ardo et al., 79 a low hardenability, and would be expected to develop a fine FP microstructure. This concept is supported by experimental data (Fig. 12). 78
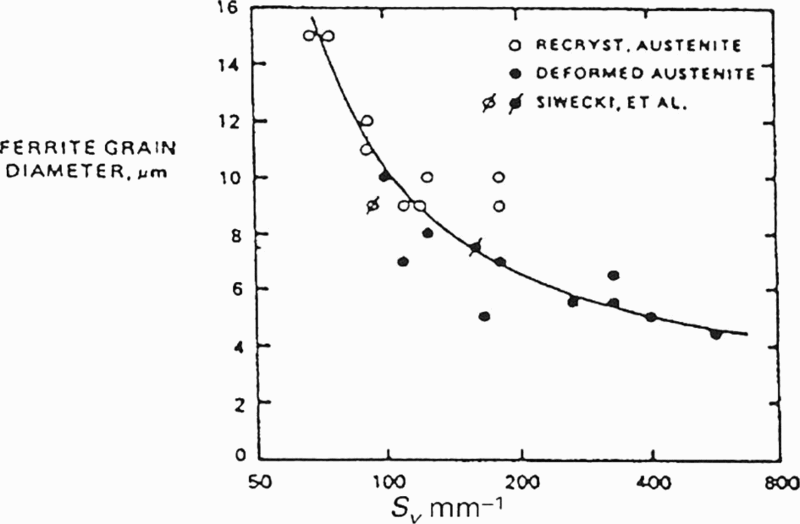
Ferrite grain sizes produced from recrystallised and unrecrystallised austenite at various S v values (after Priestner and Rios 78 )
Final deformation in the low temperature austenite is the temperature range within which complete static recrystallisation no longer takes place between rolling passes, leading to the retention of workhardening.
Basis of modelling (TMCP)
Jonas and Sellars 43 explain succinctly that ‘TMCP in involves the production of specific microstructures, which are associated with particular mechanical and physical properties. In this way, TMCP differs from traditional deformation processing, which is generally concerned with reductions in thickness and with developing desirable changes in shape. TMCP involves the control and interaction of the following fundamental mechanisms: dislocation glide and climb, recrystallization, grain growth, phase transformation, precipitation, particle coarsening, particle pinning and solute drag. The interest in many of these structural changes is whether they take place dynamically (i.e. during deformation) or statically (i.e. after deformation). TMCP is concerned with the synthesis of these fundamental mechanisms. Of particular interest to microalloyed steels, is the TMCP operation concerned with grain refinement for high strength and toughness. Two contrasting, but complementary approaches have been taken; laboratory simulation and computer modelling. The former involve compression, tensile or torsion testing. Compression testers are useful for the determination of the kinetics of recrystallization or precipitation, while tensile machines are used to measure hot ductility, leading to the determination of the temperature and strain rate ranges associated with optimum workability. Computer modelling aims to quantify the fundamental mechanisms involved in TMCP, in terms of the variables of temperature, strain, strain rate and time, and to incorporate them within an overall mathematical description of industrial forming processes. The inclusion of the parameters of microstructural features is now widely accepted as essential, both for off-line optimization of processing conditions, and for on-line control. In the case of hot rolling of steels, the modelling process involves sequential deformation passes taking place over a range of temperatures, rolling speeds, and interpass times.’
As explained by Sellars, 42 ‘during the hot deformation process itself, e.g. a rolling pass, work hardening takes place but it is balanced by the dynamic softening processes (i.e. during deformation) of recovery and recrystallization. These processes, which are thermally activated, lead to a flow stress that depends on strain rate and temperature, as well as strain. The microstructural changes taking place within the material result in an increase in dislocation density with strain, causing dynamic recrystallisation, which takes place repeatedly as new recrystallised grains are themselves work hardened. These dynamic microstructural changes leave the metal in an unstable state and provide the driving force for static recovery and static recrystallisation to take place after the deformation pass. Static recrystallisation may be followed by grain growth, if the temperature is sufficiently high’. Sellars 42 poses two important questions, which must be answered to apply the above principles to commercial practices: (a) how long does recrystallization take after a deformation pass; and (b) what grain size is produced by recrystallization and grain growth? The answers determine the microstructure of the material entering the next and subsequent passes and hence influence the flow stress of the material and the working forces required. Eventually they determine the microstructure and properties of hot worked products.’ Details of the application of this approach is given by Sellars 40 and in a shorter form by Jonas and Sellars. 43
Depending on the eventual product, after casting and solidifying into an ingot, the steel is further processed by rolling (or forging). The rolling process requires the ingot to be reheated (soaked) before primary rolling to a smaller intermediate size (billet), which is cooled to ambient temperature. Normally, the steel is finish rolled in a different mill at a later time, and it is at this stage that the controlled rolling process is undertaken.
To make the forming process easier, relatively high soaking temperatures are traditionally employed for primary rolling, which causes considerable grain growth. Therefore, the first step in controlled rolling is to control the austenite grain size, which increases exponentially with temperature, during reheating. The temperature should be sufficient to take into solution the microalloying elements, as discussed in detail in a later section.
From a commercial aspect, the modelling must relate to the stock soaking temperature and time; the rolling schedule in terms of rolling velocity, deformation per pass, temperature at entry of rolling stock to each pass, interval between passes and final rolling temperature; and the cooling rate following rolling.
Vervynckt et al. 7 describe T nr the non-recrystallisation temperature, as representing the start of the inhibition of complete static recrystallisation, during cooling between rolling passes. They also considered the experimental methods for the determination of T nr and note that most measurements of this parameter relate to plate mill schedules, which are easier to simulate by laboratory test methods due to their long interpass times of 10–20 s, compared to strip mills. They consider that new experimental methods are essential to determine T nr for strip mills, with their short (1 s) interpass times. In MA steels, the initiation of SIP, which is the normally considered as the nucleation of carbides, nitrides and/or carbonitrides on dislocations introduced by deformation, has the ability to suspend both static and dynamic recrystallisation. In other words, SIP, strongly influences T nr. He also points out that continuing deformation also leads to coarsening of existing precipitates and that ‘fresh’ precipitates are generally considered to be necessary for preventing recrystallisation. This effect is important in modelling of rolling operations because the interruption of both static and dynamic recrystallisation leads to increases in rolling loads, whereas the initiation of dynamic recrystallisation in the absence of precipitation results in a sudden decrease in rolling load. It should be noted that the models developed for controlled rolling processes, which involve precipitation, normally do not include coherent precipitation or interphase precipitation, but only heterogeneous nucleation of particles on dislocations (SIP).
A different approach has been adopted by Matlock and Speer. 80 They started from the product, in their example, long bars, and then developed a microalloying and processing strategy for their manufacture. In this way, they considered that it was more likely that strategies that are less successful could be avoided. While this is a far less rigorous approach than the modelling described above, it does have the merit of being applicable to a very wide range of steel compositions and processes, associated with microalloying. These include thermomechanical processing of steel bars, which involves forging, and the production of reinforcing bars. Matlock and Speer 80 also considered the process route of a range of products, such as automotive springs and components for automotive engines and transmission systems, which require quenching and tempering and high temperature carburising.
Solubility of particles that control grain size and provide dispersion hardening
As mentioned earlier, the soaking temperature, before rough rolling, must be sufficient to take into solution the microalloying elements. In MA steels, the most important particles are carbides and nitrides of the transition metals, niobium, titanium, vanadium and to a considerably lesser extent, zirconium. These are often present as carbonitrides. In certain cases, oxides of titanium, and sulphides are of importance, as are aluminium nitride precipitates. During processing, the lowest temperature for taking into solution the particles precipitated on casting, which are later to control grain size and dispersion strengthening, is determined by their solubility in iron as a function of temperature and time. Matlock and Speer, 80 in defining a strategy for the application of microalloying to a range of products, summarised the main objectives of adding niobium, titanium and vanadium to steels with a spectrum of carbon contents, relevant to long products. For example, they considered that the main precipitates were likely to be NbC, V(C,N) or TiN. NbC precipitates are expected at reaustenitising temperatures in virtually all heating treating applications. Solute niobium, remaining in solution in austenite, may also contribute to subsequent formation of nanopreciptates or clusters,51–57 and finer dispersion strengthening precipitates in ferrite. As discussed below, vanadium exhibits considerably greater solubility in austenite than niobium or titanium, and carbonitrides are only expected to form at the lowest austenite temperatures in alloys containing relatively high levels of vanadium, carbon and/or nitrogen. Austenite grain refinement is therefore less likely to occur in leaner alloys, but vanadium is available to precipitate in ferrite over a wide variety of steels, across the entire spectrum of carbon concentrations and processing temperatures. TiN is very stable and usually precipitates at high temperatures in the austenite phase and may be useful in resisting subsequent austenite grain coarsening, when added as a small addition. 80 The basis for most of the conclusions made by Matlock and Speer 80 lay with the solubility equations appropriate for carbides and nitrides of niobium, titanium and vanadium in austenite.
Solubility equations allowing the temperature of compounds in a solvent to be estimated, for example, zirconium carbide in austenite,
13
are normally described in the form of an Arrhenius equation. This gives the dependence of the rate constant K of chemical reactions on the temperature T (in absolute temperature, kelvin) and activation energy E
a, as shown below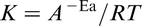
In MA steels, the microalloying element, M is often combined with an interstitial X, to give a compound, MX, some or all of which, dissolves in austenite as the temperature is raised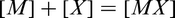
The rate constant K in equation (2) is now described as an equilibrium constant for the reaction given by equation (3). In practice, the concentrations of M and X are normally low, being < 1% and therefore may be considered as having an ideal solution behaviour. M and X are expressed in terms of the weight percentage of the alloying element present in the steel chemical composition. This allows equation (1) to be expressed as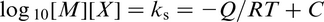
Empirical Arrhenius equations have been determined for many of the important refractory carbides and nitrides known to form in steels, but similar equations for sulphides have not been found. Unlike the solubility equations of transition metal carbides and nitrides of niobium, titanium or vanadium in austenite, the corresponding equations of zirconium are almost entirely due to one source and have some shortcomings.
Table 4 summarises selected Q and C values, collated from the literature, for the relevant grain boundary pinning and dispersion hardening compounds found in MA steel. Taylor 81 has calculated values for carbides in ferrite. Examples of solubility diagrams for NbC, VN and VN in austenite over the temperature range relevant to TMP, and based on the data in Table 5, are given in Fig. 13, taken from Matlock and Speer. 80 For more details of the solubilities, the reader is referred to the reviews on the role of individual elements in MA steels, 8–14 and the book by Gladman, 3 which also provides examples of some mutually soluble carbonitrides.
Selected solubility parameters in austenite of carbides and nitrides found in MA steels
Use with caution. 13
A comparison of these carbide and nitride solubilities is shown in Fig. 14. Here, it can be seen that, while zirconium carbides and nitrides show little solubility in austenite, and often nucleate while the steel is still molten, both zirconium and titanium precipitate in austenite, as do niobium and vanadium, both as carbides and nitrides.
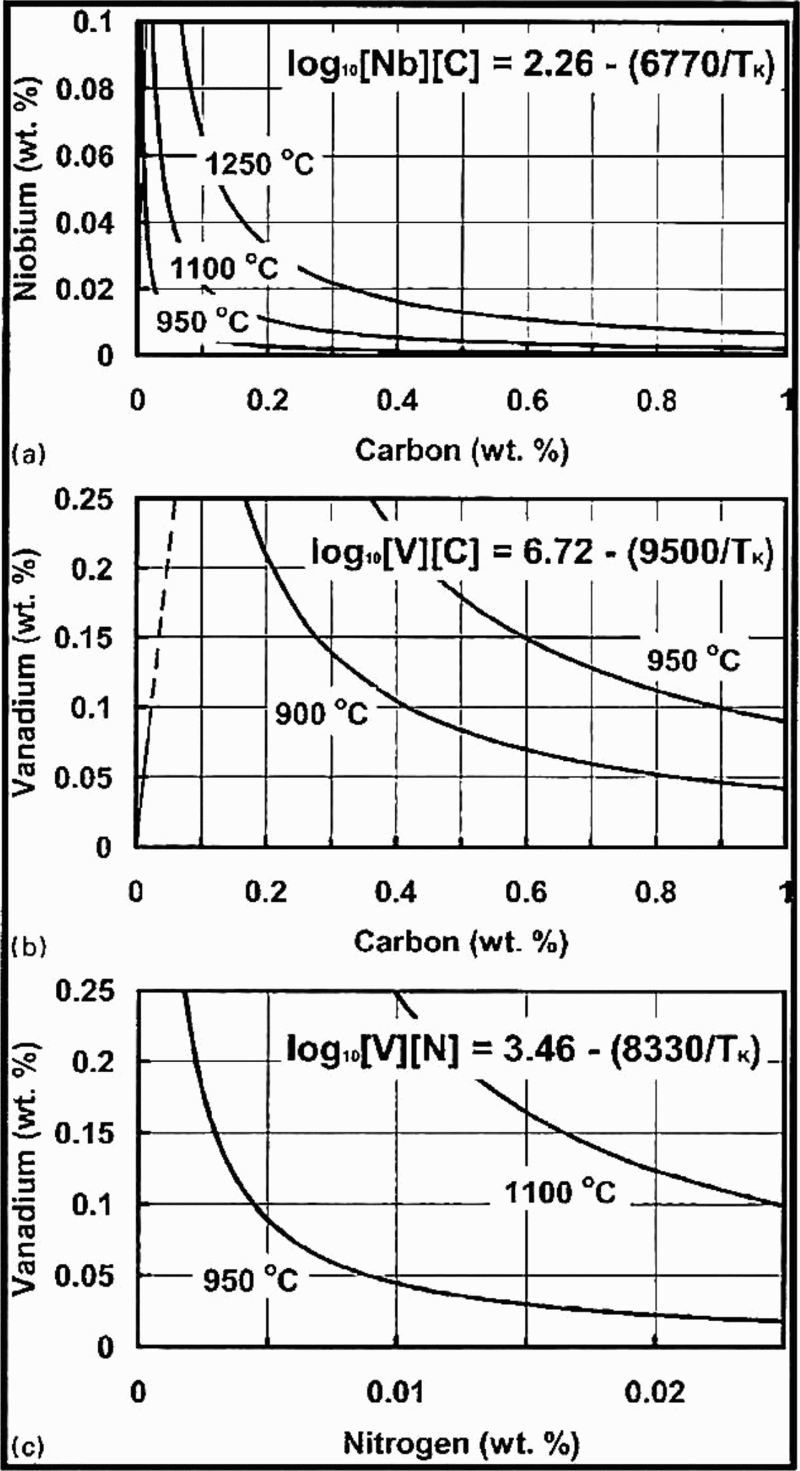
Calculated solubility diagrams at temperatures associated with thermomechanical processing for a NbC, b VC and c VN (after Matlock and Speer 80 )
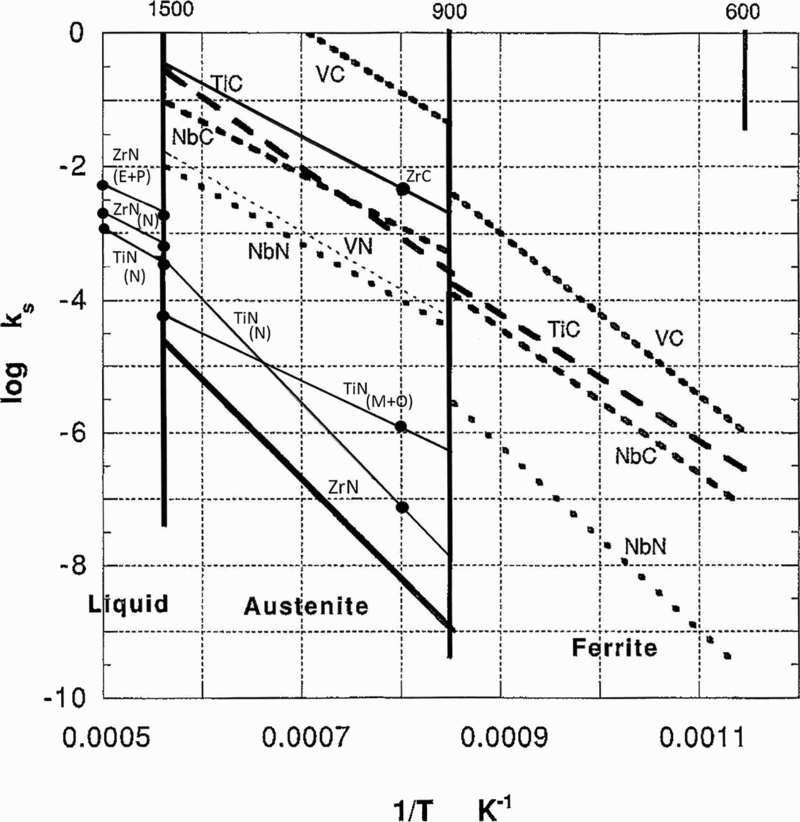
Comparison of solubility products of transition metal carbides and nitrides found in microalloyed (after Gladman 3 )
In general, nitrides are more soluble in austenite than carbides. Zirconium and titanium nitrides are the least soluble, while vanadium carbide is the most soluble. In ferrite, vanadium carbide is the most soluble, followed by titanium carbide and then niobium carbide, all of which, with high carbon/nitrogen ratios, could be important sources of dispersion hardening.
Narita 83 has given data on the recovery of the elements in groups IVA and VA, which are reproduced in Table 6. Both niobium and vanadium can be seen to have a higher per cent recovery than titanium and zirconium.
Recovery of elements in groups IVA and VA
Vervynckt et al. 7 considered that ‘for most steel grades, Tnr is determined by the niobium and carbon contents. One the other hand, titanium forms a very stable compound, TiN, which may remain undissolved in austenite. This compound consequently limits austenite grain growth at relatively high soaking temperatures and also restricts nitrogen from forming Nb (C, N), enabling dissolution of NbC to occur more readily.’
A term of importance in discussing particle pinning of grain boundaries is the austenite grain coarsening temperature T
c, which is the temperature where the pinning effect becomes ineffective. This condition considerably restricts grain growth. As would be expected, T
c is significantly lower than the solution temperature of the precipitate. There have been several studies
49,84–87
of the effect of carbides and nitrides on T
c. Figure 15, taken from Cuddy and Raley,
86
illustrates some results that clearly show the importance of NbCN and AlN compared with VC, which is confirmed in more recent work.
3
As a result of experimental studies, linear relations were obtained between T
c and the temperature for complete solution of the microalloying carbide or nitride T. This lead to the relationship (°C)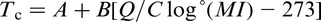
M and I are the metal and interstitial, A and B are the intercept and slope of the line segments in Fig. 15, while Q and C are constants, such as those given in Table 5.
Using these data for Q and C for AlN and NbC, the latter labelled NbCN by Cuddy and Raley, 86 who give the respective constants A and B as 285 and 460°C and 0.535 and 0.569 respectively, to allow the calculation of T c. From the work of Hall, 30 Petch 31 and Zener, 88 it is evident that ferrite grain size and the size and volume fraction of precipitated particles are of seminal importance in determining the mechanical properties of polycrystals. The volume fraction of particles is initially controlled by their solubility in austenite, where most of the deformation, which determines the final size and shape of the steel, is scheduled.
As acknowledged by Martin,
89
it is well established that dispersed, hard, incoherent particles can either retard or accelerate recrystallisation of a metallic matrix, and this was affirmed by the work of Docherty and Martin.
90
Zener
88
was the first to devise a relationship involving a dispersion of particles and the retarding force that they exerted on a grain boundary. This effect is known as the Zener drag after his original analysis and was first published by Smith; Zener proposed that the driving pressure for grain growth due the curvature of the grain boundary would be counteracted by a pinning (drag) pressure exerted by the particles on the boundary. Consequently, normal grain growth would be completely inhibited when the average grain size reached a critical maximum grain radius, also known as the Zener limit R
c, and is given by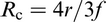
He considered that both grains and particles could be approximate to spheres.
In its general form, the Zener equation is given as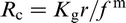
The model by Zener 88 has been shown to overestimate R when compared with experimental data overestimate R c (Ref. 1) when compared with other data. 91–93 The most extensive consideration of the many modifications proposed to the Zener equation has been undertaken by Manohar et al., 93 who examined in detail some 32 models published up to 1987.
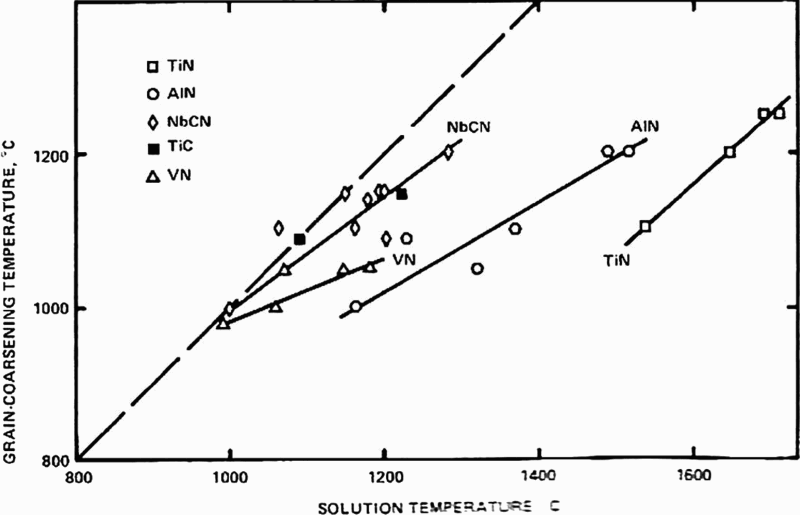
Relation between observed grain coarsening temperature of austenite and computed temperature for complete dissolution of microalloying carbides and nitrides in austenite 86
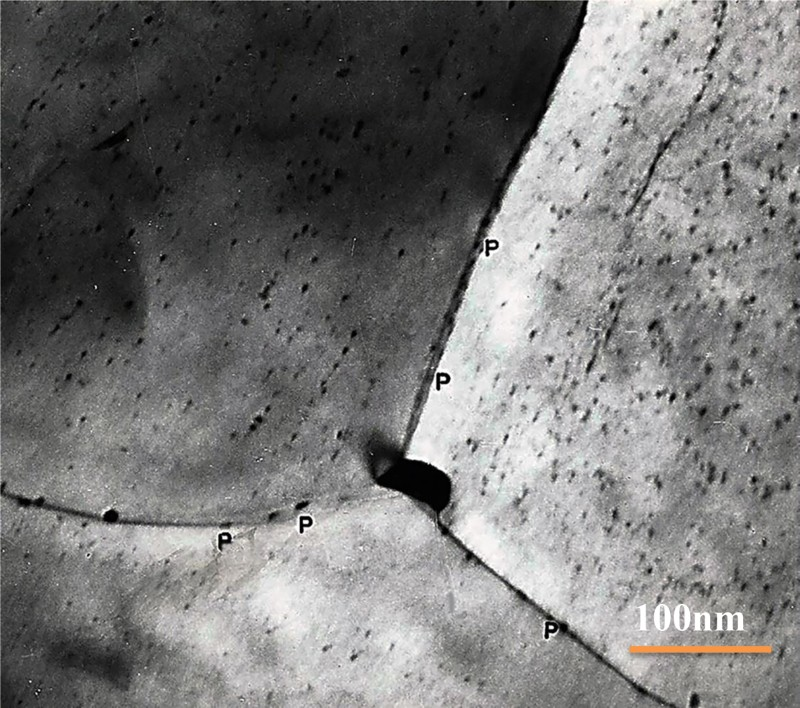
Thin foil of vanadium microalloyed steel, with 80 nm long particle at triple intersection of boundaries and smaller precipitates, ∼12 nm, at P, on boundaries, also smaller particles within grains, some associated with out of contrast dislocations
In general, the pinning of subgrain and high angle grain boundaries has been shown to occur when the particle radius r is in the size range of 30–800 nm and particle volume fraction f < 0.01. While data for R c and, to some extent, r are available, no reliable data on volume fraction of precipitates in these steels have been published. 13 Figures 16 and 17 show the effect of particles pinning grain boundaries. There also exists a body of experimental evidence to show that in steels, particles, particularly oxides and carbides >0.5 μm in length, with interparticle spacings, also >0.5 μm, can lead to acceleration of recrystallisation due to nucleation of new grains at carbide particles 94,95 and oxide slag inclusions. 96 These particles are assumed to create lattice curvature at particle/matrix interfaces in the deformed matrix, which enhances recrystallisation and gives rise to accelerated recrystallisation, also known as particle stimulated nucleation. 91
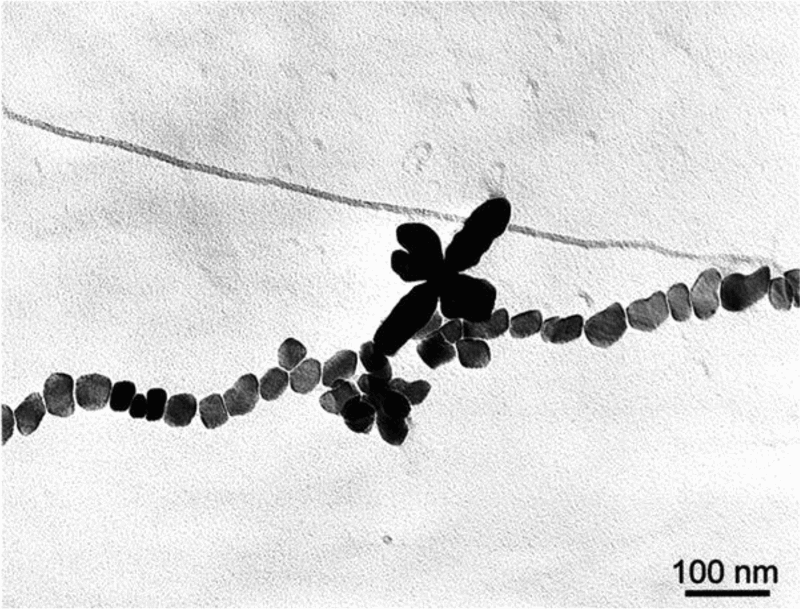
Carbon replica of vanadium carbonitride particles nucleated on prior austenite grain boundary 187
Precipitation and recrystallisation
As mentioned above, while restriction of grain growth by particle pinning initially takes place during the soaking stage of the rolling schedule, an equally important aspect of precipitation is that during the rolling, most of it occurs in the austenite phase in niobium steels. In vanadium steels, precipitation in ferrite also is important (Fig. 18). The inter-relationship between hot deformation and concurrent precipitation has been conveniently summarised by Pandit et al., 97 reproduced in part here: ‘The presence of precipitates increases the non-recrystallisation temperatures (Tnr), which is of significance in deciding the reheating temperature for hot rolling. 98 The presence of precipitates in austenite, increases the flow stress of the material and hence the rolling loads. Niobium is the most potent of the microalloying elements in retarding austenite recrystallisation through solute drag 98 and/or by SIP’. 99,100 Figure 19 shows that only NbC can have high supersaturations over a large portion of the typical hot rolling range. 6 It is well established that the onset of precipitation is greatly enhanced by prior deformation. A comparison of the precipitation kinetics of strain induced NbC particles with those in undeformed austenite reveals that at least two orders of magnitude difference occurs between the two, over a given temperature range. 101 This may be attributed to the presence of dislocations, deformation bands, subgrain structure and twins, the well known potent sites for nucleation that accelerate the precipitation process in the deformed microstructure. 101,102 The kinetics of SIP of niobium in MA steels has been extensively investigated for over 25 years by Sellars's group, who have compared their experimental data with modelling predictions with increasing success. 41,42,101–106 Recently, Nöhrer et al., 107 using a quenching dilatometer and TEM and APT techniques, considered in detail the influence of deformation on the behaviour of niobium precipitation in an MA steel containing 0.20C–1.29Mn–0.029Nb–0.035Al–0.004N and found that this was different in the austenite and ferrite regions. The steels were solution treated at 1250°C, cooled to a deformation temperature of 700°C in the austenite and ferrite region, held for 7 min to partially transform to ferrite, before being deformed to true strains of 0.7, 0.2 or 0.05, at a strain rate of 0.1 s− 1. After a second hold, the samples were gas quenched to room temperature (RT) within 5 s. It was found that the niobium precipitates nucleated as carbonitrides, taking up the total available nitrogen. Following deformation, with longer dwell times, increasing carbon levels were detected in the niobium precipitates. A higher volume fraction of precipitates, with a higher carbon content, was found in ferrite compared to austenite. With increasing strain, the dislocation density increased, resulting in a higher volume fraction of strain induced niobium precipitates, not through accelerated precipitate growth.

Dark field transmission electron micrograph showing dislocations interacting with precipitates in microalloyed steel
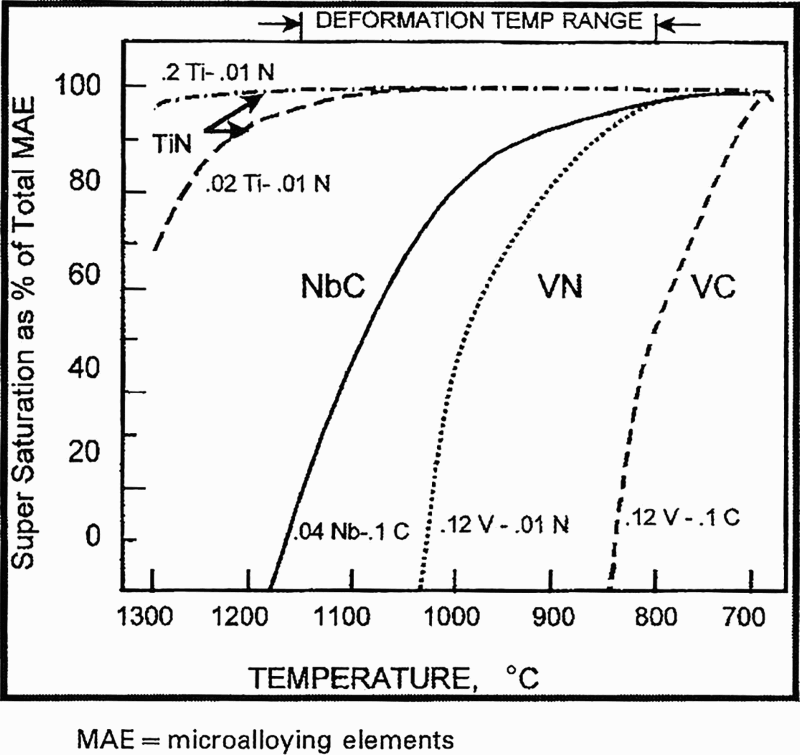
Precipitate potential for various microalloying systems 6
While an abundance of research has been reported on the SIP behaviour of Nb (C,N), less consideration has been given to the SIP of TiN and V(C,N) in austenite. The evolution of SIP of vanadium precipitates in an 0.20%C steel at 600 and 700°C was considered by Nohrer et al. 108 using the same techniques mentioned above. 107 They observed that the initial V(C,N) precipitates were higher in nitrogen than carbon, and this changed with increasing dwell time and occurred faster at 700°C, as in their niobium steel study. 107 Gomez et al. 109 considered more complex additions in the MA steels, e.g. 0.034Nb–0.004N (steel a), 0.092V–0.0065N (steel b), 0.018Ti–0.008N (steel c) and 0.037Al–0.010N (steel d), on recrystallisation/precipitation interactions, using hot torsion tests. Their results showed that steels a and b exhibited long inhibition plateaux, while steel d displayed a very short plateau; steel c did not show any plateau. This was interpreted as indicating that niobium and vanadium precipitates (nitrides and carbides) can inhibit the static recrystallisation, but this does not occur for the aluminium and titanium steels, which formed only nitrides. Recrystallisation–precipitation–time–temperature diagrams showed the interaction between both phenomena, along with the SIP kinetics and precipitate coarsening. Studies by TEM, based on the average precipitate size, found that AlN particles (87 nm) nucleated and grew faster than NbCN (22 nm) or VN (10.5 nm). TiN (1250 nm) in this work had a size dependent only on the solution temperature of 1300°C. The effect of a titanium addition on SIP of NbC in simulated deformed 0.047%Nb and 0.043Nb–0.016Ti steels was explored by Hong et al. 110 using two-stage interrupted compression tests. They found that the size of NbC formed in the Nb–Ti steel was smaller than in the Nb steel and that the precipitation start time in the Nb–Ti steel was delayed compared to that of the Nb steel. After reheating the Nb–Ti steel at 1250°C, undissolved Ti rich (Ti, Nb)(C,N) particles were located at prior austenite grain boundaries, resulting in a finer austenite grain size of 130 μm, compared with 180 μm for the Nb steel.
Medina 111 studied the influence of strain on precipitate nucleation in austenite comparing three Nb–V–N MA steels. Precipitation start–time–temperature diagrams were determined by hot torsion tests, while corresponding increases in dislocation density and the driving force for precipitation ΔG v were calculated from established equations. The results show that SIP in austenite is transformed from heterogeneous nucleation on dislocation nodes, when ΔG v is small ( − 1.8 × 10− 9 J m− 3), to homogeneous nucleation as the microalloying content, and therefore, ΔG v both increased ( − 2.48 × 10− 9 J m− 3). ΔG v was obtained by considering that niobium forms as carbonitrides and vanadium as nitrides. However, in many cases, (Nb,V)(C,N), of varying compositions, after removing from different stages in a process, have been characterised. In this case, no such work was undertaken by Medina. 111 It is important that the assumption that Nb(C, N) and VN have separate roles in this work is confirmed experimentally.
Two more complex steels of compositions, i.e. Nb–V, 0.19C–1.5Mn–0.44Si–0.1Cr–0.125V–0.035Nb–88 ppm N–0.008Al, and Ti–V, 0.18C–1.5Mn–0.44Si–0.09Cr–0.13V–0.036Ti–80 ppm N–0.008Al, were compared by Pandit et al., 97 who carried out mechanical relaxation tests using a Gleeble 1500 thermomechanical simulator. Holding specimens at the reheating temperature of 1200°C for 5 min was sufficient to dissolve all the niobium, but only part of the titanium. Deformation was carried out in austenite, between 1090 and 900°C. Both Datta and Sellars 102 and Liu and Jonas 112 suggested that SIP in deformed austenite occurs mostly on dislocations and sub-boundaries, which implies that the austenite grain size per se is not a dominant factor in influencing precipitation kinetics; Pandit et al. 97 were able to show from their data (Fig. 20) that the precipitation kinetics of the Ti–V steel was more sluggish than the Nb–V steel.
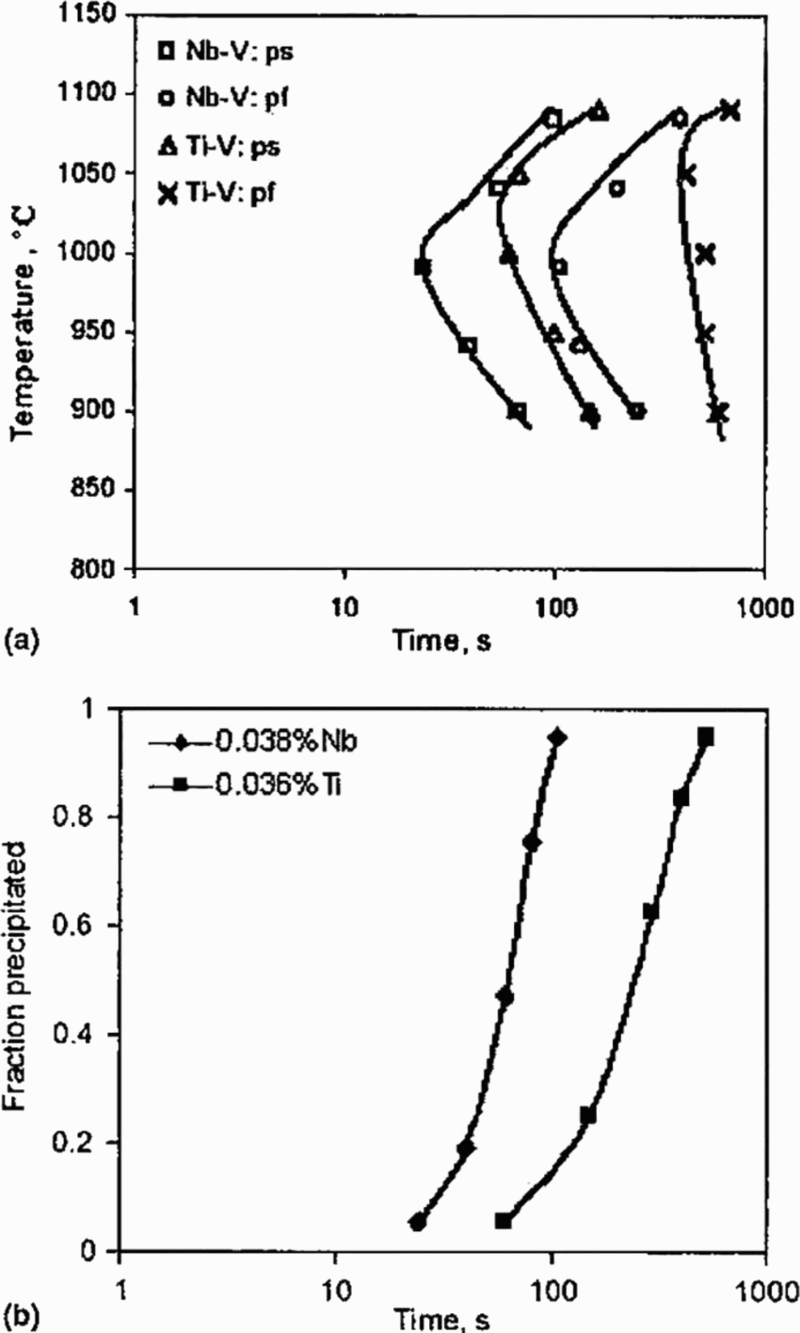
Comparison of precipitation kinetics for Nb–V and Ti–V steels 97 (ps: precipitate start; pf: precipitate finish)
Multiple additions of niobium, titanium, vanadium zirconium and nitrogen in MA steels
The year 1980 was considered by DeArdo et al. 113 to be a watershed for MA steels. Before 1980, MA steels were of low hardenability with FP microstructures and, after air cooling following hot rolling, had σy values of ∼420 MPa. Around 1980, both the linepipe and automotive industries required steels with higher strengths than could readily be obtained with FP microstructures. Increasing the microalloying content to enhance the dispersion strengthening contribution from the range 50 to 80 MPa, recorded for FP steels, seemed limited. This magnitude is comparable to the strengthening conferred by solid solution strengthening, but significantly below of 100–480 MPa levels, which can be obtained from dislocation strengthening. 1,2,114,115
One of the main developments that has occurred over the past 25 years to raise the yield strength is the increasing tendency for steelmakers to alloy with more than one of the niobium, titanium, vanadium trio of transition metals, often with a deliberate higher nitrogen levels than the normal range of 0.003–0.005%N. This has given rise to fine ferrite grain sizes of < 10 μm and to complex particles, which may precipitate in either or both the austenite and ferrite phases, depending on the chemical composition of the steel and the details of the TMP route adopted. However, multiple additions are not a recent move, as they were considered over 45 years ago by, for example, Heisterkamp et al. 116 They investigated 19 steels, 14 of which contained zirconium, with either single additions at levels of nominally 0.043, 0.057, 0.072% or in combination with two levels of niobium, 0.020 or 0.035%. These steels were compared with five vanadium–zirconium steels arranged in two groups: one of three steels based on 0.030%V containing respectively 0.019 and 0.035% and 0.057%Zr and a second of two steels with 0.049%V, and 0.053 or 0.092%Zr. The steels were examined in the as rolled and normalised conditions. Little effect of zirconium additions was found on the strength of the niobium–zirconium steels, the as rolled strip having σy of ∼500 MPa, which is ∼20% greater than the 420 MPa mentioned above. Furthermore, a decrease from 460 to ∼420 MPa was found when a zirconium addition was present with 0.030–0.050% vanadium.
In controlled rolled MA steels, some attention has been paid to the combination of additions of titanium and vanadium, titanium and niobium or titanium and niobium and vanadium, with the expectation that the potential of each element will be fully exploited. Most of the work discussed below emphasise that titanium additions are normally made as hypostoichiometric relative to nitrogen, i.e. < 3.4. This ratio is discussed by Crowther and Morrison, 117 who explained the effect that titanium additions were observed to have with Al–V and Al–Nb MA steels, particularly with regard to loss of strength. This was purported to be due to changes in the dispersion strengthening associated with modifications to niobium precipitates.
Strid and Easterling 118 used STEM-EDX microanalysis to understand the influence on microstructure, of precipitate compositions, morphologies and sizes in hot rolled MA steels containing Ti, Ti–V and Ti–Nb, with different levels of nitrogen. They found that the median ferrite grain sizes in the Ti–Nb steels, 29 and 32.5 μm, was greater than in the Ti (∼16 μm) and Ti–V (13 μm) steels. In addition, the median particle size, 19 nm, of an Ti–Nb–0.010N steel containing predominantly nitrides showed a coarser size distribution than in a Ti–Nb–0.006N steel, where the median particle size of predominantly carbides, decreased to 10 nm. Increasing the aluminium content of their Ti and Ti–V steels from 0.022 to 0.072 wt-% resulted in an increase in the particle size from ∼8 to ∼12 nm. These changes had only a minor influence on the yield strength, which reached 377 MPa for the steels with Ti and Ti–V additions, but was not given for those with Ti–Nb. Strid and Easterling 118 were unable to quantify the C or N levels in this work, and therefore, details of the chemical compositions of the particles were restricted to the transition metals. This problem was overcome to some extent by He and Baker, 119 who were perhaps the first to use both TEM-EDX, in an EM400, and electron energy loss spectroscopy (EELS) in an HB5 instrument, to characterise their carbonitride precipitates present in an MA steel. They attempted to elucidate the problems associated with additions of titanium in MA steels, which had been reported to have differing effects when used with additions of niobium compared to vanadium. Following controlled rolling, three steels containing nominally 0.10C–1.4Mn–0.005N–0.017Nb, with respectively < 0.005, 0.010 or 0.022Ti, were examined. For the different titanium additions, giving different Ti/N ratios, a distinct size variation of the Ti–Nb carbides and nitrides was observed, especially for the coarser particles. Figure 21a includes large Ti–Nb particles in an 0.01%Ti–Nb steel, with many smaller Nb–rich spheroids, while an EELS spectrum, Fig. 21b collected from the centre of a ∼20 nm Nb rich spheroid, shows the niobium, carbon, nitrogen and titanium edges. 119 Using a similar range of techniques to He and Baker, 119 Subramanian and Weatherly 120 investigated the precipitation behaviour in Ti–V and Ti–Nb steels, but containing a higher carbon level of 0.426. The presence of complex precipitates in as cast blooms was confirmed; in the Ti–V steel, nitrides (TixV1 − x)N were observed followed at lower temperatures by carbides (V4C3); in the Ti–Nb linepipe steel processed from slabs, dense well dispersed mixed nitrides (TixNb1 − x), on which epitaxial growth of Nb rich mixed carbides, were observed. The authors considered that epitaxial growth obviated the need for SIP of mixed carbides. Here, the increase in the volume fraction of precipitates increased the Zener drag force on boundary mobility, thereby retarding recrystallisation. Similar results were found by He and Baker 121–124 studying controlled rolled ∼0.10C steels containing additions of Nb–Ti–N or Nb–Ti–Zr–N. They observed NbC or Nb(C,N) caps or coatings to nucleate on (Ti,Nb)N core particles (Fig. 22), which reduced the niobium available for both grain refinement and dispersion strengthening. A 0.016Zr addition resulted in complex nitrides >100 nm size of Ti–Zr and Ti–Nb–Zr, which appeared to have no effect on < 100 nm Ti–Nb carbonitrides, also present. All the steels had σy values ∼400 MPa with 50 J IIT of − 80 to 100°C. The presence of caps nucleating on more stable core precipitates has been reported more recently. Caps of Nb rich (Nb,Ti)C were also observed to nucleate heterogeneously on undissolved (Ti, Nb)(C,N) core particles by Hong et al. 110 and by Grajcar. 125
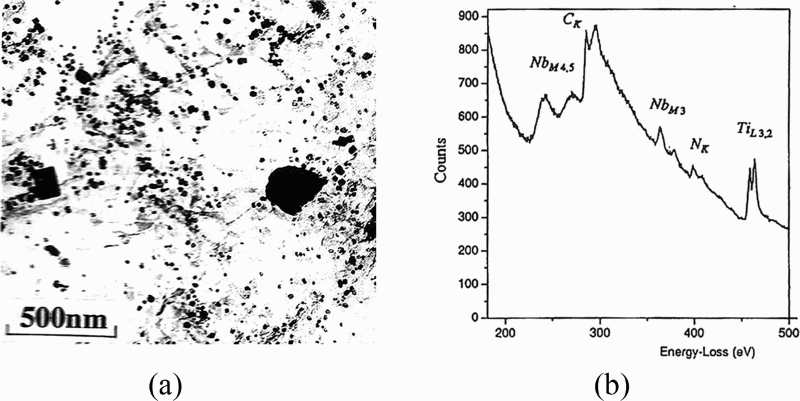
a Ti–Nb particles in 0.01%Ti, showing smaller Nb rich spheroids and b EELS spectrum collected from centre of ∼20 nm Nb rich spheroid showing niobium, carbon nitrogen and titanium edges 119

a four caps formed on sides of cuboid core with probability that there are also caps on top and bottom faces and b cruciform particle with four arms nucleating and growing from core
In another work, Beres et al. 126 found that a 0.03C–0.08Nb–0.01Ti 20 mm thick plate steel ‘produced complex agglomerates with a cubic TiN seed crystal overgrown by a cubic NbC particle’. In some of the studies where core/caps particles were observed, the choice of reheating temperature before processing has been too low to take all of the core precipitate, normally titanium or zirconium based, into solution. While the resultant conglomerate may affect the rate of grain growth, in general, these particles should be avoided through relating the steel composition to higher reheating temperatures.
Courtois et al., 127 who studied model Fe–(Nb,C) and Fe–(Nb,C,N) ferritic alloys, showed that ‘the addition of nitrogen lead to a complex precipitation sequence, with the co-existence of two populations of particles, pure nitrides and homogeneous carbonitrides, respectively.’ In another work, a comparison of precipitates in Nb–Ti and Nb–Ti–V MA X80 pipeline steels produced as 18.4 mm thick strip was undertaken by Li et al. 128 Their microstructures consisted of quasi-acicular ferrite (AF).
Perhaps the first researchers to consider moving from F-P to A-F microstructures were Smith and Coldren, 129 who investigated the microstructure and properties of 36 laboratory steels. Among the most successful were steels based on a composition of 0.4C–1.6Mn–0.09Nb–0.2Mo, FR at 870°C, spray cooled at ∼22°C s− 1 to a simulated coiling temperature of 635°C, the approximate temperature for AF formation. This schedule resulted in a yield strength of ∼570 MPa and one-half size Charpy transition temperature of − 60°C.
Some of the more recent works, which lead to the gradual raising of the yield strength by controlled rolling MA steels, was summarised by Ji et al. 130 Campos et al. 131,132 developed yield stress of 530–608 MPa respectively using nominal chemical compositions of 0.12C–0.057Nb–0.049Ti and 0.11C–0.04Nb–0.11Ti. In addition, the precipitation behaviour in a 0.07C–0.086Nb–0.047Ti steel was investigated by Charleux et al., 133 who obtained σy levels of 650 MPa in a plate. Misra et al. 134 developed a steel with σy of 770 MPa, from a complex steel composition of 0.06C, 0.07–0.09Nb, 0.065–0.085Ti, 0.1–0.2Mo and 0.0005–0.001B.
As pointed out by Ji et al.,
130
these highly alloyed steels detailed above achieved their yield strength at a relatively high cost. The recent approach of Ji et al.
− 1
compared two laboratory steels of compositions based on 0.09C–0.11Ti and 0.05C–0.025Nb–0.11Ti. Both steels reached σy values of >700 MPa with >20% elongation and good toughness; it should be pointed out that one steel was without a niobium addition. The plate FRT was controlled to 910°C and then cooled at 80°C s− 1 to the coiling temperature of 620°C, using an ultrafast cooling technique, producing a microstructure that was predominantly of massive ferrite and AF, with ∼3.2 nm TiC precipitates in the Ti steel and ∼5.0 nm (Ti,Nb)C particles in the Ti–Nb steel. This kind of microstructure has been further considered in a series of papers involving Shan and co-workers,
135–138
who explored the AF route to achieving X60 to X100 pipeline steels. The laboratory developed AF pipeline steel was based on their CCT diagrams of an experimental steel, which was vacuum melted, reheated at 1150°C for 50 min, started rolling T
s, at 1050°C and finished rolling T
f at 750°C, starting from a thickness of 70 mm and reducing in seven steps to 7 mm, then cooled to a finish cooling temperature T
c of 600°C at a cooling rate V
c of 20°C s− 1 in order to obtain the AF microstructure. The steel was finally held at 600°C for 1 h and furnace cooled to RT, to simulate the coiling process. The commercial AF pipeline steel was reheated at 1180°C for 230 min and then rolled in two stages, rough rolling and finish rolling. The rough rolling stage for the steel reduced from a thickness of 230–40 mm, in seven steps with T
s 1130°C; then, the finish rolling stage started at 1020°C with T
f at 840°C, reducing from 40 to 7 mm in another seven steps. Finally, the steel was cooled to 600°C at 20°C s− 1 for coiling and then air cooled to RT. With the laboratory steel,
134
which had a nominal composition of 0.07C–0.9Mn–0.04Nb–0.04V–0.015Ti–0.004N, they found that the properties were dependent on T
s, T
f, T
c and V
c. Optimum mechanical properties were obtained when T
s was ∼1100°C, T
f was 890°C, T
c was 520°C and V
c was 30°C s− 1. Niobium in the steel was considered to have a stronger influence on T
s compared with vanadium and titanium, in controlling mechanical properties; Nb carbonitrides form in the range close to T
s of 1070–1120°C; V carbonitrides will mostly dissolve in γ-Fe ∼900°C, while the formation temperature of Ti carbonitrides is far higher than T
s. Thus, an increase in T
s will mainly place more niobium in the solution, which raises σy to a maximum of 561 MPa. Regression equations, which described the relationships between σy and elongation El and the processing parameters, were produced from the data obtained from the laboratory steels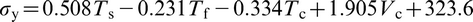
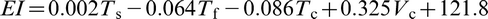
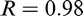
A comparison made between the same laboratory X90 grade AF steel, containing 0.025C–0.058Nb/Ti and 0.0062N, with a commercial X70 grade containing 0.08C–0.095Nb/Ti and 0.005N. It was concluded that the higher σy (626/530 MPa) and better toughness (at − 100°C, 137/108 J) in the acicular steel resulted from its finer grain size and higher dislocation density and sub-boundary content. 138 The above laboratory X90 grade steel was processed to produce either an AF or an ultrafine grain microstructure, which allowed a comparison to be made of the two microstructures and properties. The dispersed (Ti,Nb)(C,N) precipitates, together with a high density of tangled dislocations and the finer cleavage facet size ferrite bundle, in the AF steel was considered to be a better candidate microstructure for oil and gas pipeline steels than the ultrafine ferrite steel. 136 In their review, Beladi et al. 139 have also considered alternative methods to develop high strength steels at a lower cost. Their most promising route to achieving this goal was by reducing the alloy content of the steel and relying on a reduction in the grain size to provide higher strength and toughness. It is pointed out 139 that the Hall–Petch equation (equation (1)) predicts that a reduction in the average grain size from 5 to 1 μm will increase σy by ∼350 MPa. However, using conventional industrial approaches, the level of ferrite refinement has been found historically to be limited to 5–10 μm. Nowadays, several thermomechanical processes have been developed to produce ferrite grain sizes of ≤ 1–3 μm, ranging from extreme thermal and deformation cycles to more typical thermomechanical processes. Of a number of routes established to achieve a fine grain size in the laboratory, dynamic strain induced transformation (DSIT) was considered by Beladi et al. 139 as the most promising method for adapting to commercial practice. Dynamic strain induced transformation is based on the discovery, by Hodgson et al., 140 for the formation of ultrafine grains during the hot deformation of thin strip. This development requires deformation to large strains within the Ae 3 to Ar 1 temperature range for a given alloy, followed by rapid cooling. 141,142 The formation of ultrafine ferrite involves the dynamic transformation of a significant volume fraction of austenite to ferrite, which initially occurred at an early stage of deformation, at prior austenite grain boundaries, and was followed by intragranular nucleation, as in controlled rolling. 139 However, DSIT arises from the introduction of extensive extra intragranular nucleation sites, which are not present in conventional controlled rolling. They were found in an 0.17C–1.5Mn–0.02V steel. 143 Priestner and de los Rios 78,144 were among the first to recognise that extra grain refinement may be possible if the transformation could be induced during the deformation process. The final microstructure of DSIT low carbon steels usually consists of fine equiaxed ferrite grains ( < 2 μm) and cementite (typically 0.05–0.2 μm) in the grain interiors and at the grain boundaries (Fig. 23). Most of the yield strength values in Fig. 24, collated by Beladi et al., 139 lie within the 400–700 MPa range, the upper end being beyond what most FP MA steels achieve. However, the commercial exploitation of the DSIT route is still a work in progress. 142,145 Recently, a guide to industrial production of ultrafine grain MA steel was published. 146 It is based on DSIT, using experimental data to aid the development of a multiscale computer model, which predicted the mechanical response of the microstructure in a hot rolling process. Two steel grades were examined: 0.07C–1.36Mn–0.06Nb–0.03Ti–0.098N–0.003B and 0.08C–1.67Mn–0.06Nb–0.018Ti–0.0316N–0.26Mo. Niobium was the principal alloying element used to control the dynamic transformation of austenite, while the additions of B or Mo increased significantly the hardenability, thereby expanding the thermomechanical processing window. However, while these steels can achieve higher σy levels than FP MA steels, they still rely on expensive alloy additions. Others claim that it is possible to obtain an ultrafine grain microstructure without such alloying, 147 but it is not known if this route is used commercially. As mentioned above, small amounts of boron when added to ultralow carbon steels promoted bainite by suppressing the formation of polygonal ferrite.

Ultrafine ferrite on rolled strip surface in low carbon steel formed through DSIT route using single pass rolling (after Hodgson et al. 141 )
Effect of microstructure on mechanical properties of UFF steels produced through DSIT mechanism (after Beladi et al. 139 )
Tamehiro et al., 148 in studying TMP with accelerated cooling, considered the effect of a combined addition of boron and a grain refining element such as niobium, titanium or vanadium in very low carbon steels, in terms of mechanical properties and microstructures. They found that, although boron, as a single addition, has little effect on the properties, the combined addition with niobium improves the balance of strength and toughness. Niobium additions to boron steels suppressed the precipitation of Fe23(C, B)6 and strongly retarded the γ–α transformation, forming a fine grained bainitic structure. Titanium has been found to have the same effect as that of niobium, but vanadium does not. The strengthening and toughening mechanism by the combined addition of niobium and boron was also investigated by examining the effect of alloying elements on the minimum recrystallisation temperature of austenite during rolling, and γ–α transformation behaviour. 148
High strength/bainitic/martensitic MA steels
Acicular ferrite and conventional bainite (CB) have been mentioned in the previous section, but the difference between these two phases needs to be clarified. 149,150 Fig. 25 shows a large austenite grain size containing a relatively high number density of intragranular nucleation sites, which leads to a microstructure consisting predominantly of AF. By comparison, a small austenite grain size, containing a relatively high number density of grain boundary nucleation sites, leads to a microstructure consisting predominantly of CB. 149 As indicated by Zhang and Boyd, 150 ‘CB nucleates at prior austenite boundaries and grows as packets of parallel ferrite plates, appearing as laths in 2D, which have similar crystallographic orientation and a high uniform dislocation density. The length of the ferrite laths is limited by impingement with other packets, and carbon-enriched interlath austenite becomes a martensite–austenite (M/A) constituent.’ By contrast, AF can be nucleated by inclusions and grows as randomly oriented ferrite laths, or groups of laths, which contain a high dislocation density, and the MA usually occurs as discrete particles. This less organised aspect provides the potential for AF combining high strength with high toughness, and is the reason of some for developing AF microstructures. 151 A crack, therefore, has to follow a more tortuous path through an AF microstructure, thereby leading to an improvement in toughness without compromising strength. 151,152 Bainite has traditionally been separated into two groups (Fig. 26): granular bainite, often described as lower bainite, which consists of sheaves of elongated ferrite crystals with low misorientations and a high dislocation density, containing roughly equiaxed islands of M/A microconstituent; and lath bainite, often described as upper bainite, which consists of packets of parallel ferrite laths (or plates) separated by low angle boundaries and containing very high dislocation densities. In contrast to granular bainite, the M/A particles retained between the ferrite crystals in lath bainite have an acicular morphology. 153
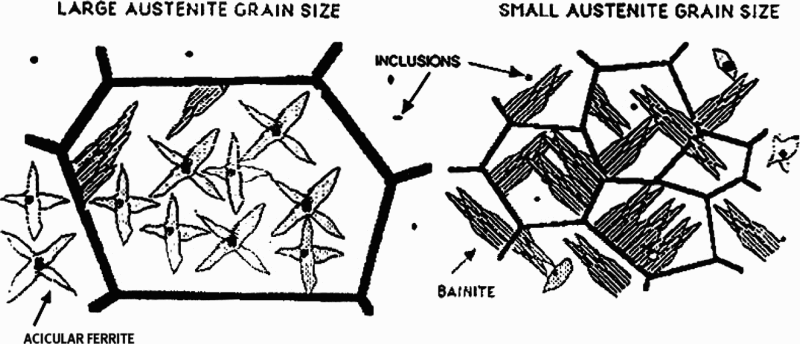
Effect of austenite grain size in determining whether microstructure is predominantly acicular ferrite or bainite 149
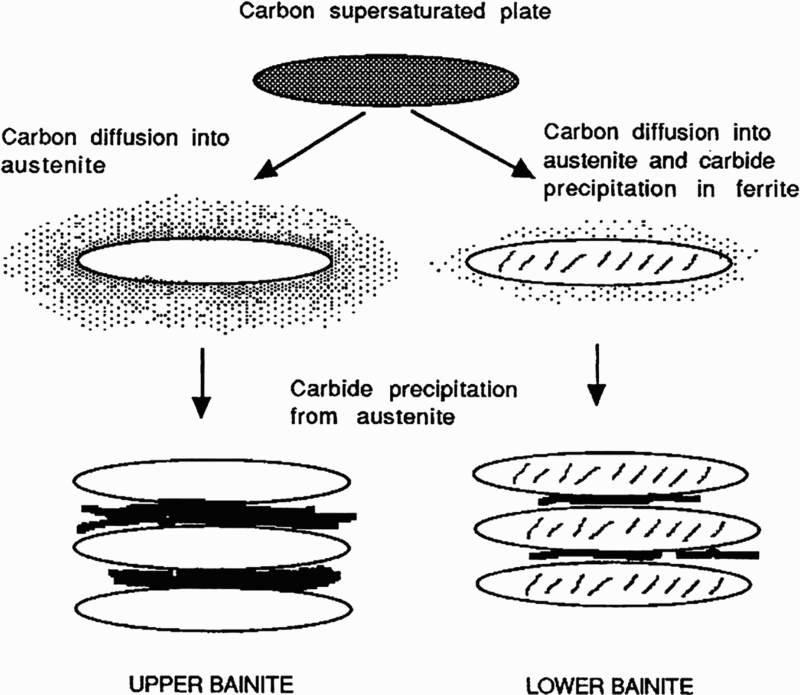
Schematic representation of transition between upper and lower bainite 149
The M/A particles can significantly affect fracture toughness due to the brittle nature of martensite. Not only is the phase fraction of importance but also the M/A morphology (Fig. 27). Most of the detailed studies of the morphology of M/A phase have been conducted on weld heat affected zone (HAZ) microstructures, for example Refs. 154–156. Li and Baker 155 reported four different morphologies of M/A particles, including blocky-like particles, or islands, connected or nearly connected particles along prior austenite grain boundaries, elongated stringer particles along bainitic ferrite laths and M/A-C particles, consisting of M/A and a second phase, i.e. carbide and ferrite. The M/A islands were reported with sizes between 0.5 and 5 μm, and elongated particles were found with a length of up to 10 μm and a width between 0.2 and 2 μm. 155,156 In addition to cracks initiated by M/A constituents, it is also reported that M/A particles can delay the growth of cracks. 156 This observation indicates a very complex behaviour of the influence of M/A particles on fracture. More recently, Reichert et al. 156 have investigated the formation of M/A in X80 linepipe steel using a Gleeble simulator.

SEM micrographs of simulated ICCGHAZ microstructures a showing void between two closely spaced MA particles: two cracks at C have not propagated. b MA + upper bainitic structure c crack stopped by MA particle 155
While it is accepted that bainite grows via a displacive mechanism, there is still much discussion on the diffusion or diffusionless nature of bainite. Caballero et al. 157 used APT to track the atom distributions during the bainite reaction in steels with different carbon and silicon contents. The steels were transformed over a wide range of temperatures (200–525°C) to elucidate the role of reaction rate and diffusion in the formation of bainite with and without cementite precipitation. From their studies, they concluded that the transformation is diffusionless, but carbon atoms partition and form clusters in the residual austenite (or precipitate as carbides), shortly after growth is arrested. The precipitation of carbides is therefore a secondary event.
The means of achieving these bainitic/martensitic microstructures in MA steels, is by water cooling after hot rolling. For processing plate, interrupted accelerated cooling and IDQ were introduced in plate mills. 63–65 As recounted by DeArdo et al. 6 , ‘run-out table water spray, cooling to the coiling temperature, had been in practice in strip mills since the 1960s, but not as a microstructural control tool for increasing strength.’ To achieve a yield strength ∼700 MPa, steels should have an adequate hardenability (carbon equivalent CE>0.50, P cm>0.20) and be controlled rolled, followed by IDQ to below the bainite start B s temperature of the pancaked austenite. With controlled rolled FP steels having a maximum yield strength of < 700 MPa, the way forward to higher strengths lay with steels having lower transformation microstructures, such as non-polygonal ferrite, AF, the bainites (B) and martensite (M), either individually or in combination. 6
While processing using accelerated cooling after controlled rolling, normally had the aim of developing AF/bainitic microstructures, other studies have been conducted on steels having sufficient alloy content to produce martensitic microstructures following direct quenched (DQ). Two papers published in the early 1990s provide good descriptions, and a collection of references pertinent to this route, for achieving proof stress levels of up to 1100 MPa. Taylor and Hansen 158 studied the structure and mechanical properties of a series of TMP-DQ martensitic laboratory steels, 0.1C–1.4Mn–0.5Mo–0.035Ti–0.0096N–B containing 0–0.24V. Titanium was added to stabilise the nitrogen as TiN. Figure 28 shows schematically the three processing routes investigated, together with reheated and quenched samples, to simulate the standard production practice for Q&T steels. This latter process involved austenitising at 920°C for 1 h and WQ as for the DQ samples. The average cooling rate, between 800 and 500°C, at the plate centre during cooling, varied from 45 and 52°C s− 1.
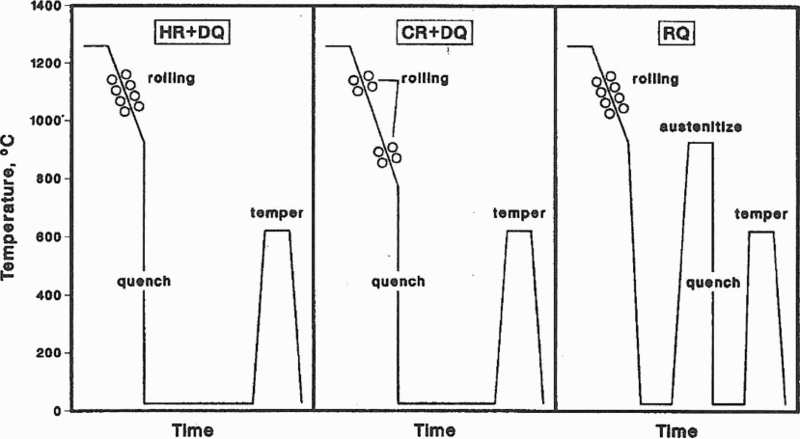
Schematic temperature/time profiles for hot rolled and direct quenched (HR+DQ), control rolled and direct quenched (CR+DQ) and reheat quenched (RQ) laboratory plates 158
Figure 29 shows that, after tempering at 620°C for hours, the vanadium containing steels exhibited markedly higher strengths than their vanadium free counterparts, while overall, the CR+DQ steels produced the best combination of strength and toughness. This was attributed to the precipitation during tempering of mainly spherical 2–4 nm diameter (46%V, 52%Mo, 3%Ti) carbides, providing an estimated 100 MPa/0.1 wt-%V, over the range of vanadium additions investigated.
The effect of variations in process route and processing variables is also well illustrated in the paper by Weiss and Thompson, 159 who studied similar steels to Taylor and Hansen, 158 with either 0.12C or 0.19C and 1.4Mn, 0.3Mo, ∼0.002B, 0.02–0.03Ti and 0.006N with and without 0.06V. Their goal was to vary the condition of the austenite phase before quenching by changing the process route, in a similar manner to Taylor and Hansen, 158 except the cooling rate during DQ, at ∼25°C s− 1, was lower. Figure 29 compares both sets of data in terms of strength and toughness, which clearly show the advantage of the CR+DQ process. Here, the precipitates in the 625°C tempered state were characterised by select area electron diffraction (SAED) as cementite and Ti(C,N), considered to be remaining out of solution following heating before rolling. As no EDX analysis was undertaken, the Ti(C,N) was probably what Taylor and Hansen 158 analysed as (V,Mo,Ti)C, as these particles in both papers are the same size order of ∼5 nm. More recently, with a steel microstructure of martensite and a little AF, Xie et al. 17 obtained a yield stress of 1144 MPa combined with an impact absorbed energy of one-fourth size Charpy specimen of 17 J at − 20°C. The laboratory steel containing (0.08–0.11)C–0.4Si–1.0 to 1.9Mn–0.8Cr–0.12(Nb+V + Ti)–0.0013B was soaked at 1200°C, air cooled to 950°C, rolled to 6 mm, FR at 840°C, water cooled at 80°C s− 1 to ∼260–280°C, followed by furnace cooling to RT.
However, while martensitic steels achieve significantly higher strengths, but with some loss of toughness, than FP steels, the additional alloying costs, together with those associated with processing via controlled rolling followed by quenching and tempering, which is not an online process for most plate thicknesses, have significant economic demerits.
The advantages of a bainitic microstructure on toughness have been discussed by Edmonds and Cochrane. 160 Over the past two decades, the formation of bainite has been recognised as a diffusion assisted, shear type (displacive) transformation, with excess carbon partitioning by diffusion to highly concentrated islands of austenite, A, in the B–F microstructure, either within the ferrite grains or between the grains. In addition, a second form of MA is caused by incomplete transformation of A to B, but with a lower carbon content. Therefore, there are two sources of MA in DQ steels: that from carbon partitioning during the B–F formation and that associated with untransformed austenite. Owing to the enrichment factor, one might expect the former to be of higher carbon content than the latter. These islands would subsequently transform to a high carbon martensite on cooling. 11,150 Such microstructures are also known to show improved toughness. 152,160
The early work at the British Iron and Steel Association, Sheffield, circa 1970, reported the advantages of superior strength while maintaining good impact resistance of MA steels that were rapidly cooled immediately after hot deformation, compared with the same steels that had been air cooled after rolling. 161–163 This change in processing route produced a different microstructure from the FP associated with conventional controlled rolling. 63–65 These non-FP steels were originally developed as laboratory experimental steels, at a time when both nitrogen additions ≥ 0.005 and the development of bainitic microstructures were generally regarded as deleterious to toughness. Tither et al. 163 concluded that, by utilising the direct quenching process, the near maximum secondary hardening potential of steels could be achieved.
More recently, several research groups have sought to raise the yield strength of MA steels without producing deleterious effects on toughness and ductility. Steels containing separate additions of MA steels containing vanadium and nitrogen were investigated by Lagneborg et al., 10,11,164,165 who explored the effect of both nitrogen and cooling rate on properties. They considered a process based on a repeated recrystallisation of austenite. Figure 30 shows that ‘the steel with higher nitrogen content is significantly stronger than that with a low nitrogen processed in the same manner, although with some sacrifice of toughness. It can be seen that the strengthening potential of vanadium can be effectively utilised only at higher levels of nitrogen and that increased cooling rates have a profound effect, especially at these higher levels.’ 11 In these steels, the titanium addition forming TiN will influence the austenite grain size, while the remaining nitrogen combines with vanadium to form V(C,N), which is designed to nucleate in ferrite during cooling after rolling. The finishing accelerated cooling temperature and cooling rate have a significant influence on the properties of MA steels, normally increasing yield strength when finishing accelerated cooling temperature decreased to 500°C or lower. In Ti–V–N and Ti–V–Nb steels, as the cooling rate increased from 5 to 15°C s− 1, a greater volume fraction of both bainite and small particles with a reduced particle spacing was observed, the latter producing an increased dispersion strengthening contribution to the yield strength. 11,166 Further exploratory laboratory work by Siwecki et al. 167 was centred on dilatometry, with the aim of producing yield strengths >600 MPa in 8 mm strip, directly in the mill. To stimulate bainite formation and simultaneously avoid ferrite/pearlite, a composition based on 1.4Mn–1.0Cr–0.25Mo was chosen to provide adequate hardenability combined with a cooling rate of 30°C s− 1 between 600 and 400°C. Satisfying these conditions for a steel with 0.08V and 0.010–0.020N gave yield strengths between740 and 790 MPa by the simulations. Raising the chromium level from 1 to 2%, with the same compositions, increased the yield strengths to 848–880 MPa, or alternatively allowed a lower cooling rate of 20°C s− 1, which is associated with 12 mm thick strip production in commercial mills, to give yield strengths reaching 780 MPa. These strengths developed due to the arresting of the normal softening effect due to tempering of bainite during coiling, resulting in a ferrite lath width of 0.5–1.0 μm, in which a high dislocation density was retained by a dense precipitation of V(C,N) in the ferrite. A recent paper from the same group 168 extended these laboratory simulations further in the full scale commercial hot rolling of 8 mm thick strip to give yield/proof strengths >700 MPa. These targets were all met or exceeded for the selected single trial steel composition: 0.042C–1.55Mn–1.0Cr–0.30Mo–0.024Al–0.012N–0.079V. The additional requirements of good ductility (total El>11%), toughness down to less than or equal to − 40°C and adequate weldability were achieved.
Effect of nitrogen, vanadium and cooling rate on yield stress and impact toughness of Ti–V–Nb–N steels after rapid cooling process 11
A series of papers by Misra et al. 17,134,169,170 have made significant contributions to understanding the development of microstructures essential to attain yield strengths eventually reaching >1000 MPa. This σy permits the use of a reduced strip thickness in automotive weight reduction applications and in pipeline. Microalloyed steels 134 in the composition range of 0.05C–1.5Mn–(0.1–0.2)Mo–(0.07–0.09)Nb–(0.065–0.085)Ti–(0.0005–0.0010)B were reheated at ∼1325°C, finished rolled at ∼900°C and coiled at ∼600°C to give a ferrite–bainite microstructure. The precipitates in the final state, which were characterised from foils and replicas, included 120–400 nm (Ti,Nb)N, 10–120 nm, spherical or plates (Nb,Ti)C and 3–5 nm needle-like (Nb,Ti)C. It is quite possible that the precipitates were carbonitrides, as parallel EELS (PEELS) analysis was not available to analyse carbon and nitrogen in the particles. The high yield strengths of ∼770 MPa were attributed to the presence of a fine grain size in a mixed ferrite ∼10% bainite microstructure containing an estimated dislocation density of 10 14 m− 2 retained by a fine dispersion of (Nb,Ti)C precipitates. The observed good toughness, of 50 J at − 40°C, was believed to be a consequence of the fine grain sizes of 3–5 μm and a bainitic low temperature transformation product. Another study of steels for pipelines, with similar carbon and niobium levels, a slightly lower titanium and with 0.3–0.4Cr replacing molybdenum, included an examination of the welded steel. 169 The type and size range of precipitates were comparable to that above, 134 and found in the parent plate, HAZ and weld metal, together with small areas of degenerate pearlite/upper bainite, M and MA, all as microphases, which contributed to the yield strength of ∼700 MPa.
Achieving even higher yield strengths, ∼1000 MPa, is possible, but at an increased cost in both alloying and processing. 170 A steel with a nominal composition of 0.12C–0.5Si–1.4Mn–0.45(Mo+Nb+Ti+V)–0.00015B was hot rolled to 6 mm thick strip and coiled at 600°C. It was then subjected to high frequency induction tempering at 600°C, following quenching at ∼40°C s− 1 from 900°C. This produced a duplex microstructure of lath M and CB laths. Less than 10 nm (Nb,V)C and ∼5 nm VC particles were identified by field emission gun TEM-EDX, the latter considered to precipitate in ferrite, obeying the Baker–Nutting orientation relationship. 60 Recently, a novel low carbon Nb–V–Ti–Cr MA bainitic steel with yield strength of 1000 MPa and excellent low temperature toughness was successfully processed. The experimental results obtained by Xie et al. 17 indicated that the steel subjected to a cooling rate of 65°C s− 1 and a coiling temperature of 380–400°C (above M s of 372°C) and finally furnace cooling to RT, resulted in superior mechanical properties. The ultrahigh yield strength of 1058 MPa was attributed to the transformation strengthening from CB and precipitation hardening from nanoscale (Nb,Ti)C precipitates. The Charpy impact absorbed energy of 24 J at − 20°C was associated with CB and AF, together with a high fraction of large misorientation grain boundaries, obtained by controlling the coiling temperature.
Similar laboratory steels to Misra et al. 170 investigated by Yi et al., 171–174 who achieved σy of 700 MPa in bainitic high strength steel containing 0.15C–0.18Ti through conventional rolling, 171 and in a steels with 0.08C and 0.06–0.08Nb+V + Ti, rolling to 4 mm strip, FRT at 800°C and cooling at 40°C s− 1 to a coiling temperature between 560 and 350°C. The authors claimed that coherent precipitation occurred during coiling, which, along with a ferrite grain size of 2–6 μm, contributed to the strength. The Nb–Ti precipitates were identified by EDX. 172 However, it is unlikely that these particles were coherent, and their method of identification was not given. A later work by Yi et al. 173 , using a steel containing 0.04C–0.09Ti–0.2Mo, raised the yield strength to ∼800 MPa, through dispersion strengthening by nanometre carbides.
The quest for stronger and tougher steels has resulted in the recent application of both APT and electron backscatter diffraction (EBSD) techniques to further the understanding of non-polygonal transformation products. The transformation behaviour of a low carbon Mo–Nb linepipe steel and the corresponding transformation product microstructures have been investigated by Cizek et al. 175 using deformation dilatometry. They found that heavy deformation of the parent austenite caused a significant expansion of the polygonal ferrite transformation field in the CCT diagram, as well as a shift in the non-equilibrium ferrite transformation fields, toward higher cooling rates. In addition, the austenite deformation resulted in a pronounced refinement in both the effective grain (sheaf/packet) size and substructure unit size of the non-equilibrium ferrite microstructures. This was confirmed in a recent research investigating a 0.01Ti–0.039Nb–0.0046N steel by Liang and DeArdo 176 who used EBSD images to quantify grain and subgrain morphology.
Superbainitic (SB) steel
A development of the bainite story, which, however, is marginal but pertinent to MA steels, is the development of ‘superbainitic’ microstructures, which can also result in strength levels of ∼1000 MPa. According to Garcia-Mateo et al., 177 the bainite obtained by transformation at very low temperatures is the hardest ever, has considerable ductility (almost all of it uniform), does not require mechanical processing and does not require rapid cooling. Therefore, the steel after heat treatment does not have long range residual stresses. Nominally, it is very cheap to produce and has uniform properties in large sections. Steel transformed into carbide free bainite can meet these criteria. Super bainitic steel comprises 50–90% bainite, the rest being austenite. The excess carbon remains within the bainitic ferrite at a concentration beyond that consistent with equilibrium; there is also partial partitioning of carbon into the residual austenite. The microstructure has very fine bainite platelets with an average bainite platelet thickness < 40 nm. One composition is 0.6–1.1C, 1.5–2.0Si, 0.5–1.8Mn, ≤ 3Ni, 1.0–1.5Cr, 0.2–0.5Mo, 0.1–0.2V, balance iron, save for incidental impurities. The content of silicon and or aluminium is sufficient to render the bainite substantially carbide free. Of the various processes used, one includes the fast cooling from austenite to avoid transformation to pearlite, and transforming the steel to bainite at a temperature in the range of 190–250°C, by holding between ∼8 h and 3 days. Owing to the combination of ultrahigh hardness and ductility, a major application for super bainite is armour, which is included in the patent. Friction stir welding is one means being explored to join SB steel plates and to other steels of other compositions. The topic of SB steels is discussed in more detail by Bhadeshia 178 and recently extended by Caballero et al. 179 and Avishan et al., 180 who explored and detailed the microstructures of high carbon steels, some with microalloying additions of niobium or vanadium to produce nanocrystalline bainitic ferrite, with plates a few tens of nanometres in thickness.
Thin slab direct charge processing
The background to the TSDC processes, also known as thin slab direct rolling (TSDR) and compact strip production, which originated in 1989 in Crawfordsville, IN, USA, has been very well described by Glodowski. 181 ‘It appears that the commercial (TSDR) process preceded any academic research, and steady progress has been made over the past 25 years. Initially, the emphasis was on the obvious economic and ecological benefits gained by retaining part of the heat of casting in the slabs through to the rolling process, and casting to a “near net shape,” thereby reducing the amount of hot working necessary to reduce the material to final gauge. Conventional microalloying approaches for high-strength, low-alloy (HSLA) strip steels presented some processing complications that were new and required some alternative solutions. For many mills, the vanadium and nitrogen alloying system has proven to be highly effective and compatible with the processing peculiarities of the thin-slab-casting and direct-charging process. The thin-slab-casting, direct-charging and finish-rolling process has a number of characteristics that vary from conventional blast-furnace/BOF/slab-cast/slab-reheat/roughing-mill/finishing-mill routings. Figure 31 shows these, plus the metallurgical mechanisms that operate at different stages of the TSDR process and ASP process. 181 First, steel making often uses electric-arc-furnace (EAF) melting from predominately scrap charges, and the refining process takes place in separate ladle-furnace operations. Second, the casting process involves rapid cooling, necessary for high-volume production in thin slabs from 50 to 100 mm in thickness. Third, the slab is directly charged into a tunnel holding furnace without undergoing a γ → α phase transformation. Fourth, the tunnel furnace is limited in reheating capabilities, typically from to 1100 to 1150°C maximum. Finally, the 50-mm slab will enter directly into a 5- or 6-stand finishing mill without undergoing a roughing mill reduction. When processing thicker slabs, 80–100 mm slab casters will have one or two roughing mills prior to a 5-stand finishing mill. In those cases, a transfer system of coil boxes or holding furnaces will be installed after the roughing mills to equalise temperatures before entering the finishing-mill stands. This step provides a speed “break” between the start and finish rolling speeds, to allow more reasonable entry speeds in the first reduction pass. The remainder of the process, from finish rolling through a run-out table usually equipped with significant accelerated-cooling capability and into a down coiling system, will be similar to existing modern strip mills. Many of the mills have the capability to approach 1-mm final thickness, although most commonly the final gauge will be 1.5-mm or larger.’ The changes in the TSDR technology over a period of 20 years to 2010 has been recounted by Klinkenberg et al., 183 who also list the wide variety of steels now produced by this route.
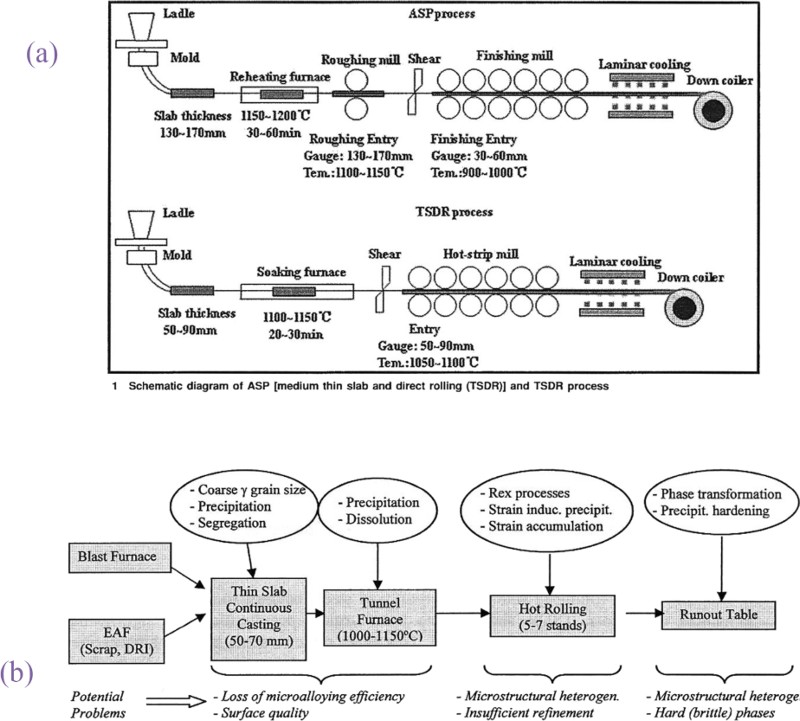
The TSDR route has attracted a plethora of research activity over the past 15 years. Much of this has been concerned with optimising production parameters and relating to them properties through microstructure. In particular, the evolution to precipitates associated with the various stages of the process has been investigated, and the development of coarse grained austenite received attention. These topics are considered in more detail below.
A particular theme running through an excellent review of 52 papers, published up to 2005, which are considered in some depth by Rodriguez-Ibabe, 184 is the dependence on microstructure of the production parameters of TSDR technology route. He considered this route particularly attractive to MA steels. This is because the microstructural development during TSDR, and the mechanical properties of the final coil, differs from equivalent steels conventionally controlled rolled. In TSDR, slabs are thinner, compared with 200–250 mm in conventional controlled rolling, while casting speeds are higher at 3.5–6.0 m min− 1, compared with 0.75–1.25 m min− 1. Consequently, the thin slabs solidify much quicker, show less segregation, smaller inclusion sizes and a more homogeneous microstructure. In addition, when scrap based electric arc furnace routes are used, thin slabs will have a very coarse as cast γ grain size before hot rolling and experience a smaller total reduction during rolling and higher nitrogen and residual element concentrations. Some of these aspects can result in a loss of microalloying efficiency as a consequence of premature precipitation before rolling. This is especially the case with titanium additions, and niobium steels must be considered with care. However, a different view was expressed by Bruns and Kasper, 185 who, together with Priestner and Zhou, 186 published some of the earliest studies describing the laboratory simulation, which resulted in the TSDR microstructural evolution of Nb–Ti MA steels. The former 185 found that direct charging resulted in a greater dissolution of niobium and titanium compared with cold charging, leading to more precipitation of (Nb,Ti)(C,N), with a consequential greater strength in the final product.
A series of papers associated with Li 187–191 also used laboratory simulation to study several vanadium based MA steels, which were vacuum melted, cast into moulds and controlled cooled at 3.5°C s− 1, transferred to an equalisation furnace and held at 1050, 1100 or 1200°C for between 30 and 65 min, before rolling to 7 mm strip with five passes. After rolling, the strip was cooled under water sprays to simulate run-out table cooling, with a cooling rate of 18°C s− 1, and the aim end water temperature was 600°C. Following cooling, the strip was immediately transferred to a furnace set at 600°C and slow cooled to simulate coiling. Samples were quenched after casting, after equalisation, after the fourth rolling pass and after coiling, to follow the evolution of both precipitation and mechanical and impact properties. Using this TSDR simulation in an investigation of the effect of a 0.008Ti addition to a 0.10Vsteel, Li et al. 187 reported that, for both V and V–Ti steels, the average austenite grain size in the one-fourth thickness position of the as cast ingot was ∼1 mm. This was reduced to a ferrite grain size of 4.8–7.2 μm in the final strip. After equalisation at 1050°C, precipitation of VN in austenite in the V steel was replaced by (V,Ti)N in the V–Ti steel. Both types of precipitates, which were in the range of 2–10 nm, made a contribution of >90 MPa to dispersion strengthening. However, the yield strength of the V–Ti steel was lowered by the removal of a significant fraction of vanadium and nitrogen to form (V,Ti)N in austenite, thereby reducing the volume fraction of V(C,N) particles precipitated in ferrite. A similar situation occurred with V–Nb–Ti steels, with the addition of Ti resulting in a reduction of the yield strength due to precipitation in austenite of nanosized particles of (V,Nb,Ti)N, as characterised by both EDX and PEELS. 190,191 These transition metal nitrides did not coarsen during the final stages of processing, retaining a size of ∼7 nm.
More recently, a TSDR process, similar to that described by Glodowski, 181 called the Angang strip production process, 182 has been used with medium thin slab of 170 mm, by installing a roughing mill after the reheating furnace and before the finishing mill. Sha and Sun 182,192–194 studied commercial sized slabs of an 0.04C–0.06Nb–0.30V–0.014Ti–0.23Mo–0.0032N MA steel in the as cast condition. The slower cooling rate of the 170 mm slab influenced the secondary dendrite arm spacing and the austenite grain sizes (320–2200 μm), which were slightly larger compared to the microstructure of specimens taken from a corresponding TSDC route processing of 53 mm thick slab (150–2000 μm). Dendritic precipitates were mainly niobium rich carbonitrides, while large cubic (150–600 nm) and fine cubic (10–40 nm) particles were characterised as titanium rich carbonitrides. The authors considered that vanadium was not detected in the complex precipitates due to the low nitrogen level in the steel. The refinement of the coarse grained austenite in the same steel, during roughing rolling after simulated equalisation, and more recently, after preliminary rolling in a roughing mill installed between the reheating furnace and the finishing mill, was also examined by Sha et al. 182 The microstructural homogeneity using the Angang strip production route was found to be better than the TDSR route without additional rolling, due to a higher reheating and entry temperatures and a larger reduction with the thicker slab. The coarse grained austenite (∼1 mm) that developed during equalisation (compared with ∼250 μm in as reheated) in the Nb–V–Ti steels was modelled by Sha et al. 194 This followed the austenite grain size distribution method used by Gao and Baker 195,196 and Zhang and Baker, 197 who considered abnormal austenite grain growth, an approach also developed by Uranga et al. 198 for their model, which they later expanded to include the carbon and nitrogen content of MA steels. 199 The modelling, involving the same processes discussed previously, 97–106 was used to alleviate microstructural heterogeneities in the final product. These heterogeneities are thought to be associated with the coarse austenite grains, which developed through the processing parameters not perfectly matching, leading to unrecrystallised regions after the rolling process, particularly in niobium steels. All these authors 184,195–199 concluded that their models highlighted production pitfalls, which could be avoided, supporting the view that strict control of grain size at each stage of the TDSR process is essential.
The application of the TDSR process for thin slab production for pipeline MA steels, reducing 50–90 mm slab to 6–12.5 mm thick strip, was reported by Reip et al. 200 Two steels containing nominally 0.06C–1.35Mn–0.074V–0.04Nb–0.0065N with 0.01Ti (steel a) and 0.002Ti (steel b) were processed in a commercial caster. After processing, both steels exhibited very good notch toughness of 200–400 J cm− 2 at − 40°C, and steel a had σy of 680 MPa, which was 100 MPa higher than steel b. The excellent toughness of both steels was attributed to the fine grain sizes of 2.6 and 6 μm respectively.
It has been shown by Lee et al. 151 that increasing the dislocation density opposed the formation of AF and bainite through a mechanical stabilisation effect. The results of Reip et al. 200 suggest that the exclusion of titanium in steel b favours the formation of AF and bainite. This maybe be due to the higher percentage of dissolved niobium, which supports the formation of AF and bainite, because titanium, precipitated as TiN, may provide nucleation sites for precipitation of Nb(C,N) during the final phase of rolling. 201 Further work was devoted to comparing fine scale precipitates in high strength industrially processed trial steels containing Ti–Nb and Ti–Nb–Mo–V additions. This focused on the precipitate characterisation based on crystallography, through some careful analysis of electron diffraction patterns gained from thin foils. 202 As the nitrogen content of the steels was not given, it seems that, based on lattice parameter determination, they were all considered to be carbides.
An investigation was undertaken by Nagata et al. 203 to assess the potential of obtaining and utilising titanium nitride refinement, primarily for austenite grain size control, through the increased post-solidification cooling rates associated with the TSDC process. They compared eight commercially TSDC cast steels with a conventionally cast (CC) steel. The steels had titanium contents in the range of 0.009–0.048% and Ti/N ratios between 0.6 and 6. The precipitation behaviour was explored with extensive TiN particle size distributions evaluated by TEM. Despite designing the steels to prevent precipitation in the liquid state, TiN cubic particles of ∼1 μm were found in some steels after casting.
The hyperstoichiometric steels (excess titanium) exhibited cubic TiN particles of mean size < 40 nm after casting and soaking, but showing dramatic coarsening to >90 nm after hot rolling. Hypostoichiometric steels, which contain excess carbon and/or nitrogen, exhibited no coarsening in the end product. Furthermore, comparison of the particle sizes of the TSDR and CC steels showed a shift to larger sizes and greater relative frequency for the particles in the CC steel.
Weldability of MA steels
The weldability of MA steels is an extensive topic in its own right and has been addressed in many publications, including several of the reviews discussed here
4,8,9,11,13
and parts of the book by Easterling.
204
As expressed by Hart,
205
‘the opportunities that microalloying gives for improving weldability of microalloyed steels, by reducing carbon content was fairly quickly recognised and became another major driving force for the development and understanding.’ In addition, many modern steels have low sulphur, and generally, nitrogen is < 4 ppm. However, rapid cooling of regions beside the weld (HAZ) frequently produces martensitic hard zones leading to local increases in hardness, which can result in lower toughness. It is a common practice to specify a maximum hardness that can be tolerated in the HAZ. As the hardness of martensite is related to the carbon content of the steel, this has a profound influence on hardenability. Other elements affecting hardenability are considered in the CE equations deduced from multiple regression analysis, such as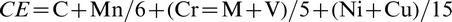
As pointed out by Lagneborg et al., 11 ‘nitrogen levels in plain C–Mn steels show a strong correlation with coarse-grained HAZ toughness, where higher N-levels are clearly deleterious, showing a marked increase in Charpy Tc, which is reflected in maximum permitted contents of nitrogen in various steels specifications. However, the presence of the nitride forming elements Al, Ti, Zr, Nb, V, can mitigate considerably the negative effect of nitrogen, indicated by the relaxation of the limit of nitrogen in the presence of these elements.’ Much attention has been paid to steels containing titanium and nitrogen and how these elements interact in combination with vanadium, niobium and zirconium.
In a review on the status of microalloying elements in plate used for the construction of naval ships, by McPherson, 206 it is recognised that the potential problem area tends to be the HAZ toughness. A tentative relationship exists between the HAZ toughness effect of the microalloying element and its effect on the susceptibility to induce transverse slab cracking. Overall, it was found that the benefits improve in the order of niobium, vanadium and titanium. If cost is factored into these effects, then the microalloying element with the most potentially positive benefits is titanium. 206
In titanium MA steels, the effects of Ti and N contents in improving the HAZ toughness through the pinning of austenite grains by TiN particles have been mentioned earlier and is well established. 207–210 TiN particle size and volume fraction both influence toughness. 211,212 With hyperstoichiometric titanium additions, coarse TiN particles (side length>0.5 μm), form in the liquid state and, above a critical number density, can act as fracture initiation sites when ferrite grain sizes are >9 μm. 212 It is recommended that the contents of Ti and N should be kept close to the stoichiometric ratio Ti/N = 3.42. Another view is that soluble nitrogen should be minimised. Mixed additions of Nb–Ti–V in MA steels were considered by Adrian and Pickering 213 and by Hamada et al., 214 as a function of nitrogen content. Both concluded that high nitrogen, 0.018 and 0.0084% respectively, was advantageous, resulting in the substantial refinement of austenite grains, 213,214 and in the case of HAZ simulation, an austenite grain size of ∼30 μm was produced by ∼10 nm (Nb,Ti)N precipitates. The effects of zirconium, along with those of titanium and vanadium, on submerged arc deposits were compared by Koukabi et al., 215 who concluded that vanadium had a greater effect on increasing toughness in both the as welded and stress relieved welds than did zirconium. It is well established that both TiN and ZrN particles have little solubility in austenite, as seen in Fig. 14. Zirconium additions were expected to behave similarly to titanium, but Koukabi et al. 215 found that ZrN particles did not promote AF, and the weld toughness was only slightly improved due, they considered, to the removal of the soluble nitrogen. The observations of Koukabi et al. 215 were later explained by Chai et al., 216 who suggested that the equilibrium number and composition of the inclusion phases changed with the zirconium addition from Ti2O3 to ZrO2. When the zirconium content was lower than 0.0085%, the inclusions were mainly Ti2O3 and ZrO2, and the oxides shared the same proportion of ∼0.0045%Zr content. The role played by ZrO2 is also supported by the work of Guo et al. 217
A study by Wan et al. 218 of simulated HAZ in Ti–Zr steels also found in situ evidence for AF nucleating on Zr–Ti oxides containing Al, Mg and Mn when the austenite grain size was large, 100–200 μm, but bainitic plate packets, nucleated on different sites of smaller, ∼50 μm, austenite grains.
While inclusions in steels are usually classed as oxides and sulphides, Koseki and Thewlis 219 also include large nitrides, as discussed above. Inclusions are normally regarded as being deleterious to toughness, but over the past three decades, inclusion assisted microstructure control has become a key technology in the improvement of toughness of weld and HAZ regions. In addition to pinning austenite grain boundaries, it is well documented that inclusions nucleate intragranular AF. 219–221 As discussed previously, the development of a microstructure with a high proportion of AF is recognised as means of generating steels with high strength and good toughness. This approach had its birth in attempts to improve the microstructure of welds, where toughness problems were mitigated by inclusion nucleated AF. The formation of AF is closely related to oxygen content of the weld metal and the type, volume fraction and size distribution of the inclusions. 221,222 It has been reported that TiO and TiN, if oriented correctly, have good coherence with ferrite, when the two lattices exhibit a low misfit δ. 223
Mills et al. 222 identified a titanium rich phase, which they considered to be a mixture of TiO and TiN with other phases, on the surface of a glassy manganese silicate and on galaxite (Al2O3, MnO). Both TiO and galaxite have a low δ, ∼3%, with ferrite, which is considered to be the reason that these phases nucleate AF. Several other phases have been collected by Koseki and Thewlis 219 showing low δ values, with < 5% mismatch. In addition to low misfit, the number density of inclusions N AF, which act as AF nucleation, is important.
More recently, the AF formation potency of Ti–rare earth metal–Zr (TRZ) complex oxides has been investigated in the simulated HAZ of low carbon steel studied by Nako et al.
224
The TRZ complex oxide shows a higher AF formation potency than either titanium or aluminium oxides. Acicular ferrite crystals nucleate on the interface between austenite and the Zr rich oxide phase. Good lattice coherence through the orientation relationship
While the welding of MA steels by the standard techniques is well established, less has been published on the application of FSW to this group of steels. 225,226 Friction stir welding, which was invented by TWI in the early 1990s, was originally developed for joining Al alloys. 227–231 Friction stir welding is a solid state joining process (the metal is not melted) that uses a third body tool to join two facing surfaces. Heat is generated between the tool and material, which leads to a very soft region near the FSW tool (Fig. 32). It then mechanically intermixes the two pieces of metal at the place of the joint, after which the softened metal (due to the elevated temperature) can be joined using mechanical pressure that is applied by the tool. 232 Friction stir welding is now a mature process for welding several alloys, including MA steels. The advantages of FSW include improved mechanical and metallurgical weld properties, rapid joining speeds, elimination of weld cracking and porosity and no need for shielding gas or filler metal, together with normally resulting in lower residual stresses than fusion welding. 227–233 In addition, FSW is a ‘green technology’ in that it produces no arc radiation, no significant fumes and no hazardous waste. The possibility of joining dissimilar metals and alloys, such as steels and aluminium, is now being actively explored. 225 There are several publications describing FSW of ferritic steels, where additional advantages include lower heat inputs, with, it is claimed, fewer metallurgical changes in the HAZ and also the potential for joining dissimilar steels. 234–236 The majority of the material joined by the FSW process has a thickness of ≤ 10 mm. The plate thickness that can be welded is limited by the length of the tool, which, for BN pins, often the pin of choice for FSW of steels, is ∼8 mm. 237,238 However, the severe stresses and high temperatures experienced by the tool materials pose a formidable challenge for the development of cost effective and durable tools for the FSW of hard alloys. As a result, the initial commercial applications of FSW to steels are likely to come from specialised, niche areas where fusion welding clearly has serious difficulties, provided the required structure and properties can be achieved by FSW. 239 While much of the earlier FSW research was concerned with details of the process and modelling of flow, 227–239 the role of microstructural evolution is now beginning to be addressed in more detail, 240–245 ‘as an understanding would enable a prediction of welding properties resulting from the microstructures’. 243 As detailed by De et al., 239 compared to fusion welding, microstructure is affected by several factors. The peak temperatures for FSW are lower, and therefore, the grain size of austenite that forms in the HAZ is often smaller than after fusion welding. Furthermore, severe deformation and flow of plasticised steel alter the stability of austenite in the thermomechanically affected zone (TMAZ) because of the stored energy, which affects its decomposition to daughter phases. In contrast, the austenite formed during cooling of molten weld metal in fusion welding is hardly affected by any stored energy. Because deformation in the stir zone (SZ) varies with position, this influences the local austenite transformation kinetics, as in thermomechanically processed MA steels, the modelling of which was considered earlier. 239 Therefore, microstructures are expected and found to vary considerably in different parts of the SZ.
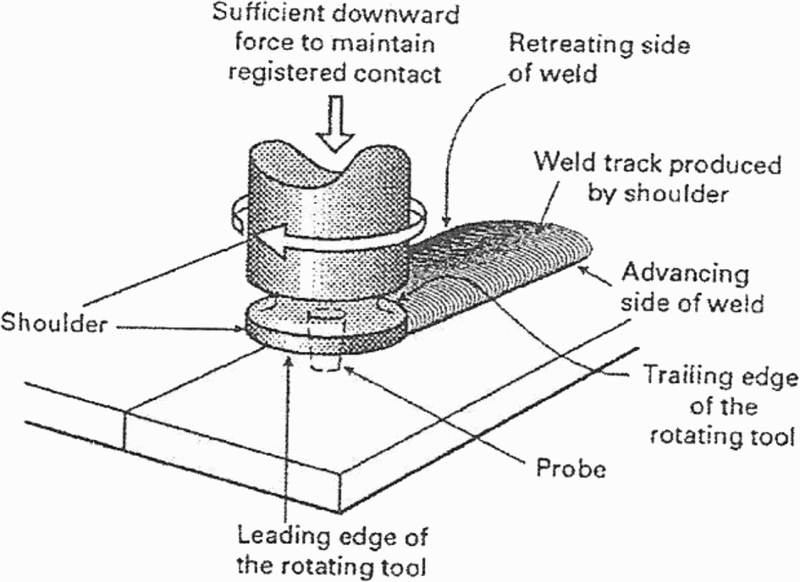
Schematic diagram of friction stir butt welding 233
Electron backscatter diffraction produces orientation maps, which provide vital information on grain misorientation and grain size from the base metal, HAZ, TMAZ and the SZ of FSW. This application of EBSD has been used to good effect by Davies et al., 246 studying deformation textures in FSW Ti–6Al–4V alloy, who showed that the texture of the Ti α hcp fully lamellar microstructure was inherited from the shear texture of the high temperature β phase. This does not yet appear to have been investigated for the γ → α transformation in FSW MA steels. Cho et al. 243 were among the first to apply EBSD obtained with a field emission SEM to study the microstructural evolution of a FSW high strength linepipe steel. They found that, in the SZ, the structure morphology, texture and misorientation angle were completely different from other FSW regions. Figure 33 shows the changes in grain size and the fraction of AF across the weld section, observed in the SZ. An increase in hardness in the SZ, also recorded by Lienert et al., 234 was attributed to acicular shaped bainitic ferrite, observed by TEM. Mirinov et al. 242 found that the material in the SZ of pure iron had a prominent (112) [111] shear texture with the shear direction aligned approximately tangential with the shear surface, while the work of Cho et al. 243 revealed only a weak shear texture.
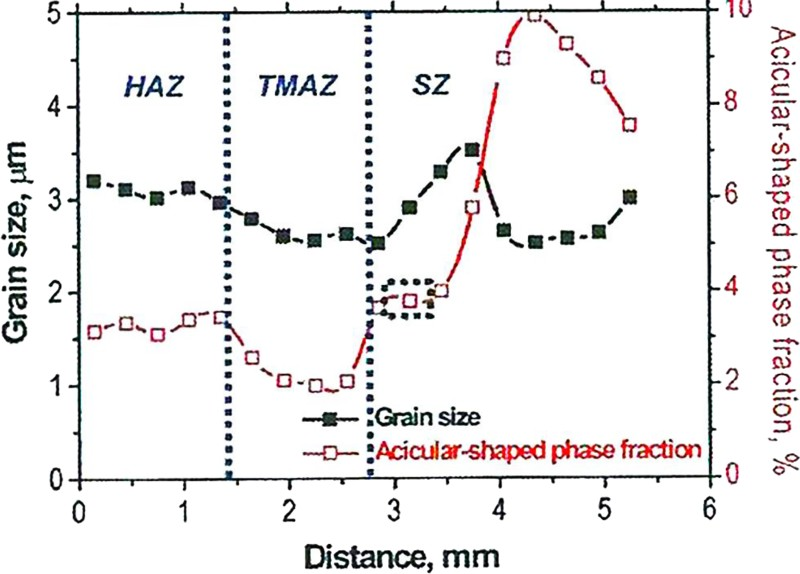
Profiles of grain size and acicular shaped phase fraction 243
An FSW method developed for thicker plate, >12 mm, with applications, for example, in shipbuilding, involves passing the tool sequentially on both sides of the abutted plates. 247,248 This gives rise to a complex interaction zone (IZ) where the deepest resolidified metal created by the first pass is remelted by the second pass on the reverse side of the plate (Fig. 34). The HAZs are also affected by the plate reverse side FSW pass. Baker and McPherson 249 have investigated in detail the variation of microstructure and the related microhardness in different parts of an FSW EH 46 MA steel. They found a decrease in microhardness in the IZ relative to the HAZ and TMAZ. It was also noted that a significant variation in the IZ microstructure over small distances occurred, as predicted by De et al. 239 Packets of laths beside polygonal ferrite grains (Fig. 35), some containing dislocations pinned by particles, were also observed. It was considered that SIP occurred in the TMAZ. Rahimi et al. 250 used EBSD, after the manner of Gourgues et al., 251 to assess the crystallographic features of the EN46 steel. Their EBSD data 250 show that the IZ underwent deformation in ferrite, since the pole figure shows bcc shear texture. In addition, the IZ has ultrafine grain size (i.e. ≤ 1 μm) in which the misorientation distribution fits in well with the MacKenzie distribution, implying that the ultrafine grain ferrite is recrystallised. Data (TEM) collected from the IZ shows dislocation cell structures, which agrees well with the EBSD data. However, in the TMAZ, the EBSD data show (111) pole figures in the bcc ferrite similar to (011) fcc shear texture, implying that the deformation during FSW occurred in austenite. The data also show a significantly high fraction of substructures that are scattered from the MacKenzie distribution. These can be due mainly to low angle grain boundaries between ferrite grains that are originating from the same austenite grain. 250 The study of microstructure using TEM and related EBSD is at a relatively early stage, and it is expected that more will be forthcoming in the future.

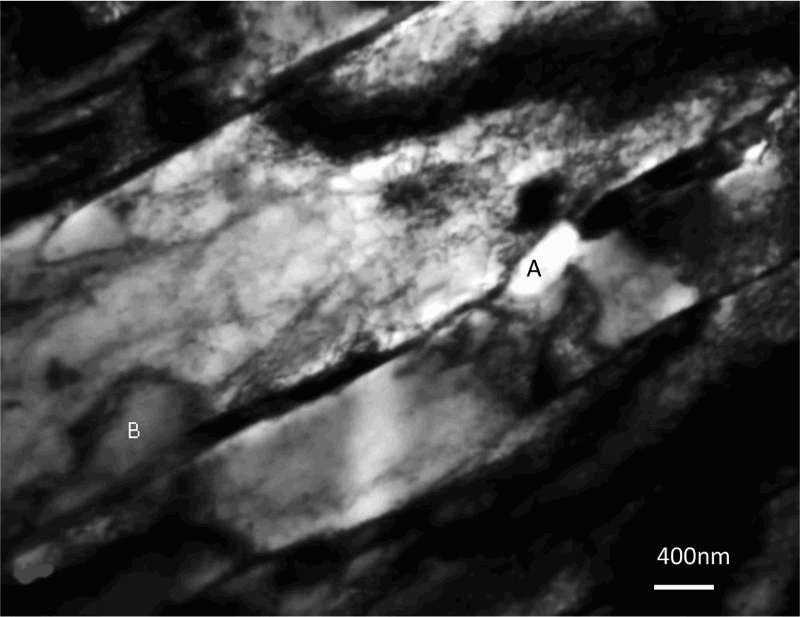
Image (TEM) of IZ of double sided FSW EH 46 microalloyed steel, showing laths containing pinned dislocations and precipitates growing from lath boundaries at A and B 249
Strength, toughness and ductility
14.1Components of strength
For many years, the possibility of improving the properties of polycrystalline materials through understanding and quantifying the microstructural components that determine the properties has been a major goal of materials scientists. 30,31 This topic has been explored to a much greater depth in the case of MA steels than in perhaps any other material. One of the main reasons, has been the challenge posed by such a complex microstructure, which involves contributions from solid solution hardening, precipitate dispersion strengthening, dislocation densities, grain and subgrain size, the Peierls–Nabarro stress and a possible texture contribution. 2,28,29,252–257 In addition, the effect of the interaction of some of these components, giving rise to superposition, which is considered below, must be addressed. 114,257
It has long been appreciated that a fine grain size is associated with superior properties. In the eighteenth century, separately Reaumur 258 and Grignon 259 noted that, as revealed on fracture surfaces, excellent metals were distinguished from mediocre ones by the fineness of their grains. This historical background was initially described by Desch 260 and more recently by Armstrong. 261
The measurement of a fine grain size necessitated optical microscopy, which leads to the study of microstructure normally described as ‘metallography’. The earliest systematic study is generally attributed to H. C. Sorby, who, in 1849, extended a technique to geological specimens of meteorites, taught to him by a Manchester surgeon, W. G. Williamson. 262 Sorby adapted his techniques to the examination of iron and steel products of his native city, Sheffield. Many years later, W. L. Bragg who had long held the view that grain size influenced the strength of polycrystals, set Orowan in the late 1940s, with two research fellows, Hall and Petch, to systematically study this effect in mild steels. 263
The Hall–Petch relationship, which was initially concerned with FP steels, conveniently allows σy, the lower yield stress (or often in practice, the 0.2% proof stress) to be related to individual components of strengthening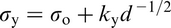
As can be seen in Fig. 36, based on the work of Morrison, 264 when σy is plotted against a function of the grain size d − 1/2, a straight line relationship is obtained over a range carbon contents in steels. The intercept on the ordinate axis σo and the slope of the straight line k y are experimental constants. Hall and Petch proved that strengthening was therefore due to a reduction of grain size together with an increase in the factors that determined σo. The latter, which is often referred to as the friction stress, 29 results from the features that arrested the motion of dislocations across the grains. Problems associated with the determination of σo have been discussed in detail by Baker. 255 These included strain fields from misfitting solute atoms in solid solution σss, precipitates σp and other ‘forest’ dislocations σd. Other factors are subgrain sizes σsg and texture σt plus the basic lattice Peierls–Nabarro component σi.
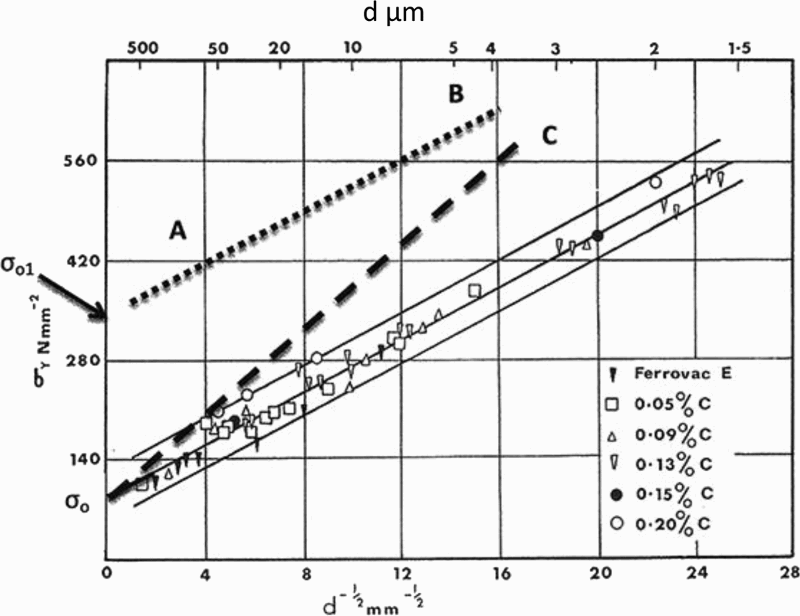
Relationship between lower yield stress σy and grain size d expressed as d − 1/2for steels containing different carbon levels; dotted line A–B shows situation when σo is increased to σo1 through dispersion strengthening component, while dashed line CD shows situation when k y is increased (after Morrison 264 )
The linear summation of these components leads to the relationship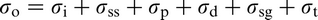
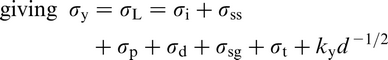
Each of these components is now considered separately.
Solid solution strengthening σss
Mechanisms of solid solution strengthening in MA steels are dealt with in detail by Gladman.
3
The contribution of σss to σy, based on data provided by Gladman,
3
updated from Pickering and Gladman,
252
obtained from regression equations where the alloying elements are given in weight per cent, can be expressed as (MPa)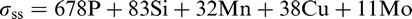
14.1.2Dispersion strengthening σp
Detailed accounts of dispersion strengthening, otherwise known as precipitation hardening, are found in several publications, 265–267 while others deal specifically with MA steels. 268,269
As mentioned previously, while vanadium carbides can precipitate coherently in ferrite, it is generally agreed that, in commercially processed niobium MA steels, the incoherent and the SIP in austenite provide the main contributions to σp. In many cases, it is not possible to separate the contributions of σp and σd. This is because deformation is introduced in the temperature range where precipitation commences and is known to lower this temperature from the situation where precipitation of the same particles starts in the absence of strain. Briefly, strengthening takes place when particles impede the motion of dislocations, 265–269 and the overall effect is mainly due to the particle radius r and the particle volume fraction f. Dislocations can shear coherent (soft) particles, and σp ≈ (fr)1/2, or dislocations can loop around the incoherent (hard) particles, where σp ≈ f 1/2/r.
Goldschmidt 270 found extensive mutual solubility of all the carbides and nitrides collated in Table 5, which formed carbonitrides. Examples are the TiN–NbN system 122 and the ZrC and ZrN system. 123 The exception is VC and VN with ZrC and ZrN, attributed to the difference in the sizes of the Zr and V atoms, which are given in Table 7.This also has implications for ternary systems such as TiN–ZrN–VN and ZrN–NbN–VN, where a miscibility bay should occur, in contrast to complete solution systems such as TiN–VN–NbN, or similar mixed carbides or carbides and nitrides. Therefore, in most MA steels, which contain both carbon and nitrogen in their chemical composition, the strengthening precipitates will be carbonitrides of varying C/N ratios.
The BNOR was not found in the detailed and careful crystallographic study undertaken by Epicer et al. 271 They used a model Fe–V–C alloy and found the V6C5 precipitates, which exhibited an ordered monoclinic form, also characterised by Hollox et al. 272 Furthermore, their survey of the literature found no conclusive evidence for the existence of precipitates with the V4C3 structure. 271 This is surprising, as many publications have included SAED patterns, matching the fcc:bcc BNOR 60 for transition metal carbides and nitrides, sometimes described as the Bain orientation, with the correct lattice parameters. 273–278,280 Baker and Nutting orientation relationship has been shown to apply to incoherent Nb, V, Ti carbides, nitrides and carbonitrides precipitated in ferrite, including interphase precipitation. 277–284 It should be pointed out that both the composition of the model alloy studied by Epicer et al. 271 and the heat treatment conditions are unrepresentative of MA steels. It would be edifying if these authors applied the same rigour to vanadium carbonitrides precipitated in commercial controlled rolled MA steels. Here, however, the main point is to confirm whether the V6C5 precipitates are sheared or looped by dislocations, as this leads to different estimates of strengthening. There is still disagreement on the subject on the role of solute atoms, particularly niobium, versus SIP of nitrides and carbonitrides, on the kinetics of austenite recrystallisation, discussed previously, and also whether or not carbides, nitrides or carbonitrides form coherently or incoherently in ferrite. 51–57,131–133 Almost invariably, particles characterised in MA steels are incoherent. This is supported in a recent APT study on vanadium steels, which found 5–20 nm size, randomly distributed V(C,N) precipitates, with a BNOR, in ferrite. Interphase precipitates formed in a region of higher carbon. 280
A more detailed expression derived from the initial work of Orowan,
285
developed by Ashby
286
and others,
4
leads to an expression that allows quantification of σp (MPa)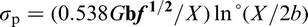
For MA steels, with G = 80 300 MPa, 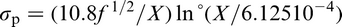
σp values of 50–100 MPa are typical of niobium steels of the 1980s. Kostryzhev et al. 54 have collated σp values from eight sources, reporting on Nb and NbTi MA steels, which range from 20 to 230 MPa, and point out that, for the same steel using different TMP routes, variations up to 4.5 times have been found. However, some of these data may include a contribution from dislocation strengthening. Recently, Yen et al., 281 studying interphase precipitation of TiC, estimated σp values to be ∼300 MPa, in steels with σy of 780 MPa.
One of the main difficulties in calculating σp is the estimation, based on experimental determination of the volume fraction f, of precipitates involved. Often, extraction carbon replicas have been employed. The problem is to determine the depth of steel, which has been etched to provide on the replica in two-dimensional, the particle volume fraction distributed in three-dimensional. A solution to the problem was given by Ubhi and Baker 287 and discussed further by Baker. 255,288 Using the extraction carbon replica technique, one study noted that few particles ≤ 4 nm were recorded. 124 MacKenzie et al. 289 repeated the work using a focused ion liftout technique, followed by low ion energy milling. This allowed the quantitative analysis of ∼1 nm particles of VN. How much the loss of particles between extraction carbon replicas and the above technique, in terms of volume fraction and influence on σp, is not known, as this work is probably the only record of this observation.
Dislocation strengthening σd
Motion of dislocations (mobile) is impeded by interaction with both other mobile dislocations and with immobile dislocations already present in the grains, usually described as forest dislocations. These may be present along with precipitates, as often in the case of as rolled MA steels. In the former case, σd can be calculated based on the equation first derived by Taylor
290
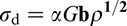
Based on the modifications to the Hall–Petch equation, many methods have been developed to estimate the lower yield stress σy from microstructure measurements of particle size and dislocation density. These have met with some success in MA steels when dislocation densities where relatively low. Modifications to equation (11) have been proposed when dislocation densities are high, >10 15 m− 2, due to the strong precipitate–dislocation interaction resulting in superpositioning, discussed below.
Grain size component
Figure 36 can be used as a guide to the possible strengthening routes for MA steels with a FP microstructure. Thus, for a grain size of 50 μm, tested at 300 K, the data give a corresponding σy of ∼160 MPa. For a small ferrite grain size of 5 μm, σy increases to 350 MPa, emphasising the importance of grain size as a method of increasing strength in polycrystals. Figure 36 also shows that strength can be increased by displacing σo to the dotted line A–B. For a grain size of 5 μm, σy increases to 600 MPa. This is achievable by controlled rolled microalloy steels. In theory, changing the slope k y, as shown in Fig. 36 for the dashed line σo–C, will also increase σy, although this is not normally a practicable route. The Hall–Petch relation has been found experimentally to hold for materials with grain sizes ranging from 1 mm to 1 μm. However, experiments on many nanocrystalline materials demonstrated that, if the grains reached the critical grain size, which is typically ∼10 nm, σy would either remain constant or decrease with decreasing grain size. 292
A number of different mechanisms have been proposed for this relation, which fall into four categories: dislocation based diffusion based grain boundary shearing based two-phase based.
293
Manufacturing engineering materials, such as MA steels, with this ideal grain size, is difficult because only thin films can be reliably produced with grains of this size.
However, MA steels comprising of subgrains, having misorientations θ of < 8°, are well documented and include AF and bainitic microstructures. DeArdo et al. 6 comment that ‘as there are relatively few high angle boundaries, beyond the prior austenite boundaries, Hall–Petch strengthening will not be important.’ However, the friction stress term σo, envisaged by Petch, 31 included a contribution from dislocations, as seen in equations (11) and (12). Equations dealing with substructures, sometimes described as cells, are available and discussed in detail by Embury 294 and Thompson. 295 Both provide a comprehensive list of references to relevant early research, extended by Castrofernandez and Sellars. 296
σsg is usually related to the subgrain size l by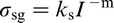
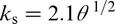
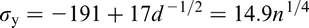
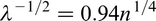
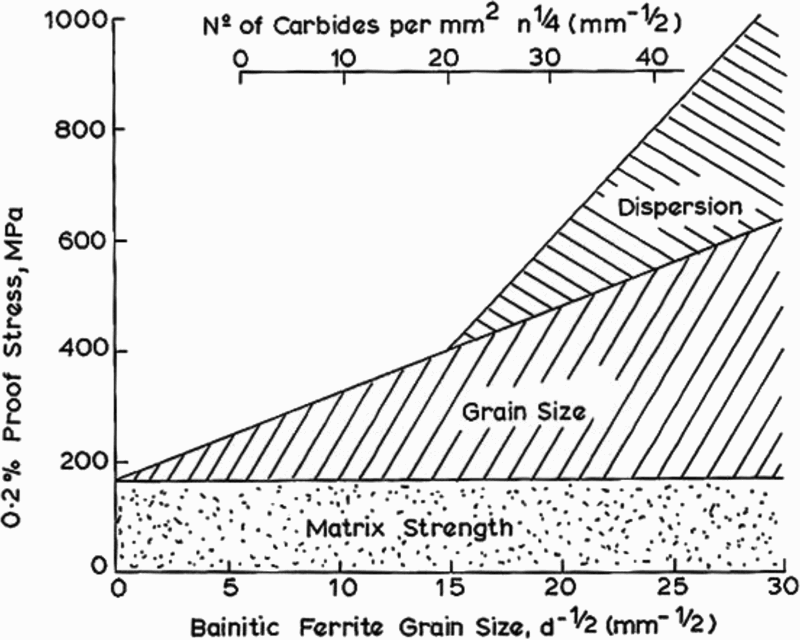
Strengthening effects in low carbon bainite 254
Summation of strengthening components
One of the earliest examples of successful linear additive strengthening of MA steels comprising grain, subgrains and precipitation was by Mangonon and Heitmann, 297 extended more recently by Morales et al., 298 who considered the same method for their linepipe steel, which was comprised of AF and PF microstructures.
The summation of these components leads to the relationship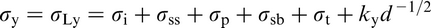
In a reassessment of results obtained during research on niobium steels for microstructures containing high dislocation densities, the initial comparison of the estimated σLy, by linear addition, and experimental data σM was poor.
256
With a second approach, taking into account superposition,
114,256
the components were divided into two groups, σA and σd, and then used in a relationship based on the square root of the sum of the squares of σA and σd
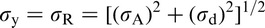
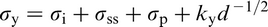
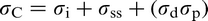
Atomic radius of elements used in microalloyed steels and per cent difference from iron
Comparison of σy determined experimentally σM (Ref. 114) and calculated from microstructure 114
As can be seen, comparing the data for σL with σC and σR, both approaches are superior to the linear summation, and σC data are slightly closer to σM when the dislocation density is lower. However, for high dislocation densities produced by, for example, low controlled rolling finishing temperatures, σR data for steels S5 and S25 gives a better agreement with that of σM. For this reason, the summation method recommended by Irvine and Baker
114
was used in recent work by Carretero Olalla et al.
115
analysing the strengthening mechanisms in MA line pipe steel, with satisfactory results. It is interesting to note that the difference between σM and σR values in Table 8 is 7 to 27 MPa, i.e. 1.0–5.1%. In general, obtaining an estimated σy from the microstructural features considered above to within ≤ 50 MPa, i.e. ∼10%, compared to σM, is acceptable. However, the determination of σy only from microstructural measurements is tedious. In work where the details of individual components was of less interest
189,303
and the dislocation densities relatively low, the combined σp and σd was estimated using equation (23)
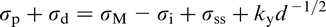
There are alternative ways of determining strengthening components, other than by microstructural measurements. 304 Speich 305 used resistivity measurements to follow precipitation in alloy steels during tempering treatments to good effect. The same approach was used by Hall and Baker 306 studying precipitation of vanadium carbide in controlled rolled steels. The accuracy of ± 10% Fig. 38 was similar to that obtained based on microstructural estimates, but no less tedious, and an example of the summation of the components is given in Fig. 39.
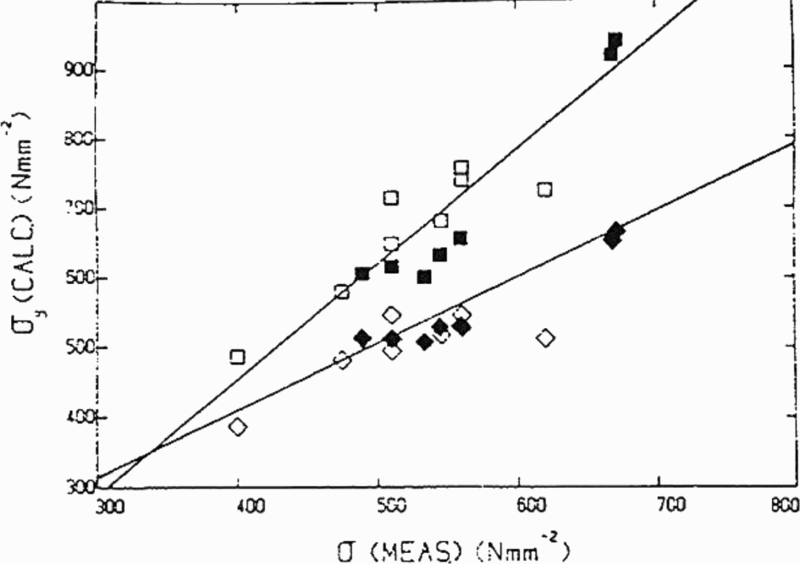
Comparison of measured and calculated yield stress data: ♦ σL (Ref. 114); ▪ σR (Ref. 114); ▪ σL (Ref. 256); ▵ σR (Ref. 256)
Hornbogen and Staniek 253,307 pointed out that, whereas the breakdown of coherence to develop incoherent precipitates via partially coherent precipitates 308 may involve the generation of dislocations, the presence of coherent precipitates alone does not. This led them to conclude that a superposition of coherent particles and dislocations was not relevant and that linear addition of estimated strengthening components gave a satisfactory agreement with the measured yield strength.
Recently, a three-dimensional analytical model of precipitate strengthening with superposition laws was developed, and characterisation parameters were obtained by local electrode atom probe tomography. 309 This allowed a detailed calculation of yield strength of secondary hardening at different stages of aging due to multiple precipitates of Cu and M2C in a BA160 steel to be made. The model predicted σy as a function of aging time at 450°C within ± 13% of σM. The authors state that this is the first time that the prediction of the yield strength directly from the microstructure of a multiphase steel was shown to be feasible.
Another recent study systematically investigated the stress–strain behaviour of ferrite and bainite with nanosized vanadium carbides in low carbon steels. 310 The ferrite samples were obtained through austenite–ferrite transformation accompanied with interphase precipitation, and the bainite samples were via austenite–bainite transformation with subsequent aging. The contributions from solute atoms and grain boundaries were simply added, whereas those from dislocations and precipitates were treated by taking the square root of the sum of the squares of two values, giving σN. This is a different approach from that given by equations (20) and (21). From the data they give, σL compares well with σM for ferrite, ≤ 9.6% over estimate, while comparison with σN is 9.6–19.2% under estimated. For bainite, σN ≤ 9.4% underestimate is superior to σL summation, which is 19–37% overestimated. 310 Kostryzev et al., 311 who used a linear addition of components to quantify the strengthening components of Nb–Ti FP thermomechanically processed Nb–Ti steels, also obtained disagreements between measured and calculated σy values, which could reach 54%. These were attributed to overestimates in solid solution and precipitation contributions, and the errors increased with decreasing deformation temperature.
Despite the detailed consideration of the models used in calculating the individual components strengthening and the care taken to ensure the highest accuracy from microstructural parameters, 115,256,288,309–311 such as the foil thickness required for dislocation density, determination for σd and precipitate volume fraction f needed for estimates of σp, no improvement on the accuracy of the estimate in σy from microstructural measurements has been achieved. It is apparent that the calculated σy has, at best, a ± 90% accuracy compared with σM. The error is often due to the inhomogeneous microstructure and the high magnifications required to image small particles and dislocations. This has implications for the determination of ρ and f.
Toughness
For many commercial applications of MA steels, good toughness is the over-riding property requirement. The development of a small grain size is the only means of making a simultaneous improvement in strength and toughness. Toughness is normally considered in terms of the energy absorbed during deformation by impact and, in MA steels, is defined by the temperature at which the fracture mode undergoes a ductile to brittle transition, or some fixed value of absorbed energy, for example 55 J, for a Charpy test specimen of given cross-sectional area. In 1947, Barr and Tipper,
312,313
working at Colvilles in Scotland on the Liberty ships facture problems,
314
showed that the ductile–brittle transition temperature was raised by an increase in the ferrite grain size. Influenced by this work, Petch,
315,316
after dealing with yield and cleavage strength–grain size relationships, turned his attention to fracture toughness and, in an initial study, found a similar relationship with grain size to that of strength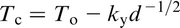
This classic work did not take into account the influence of carbide films, which are normally present at grain boundaries. It is generally agreed that McMahon and Cohen
317
were among the first to appreciate the importance of the role of carbide films at grain boundaries in the fracture process in carbon steels. Detailed measurements on carbide film parameters were undertaken by Mintz et al.
318,319
to obtain statistically significant quantitative data to test the models of Smith,
320
Almond et al.
321
and Knott and Curry.
322
These data showed that, over the grain size range of 1–14mm− 1/2 (d = 10–500 μm), the change in T
c was 160°C, whereas the greatest change that can be produced by changes in carbide thickness alone was 60°C. These data provided support for the conclusions of Almond et al.
321
. The above publications were among those which included ideas leading Petch
323
to revisit the topic some 25 years later than his earlier work and to incorporate both grain size and carbide thickness into his model. The results in Fig. 40, where the cleavage strength for fracture σc is plotted against d
− 1/2 for a range of carbide thicknesses t. The line OA, Fig. 41 shows that σc is directly proportional to d
− 1/2 and unaffected by carbide. At the other extreme, with a fine grain size and a coarse carbide, where t equals 5 μm, the carbide has a dominant influence on σc. The region between these two extremes shows the strong influence of grain size. Petch pointed out that, over the grain size range of 3–10 mm− 1/2, i.e. 100–10 μm, and with some carbide refinement concurrent with grain refinement, equation (3) can normally be used. However, when the grain size is finer (as is sometimes the case in MA steels), then the equation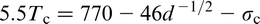
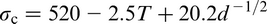
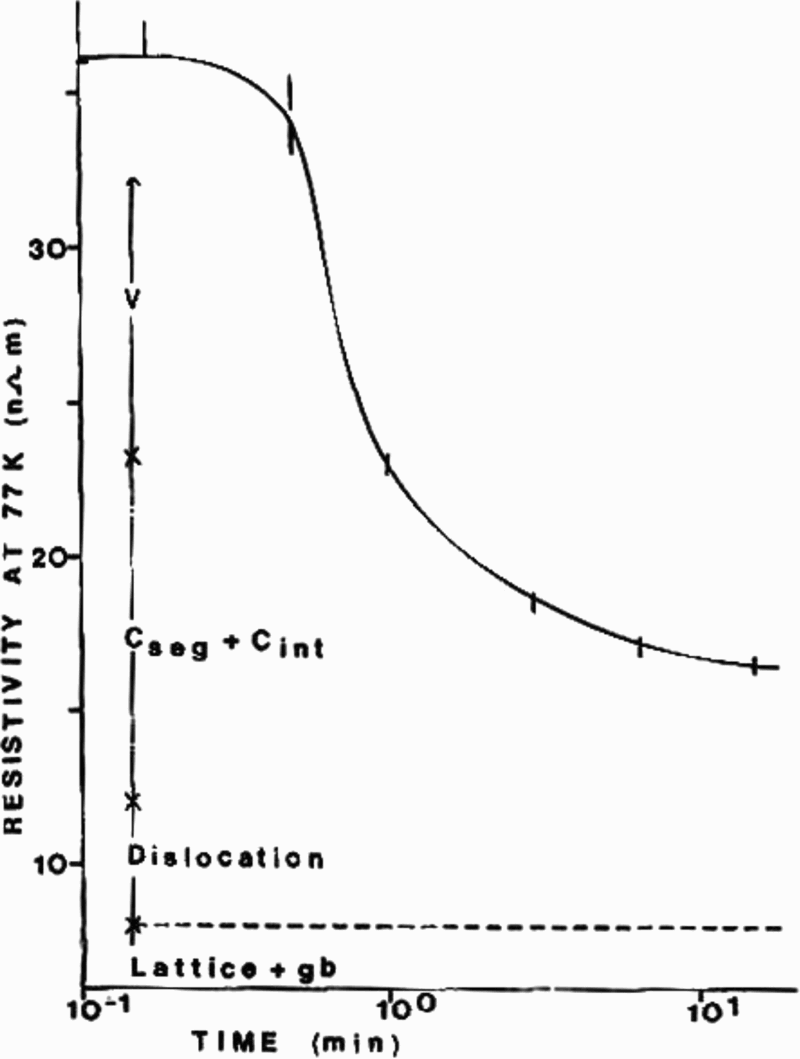
Comparison between calculated resistivity contributions for Fe–C–V alloy, totalling 32 nΩ m, and experimental value of 36 nΩ m (Ref. 255)
Bingley, 325 in a more extensive paper, studying both laboratory and commercial MA steels, confirmed the earlier d 20 analysis. 324 .
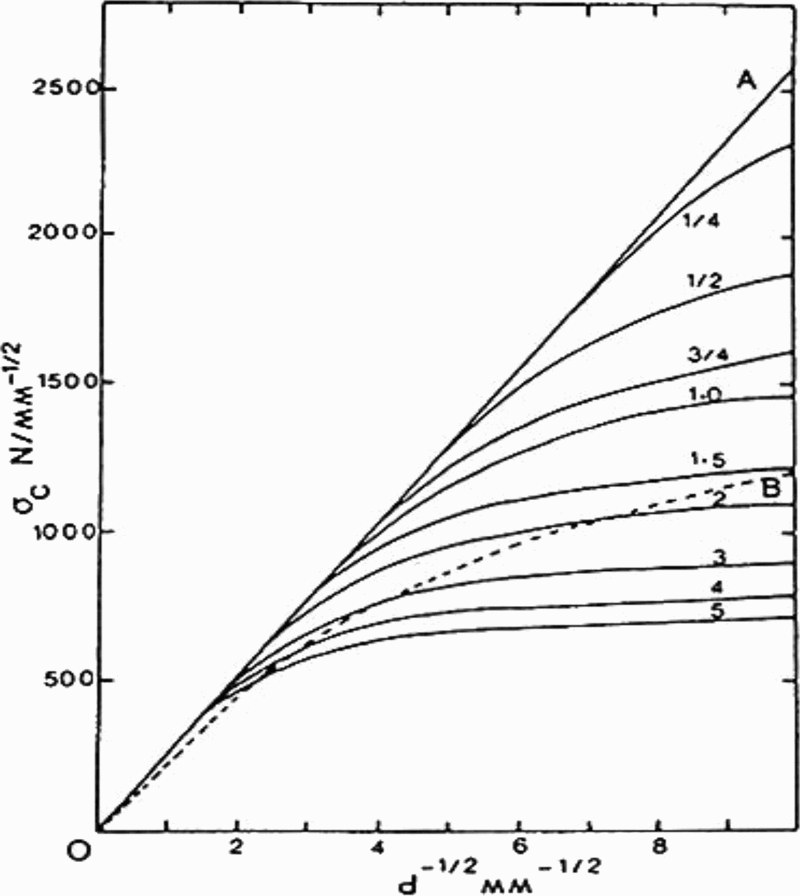
Cleavage strength for fracture at yield σc versus d − 1/2 for various carbide thicknesses t (μm) 323
Ductility
Ductility at ambient temperatures is normally recorded following tensile testing, as elongation percentage and reduction of area. A multiple regression analysis of 60 casts of carbon steels containing ≤ 0.25%C, some with Nb and Al additions, was undertaken by Pickering and Gladman
252
to quantify the factors that control the properties, including RA%. This analysis resulted equation (27)
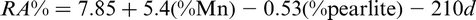
Figure 42 shows the calculated RA% plotted against the observed RA%. The correlation factor is 78%.
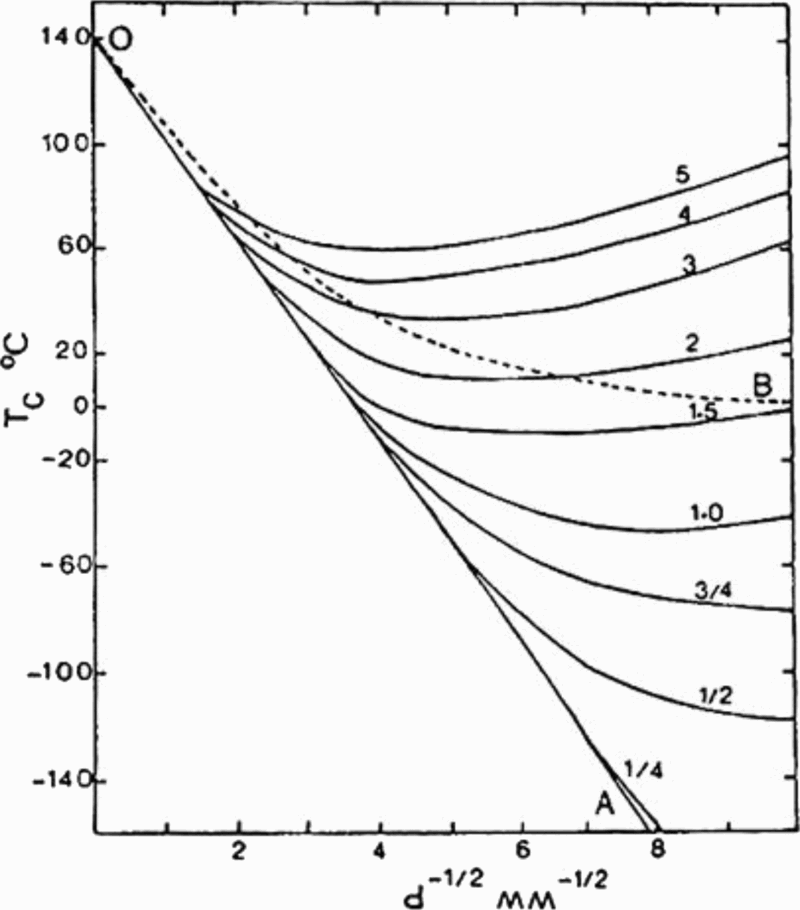
Calculated T c data for various values of t and d − 1/2 (Ref. 323)
A more recent regression analysis by Mintz et al.
326
from tensile tests undertaken over a range of temperatures and strain rates produced equation (28). This predicted a min. RA% with an accuracy of ± 12% (95% confidence limit)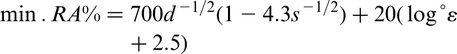
The importance of a small ferrite grain size and limiting the grain boundary carbide thickness are well established means of controlling the toughness to acceptable levels in MA steels from +100 to − 100°C. These parameters can also influence hot ductility, which is important in processing MA steels. Hot ductility testing was originally undertaken by torsion testing, 327–329 as this method was considered to provide a good simulation of hot rolling. For investigating continuous casting failures and predicting the likelihood of transverse cracking during the unbending operation, hot tensile testing, often using a Gleeble machine, is the preferred method. 326,330–335 The sample is cooled at the rate experienced by the surface of the strand during the continuous casting operation, ∼60 K min− 1, and is strained at rates between 10− 3 and 10− 4 s− l, which are chosen to simulate those associated with straightening. 326 The variation in hot ductility with temperature is obtained by plotting reduction in area RA% versus test temperature, as in Fig. 43. For both the cast and wrought states, a decrease in hot ductility, described as a ductility trough, occurs between 700 and ∼1200°C; intergranular fracture is observed. The ductility trough has implications, both for normal continuous cast, and for thin slab cast steels. 326,329–334 Several explanations have been suggested for the ductility trough, including failure within a thin film of ferrite at the austenite grain boundaries, or SIP of carbonitrides, which pin austenite grain boundaries and therefore reduce boundary mobility, and in niobium steels, precipitate free zones. 329 Fig. 43, taken from the work of Crowther and Mintz, 331 shows that refining the austenite grain size is generally beneficial to hot ductility, as it reduces the width of the trough and increases the minimum RA%. They showed that decreasing the austenite grain size from 350 to 70 μm increased the RA by 20%.
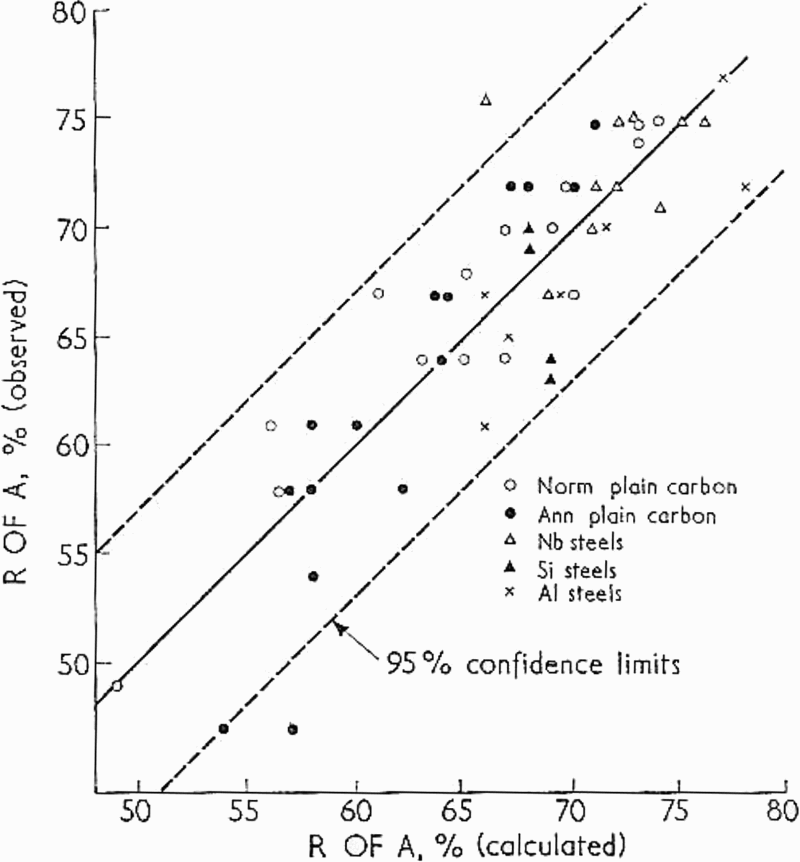
Observed and calculated reduction of area 252
Over many years, Mintz and co-workers have conducted extensive investigations to understand the role of the transition metals, Nb, Ti and V, together with Al, N and C, on hot ductility of steels. 326,331,333,334 It is well established that increasing the aluminium content of carbon–manganese steels causes a progressive deterioration in hot ductility. 14 During the γ → α transformation, precipitation of AlN occurs preferentially in ferrite, especially under deformation conditions. Microvoid nucleation at the embrittling AlN particles is one explanation for the impairment of the hot ductility. Niobium steels are known to have hot ductility problems. On the other hand, aluminium additions to niobium steels are thought to refine Nb(C,N) grain boundary pinning precipitates, which mitigates loss in ductility.
Care must be taken with titanium additions. As seen in Fig. 44, adding 0.02–0.03%Ti to the C–Mn steels detailed in Table 9, can have a beneficial effect on hot ductility, 331 by reducing both the depth and width of the trough. This is due to the restrained effect on austenite grain growth of coarse TiN particles still remaining out of solution, after soaking at 1350°C. For steels 4–8 in Table 9, it was considered that the suppression of AlN precipitation, due to removal of nitrogen to form the more stable nitride, TiN, resulted in improved hot ductility (Fig. 44). However, if due to increased cooling rates during the simulation, fine TiN particles precipitate restricting particle growth, the beneficial influence of the titanium addition is lost. Therefore, variation in cooling rates during hot tensile testing, to simulate as close as possible the conditions pertaining to the continuous casting operation, have been an important objective in this research. 326,335 A major concern in simulating the continuous casting process with a Gleeble machine is in deciding the thermal programme, which will ensure that the appropriate RA% is recorded. 336,337 Introducing a cooling regime similar to that undergone by the strand during commercial continuous casting has been shown for low C low Ti containing C–Mn–Nb steels, always to result in an improvement in hot ductility below 900°C. When the test RA% is >40%, the steel should not be susceptible to this cracking problem. 335 The addition of zirconium can also reduce transverse cracking, but cannot be added to niobium grades to be used in the as rolled or normalised conditions, since strength and toughness are impaired. However, zirconium has been successfully added to Q&T grades. 338
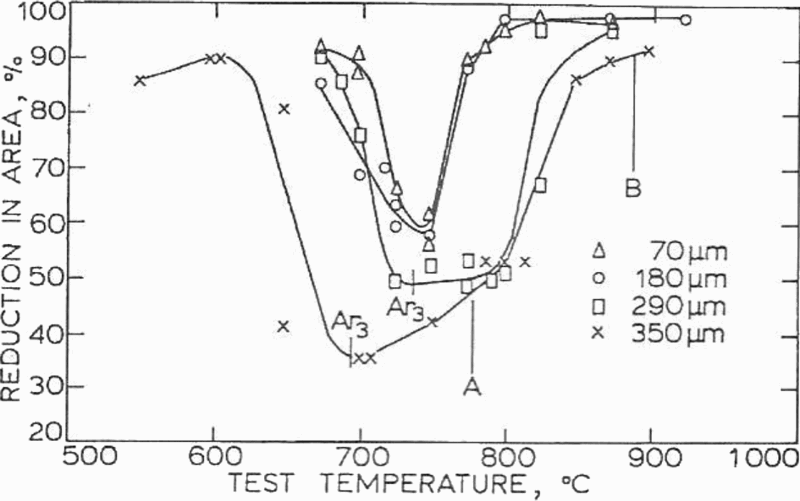
Hot ductility curve for 0.19%C steel after normalising and heating directly to test temperature 331
A general requirement of continuous cast MA steels, is that while the composition is important for avoiding cracking following processing, the choice of alloying elements must be such that the strength and toughness of the final product meets specification. Where the as cast slab is to be cooled to ambient before reheating for controlled rolling, then many of the precipitates formed during casting will be taken into solution and the microstructure controlled as discussed in the previous sections. However, if the TSDC route is to be followed, where the reheating temperature and time are limited, then steel compositions may require modification. Vanadium steels, with the lowest solution temperatures of carbides, nitrides and carbonitrides of the conventional alloy transition metals used for microalloying, may have advantages over niobium steels. 11 Roy et al. 339 modelled the effect of segregation on the stability of microalloy precipitates and on the size distributions at different regions (solute rich and solute depleted), which is not well understood. They compared two as cast slabs: 0.09C–1.42Mn–0.035Al–0.050Nb–0.019Ti–0.05V (A) and 0.07C–1.20Mn–0.034Al–0.041Ti (B). The nitrogen level was not given.
Cuboidal TiN, cruciform and cuboidal (Nb,Ti)(C,N), and spherical NbC and VC, precipitates, which had the greatest volume fraction, were characterised and varied with position in slab A. TiN precipitates, with various morphologies and sizes were found in slab B. The model proposed for microsegregation, which was a feature of the slabs microstructure, gave a satisfactory agreement between experimental observations and predictions for precipitate size distribution and the amount of precipitates in the interdendritic and dendrite centre regions of segregated slabs. It was concluded that this type of model may help in avoiding hot cracking, in the selection of soaking time and temperature and predicting austenite grain size during soaking and in designing the rolling schedule for achieving maximum benefit from the microalloying precipitates. 339
While problems deriving from residual elements are not specific to MA steels, they should be touched upon. A significant proportion of recycled steel (scrap) is now part of the normal charge load during steelmaking, and it is accepted that the level of residuals in steels is increasing. Basic oxygen steel uses around 10–12% scrap only, while electric arc ˜75% recycled scrap. Residual elements, such as copper, nickel, tin, antimony and arsenic, at trace levels, have long been recognised as leading to potential hot shortness problems. They are known to concentrate in the layer of metal immediately beside the oxide scale. These are of particular concern in tube making and forging of carbon steels. This subscale enrichment can eventually lead to the precipitation of a phase, which is molten at temperatures <950°C.
As explained in a survey by Melford,
340
it is more often the interaction between a combination of trace elements that has to be understood if their net influence on high temperature mechanical properties is to be evaluated. An earlier paper,
341
Melford produced a useful guide to the limiting residual contents that could be tolerated in the form of the expression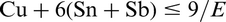
E = average concentration of residual in the subscale/bulk residual concentration
A literature search has not produced any extension of this work to MA steels.
Summary
Over the 56 years, since the first publication by Beiser, 24 generally acknowledged to bring niobium steels to the attention of the world, much progress has been made. The yield strength of mild steels ∼250 MPa was raised up to 400 MPa with an addition of 0.03%Nb. The introduction of controlled rolling made these strength levels possible, together with good toughness in weldable steels. Many of the ideas of using accelerated cooling and modifying steels compositions, to produce bainitic and acicular, with strengths above 600 MPa, were known from laboratory research in the 1970s. Since this time, the expansion of linepipe production and the automobile industry requiring high strength and toughness in thinner sheet, to name two examples, transferred these possibilities to the production line. The use of multiple additions of niobium, titanium and vanadium, sometimes with a high nitrogen content, is now commonplace, while processing of MA steels using thin slab direct charging technology is on the increase, as is the application of FSW. The recognition of the role of microstructure in achieving these improved properties has been at the root of many of these advances. Electron microscopes, coupled with an array of analytical equipment, now allows far more detailed information to be processed quicker and with user friendly software, than even a decade ago, but at an increasing cost. While FP MA steels are expected to provide the bulk production, the future may see the development of a spectrum of MA steels, not only dictated by economics but also tailor made for the processing route. This could well be the case for both thin slab cast direct charge steels, with their restricted reheating and rolling passes, and friction stir welded steels, with very inhomogeneous microstructures in the SZ, especially for thicker plate, where a double pass is used.
Postcript
The author has been involved with MA steels for over 50 years and has been associated with many of those who have made major contributions to this field. Initially, this was during the time at Hinxton Hall, the Tube Investments Research Laboratory, when we were involved in 30 Oak, a steel produced by Round Oak Steelworks and purported to have a lower yield stress of 30 tsi (∼400 MPa), which was true, at ≤ 10 mm thickness. As an undergraduate at Sheffield University, E. O. Hall taught me physics and J. H. Woodhead and Professor (later Sir) R. W. K. Honeycombe, metallurgy. The latter later invited me to write a review for Science Progress (Ref. 1), and I was included in those who presented work at a celebration of his 70th birthday (Ref. 2). I met W. B. Morrison at Colvilles Steelworks in the mid-1960s, having previously visited Swinden Laboratories of United Steels, to talk with (Professor) T. Gladman and (Professor) F. B. Pickering about AlN precipitation, which I had been taught to SAED index by (Professor) D. W. Pashley, who with (Sir) J. W. Menter, introduced me to TEM. Professor N. J. Petch joined the Metallurgy Department of Strathclyde University in the early 1970s. We had many discussions, and he became a firm friend. Dr N. A. McPherson undertook PhD work with me, as did Drs N. Gao, K. He and Y. Li, who continued in the Group as post-doctoral research fellows. All of us benefitted from many years of collaboration with Professor A. J. Craven, University of Glasgow, through PEELS analysis of nanoparticles.
