Abstract
The present work critically reviews the scientific and patent literature on low melting bismuth based oxide glass frits in materials for electronics, sensors and related applications such as sealing glasses, solar cells, architectural and automotive glass, the main motivation being to replace lead based materials by environmentally more benign ones. Due to similar glass forming properties of Bi and Pb, Bi based glasses are the closest ‘drop-in’ alternative for lead bearing formulations, and are therefore actually replacing them in many applications, helped also by previous experience with Bi containing materials in thick film technology and component metallisations. The outstanding issues are discussed, e.g. matching the lowest processing temperatures achieved by the classical lead based glasses without sacrificing durability and stability, as well as stability versus chemical reduction. Finally, consideration is also given to special ‘heavy’ glasses (often containing Bi and Pb together) that are useful in fields such as optics, superconductors and nuclear technology, as well as to specific Bi2O3 containing crystalline compounds.
Introduction
Low melting glasses in electronics and other applications
As for ceramics, inorganic glasses, glass–ceramic and glaze materials have long gone beyond their traditional uses to address a wide array of modern technological challenges,2 – 43 in fields such as electrical engineering/electronics/sensors,9 – 32 solar energy,42, 43 protective and decorative coatings,20, 27 – 35 optics/optical telecommunications,36, 37 structural mechanics,8 medical,38 nuclear technology,6, 7 superconductors39 and microfluidics.40, 41
Owing to performance and cost criteria, most standard glasses have relatively high softening points. However, there are many technological applications where a low softening temperature is required, in order to lower energy expenditure, avoid damaging devices in contact with the glass during processing or ensure compatibility with other materials:
hermetic sealing of packages, lamps, electrical feedthroughs and semiconductor devices13, 14, 16, 17, 19, 44, 45
hermetic sealing and mechanical attachment of sensors23, 27 (Fig. 1)
overglazing of automotive, packaging and architectural glass33, 34, 46 – 48
photovoltaic (PV) solar cell technology – conductors and contacts42, 43, 49 – 53
enamelling of aluminium in architecture and home appliances35, 54 – 58
thick film (TF) electronics and other devices21, 22, 24, 25, 27, 59 on various substrates:60 glasses for TF resistor (TFR),61, 62 conductor,63, 64 overglaze, dielectric65 and sealing15 – 19 materials (Fig. 1, the section on ‘PbO in low melting frits and TF technology’); especially, special low firing compositions for fabrication of circuits and sensors on glass or metals.1, 28, 66 – 72
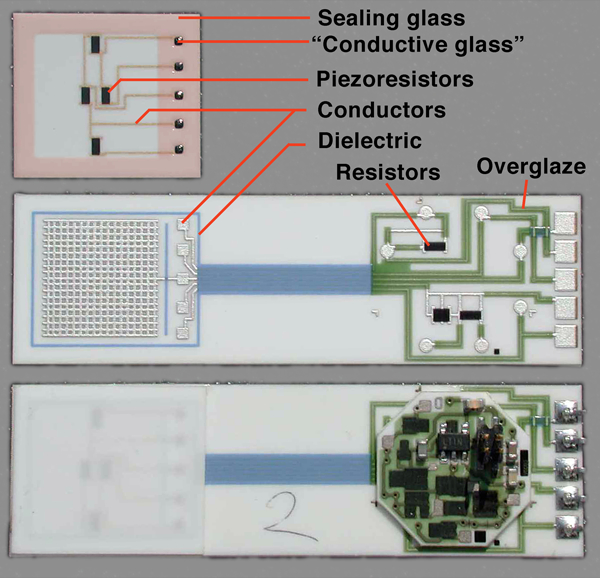
Example TF circuit, piezoresistive pressure sensor27, showing typical involved materials: reddish tint added to sealing glass to enhance visibility; ‘conductive glass’ seal = low firing TFR composition
For these applications, glasses are often formulated as frits (e.g. finely divided powder), which may be applied, dispersed in a suitable medium, onto a substrate by various methods such as slip casting, screen printing, roller/curtain coating, spraying, dispensing and electrophoresis, or as preforms for sealing. Classically, the aforementioned applications have to a great extent used lead based glasses, which have a rather unique combination of desirable properties,10 – 12 as will be discussed hereafter in the section on ‘PbO in low melting frits and TF technology’. Table 1 compiles the compositions and melting points/processing temperatures of selected classical low melting lead based glasses.
Representative compositions (cation-%) of low melting lead based glasses [Temperatures = melting points (eutectics) or processing temperatures (others)]
*Compositions on a cation basis, i.e. LiO0·5, NaO0·5, PbO, ZnO, BiO1·5, AlO1·5, BO1·5, SiO2, etc.
Figure 1 shows a TF integrated pressure sensor27 that illustrates many of the aforementioned applications: hermetic sealing of the sensing membrane combined with mechanical attachment and electrical contact, encapsulation through a hermetic dielectric of a wetted surface, conductors, resistors and overglazes.
While most low melting glasses are used on a substrate or for sealing, there are several significant ‘bulk’ applications of low melting or relatively low melting glasses:
Main performance criteria
The performance criteria in selecting a low melting glass depend on the application. A short overview is given in Table 2 (layers and sealing) and Table 3 (bulk) for the aforementioned applications. In contrast to most ‘film’ applications, most of the ‘bulk’ examples listed above depend specifically on the presence in the glass of HMOs, rather than just require a reliable low melting glass. In optics, heavy, polarisable cations such as Pb2+ and Bi3+ impart to the glass a high diffraction index, a high dispersion, strong nonlinear effects and better IR transmission,12, 36, 112,whereas CRT and gamma radiation shielding benefits from the strong absorption of ionising radiation by heavy elements.11, 129, 130
Requirements for (relatively) low melting glasses for layers and seals (‘−’ = normally not important; ‘+’ = significant; ‘++’ = critical)
*(a) Critical for sealing organic parts and semiconductors/thin film devices; (b) match not critical for thin layers on planar substrates – avoid tensile stresses; (c) stress relaxation→risk of signal drift; (d) depends on environment; protection of seal with organics sometimes possible; (e) often significant due to seal overlapping conductor tracks (see Fig. 1); (f) critical to avoid degradation;45 (g) difficult combination of low process temperature and low thermal expansion, especially directly on chip, achieved through fillers;18 (h) surface states in semiconductors also important29, 30 – reduce/avoid alkalis, which are mobile under electric field; (i) important on sensitive substrates/other layers, e.g. glass, metals, prefired TFRs; (j) critical for underwater applications27 or for automotive.48 (k) needed for acid planting baths.
Requirements for low melting ‘bulk’ glasses (‘−’ = normally not important; ‘+’ = significant; ‘++’ = critical)
*(a) Minimal volatilisation of toxic/radioactive compounds89, 95 and stresses in large parts/bonds; (b) good transparency and high refractive index; (c) minimal leaching of toxic and radioactive components; (d) browning of glass unimportant for tube part; shielding against X-rays required; (e) depending on application: high refractive index, IR transparency, nonlinearity, luminescence efficiency; (f) conservation of transparency despite high radiation doses.
PbO in low melting frits and TF technology
Thick film electronics makes wide use of glassy compounds, used as main components of overglazes, permanent binders for dielectrics and resistors, and also as frits/adhesion promoters for conductors.21, 22, 24, 59, 61 – 65 Note that the technology and materials are very similar for other applications such as architectural/automotive/solar cell overglazes and conductors.
For conductors, resistors and overglazes (and relatively old dielectrics), most classical low melting frits are based on the PbO–B2O3 (lead borate) system, with mainly SiO2, ZnO and Al2O3 additions. Several phase diagrams and property maps exist for these systems.21, ,91 – 93, 136 – 144
Table 1 gives several representative ‘traditional’ glass compositions, compared with that of traditional leaded ‘crystal’ glass89 and some representative eutectic compositions in the phase diagrams. Throughout this work, compositions are given on a cation basis unless specified otherwise, as by Dumbaugh and Lapp;36 this convention facilitates comparison with Bi2O3 based glasses when PbO is replaced by approximately equimolar amounts of ‘BiO1·5’.
By altering the composition, the properties, especially the processing temperature and the tendency to crystallise, can be easily and reliably tuned. Low melting glasses in this system, which have a composition relatively close to the PbO–B2O3 binary eutectic, allow a reduction in binder glass amount in TF conductors, and/or a decrease of processing temperatures down to ∼400°C for low temperature TF conductors, dielectrics, overglazes and resistors,67, 72, 85, 86 for glass sealing (‘solder glasses’) of cathode ray tubes (CRTs) and flat panel displays (FPDs), or for glass encapsulation of semiconductor devices.15, 16, 73, 74
Low melting glasses in the lead zinc borosilicate system can be formulated as essentially ‘stable’, e.g. with little or no crystallisation during firing or sealing, or devitrifying, e.g. forming significant amounts of crystalline phase and thus conserving dimensional stability upon later reheating. These latter crystallising glasses are referred to as glass–ceramics, vitroceramics, or ‘cements’ in glass sealing parlance. In these compositions, devitrification is usually favoured by high amounts of ZnO, whereas B2O3, SiO2 and Al2O3 tend to stabilise the glassy state (Table 1). To achieve even lower processing temperatures and/or promote wetting, compounds such as CuO, Fe2O3, Bi2O3, V2O5, WO3, MoO3 and fluorine (batched as CaF2, PbF2, ZnF2, BiF3, etc.) can be added to the glass formulation.19,75,145 75, 145 – 152 Interestingly, fluorine, which is effective in lowering the processing temperature, was found to have better compatibility with glasses where a sizeable amount of PbO was replaced by Bi2O3.149 Very low processing temperatures may be reached by glasses largely based on PbO–TeO2,45 PbO–V2O5 145, 147, 148 and especially SnO–SnF2–PbO–PbF2–P2O5.153
Glass frits are often used in conjunction with other materials that act as fillers (Table 4): insulating powders for dielectrics/overglazes/encapsulation/sealing glasses,18, 67, 145, 147, 149 – 152, 154 conductive oxides for resistors,61, 62, 155 – 157 metal powders and adhesion promoters for conductors,63, 64, 88, 158 – 160 pigments, etc. Even for applications such as sealing, encapsulation or TF overglazes, where they are not intrinsically required, fillers are often found necessary or advantageous in practice, mainly to adjust the coefficient of thermal expansion (CTE) of the deposited material to that of the substrate(s) (see Donald’s review20 for an extensive list of filler CTEs). The filler can also be used as a nucleating agent to better control the crystallisation process of a devitrifying glass. Alternatively or additionally, chemical and mechanical stabilisation of a glass can be obtained by reaction with the filler; an example is the reaction of lead bearing glass with TiO2 and MoO3,161, 162 yielding both an increase of the filler volume (by formation of PbTiO3/PbMoO4) and of the glass softening point (by the resulting depletion of glass PbO content).
Representative materials/fillers used in conjunction with glass frits
Correctly formulated, both stable and devitrifying lead based glasses achieve an excellent combination of very consistent and reliable properties, relatively large processing windows, acceptable corrosion resistance and low processing temperatures, all this without requiring, in their composition, alkaline oxides, which are detrimental for insulating properties (due to the mobility of alkaline ions under electric fields) and chemical durability, and impart a high CTE that is deleterious in most cases (except for substrates with CTE greater than ∼10 ppm K−1). Owing to these advantages, which have been recognised for a long time (see the section on ‘Lead and bismuth in glass: a historical perspective’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1), these lead based glasses have achieved widespread use, and have been the object of extensive studies and reviews.10 – 12
An overview of the current status of commercial TF compositions is given in Table 5; modern multilayer dielectric compositions such as ESL 4913 are commonly lead free,163 and recently introduced (relatively) low melting overglaze materials use Bi2O3 instead of PbO. Surprisingly, even an old composition such as Ag/Pd conductor DP 9473 uses a Bi2O3 based glass.164
Qualitative composition (+++ = high, ++ = medium, + = low, ? = very low or absent) of commercial TF inks (T f = firing temperature): dielectrics167 (compared with LTCC),168 – 170 conductor164 and resistor.85 Boron most likely present in all these compositions, but not always detectable by the analysis methods – mentioned where explicitly formulated/detected
*Used firing temperature for sample processing, i.e. not necessarily the one specified by the manufacturer.
†LTCC: low temperature cofired ceramic.
‡DP: DuPont Microcircuit Materials (Bristol, UK); ESL: ElectroScience Laboratories (King of Prussia, PA, USA); Her: Heraeus Precious Metals, Thick-film division (Hanau, DE).
§Glass part only.
The trend away from lead
In recent times, there is a trend towards removing lead from electronic materials due to its toxicity (see the section on ‘Toxicity of elements in glasses’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1), a move spurred by the enactment of the European Union RoHS (Restriction of Hazardous Substances) directive.171 This has already largely taken place in the field of metallic solders, where the Sn–Ag–Cu alloy (‘SAC’) has become the standard to replace the classical Sn–Pb–(Ag) eutectic.172 Although glasses in electronics are mentioned under the list of exemptions,173 the directive requires this list to be periodically reviewed in the future, and further restrictions on the use of lead bearing glasses are therefore likely in the medium term. Moreover, cadmium, also a popular addition to low melting glass frits, must be abandoned.
In contrast to the case of metallic lead and its simple, relatively soluble compounds such as litharge and massicot (PbO), minium (Pb3O4) and ceruse (2PbCO3.Pb(OH)2), lead in well prepared glasses and glazes was traditionally considered to be stably bound, and therefore not a health concern, provided the composition was well formulated, and, for glazes, sufficiently fired.174 This perception drastically changed after the widely publicised 1991 work of Graziano and Blum,175 who demonstrated that important amounts of lead could be leached out of ‘crystal’ glass over time by (acidic) wines and spirits. This alarming report was later somewhat contradicted by more realistic tests,176 while other research177 showed that water dredged from Pb contaminated sediments remained well within the official limits, and, more to our point, that lead bearing low temperature co-fired ceramic (LTCC) compositions could even exhibit good biocompatibility.178, 179 However, although human exposure to lead and corresponding blood levels have drastically dropped in recent times, the ongoing controversy over the effects of low lead levels, especially for children,89 will likely generate additional regulatory pressure on its uses (see the section on ‘Toxicity of elements in glasses’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1). In the case of ‘crystal’ glass, this has led to research activity towards lead free substitutes,90 which showed that most of the properties of original ‘crystal’ could be largely duplicated (although the working range was somewhat smaller), while guaranteeing minimal leaching of potentially dangerous substances.
The situation of glasses in electronics might seem less critical, due to the smaller volumes involved and to the fact that contact with foodstuffs or beverages is (usually) not specified. However, electronic glasses often require lower processing temperatures and only little or no alkali ions are tolerable when good insulating properties are required. Therefore, they can contain much higher amounts of lead than ‘crystal’ (⩾24 mass-%): ∼65% for classical 850°C firing TFRs62 and up to ∼85 mass-% for sealing glasses12, 15, 16, 73, 74, 150 and low firing TFRs85, 86 (Table 1). This results in much lower stability against dissolution in acids,140, 141 which again raises the issue of contamination of groundwater from disposed electronics waste. Moreover, very low temperature electronic encapsulant and sealing glasses may contain even more dangerous metals such as Cd and Tl.165 Therefore, especially for these applications, alternative materials are needed. Finally, even if the final product may be considered stable, occupational exposure during processing is always a concern.
In TF electronics, removal of lead started in the 1980s with multilayer dielectrics, where traditional ceramic filled glass formulations have given way to crystallisable types, which can be formulated lead free.163 More recently, there has been an effort to remove lead from frits in conductors,180 overglazes181 and sealing glasses,182 and commercial lead free compositions have become widely available (see Table 5). However, resistors (and to some extent sealing glasses and overglazes) have lagged behind in this trend, due to the exceptionally easy processing of lead based glasses and the considerable development work required for entirely new TFR series. Lead free glasses were widely used in the 1970s to 1980s, including for resistors, due to the then considerable development of nitrogen firing TF systems.183 – 189 However, these materials have largely fallen into disfavour, due mainly to performance and reliability problems, especially in ensuring proper organic vehicle burnout.190
While ‘lead free’ is an important aspect in the present review, glasses containing both Bi2O3 and PbO are also included, as they are relevant for specialised applications.
Low melting frits: alternative systems
After the ‘classical’ lead based glasses (see previous sections), a short discussion of the potential oxide glass substitutes based on elements other than bismuth is given in this section. The reader is referred to other reviews for halide, chalcogenide (non-oxide) and chalcohalide glasses.37, 191 – 194
Borate/borosilicate/silicate glasses
Several glass systems have been proposed to replace lead bearing frits. In the case of multilayer dielectrics for ‘standard’ (firing at 850–900°C) TF technology, crystallising glasses containing mainly CaO–Al2O3–B2O3–SiO2, forming phases such as anorthite or celsian, have largely displaced lead bearing types in both screen printed 850°C firing multilayer dielectrics163 and LTCC,9, 168, 169 with mostly improved performance, and therefore provide a satisfactory solution.
A complete lead free cofireable TFR+LTCC system for processing at 900°C has been implemented by Bosch in its production of car engine control units (ECUs).169 This system is based on two glasses: an anorthite crystallising CaO–Al2O3–B2O3–SiO2 glass (as in the case of dielectrics),163 and a lower melting Na2O–B2O3–SiO2 one, which probably acts as a binder. In spite of its good properties, its extension to a general purpose TFR system would be unlikely, as the complicated reactions involved in its processing require a very rigid and tightly controlled manufacturing process: as the resistor has a higher CTE than the substrate, it has to be co-fired with its overglaze, which imparts a protective compressive stress. Such very standardised processes may probably also be used by the chip resistor manufacturers to produce lead free components.
There have been some attempts at making general purpose TFRs based on similar glasses, with RuO2 195 – 198 and ruthenate perovskites,197, 199 – 202 or pyrochlores82, 203 as conducting phases, which have partly resulted in promising properties, albeit with problems of high process sensitivity and the requirement of a large amount of, expensive, conducting RuO2.
The high encountered process sensitivity is expected, as these glasses tend to be not so ‘well behaved’29, 198, 204 – 207 as lead based ones, which may be formulated to be virtually non-crystallising;45, 73 the glass forming range and stability of the lead free glasses is in general more limited, and the processing range is restricted to relatively higher temperature applications than for lead bearing ones, or other properties such as CTE matching and durability are compromised. Therefore, the abovementioned lead free glasses are not applicable to compositions requiring very low processing temperatures in applications such as low firing TFR overglazes and sealing glasses in flat screens.206, 207
Nevertheless, silicate, borosilicate or borophosphate glasses have found large scale low temperature applications such as the overglazing of architectural and automobile glass,34, 46, 47 and enamelling of aluminium.35, 47, 54 – 57, 208 In these applications, the processing window between sufficient melting of the glass and degradation of the substrate is narrow, and firing schedules are tightly controlled, so a very wide stability range against crystallisation is not necessary. Moreover, significant amounts of alkali oxides, which are detrimental for insulator dielectric applications, are tolerated within the limits set by their detrimental effect on corrosion resistance; in enamels for aluminium, they impart a desirable high CTE to the glass.
Phosphate glasses
Phosphate glasses209, 210 are an interesting alternative, as they usually have low working temperatures. On the other hand, high CTE and water absorption are potential issues. An example low melting system is Na2O–Cu2O–CuO–P2O5,211 but chemical durability is only passable and it contains a high alkali content, limiting its use in electronics.
Many promising phosphate glasses are based on/derived from the ZnO P2O5 system, with additives such as B2O3, SiO2, MgO, CaO, Al2O3, Fe2O3, V2O5 and Nb2O5.97, 212 – 216 For instance, Nb2O5 additions were claimed to allow sealing glasses with processing temperatures as low as 500°C, while retaining good durability and moderate CTE values.216 Explorative TFRs have also been formulated with such glasses, yielding, however, compatibility problems with Ag terminations.214, 216 As in borosilicates, a good combination of low processing temperature, stability and durability is imparted by PbO, and corresponding lead iron phosphate glasses have drawn interest for vitrification of high level radioactive waste; avoidance of PbO is possible for this application if somewhat higher processing temperatures can be accepted.6, 95, 97, 98
Glasses based on divalent tin
A major breakthrough towards low melting phosphate frits was achieved with the SnO–ZnO–P2O5 system.182, 217 SnO, with Sn in the unusual +2 oxidation state, seems to behave in a similar manner as PbO, without the toxicity problems. In fact, comparing simple binary SnO, PbO and ZnO phosphate glasses, SnO gives the lowest glass transition temperatures, in the order SnO<PbO<ZnO.218, 219 Thus, SnO–ZnO–P2O5 glasses (with more SnO than ZnO) can achieve flow characteristics similar to those of traditional lead based frits,182 while remaining lead and alkali free and having acceptable chemical durability. A recent review219 of SnO based glasses shows that low melting properties are also found in tin(II) borate and silicate glasses, and, like PbO, SnO allows very wide glass forming ranges with the glass forming oxides, because it can partly behave as a glass former at high concentrations. Substituting part of the O2− anions by F− or Cl−,220, 221 can further reduce processing temperatures (usually at the expense of durability, greatly improved by additions of none other than Pb),153 while posing less migration problems than the alkali ions often present in other low melting glass compositions.
Although these glasses seem very promising, there are issues about their rather large thermal expansion,182 mediocre adhesion to silicates such as float glass222 and mechanical properties.150 Moreover, the +2 valence state of Sn, which is not stable in ambient air, raises two important processing issues. First, processing in air is preferable (cost and burnout of the organic vehicle), but can oxidise Sn2+ to Sn4+, leading to devitrification and halting densification. This issue can be solved by replacing some of the SnO with low valence oxides of transition metals such as Mn, Co and Fe, which would protect Sn2+ by acting as buffers that stabilise the oxygen activity in the glass to low values while being preferentially oxidised, as has been patented for Mn.223 This, however, raises the second issue: such glasses, once they achieve densification, have a reducing character for anything they encapsulate, as evidenced by the tendency of Cu ions to be reduced to metal.219 Although this opens up interesting applications such as base metal TFs, compatibility with some applications such as existing RuO2/ruthenate based TFRs will be problematic, due to likely reduction of the Ru compounds to metal (2SnO+RuO2→2SnO2+Ru). Finally, the presence of metals in several coexisting valence states can degrade the insulating characteristics of dielectrics based on these glasses.224 – 228 One interesting open point relevant for this work is the possible substitution of Pb by Bi as an additive to achieve water durable ultra low melting tin fluorophosphate glasses,153, 220 i.e. whether Bi oxifluoride in glass is first at all thermodynamically compatible with Sn2+ (not reduced to metal) and, if this is the case, yields similar improvements in durability as Pb while maintaining a low processing temperature.
Other systems
Finally, other more ‘exotic’ systems must be mentioned, such as glasses containing important amounts of TeO2, V2O5, Nb2O5, Ta2O5, MoO3 and WO3, with TeO2, V2O5 and MoO3 giving especially low melting compositions.45, 73, 113, 120, 123, 126, 229 – 241 Although toxicity of V2O5 is a cause of concern (the section on ‘Toxicity of elements in glasses’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1), these oxides are useful as additives in small amounts, to improve adhesion, wetting and durability, suppress crystallisation in glasses and reduce working temperatures.35, 56 – 58, 146, 198, 207, 216, 222, 242
Discussion
From the above considerations, one can conclude that replacement of lead based frits by the abovementioned systems, of which several examples are summarised in Table 6, may be achieved for applications not requiring a too demanding combination of good insulating properties (e.g. alkali free), wide processing window, high durability and low processing temperatures; enamelling/overglazing aluminium and glass for protective, functional and decorative purposes, as well as TF dielectric and LTCC compositions, are good examples of successful large scale replacement of lead bearing glasses by borosilicate/silicate compositions. However, durability is often problematic if low processing temperatures are specified.33
Some low melting lead free glass systems (without Bi), with typical glass transition temperature T g [R2O = (Li,Na,K)2O; RO = (Ca,Sr,Ba,Zn)O]
In electronics, mass produced chip resistors and co-fired LTCC devices including resistors may also be manufactured lead free using similar glasses. However, it would be difficult to achieve a general purpose TF system with a comfortable processing window using these materials.
Phosphate and SnO based glasses, especially those derived from the SnO–ZnO–P2O5 system modified with transition metal oxides, are very promising, and their flow characteristics can resemble those of lead based frits, but they represent a very radical departure from the heretofore applied chemistry, especially due to their intrinsically reducing character. This may lead, through the likely resulting presence of mixed valence transition metal oxides, to degradation of the insulating properties of dielectrics. Also, TFRs, currently based on (most likely incompatible) RuO2 would have to be formulated anew, using compatible conductive phases based on compounds such as reduced/doped SnO2, Fe3O4, MoO2 and WO2. Finally, the high water affinity of phosphate glasses209 is an issue which cannot be ignored if well defined, high reliability electronic materials are to be manufactured.
Bismuth glasses
Introduction
In contrast to the abovementioned lead free glasses, Bi2O3 appears a quite promising ‘drop in’ replacement for PbO, as also evidenced by comparing the commercial lead free and lead based TF overglazes (Table 5). The intentional use of bismuth in glasses is by far not as old as that of lead (the section on ‘Lead and bismuth in glass: a historical perspective’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1), but the similarity of Bi2O3 and PbO was immediately noticed in the early studies;129, 244 – 247 akin to PbO, Bi2O3 belongs to the class of ‘conditional glass formers’: while it does not by itself readily form a glass, it can be incorporated in very large quantities in the classical glass forming oxides SiO2, B2O3 and P2O5 and GeO2,2, 110, 245, 246, 248 where it acts as a glass modifier at low concentrations, but partly as a glass former at higher ones. These glasses may in turn incorporate, under standard glassmaking conditions, large amounts of alkaline earth (especially SrO and BaO) and transition metal oxides (e.g. ZnO, Fe2O3, CuOy, MnOy, CoOy), as well as PbO, with small additions of enhancing vitrification.73, 122, 129, 246, 248 – 272 Other possible additives are alkalies247, 248 and rare earths.273 – 278 Vitrification in different systems is detailed more fully in the following section. Representative compositions are given in Table 7, and a system property reference index of studied systems is given in Table 8 for borates, Table 9 for silicates, germanates and phosphates, Table 10 for other systems and Table 11 for binary systems without network formers; systems with several network formers are attributed on a following priority basis: B2O3, SiO2, GeO2, TeO2, V2O5 and MoO3.
Bismuth glass compositions, in cation mole percentage
*Ln: lanthanide mix.
†Only sum Si+Al+Ba given in reference – ‘educated guess’ according to Table 18.
‡Fluorine expressed as replaced oxygen fraction, i.e. F2/(O+F2); usually inaccurate due to volatilisation losses.
§Possible error in paper – bottom formulation more likely.
System property index: borates
†Also +Sb fining agent.
‡Error in Kim et al. 260 – T g/T liquidus≈2/3, authors used °C instead of K.
System property index: silicates, germanates and phosphates
*Also with fluoride additions.
System property index: tellurites, vanadates, molybdates and other
*Binary systems without glass formers: see Table 11.
†Probable Al2O3 contamination from crucible.
Glasses without standard network formers: binary systems
*Vitrification range; X = none found.
†Probable Al2O3 contamination from crucible.
One fortunate difference with lead is the much lower toxicity of bismuth, which compares well in this respect with other potential substitutes, as discussed in the section on ‘Toxicity of elements in glasses’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1. A less fortunate aspect, however, is the somewhat lower fluxing ability, as can be inferred from the higher overall bonding of Bi3+ vs. Pb2+: simple substitution of PbO with ‘BiO1·5’ leads to higher processing temperatures, as illustrated by the stable liquidus (Fig. 2) and glass transition temperatures (T g, Fig. 3). This may be seen as well on the ternary PbO–Bi2O3–B2O3 phase diagram,279 where the ternary eutectic composition lies very close to the PbO–B2O3 join, at about 45Pb+4Bi+51B on a cation basis. Therefore, most studies and developed low melting glasses are based on the Bi2O3–B2O3 binary, which combines a wide vitrification range with relatively low processing temperatures, with ZnO, SiO2 and Al2O3 being the most common additions. One must, however, note that comparison on the basis of equilibrium diagrams should be made with caution, given the slow equilibration in many Bi2O3 containing systems, attributed to mesomorphism in the melt280 and illustrated in corresponding metastable phase diagrams.280 – 282
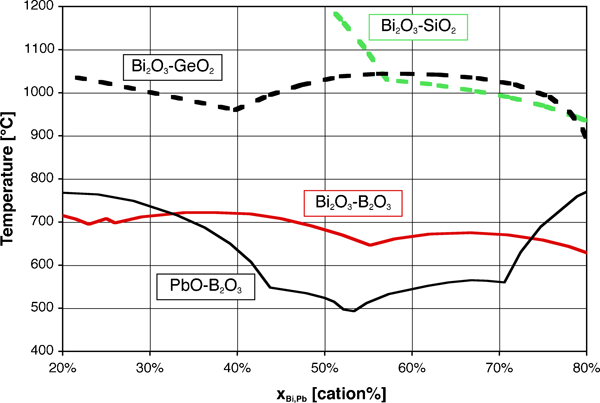
Liquidus temperatures of binary systems, redrawn from phase diagrams Bi2O3–SiO2,281 Bi2O3–GeO2 (PDC-2359), Bi2O3–B2O3 (PDC-323) and PbO–B2O3 (PDC-282)
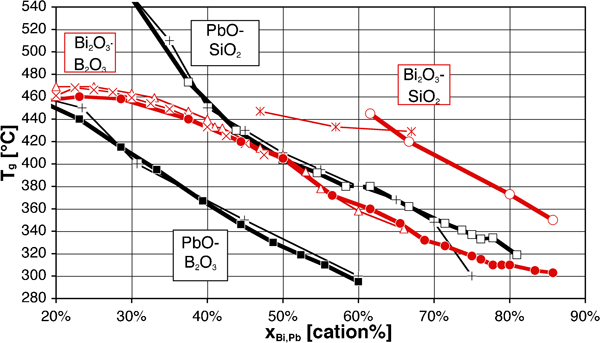
Glass transition temperatures T g of binary systems according to George et al. 248 (heavy lines), compared with other works (×: Ref. 317; Δ: Ref. 318; +: Ref. 141; *: Ref. 319)
Scientific work has been matched by technical use, the first patent dating from as early as 1945.283 In the early patents,49, 87, 146, 283 – 288 Bi2O3 was introduced in component/ceramic metallisations for its fluxing and wetting properties. The glass frits usually contained PbO and/or CdO, their elimination was at the time not an issue, and the patents gave conflicting information about how Bi2O3 should best be added to obtain maximal adhesion: included in the glass frit, ‘presintered’ with it, added separately to the paste, or even be present both in the glass and as a separate addition. Ensuring good adhesion to alumina without any alkali oxides, CdO and PbO was reported to be problematic, but possible by replacing some SiO2 by GeO2.288 Starting from 1980, a string of early Soviet patents,289 – 293 from what is now the Belarusian State Technological University, disclose a family of low melting or intermediate glass frits based on the ZnO–Bi2O3–B2O3–SiO2 system, with optional Li2O, Na2O, MgO, BaO, CuO and CdO, Al2O3 additions, and claiming a better chemical resistance and a lower CTE than analogous lead borosilicate frits. These glasses (Table 7: B80/B82/B83/B89), featuring moderate to high Bi content, processing temperatures down to ∼500°C, and designed specifically for application in electronics, overglazing and sealing,289 – 294 can truly be considered as the base for the ‘modern’ Bi based frits. More recent patents disclose usually similar compositions for glazes and enamels,295 – 302 TF conductors,250, 303 – 305 resistors251 and overglazes,306, 307 plasma display panel (PDP) dielectrics,308 conductors305, 309, 310 and low melting sealing glasses.311 – 314
The closeness of PbO and Bi2O3 may be seen by comparing, on a cation basis, some glasses taken from Table 1 (standard and low fire resistor and non-crystallising sealing compositions) with corresponding Bi based analogues (Table 7). Both types belong to the so called ‘fragile glasses’, i.e. with a strong dependence of properties on temperature around T g.253, 315
Bi based oxide glasses are already making strong inroads in commercial architectural33 and automotive overglazes,48 as well as TF compositions (Table 5). Besides these lead free substitutes, Bi2O3 based HMO glasses have found potential applications, partly together with PbO, in nuclear physics (scintillators, γ-ray shielding windows),129 – 135 optics,36, 106 – 109, 111 – 128 magnetic materials316 and glass–ceramic+ semi/superconductors.99 – 102 However, in spite of their significance, compositions based on Bi2O3 have drawn only scant attention, if mentioned at all, in classical ‘mainstream’ reviews of glasses.2 – 6, 20
Glass formation
A comparison of the vitrification ranges of Bi2O3 and PbO (and a few SnO examples) with common and uncommon glass formers, as found by various authors, is given in Table 12 for nominally binary systems, as a function of the estimated rate of cooling from the melt. This rate, indicated as a subscript for each limiting composition, is expressed in this work as a ‘quenching index’ Q, equal to the base 10 logarithm of the estimated cooling rate (K s−1):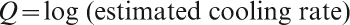
*Limits given as cation% of Bi, Pb or Sn. X: no glass-forming domain found. †Values for SnO systems (more data in review219) given in same column as PbO, in italic and with ‘Sn’ suffix. ‡Crucible types: S: SiO2 crucible; A: Al2O3; P: porcelain or similar; N: noble metal. §Assumed Bi2O3 is in fact represented as BiO1·5 in the diagrams.
Origins of discrepancies in indicated data
As seen in Table 12, some values are clearly in conflict, as exemplified by studies on air cooled gram size samples245 yielding a larger vitrification range than others on quenched ones.244 Partial volatilisation of some components, especially PbO, Bi2O3, B2O3 and P2O5, can account for some of these discrepancies, especially for quenching studies, which tend to involve small, open melts. Also, the large apparent discrepancy involving the extensive early work of Janakirama–Rao246 is tentatively attributed to the graphical representation; if Bi2O3 is taken as ‘BiO1·5’ (to make it comparable to the other oxides), a convention sometimes seen in the literature36, 112 and used in the present review, their results become closer to that of other work.
This said, the by far most common cause of extended reported vitrification ranges can be traced to small but significant amounts of SiO2, Al2O3 and other impurities (in porcelain, fireclay, etc.) leached from crucibles,118, 318 so some of the examined compositions are most likely not strictly binary. Therefore, the borate systems are marked in Table 12 by a crucible specific suffix (where specified) after the quenching index.
Binary systems
The binary Bi2O3–B2O3 system has been studied most extensively, and vitrifies easily at low cooling rates.317 A minimal amount of Bi2O3 is seen to be necessary due to the miscibility gap in the phase diagram110 (which also exists with PbO–Bi2O3 136 and many other borates), setting a practical limitation for technical purposes to above ∼19%Bi2O3, the end of the gap. It is nevertheless possible to achieve apparently homogeneous vitrification throughout this range if quenching sufficiently fast from above the gap.110
For the ‘strict’ Bi2O3–B2O3 binary, the extensive and well controlled work of Becker317 (very large melts, controlled cooling, noble metal crucibles, 20–43%Bi2O3) is deemed the most reliable for slow cooling. At intermediate cooling rates, the maximum Bi content is ∼60%,118, 244, 318 with 66% achievable for splat quenching.318 Going to twin roller quenching increases the vitrification range further, to 0–88%Bi.110, 248
The Bi2O3 rich ends of the glass forming ranges with B2O3 and SiO2 are often reported to be quite different from each other, and also from the values for PbO. However, as noticed by Dumbaugh and Lapp,36 this is due to the arbitrary selection of the ‘molecules’ PbO, Bi2O3, B2O3 and SiO2; on a cation basis, these limits (Pb versus Bi and B versus Si) become more similar, as illustrated by the results of fast quenching experiments by Stehle, George et al.,110, 248 where the four systems were examined in the same conditions; for Bi2O3–SiO2, a maximum of ∼85% Bi is obtained.
To summarise the data on binary systems with the common glass formers (B2O3, SiO2, P2O5, GeO2), the vitrification ranges (in cation-%) of Bi2O3 and PbO appear similar, although a direct comparison is difficult due to the spread in experimental data and the paucity of experiments under the same conditions. The maximum Bi2O3 content is smaller with GeO2, and even more with P2O5; the Bi2O3–P2O5 system has therefore attracted limited attention. The reverse is seen for SnO, where vitrification is especially favourable with P2O5, resulting in glasses based on the SnO–P2O5 system being another promising substitute to lead based ones (see the section on ‘Glasses based on divalent tin’).
With the unconventional network formers, vitrification is more difficult in general, and more severe quenching must be applied; comparing with PbO, vitrification appears to be more difficult for Bi2O3 with TeO2 and V2O5, while the reverse is true with MoO3. Under fast to very fast quenching, binary glasses may be obtained with Li2O, BaO, PbO, CuOy, MnOy and Ga2O3.
Complex systems with traditional network formers
As mentioned earlier, even small amounts of Al2O3 and especially SiO2 leached from the crucible considerably facilitate vitrification in the Bi2O3–B2O3 system. This synergistic vitrification is confirmed by experiments with B≈Si, where 74% Bi may be achieved by conventional melting and casting,361 i.e. much more than in either the Bi2O3–B2O3 or Bi2O3–SiO2 binary system. This is also true for low SiO2 and Al2O3 additions: a 65BiO1·5+33BO1·5+2SiO2 composition vitrifies easily when casting large plates,112 as does 70BiO1·5+25BO1·5+3SiO2+3AlO1·5 under moderate quenching.324 Such modifications are therefore very useful for low melting glasses rich in Bi2O3, as they stabilise the glass with only limited impact on processing temperature.314
Synergistic vitrification also occurs, for a given network former, when replacing on a cation basis part of the Bi2O3 with other alkaline earth and transition metal oxides. This occurs in spite of a generally poorer vitrification with these oxides (except PbO) taken individually than with Bi2O3, and is seen for borates with SrO,246, 258, 346 BaO,246, 258, 270, 349 PbO,130, 246, 279 ZnO246, 354, 355, 392 and CuOy.249 Figure 4, redrawn on a cation basis from the original studies,249, 354 illustrates the case of the Bi2O3–B2O3 binary extended with ZnO and CuOy: there is clearly an optimum in substitution that minimises the required amount of network former. This composition ‘hump’ roughly agrees with low melting zones in the corresponding ternary equilibrium diagrams.354, 356 In commercial low melting glasses based on ZnO–Bi2O3–B2O3 (Table 7, Hg–Nch–Usu), the Zn/Bi ratio, in practice also influenced by other considerations such as acid resistance, is commonly 0·2–0·7. In most cases, Zn is preferred over Cu due to the easy occurrence of mixed valence in the latter, leading to semiconductivity in Cu rich glasses249, a drawback mainly for insulators. This said, excluding PbO and alkalis, Zn and Cu apparently give the lowest melting points in the ternary phase diagrams, with the deepest eutectics somewhat below 600°C; the BaO–Bi2O3–B2O3 system, for instance,347, 348 apparently is fully solid at 600°C. Going one step further in complexity, mixing both network formers and modifier, allows a further decrease in the required amount of the former, as seen for PbO–ZnO–Bi2O3–B2O3–SiO2 glasses,256 in line with the general trend towards more difficult crystallisation of multicomponent glasses.
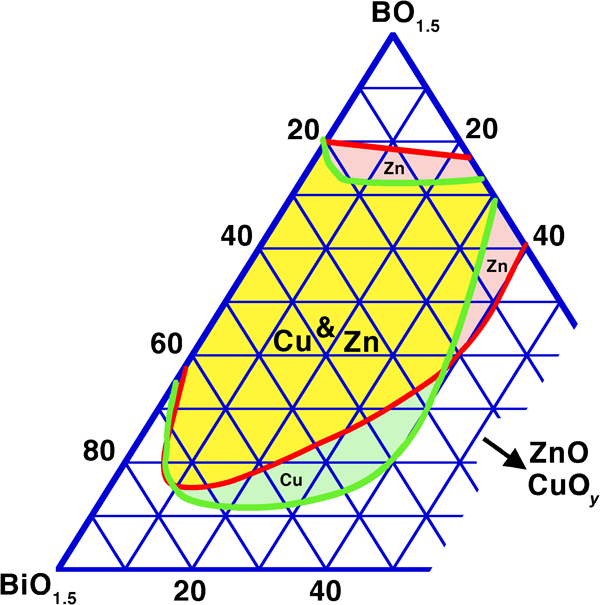
In contrast to these oxides, an extension of the glass forming range to lower B2O3 contents is not seen with the light alkali elements Li, Na and K (Fig. 5);247 the effect for small substitutions of BiO1·5 by alkalis is neutral up to ∼25% LiO0·5, then the minimum B2O3 required for vitrification rises sharply; this threshold is much lower (∼3%) for NaO0·5 and practically zero for KO0·5. Under fast cooling in the more complex R2O–ZnO–Bi2O3–B2O3 (R = Li, Na, K) system, a deleterious effect on vitrification was also observed, with a similar, but less pronounced trend of the size of the vitrification domain (Li>Na>K).392 Qualitatively, this agrees with the relatively stringent limits set on alkali content in the patents of Hasegawa et al.,313, 314 as discussed in the section on ‘Sealing and glass stability during reflow’.
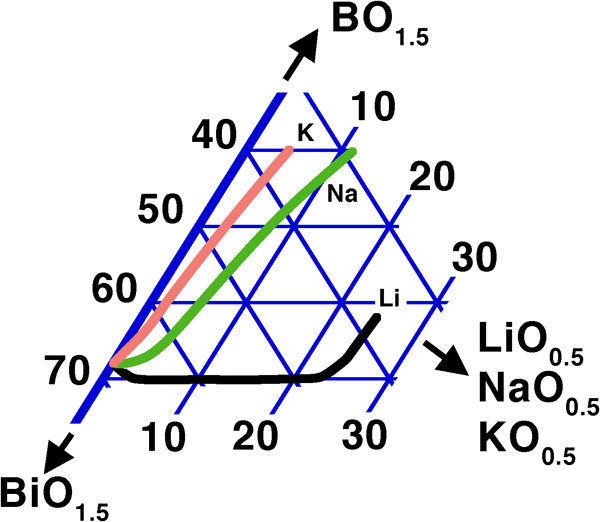
Effect of R2O on min. B2O3 in R2O–Bi2O3–B2O3 (R = Li, Na, K) systems247
While small amounts of rare earth additions are common for luminescence studies, the effect of larger quantities on vitrification Bi2O3–B2O3 have been less studied; especially, experiments where Bi2O3 is systematically replaced by Ln2O3 (Ln = rare earth and Y, except Ce) are lacking. Experiments by Pascuta et al.,276 – 278, 357 while extensive, were carried out on relatively unstable glasses with a low amount of B2O3 (20–33%BO1·5), requiring fast quenching; they could replace ∼25 to 35% of the whole glass, on a cation basis, by GdO1·5 or EuO1·5 (they achieving similar results with GeO2 as a network former374). Stabilisation of the glass was also observed with 5%Fe2O3.255 Compositions reported by this group for this and other systems255, 274, 276 – 278, 357, 374, 384, 393, 394 must be taken with caution, however, as they used very high melting temperatures (1100°C and above) in Al2O3 crucibles and their reported glass forming ranges, >90% BiO1·5 for moderate quenching, seem too high (see Table 12); their compositions are therefore likely to be shifted by reaction or volatilisation.
For more stable glasses (Bi2O3.2B2O3), vitrification is easy and therefore not affected by small Ln2O3 additions; however, strong suppression of crystallisation upon reheating was observed for 5%La2O3 and 3%Er2O3.275 In the patent literature (complex glasses based on ZnO–Bi2O3–B2O3), maximal rare earth content is reported to be at ∼4 to 8%, lower melting glasses being more sensitive.314
For the less studied silicates and germanates, a wide maximum in synergistic vitrification range is achieved when mixing Bi2O3 with PbO129, 367, 369 or PbO and some BaO107 and, while systematic studies are lacking, successful incorporation of large amounts of alkaline earth/transition metal oxides with low network former content is reported;246, 250, 251 for instance Janakirama–Rao reports facile vitrification of 31RO+62BiO1·5+7SiO2, where R = Sr, Ba, Pb or Zn, i.e with a much lower amount of SiO2 than needed for vitrification in the binary systems, and similar results when adding two oxides (CdO and WO3, PbO and MnOy, PbO and CuOy) to the Bi2O3–SiO2 binary.246 In comparison, the PbO–SiO2 system is also quite tolerant for substitution of PbO by NiO, ZnO, MnOy and FeOy,395 though a decrease of required SiO2 is not observed for small substitutions, in contrast to the Bi2O3–B2O3 system.
Data on alkali additions is not as complete as with borates. At <10 cation-%Si, R2O–Bi2O3–SiO2 is reported not to vitrify for R = Li, Na or K (as with borates), but to vitrify easily with R = Rb or Cs, even with a very large (>50%) degree of substitution of Bi by R.246
Extensions of the glass forming range by other oxides also occur with vanadates (Fe2O3 234), molybdates (PbO115, Fe2O3 234) and even gallates (PbO and CdO36). In the case of phosphates, vitrification with ZnO occurs over a wider range than with Bi2O3, but a synergistic effect is achieved nonetheless, albeit in this case with less Bi2O3 than ZnO.262
Glasses without network formers
Besides providing new insights in glass formation, glasses without traditional network formers are of interest for optical applications (the section on ‘Optics’), provided other light element oxides with strong oxygen bonding (especially Al2O3) are absent.
Although early attempts to make glasses of Bi2O3 without at least a very small amount of true network formers were unsuccessful,244, 246 several such binary systems were later successfully vitrified under twin roller quenching (Table 11), and melts with Li2O and Ga2O3 were observed to actually vitrify under relatively moderate quenching.
Adding more components facilitates glass formation, of which several examples are given in Table13. Extension of the Li2O compositions to systems such as Li2O–BaO/PbO–Bi2O3 significantly facilitates vitrification.381, 382 Khalilov107 systematically modified glasses based on Bi2O3–SiO2 with binary or more complex combinations of PbO, BaO, CdO, ZnO and MgO, under moderate quenching (cast in metallic moulds and covered with plates), and SiO2 free Bi2O3–PbO–BaO–CdO–ZnO (+optional MgO) glasses were obtained; as the mixtures were melted in Pt crucibles, contamination by SiO2, Al2O3 or B2O3 can safely be excluded. Other similar systems are SrO–PbO–Bi2O3 117 and CaO–SrO–PbO–Bi2O3–CuOy 99 (useful for processing superconductors via the glass–ceramic route [see the section on ‘Bi2O3 in (glass-)ceramics and crystals’)], and PbO–CdO–Bi2O3–Fe2O3.36 Ga2O3 was found to be particularly useful to promote glass formation, with the relatively simple PbO–Bi2O3–Ga2O3 system exhibiting easy vitrification over a wide composition range and even allowing casting of large objects.36, 106, 386
Glasses without standard network formers: complex systems
Conclusions
The following remarks may be derived from the data on glass formation:
Bi3+, akin to Pb2+, is a large, polarisable ion; both, while not vitrifying alone, behave as conditional network formers, requiring ‘abnormally’ low amounts of true network formers for successful vitrification at moderate cooling rates.
The Bi2O3–B2O3 system appears to be the most promising basis for the fabrication of stable lead free Bi2O3 glasses with low processing temperatures, but does not allow by itself processing temperatures as low as those of the corresponding PbO–B2O3 system.
In the Bi2O3–B2O3 system, adding low amounts of SiO2 and other oxides such as ZnO, Al2O3 and Fe2O3 also stabilises the glass without largely increasing processing temperatures. Further studies, however, are needed to better define glass stability as a function of composition, especially upon reheating, which is critical for TF processes (see the section on ‘Sealing and glass stability during reflow’ for some information in this regard).
Mixing several oxides of large, polarisable cations, such as Bi2O3, PbO and BaO tends to stabilise the glasses and/or lower the required amount of true network formers; adding limited amounts of other transition metal or rare earth oxides furthers this trend.
Using rapid cooling, glasses containing Bi2O3 and PbO may be formed in the complete absence of true network formers. The PbO–Bi2O3–Ga2O3 system provides an especially favourable base for such HMO glasses.
Glass structure
General considerations
Since the ‘anomalous’ ease of vitrification of Bi2O3 with standard network formers, similar to that of PbO, was noticed in early work,244 – 246 numerous studies have been devoted to elucidating the structure of Bi2O3 based glasses, using methods such as X-ray diffraction (XRD), Fourier transform infrared, Raman, electron paramagnetic/spin resonance, Mössbauer, X-ray absorption and magic angle spinning nuclear magnetic resonance (MAS-NMR) spectroscopy. A good knowledge of structural features is required to efficiently correlate glass properties with chemical data;327, 329, 399 – 401 this is especially important for borate glasses, given the different possible forms and structures assumed by borate anions.402
Bonding in crystalline compounds
To provide additional insight into the structural features of Bi2O3 based glasses, a survey of a range of relevant and related crystalline oxides, where atomic positions may be precisely determined, was carried out (the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1). Even in crystalline oxides, Bi3+ adopts a wide variety of asymmetric, disordered and often ill defined coordination polyhedra, presumably due to its high polarisability400 and stereochemically active ‘lone pair’ electrons, and bonding with oxygen is fairly covalent. An overview of idealised typical oxygen coordination shells observed around Bi3+ cations, in the crystalline oxides examined in the supplement, is given in Fig. 6, with the corresponding descriptions in Table 14. The lone pair may strongly deform the oxygen coordination shell (6-Oct33), and often replaces an oxygen anion to ‘fill’ the corresponding vacancy (3-PyM, 4-BPy/4-PyM, 5-Py14). Recently, the lone pair concept has been revisited in the light of diffraction data and spectroscopic studies of band structure, coupled with detailed computational modelling403 (see other references in the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1); the lone pair is found to stem from interaction of both metal valence s and p orbitals, mediated by oxygen 2p ones.
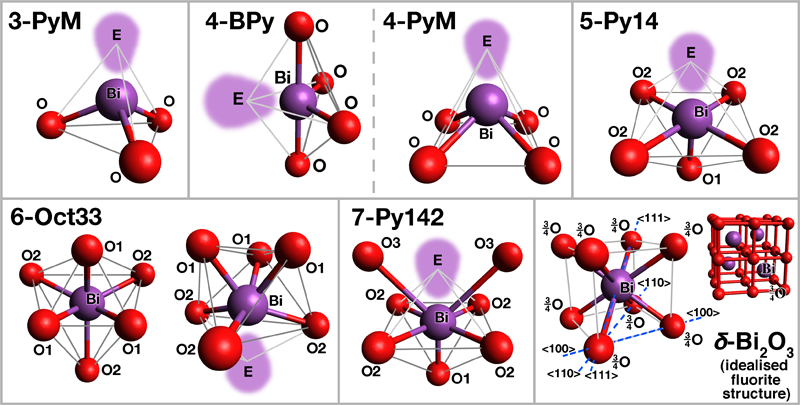
Some oxygen coordination shells around Bi observed in crystalline oxides (see section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1 and Table 14): E = Bi3+ lone pair electrons
Description of oxygen coordination shells around Bi observed in crystalline oxides (see section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1 and Fig. 6) (E stands for Bi3+ lone pair electrons)
In compounds, Bi3+ tends to have coordination number (CN) values of typically 5–7, but with very varying bond lengths and presumably strengths, with only a slight tendency to reduction to typically 5 at high Bi3+ concentrations. This reduction has little effect in practice as it only eliminates very long, weak bonds. CN = 3 is found only exceptionally, such as for a minority of Bi3+ cations in the defective sillenite γ-Bi2O3.
Pb2+ and Sn2+ are fundamentally similar to Bi3+, also being lone pair cations. However, Sn2+ has relatively well defined coordination shells and tends to low CN values, ∼3. Pb2+, being larger and more polarisable, behaves in a more similar way to Bi3+ in compounds; CN is similar to that of Bi3+ at low concentrations, but drops to 3 or 4 in Pb rich compounds. Bi3+ has more asymmetric bonding, having fairly high strongest bond valences in the range 0·8–1·3, compared to 0·6–0·7 for Pb2+ (Table S13 in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1).
Borate glasses
The binary xBi2O3+(1−x)B2O3 system has been studied most extensively; there is general agreement on several features.248, 317, 318, 350, 355, 394, 402
With addition of Bi2O3 in B2O3, the original B2O3 network, constituted of [B3O6] boroxol rings and [BO3] triangles (written BΔ, i.e. CN = 3), is initially strengthened, as in other borate glasses, by conversion of part of the BΔ groups to tetrahedral [BO4] ones (BT, CN = 4), as shown in Fig. 7. Boroxol rings persist only in compositions with very low Bi2O3 content, and disappear for x>25%. This initial increase in the degree of bonding results, as in other borate glasses , in an increase of T g (Fig. 3, Table 15) and network compacity248, 317 up to x≈20–25%.
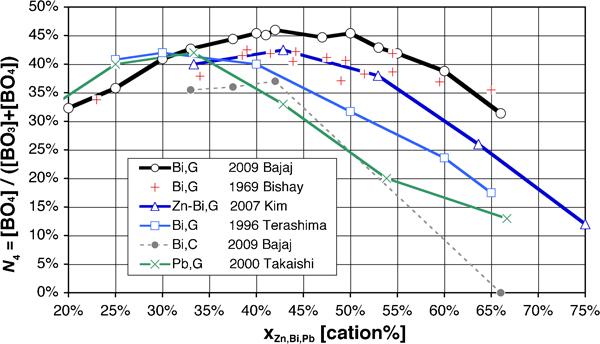
[BO4] tetrahedra fraction N 4 in boron coordination polyhedra ([BO3] and [BO4]), for Bi2O3–B2O3 glass (○: Ref. 318; +: Ref. 247; □: Ref. 327) and crystallised glass (•: Ref. 318), (ZnO.Bi2O3)–B2O3 glass (Δ: Ref. 355) and PbO–B2O3 glass (×: Ref. 397)
T g and N 4 peaks in binary x(BiO1·5,SbO1·5,PbO,SnO)–(1−x)BO1·5 glasses
*Sb2O3 (Sb3+) is close to being a true glass former – BT possibly due to observed presence of Sb5+ ions.
At low x, an ‘ideal’ modifier cation Mn+ with valence n, incorporated as its oxide MOn/2, is fully incorporated by creation of tetrahedral units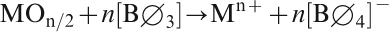
For x greater than ∼25%, N 4 further rises, but borate units are progressively depolymerised, as B–O–B linkages are replaced by weaker B–O–Bi ones, resulting in a drop of T g. This behaviour is very different from simple alkali and alkaline earth borate glasses,404, 405 where the peaks of T g and N 4 roughly coincide. PbO and SnO borate glasses also exhibit the same anomalies (Table 15), but they are much less pronounced; the exceptional structural feature in binary Bi2O3–B2O3 glasses is a very broad peak of N 4≈45%, which essentially extends throughout the ‘useful’ composition range. It is therefore assumed that Bi3+ at least partly enters the network already at low x values, and the borate network progressively adapts with increasing x.402 The same is also true, but to a lesser extent, for Pb2+ and Sn2+, given the lower discrepancy between the T g and N 4 peaks.
It is interesting to compare the maximum single bond valence range for Bi3+ (0·8–1·3) and Pb2+ (0·6–0·7) in crystalline compounds with the oxygen bonding deficit for different types of bonding and different boron coordinations, accounting for the variability of about ±0·05 seen for a given B–O bond in crystalline compounds (see Table 16 and the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1, Table S13). Allowing for total variability of two B–O bonds, i.e. ±0·1 for a given Ø, some of the strongest Pb–O bonds (0·6–0·7) may still be taken up by the bonding deficit of bridging oxygen anions bound to two borate tetrahedra (about 0·5+0·1). This is not enough for the strongest Bi–O bonds, which will therefore favour direct linkage to the ‘terminal’ oxygen anions of higher nominal valence, 1·00 or 1·25 (Table 16), the bridging oxygen ions in BΔ–Ø–BT and BT–Ø–BT links being able to take up the weaker bonds for both cations. This speculative interpretation, which still requires confirmation, agrees well with observed behaviour at low x: Bi3+ has much higher deviations from ‘ideal modifier’ behaviour of N 4 than Pb2+, and T g is similar for both cations at same x, in spite of the higher valence of Bi3+. Also, the preference of Bi3+ for higher CNs and bond disorder agrees with conservation of high N 4 values (i.e. mixed BΔ+BT) in a wide x range, compared to the more ordered crystalline phases and to PbO, which has lower valence and favours lower CNs (Fig. 7).
Nominal bonding deficit of oxygen anions in borate glasses versus structure
Anomalies in properties317, 318, 350 such as density and T g yield other hints on the structure; they are often correlated with compositions close to that of crystalline phases in the corresponding oxide systems. Based on this observation, a tendency to form local groupings in the glass similar to those that exist in the crystals was also postulated for the BaO–Bi2O3–B2O3 system,350 as typically found in borate glasses.402 Comparing data on glassy and crystallised Bi2O3–B2O3 samples on Fig. 7, however, one can see that this structural similitude progressively breaks down at high x values, where Bi2O3 becomes the dominant species and obviously assumes the function of network former, with significant amounts of O2− anions not bound to boron (i.e. only to Bi3+) identified at about x⩾65%.255, 394
In these Bi2O3 rich compositions, in spite of extensive characterisation work with well controlled samples, there are significant discrepancies in the reported N 4 values, as illustrated in Fig. 7; the work of Terashima et al. used by Dimitrov327 seems somewhat at odds with that of Bajaj et al. 318 (and previous work cited by the latter247), although the same method (MAS-NMR) was used in both cases and sample fabrication appeared to be well controlled (moderate melting temperatures and noble metal crucibles). Residual impurities318 could possibly account for some of the discrepancies, as well as thermal history (quenching rate and subsequent annealing), which significantly influences glass properties376 and even structure (see discussion on ‘polyamorphism’280, 318); interestingly, Terashima's data lies roughly halfway between Bajaj’s for glassy and crystallised samples.
Given the differences in N 4 between crystalline and glassy samples, shifts of the CN of Bi3+ in glass vs. in crystals can also be expected, but Bi3+ is less sensitive in this respect than Pb2+, as seen in the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1. In fact, most structural studies132, 255, 275, 276, 326, 352, 359, 360 in the binary or almost binary Bi2O3–B2O3 system favour retention of ‘distorted [BiO6] octahedra’ throughout the composition range, and low CN groups are not seen in Bi2O3–B2O3 glasses at least up to x≈70%. Above this level, presence of a minority of [BiO3] groups has been reported357, 394 (threshold uncertain due to likely significant contamination from the Al2O3 crucible), in line with the structure of high Bi2O3 crystalline boron sillenite Bi12(Bi0·25B0·50)O19·125 (see the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1). This agrees well with [BiO3] groups appearing together with sillenite crystallisation for about x ⩾65% in heat treated glasses.276, 394 The reported presence of [BiO3] groups at moderate Bi contents in borate, borosilicate362 and aluminoborate277, 278 glasses is doubtful, and most likely results from IR peak misassignment, expected [BiO3] peaks lying at ∼480 and 840 cm−1.276, 357, 374, 394
Substituting some of the Bi2O3 with compounds such as ZnO, PbO and BaO350, 352, 355 yields results very similar to that of Bajaj and Bishay,247, 318 as shown for Zn in Fig. 7, with only slight offsets due to the elemental substitution. Ba2+ enters the glass as a modifier,266 while Zn2+ may do the same at low concentrations,266 but forms [ZnO4] tetrahedra at high ones.122, 406
Concerning more complex systems, presence of [BiO3] groups is reported in (Li2O)–ZnO–Bi2O3–B2O3 264, 337, 406 and Li2O–Bi2O3–(B2O3)334, 335 glasses only at high Bi2O3 and low B2O3 contents, Bi3+ being otherwise present as [BiO6] only.
Compared to Bi3+, the coordination of Pb2+ is somewhat more ordered and much more dependent on x in binary borate glasses, according to XRD and MAS-NMR studies:397 Coordination number is 6 up to about x = 25%, then decreases continuously, reaching 3 for x equal or greater than ∼55%, which is matched by a corresponding decrease of average bond length from ∼300 down to 233 pm. Pb2+ is therefore roughly present as [PbO6] octahedra and behaves somewhat as a classical modifier at low x, and progressively switches at higher x, well within the vitrification range, to [PbO3] network forming trigonal pyramids (3-PyM), with fewer, stronger Pb-O bonds. However, while the average bond length found for [PbO3] agrees well with bonding in Pb rich crystalline compounds (the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1), the corresponding length for [PbO6] is clearly too long, which suggests bonding is also somewhat inhomogeneous at low x (existence of shorter bonds). This, together with N 4 being slightly less than ideal and T g starting to drop at lower x values than the CN, hints at some departure of Pb2+ from pure modifier behaviour, even at low x, albeit to a much lesser extent than Bi3+. At high x, there is also some ambiguity in the 3-PyM configuration, as there are additional, weaker bonds, as seen in the crystals;407 the configuration can be viewed as 4-PyM (more 3+1, with one longer bond), with additional, much longer ones above the pyramid apex.
Silicate and germanate glasses
The case of silicate408 and germanate231, 374, 393, 409 glasses is much simpler, as the [SiO4] and [GeO4] tetrahedra are conserved when Bi2O3 is added, with no reported formation of [GeO6] octahedra as found in the alkaline germanate glasses. The only change is gradual weakening of the original network, as attested by the continuous drop of T g with increasing x in the xBiO1·5+(1−x)(Ge,Si)O2 system, as shown in Fig. 3 for silicates. Absence of [GeO6] octahedra was also reported in complex germanate glasses, with V2O5,231 Ga2O3 359, 360 and PbO–Ga2O3 370 additions.
Bi3+ is generally reported as being present in the form of [BiO6] groups, but also as [BiO5], from X-ray absorption spectra and molecular dynamics calculations.408 Given the very high disorder around Bi3+, this difference in reported structure is probably not very significant. As for borates, no [BiO3] groups were found to high x values (80%) in Bi2O3–GeO2(–Eu2O3) glasses. On the other hand, after heat treatment and crystallisation, [BiO3], [BiO6], [GeO4] and [GeO6] groups appeared, with Bi2GeO5, Bi4Ge3O12 and unidentified peaks seen in the XRD spectra.374 Neither [BiO3] 3-PyM pyramids nor [GeO6] octahedra exist in the identified crystalline structures (the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1), but this apparent conflict may be resolved for [BiO3], assuming some of the unidentified crystalline phase is the [BiO3] containing Ge sillenite Bi12GeO20, whose formation would be expected at x = 80%, as seen in borates.276, 394 The presence of [GeO6] is more doubtful, as it is also absent in sillenite and Bi2Ge3O9 as well, but could be possible in the residual glass due to the thermal history or in an unidentified metastable phase.
The existence of [GeO6] groups in PbO–GeO2 glasses has been reported, albeit to a much lower extent than in the alkali germanates,12, 410 but more recent work409 concludes to all Ge4+ being in [GeO4] tetrahedra. There is basic consensus that Pb2+ forms [PbO3/4] (3/4-PyM) pyramids in Pb rich PbO–SiO2 407, 411, 412 and PbO–GeO2 413 glasses. On the other hand, Pb2+ coordination at lower lead contents has been questioned recently. CN = 6 was found at up to 40%PbO in PbO–GeO2,410 and progressive switch from network modifier to former behaviour (presumably [PbO6]→[PbO3/4]) up to 40% PbO in PbO–SiO2.414 Somewhat at odds with these results, Pb2+ was found to form [PbO3/4] pyramids down to 30%PbO in PbO–SiO2,412 a behaviour similar to that found in SnO–SiO2 glasses, where Sn2+ essentially appears in 3-PyM coordination, with CN only slightly increasing at low SnO contents.415
Phosphate glasses
The binary Bi2O3–P2O5 system has received only scant attention due to its limited vitrification range, which probably stems from easy crystallisation of high melting BiPO4;377 Bi2O3 is therefore mostly found as an additive (intended or as waste96, 98) in multicomponent glasses.
Replacing part of Fe3+ in a 40Fe2O3–60P2O5 glass with isovalent Bi3+ is found to effect only limited changes to the structure;228 expectedly, the phosphate groups are mostly present as Q1 pyrophosphate units,209, 210 and both Fe3+ and Bi3+ are present as hexacoordinated octahedral units. Similar incorporation as [BiO6] was determined for ZnO–Bi2O3–P2O5 262, 265, 267 and Li2O–Bi2O3–P2O5 378 glasses. As for the other systems, Bi2O3 was concluded to behave partly as a network former. In comparison, SnO in 3-PyM coordination can also enter the glass network, being able to vitrify with fully depolymerised phosphate groups.218
Gallate glasses
Ga3+ is found to form [GaO4] tetrahedral groups in HMO glasses throughout the ternary PbO–Bi2O3–Ga2O3 109, 111, 385, 386 and Bi2O3–Ga2O3–B2O3 359 359,360 systems, as well as more complex PbO–(PbF2)–Bi2O3–Ga2O3–GeO2 370, 371 compositions, with good agreement of Ga–O bond lengths with those found in crystals.109, 111 As in other HMO rich systems, Bi3+ and Pb2+ are reported to form ‘[BiO6]’ groups and [PbO3/4] (3/4-PyM) pyramids respectively, with a higher degree of disorder around Bi3+.
In these glasses, coordination around Bi3+ and Pb2+ has been examined more extensively. Assignment of Raman bands for Bi–O bonds360 agrees with the Bi3+ bonding in crystals (the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1, deformed 5-Py14 pyramids), with a short apical bond, and two groups of unequal bonds on either side of the pyramid base. The last, much weaker bond assigned in glasses to complete a ‘[BiO6] octahedron’ could actually correspond to a pair, as found in the 7-Py142 configuration often reported in crystals. These results also agree very well with detailed neutron and XRD studies of a binary 80BiO1·5+20GaO1·5 glass,109 which yield CN≈5 for Bi3+, with roughly 1, 2 and 2 oxygen anions at 213, 224 and 252 pm respectively. Obviously, CN of Bi3+ stays high even for a very Bi rich glass, influencing the CN of Pb2+ in the ternary PbO–Bi2O3–Ga2O3 system, which was found to decrease from 3·5 for Bi free glasses to 3·0 for Bi rich ones.111 This is logical, as the higher oxygen CN of Bi3+ creates a severe oxygen shortage, which is partly compensated by decrease of Pb2+ CN. Nevertheless, in all these HMO rich glasses, CN > 2 for oxygen, with CN = 3·5 for the binary 80BiO1·5+20GaO1·5 glass.
Other glasses
Vanadate231, 232 and molybdate232, 234 glasses exhibit a change of network former coordination polyhedra when adding Bi2O3:[VO5] trigonal bipyramids and [MoO6] octahedra are converted to [VO4] and [MoO4] tetrahedra respectively. Tellurates are even more complex, and tend to integrate only little Bi2O3, together with a third oxide; the original irregular [TeO4] trigonal bipyramid (4-BPy, Te4+ also being a lone pair ion) of TeO2 is partly converted, depending on Bi2O3 and other oxides, to [TeO3] (3-PyM) pyramids and [TeO3+1] polyhedra.120, 126, 237, 239, 240 Bi3+ is reported to form ‘[BiO6]’ groups, as with standard network formers.
Glasses without network formers
Structural studies on systems where mainly Bi2O3 forms the network are relatively scarce. In Li2O–Bi2O3 glasses, a disordered local structure, analogous to crystalline Bi2O4, was assumed; it was rationalised that the nominal additional oxygen was provided by Li2O, and even very atypical partial oxidation to Bi5+ (see the section on ‘Oxidation state of Bi species in glasses’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1), the rest being compensated by defects.
Structural analysis of glasses based on a nominal 89BiO1·5+11PbO formulation, probably contaminated with Al2O3 from the crucible and optionally doped with MnOy, expectedly yields coordination of Pb2+ as [PbO3/4] (3/4-PyM) groups. Bi3+ was found in the Mn free glass mainly as [BiO6] groups, with a minority of [BiO3]. However, the reported exclusive formation of [BiO3] pyramids in Mn doped glass must be taken with caution, as this does not correspond to any relevant Bi based compound.
Conclusions
Concerning the coordination of Bi3+ in glass, most IR and Raman spectroscopic studies on conclude that Bi3+ essentially forms distorted [BiO6] (CN≈6) octahedral-like configuration, with little variation of CN over a wide concentration range; a minority of [BiO3] pyramids is found only in very Bi rich compositions, in agreement with their presence in the sillenite crystalline structure (see the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1). The term ‘[BiO6] octahedron’ must be taken with proper caution, as coordination around Bi3+ tends to be ill defined; more dedicated studies109, 408 yield CN≈5 with nonuniform bond lengths, corresponding to a deformed octahedron with an oxygen vertex replaced by the Bi3+ lone pair E and thus yielding a [BiO5E] unit (Fig. 8, 5-Py14 configuration). This agrees well with assignment of Raman bands;360 the additional Raman band attributed to a further, weaker bond could in fact correspond to two such bonds, matching the 7-Py142 configuration often found in crystalline compounds.
Typical configurations/roles for dielectric glass based layers
The high CN maintained by Bi3+ to high concentrations necessitates a CN of O2− greater than 2, apparently violating the classical rules for glass formation.416 This is also true for Pb2+ and Sn2+, which have lower CN but lower valence as well. However, in contrast to Bi3+ and Sn2+, coordination around Pb2+ is seen, at least in some cases such as the PbO–Bi2O3–Ga2O3 and PbO–B2O3 systems, to be more dependent on the local environment and available oxygen to bond to.
A comparison of the analogous divalent and trivalent lone pair cations (Table 17) sheds new light into their structural features and trends. In line with original predictions,416 the rigid 3-PyM oxygen coordination shell of the lightest trivalent cation, As3+, makes it a classical network former, as is (almost) the case for Sb3+, whose coordination is somewhat less rigid.417, 418 Sn2+ is relatively rigid and favours a similar coordination, but intrinsically cannot form a glass on its own due to its valence being lower than its CN. Note that As3+, Sb3+ and Sn2+ are not stable in air, and Sn2+ tends to disproportionate into Sn0 and Sn4+.
Oxygen coordination trends of ‘lone pair’ cations versus concentration x
†4-PyM often 3+1 (1 longer bond).
‡5-Py14 often 1+2+2 (1 short, 2 medium, 2 long bonds); exceptionally 3-PyM as in sillenites.
§Variable, disordered oxygen coordination shells, more so for Bi3+ than for Pb2+.
The larger polarisable cations, Pb2+ and Bi3+ have much more variable and disorderly coordination shells, with CN behaving somewhat the same way as in crystals, i.e. being somewhat concentration dependent for Pb2+, and less so for Bi3+. Nevertheless, a tendency remains to form a limited number (3–4) of comparatively stronger bonds. One can speculatively view this inhomogeneous metal oxygen bonding as a reflection of the ambiguous behaviour of these cations, as both network formers (through the stronger metal oxygen bonds) and network modifiers (through the weak ones); considering the strong bonds only reduces the CN of oxygen, making it more comfpatible with classical rules of glass formation. More studies are clearly needed to arrive at a better definition of these complex and disordered coordination environments, possibly assisted by molecular dynamics simulations408 coupled with cation–oxygen interactions based on recent revisons403 (see also the section on ‘Coordifnation of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1) of lone pair bonding in crystals.
Oxidoreduction issues
Possible reduction of Bi3+ to metal during glass preparation,118 and later during processing, for instance through transient reducing conditions brought about by binder burnout, is even more pronounced than for Pb2+, as Bi2O3 is even less stable towards reduction than PbO.59, 419 – 422 This can be a problem for processing, especially of low melting glasses due to difficulty in burning out the organics. One must, however, mention that precious metal oxides used in TFRs, such as RuO2, are even much less stable towards reduction (see PDC-5015) than Bi2O3, so Bi2O3 reduction is not the limiting problem overall for standard air firing TF electronics.423 Finally, a moderate sensitivity to reduction actually can be beneficial in some respects, especially solderability of conductors (see the section on ‘Metallisations and TF conductors’).
Unfortunately, information about the thermodynamics of Bi2O3 (and other oxides) in glasses is rather limited: polarimetric studies were carried out424 on a borosilicate glass with very low (0·25% mol) Bi2O3 additions, but the results are not directly applicable to glasses where Bi2O3 is one of the main components, as those concerned in the present work. Nevertheless, recent reduction experiments366, 408, 425 – 427 and results of high temperature firing118, 375 do confirm easy reduction and formation of Bi0 nanoparticles, or, for glasses doped with low amounts Bi, presumably reduced species, whose nature is still subject to debate.428 – 430
Control of reduction, as in more common industrial glasses, may be achieved by ‘fining agents’, i.e. oxidoreduction buffers that inhibit reduction to Bi0 under practical firing conditions; this has been shown to be successful with low Sb, As, Ce or Cu additions,112, 114, 273, 361, 431 with CeO2 often found in the patent literature.311, 314, 432 Alternatively, using a fugitive oxidant such as KClO4 and KNO3 allows controlled reduction and precipitation of Bi0 nanoparticles to create a well defined surface plasmon resonance band.427 Additionally, the other main glass constituents, by affecting the overall basicity of the glass, will also somewhat influence the tendency of Bi3+ towards reduction.399, 430
Finally, further oxidoreduction issues involving interaction with adjacent layers, such as adhesion on metal (the section on ‘Dielectrics on metal substrates’), staining of glasses by in-diffusion from Ag conductors (the section on ‘Overglazing/enamelling’) and contacts to PV cells (the section on ‘Metallisations and TF conductors’), as well as optical properties, are discussed in the corresponding sections. Also, a strong point is made in the section on ‘Oxidation state of Bi species in glasses’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1 against, except in unusual circumstances, the occasionally reported presence of significant amounts of Bi5+ in glasses.
Applications in layer form
This section discusses in more detail the application of Bi2O3 based glasses, using TF or similar technology, to electronics, automotive and architectural glass, display panels and PVs. Uses in bulk form are discussed in the section on ‘Other applications’. The present discussion will mostly concentrate on materials covering the low processing temperature range, the main application of the Bi2O3 based glasses and the PbO based ones they should replace.
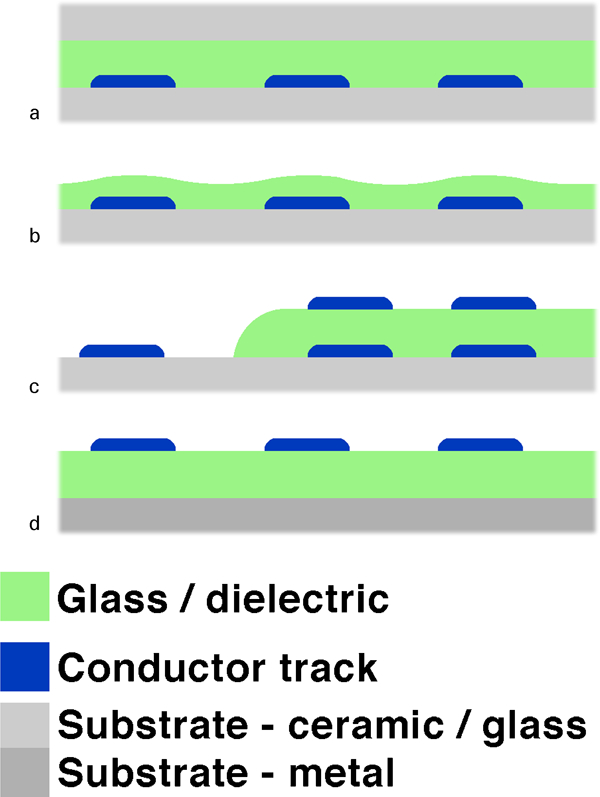
The following four sections, from ‘Sealing and glass stability during reflow’ to ‘Dielectrics on metal substrates’ discuss applications of insulating glass based layers in the four main configurations illustrated in Fig. 8, each corresponding to a specific role for the glass based layer and determining the required behaviour during firing and the insulating characteristics: sealing, overglazing/enamelling, multilayer dielectrics and dielectrics for insulating metal substrates. The section on ‘Sealing and glass stability during reflow’ also discusses glass stability upon refiring, as low temperature sealing is the most demanding application in this respect. Finally, the sections on ‘Metallisations and TF conductors’ and on ‘Glasses for TFRs’ discuss conductors/metallisations, and TFRs respectively.
Sealing and glass stability during reflow
Sealing stands apart from the other applications in that the sealing material must ideally be able to flow extensively during processing, in contrast to the other applications, where densification only is to be achieved. In the classical leaded sealing glasses, some of the PbO is often replaced by Bi2O3 to improve flowability,150, 151, 242 stability against devitrification and compatibility with temperature lowering fluoride additions,149 or even strength,152 However, as discussed in the ‘Introduction’ part of the section on ‘Bismuth glasses’, replacing most or all of the PbO by Bi2O3 results in increased viscosity,149 which may in most cases be mitigated by alkaline or fluoride additions to the extent durability and stability is not excessively degraded. As possible conductors going through the seal are usually only in a side-by-side configuration (Fig. 8a ), insulating properties should not be critical in most applications, except in PDPs, 181 181,308 where conductors are on both sides.
Sealing glass may be formulated to be either stable or devitrifying.15, 18, 74 Ideally, devitrifying seals yield the best properties, but they also tend to have tight processing requirements, limited flowability and are usually not applicable for low temperature sealing, where ‘composite’ glasses are used, i.e. stable glasses with low expansion fillers to adjust the CTE.18 In any case, extensive flow must be insured without or before crystallisation, which means sealing is arguably the most demanding application in terms of glass stability. In terms of glass formulation, this requirement tends to be in contradiction with the need for low softening temperature, as discussed hereafter.
Glass stability and devitrification in the Bi2O3–B2O3 binary system, which is the basis for most of the commercial formulations, has been the object of several studies.110, 317, 318, 326 Except the oldest study,110 they agree on a practical stability optimum near 45% BiO1·5, in agreement with the break in the liquidus temperature (Fig. 2), versus falling T g (Fig. 3); this somewhat lower optimum than Fig. 2 would suggest lies in crystallisation of the metastable phase BiBO3.317
As discussed in the section on ‘Glass formation’, small additions of other network formers and modifiers, as well as oxides such as Fe2O3 and lanthanides, hinder crystallisation; ZnO–Bi2O3–B2O3–SiO2 compositions specified in the early Soviet patents (Table 7: B80, B82 and B89), reported there as non crystallising and containing no alkalis, provide a good starting basis for low temperature sealing glasses, and the overwhelming majority of the subsequent research and patent literature report very similar compositions.
Hasegawa et al.,313, 314 in their patent applications, performed arguably the most extensive discussion, although very empirical and on a weight basis, of composition optimisation and stability in this system; the resulting restrictions, converted to a cation molar percentage basis, are given in Table 18 and discussed hereafter.
Composition ranges and ratios specified for sealing glass stability314
The indicated data are according the second application,314 which specifies a minimum amount of Al2O3 (recognising its beneficial effect on glass stability), and introduces a second, more restrictive composition range for low temperature FPD sealing with fillers, which apparently requires even better stability against crystallisation. This can be due to the filler possibly promoting crystallisation and the sealing temperature lying in the range where crystallisation is most apt to occur; sealing processes that require stronger capillary flow or firing of conductors/resistor frits occur at higher temperatures where crystallisation is less likely, i.e. slightly below or even above the liquidus.
Overall, the requirements stated in Table 18 and corresponding compositions (Table 7, Hg) correlate fairly well with glass formation (the section on ‘Glass formation’) and properties (the ‘Introduction’ part of the section on ‘Bismuth glasses’), as well as with our own preliminary refire stability experiments, performed with glass frits printed on alumina and glass and summarised in Fig. 9. The main network former is B2O3, to minimise processing temperatures, with SiO2 and preferably some Al2O3 being added to improve stability.
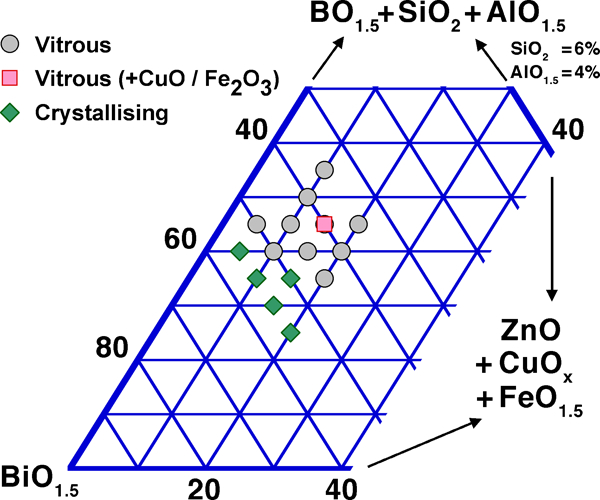
Results, in cation-%, of our experiments on stability of Bi2O3 based glasses in TF firing cycles (belt oven, 45 min total time with 10 min at peak, 400–700°C)
The beneficial effects of mixing B2O3, SiO2 and Al2O3 are illustrated by comparing with other work where either B2O3 or SiO2 were used alone;311, 355, 433 densification is reported to be problematic, with a strong tendency to crystallisation.355, 433 Crystallisation problems may also be inferred from a higher sealing temperature, in spite of a low filler volume fraction.311 In accordance with Table 18,314 using a very low amount of SiO2 in SrO–ZnO–Bi2O3–B2O3 (Nch-01), is reported to yield a devitrified seal, albeit with enough prior flow to achieve successful densification.312
Stability is improved by low levels of ZnO and alkaline earth additions, and degraded by excessive amounts, in line with vitrification studies (see e.g. Fig. 4) and other work.263 The stated maximum amounts of alkaline earths should be taken with caution, as they were added without regard to ZnO; a more pertinent test would be to add them with corresponding reduction of ZnO, i.e. keeping Zn+alkaline earths constant. Rare earths (Ln), including cerium, are stated to improve mechanical strength, an advantage compounded by CeO2 being a useful fining agent (the section on ‘Oxidoreduction issues’), and a low level of lanthanides providing stability versus devitrification.275 One not mentioned compound is Fe2O3, which is reported, together with CuOy/MnOy, to be a useful alternative to Al2O3 against devitrification in modern very low melting lead bearing sealing glasses;149, 151, 242 interestingly, Fe2O3 seems to stabilise Bi2O3 based glasses as well.255 These effects are expected to strongly depend on composition, i.e. formed crystalline phase(s) according to phase diagram, i.e. borates for low melting glasses. In higher melting, essentially Bi2O3–SiO2 glasses, crystallisation of Bi2SiO5 has been found to be promoted by Cr2O3, but inhibited by CuOy and MnOy.301
Practical sealing examples, taken from the patent literature,149, 242, 311, 314 given in Table 19, illustrate the potential of the new Bi based glasses (Hg-203/324/Usu-5) for low temperature sealing, well below the softening point of common float glass, and thus clearly demonstrates their industrial usefulness.
Composite sealing glasses (compositions: see Table 7)
*T S: Littleton softening point, at which viscosity η is 106·6 Pa s; T seal: sealing temperature.
†Values for Hg-203/324 (examples 6A and 6D) in original work dubious (too high) – confusion with volume faction (40/50%) likely.
‡Calculated assuming glass density: ∼6500 kg m−3
§LTS-1 estimated as T g+50 K; LTS-2 as flow point 30 K.
On the other hand, these examples also illustrate the remaining gap with the Pb bearing frits (LTS) for ultra low temperature sealing of electronic and optical devices at temperatures compatible with some engineering polymers, e.g. glass fibre coatings,242 in spite of these compositions not containing any alkalis. However, further studies combining fluxing additives such as F−, TeO2, V2O5, Nb2O5 and MoO3 with stabilising agents (Ln2O3, Fe2O3, etc.), i.e. optimisation similar to that of the lead bearing frits, should yield further progress in lowering achievable sealing temperatures.
Overglazing/enamelling
Low melting glassy layers (overglazes, or enamels) find a large palette of applications in electronics,291, 302, 307, 321 displays,181 automotive/architectural/container glass33, 47, 432 and cookware/dishware/chinaware,47, 56 as protective and decorative layers. Examples and properties are given in Table 20.
Overglaze/enamel/dielectric properties (glass compositions: see Table 7)
In TF electronics, overglazes protect resistors against humidity, and Ag rich conductors against electrochemical migration, thereby improving circuit reliability.63, 434 Other functions useful during processing are solder masking and protection of resistors against acid electrochemical plating baths. As for sealing, conductors are absent or lie side-by-side (Fig. 8b ); excellent electrical insulation characteristics are therefore in most cases not necessary, and minor alkali additions are usually allowed.
The choice of a TF overglaze is a balance between limiting the shift in resistor characteristics and guaranteeing a sufficient level of protection. Both standard (e.g. ESL 4771P, Table 5) and acid resistant (such as Her CL90-8325, roughly equivalent to lead bearing ESL-481) lead free overglazes are now available from most TF materials suppliers. For very low required processing temperatures, one may use the sealing glasses discussed in the previous section, adding fillers to increase strength/abrasion resistance or to match the CTE to that of the substrate, or slightly higher firing glasses.291
As for the older lead bearing glasses, acid resistant types require a higher firing temperature, ca. 600°C, than standard ones. However, extensive publications on the chemistry and properties of Bi based TFR overglazes are still lacking, although some examples are available in the patent literature (Table 20, Hy-29321 and Don-01/04307 for the ∼600°C firing types); information may be garnered, for the low firing (⩽550°C) types, from sealing glasses.289, 290, 293, 313 ,314
A related application is the overglazing of displays (FPDs), where, besides providing suitable protection, the overglaze must avoid excessive degradation of the transparent conductor, usually indium tin oxide on the glass. This is achieved using low firing glasses similar to the sealing types, with a low level of In2O3, SnO2 and SbOy additives.297, 308
Low firing TF circuits (e.g. for FPDs) mostly use pure or almost pure Ag conductors, as Pd tends to oxidise in the intermediate firing range,64 which raises the issue of staining by in- diffusion of Ag+ and probable reprecipitation of Ag0. This is prevented by Cu308, 435 and Cr306 additions, presumably through oxidoreduction mechanisms.
Compared to consumer electronics, automotive and architectural enamels are exposed to much more severe outdoors environments, such as acid rain.33, 47, 48 In a way typical of the high volume cost sensitive automotive industry, single window rim dark enamel coating fulfils multiple roles, as illustrated in Fig. 10: antistick agent during forming of the panel, and hiding of the adhesive and its protection against ultraviolet (UV) light. Moreover, to optimise costs, the enamel must fire and achieve antistick properties reliably during the heating step required for forming of the window pane (650–700°C47, 436), and be sufficiently resistant to atmospheric attack to avoid requiring extra protective layers.
Application of enamel to automotive glass, as a antistick layer during forming and b adhesive protection layer (redrawn from Sakoske47)
Such enamels have recently undergone extensive development, as attested in the patent literature.298, 299, 301, 432, 436 – 439 Firing temperatures are in the 600–675°C range suitable for glass processing. First compositions were mainly glassy, relying on pigments to provide antistick properties, but have been replaced by crystallising compositions optimised to achieve the best chemical durability. In this respect, the trend has gone from Zn borates437 to Zn/Bi silicates438 – 440 and finally to mixed Bi (Bi4Ti3O12, Bi2Ti2O7, Bi2Ti3O9) and other (Zn, V–Bi) titanates,302, 436 which are claimed to exhibit better chemical resistance to acid attack. This is generally in accordance with the investigations of Fredericci et al.,48 who report increasing acid resistance going from Zn silicate to Bi silicate to Bi titanate phases, and the facile crystallisation of titanate phases.48, 363 Other compounds with ‘acidic’ oxides such as V2O5, Nb2O5, Ta2O5, MoO3 and WO3, alone or mixed,364 may probably have similar beneficial effects on acid resistance, but the composition must also be tuned in order to ensure sufficient durability of the remaining glass phase.
When coating glass or ceramics, an important issue is the degradation of the strength of the glass substrate by application of the enamel.47, 441 This is not specific to Bi based glasses, as similar effects have also been observed by our group on ceramic TF circuits with ‘classical’ lead bearing materials.442 The implications are significant, as automotive windshields play a structurally important role in cars, and TF electronics are used in high reliability, safety critical applications.
Insulating dielectrics
As reliable crystallisable lead free dielectrics are readily available for standard 850°C firing conditions,163 Bi based glasses are mainly applied to low firing insulating layers for circuitry on glass (FPDs/PDPs)181, 308, 431 or metal.71, 323 Typical configurations are multilayer circuits and crossovers (Fig. 8c ), as well as the deposition of complete circuits on metal (D).
Dielectrics may be formulated essentially the same way as filled sealing glasses, with the difference that some sort of ‘stabilisation’ is desirable. Otherwise, the dielectric (e.g. L-Bi12, Table 20) tends to reflow upon subsequent firing of conductors and resistors. Stabilisation may be achieved by crystallisation (spontaneous or filler nucleated)312 and/or by chemical reaction with the filler, as successfully experimented with PbO bearing glasses.161, 162 Bi2O3 should react similarly, with the ‘acidic’ oxides mentioned in the previous section; tuning the reactivity may be achieved by varying the glass composition, as well as the chemistry, amount and particle size of reactive filler.
Dielectrics on metal substrates
Commercial TF dielectrics/enamels for firing onto stainless steel exist (see Table 5), but, as the porcelain enamelled steel they replace, they require high processing temperatures unsuitable for sensors or application to substrates such as titanium (limited by oxidation) or aluminium (limited by melting),68, 71 and low firing dielectrics must be used.
However, dielectrics on metal face one important issue stemming from the reducing conditions (the section on ‘Oxidoreduction issues’) at the substrate/dielectric interface; to ensure good adhesion, the formation of soft and low melting metallic Bi and Pb should be avoided. Pre-oxidation of the metal is not always possible or useful due to reasons such as manufacturing process requirements, insufficient oxide adhesion or thickness. Moreover additional reducing conditions may be generated, on alloys forming Cr2O3 scales such as stainless steels, by chromate formation (Cr3+→6+) with glass components such BaO, PbO and Bi2O3,443, 444 although Cr may also remain as Cr3+ in the glass.373 Therefore, a generally applicable approach on metallic substrates consists in including significant amounts of transition metals that may be partially reduced in the glass (Fe3+→2+, Mn4+→3+→2+, Co3+→2+, Cu2+→1+), and that are moreover not deleterious when fully reduced to metal; Co is in fact found in dielectrics for steel substrates (Table 5). As the situation is very similar to PbO, it is surmised that our previous experiments with Pb based glasses, where Fe2O3 additions were successfully applied,68, 71 may be extended to Bi based ones.
It must be mentioned no adhesion problems were encountered on Al alloys,445 in spite of the very strong reducing power of Al; it is surmised that formation of Al2O3 rapidly results in the effective stoppage of oxygen diffusion, and hence limited interaction with the substrate, a hypothesis supported by the absence of oxidoreduction buffers in commercial enamels for Al.56
The high CTE some metals, such as austenitic stainless steel (17 ppm K−1) and aluminium alloys (23) constitutes an additional issue, causing warping of thin substrates and excessive stress build-up in the dielectric. This may be corrected by suitable high expansion fillers such as quartz, cristobalite, CaF2 and AlPO4, using chemical particle coating techniques if chemical reactions must be limited. In contrast, ferritic low alloy or stainless steel (∼11 ppm K−1) and Ti alloys (8–10) are reasonably well matched with low melting Bi based frits (8–11).
As a final note, commercial lead and alkali free bilayer low firing dielectrics on aluminium substrates have recently become available,446 but no detailed studies on their composition has been carried out to date.
Metallisations and TF conductors
Metallisations and conductors for electroceramic components such as resistors, capacitors voltage limiters, for solar cells, for automotive window heaters and TF conductors fundamentally share the same technology, i.e. the starting material is a finely divided conductor powder such as Ag, Ag/Pd, Au, Cu, with oxide and/or glass frit additive that promote densification and bonding to the substrate.21, 60, 63, 64, 447 – 451 While traditional lead borosilicate frits are widely used, application of Bi based frits in conductors is hardly new, with the 1st patent going back to 1945,283 and such conductors being widely in use by the early 1970s88, 449, 452 (see also Table 5). Mixed PbO–Bi2O3–B2O3–SiO2 glass frits have also been used, and Bi2O3 is also added in its own right, without nominally being part of a glass frit,158, 159, 288, 310, 452 – 457 in order to impart good adhesion by reacting with the substrate and potentially also with the metals, as well as for promoting liquation in fritless conductors.158, 457, 458 Its reactivity with 96%Al2O3 (+3·8 mass-%SiO2 and 0·2 mass-%CaO), 99·9%Al2O3 and BeO has been extensively studied.459 Bi2O3 does reacts with Al2O3, but reaction with the SiO2 contained in the glass phase of 96%Al2O3 is dominant below 850°C. Finally, Bi2O3 (and PbO) may also be added to conductor metallisations for electroceramics that contain these elements, in order to control the chemical interactions.451 The technical importance of integrating elements in the frit or adding them separately is not clear, i.e. avoidance of patent infringement may also play an important role.
The rather low affinity to oxygen of bismuth makes it easily reduced, at least partially, during firing or soldering. It is believed that partial reduction of Bi and Cu and partition between noble metal and oxides favours the formation of bridging bonds and thereby good adhesion.88, 159, 251 Solderability is of course also favourably impacted, but aged adhesion of solder pads can be affected if reduction of bismuth by tin in solder proceeds in service,64, 449 a problem that may be mitigated by incorporation of Bi2O3 into other oxides or glass frit (initially or during firing) or by adding oxides that act as barriers against this reaction.160, 456 Bi2O3 may also lead to formation of lossy semiconducting interfacial reaction products at electrocomponent interfaces310 and, in popular Ag–Pd metallisations, reinforce the problematic oxidation of Pd to PdO at intermediate temperatures by formation of Bi2PdO4, retarding Pd reduction at high temperatures.64, 460
The above considerations are summarised in Table 21. Relatively recent work159, 180, 250, 433, 455, 456 details both fritless and fritted (Table 7, Hw-00) Bi containing Pb free silver based conductors, and such conductors have recently become much more widely available from TF materials suppliers.
Advantages and issues of Bi in conductors and component metallisations.
The case of solar cell front side Ag metallisations is rather special: the traditionally PbO bearing glass frit must dissolve the antireflective layer (normally SiNy) and ideally create a good ohmic contact with the underlying n-type silicon emitter43, 50 in a highly non-equilibrium dissolution–oxidoreduction–precipitation process. The overall reaction sequence has been shown to involve dissolution of Ag into the glass and reduction by Si to form crystallites, but the exact conduction mechanism – some direct connections or only tunnelling in a similar fashion to TFRs (next section) and the role of PbO in the frit are still somewhat elusive.43 Nevertheless, successful substitution of PbO by Bi2O3 has been demonstrated recently, with presence of Ag ions in the glass enhanced by prior incorporation into the frit as Ag2O (Jeo-1, or Jeo-1' if Al2O3 and B2O3 mistakenly inverted in the paper, as is likely)53 or by firing in pure oxygen.52 These results allow removal of lead from what has become a high volume industry.
Glasses for TFRs
Current ‘cermet’ TFR compositions consist of a granular conductive phase dispersed in a glassy insulating matrix,59, 61, 62, 461 – 465 with additives to control the electrical properties;466, 467 conduction occurs by percolation through the conductive grain network, with intergrain transport through a nanometre thin glass film, thought to occur by tunnelling, essentially accounting for the overall resistivity (Fig. 11).84, 463, 468 – 474 In classical TFR series, the conductive phase is mainly ruthenium oxide (RuO2) and/or its pyrochlore compounds with PbO and Bi2O3, lead and bismuth ruthenate (Pb2Ru2O7−y and Bi2Ru2O7), and the glassy matrix is a high lead lead borosilicate glass (Fig. 12).59, 62, 475 Ruthenates give a higher resistivity than RuO2,423 and the composition of the glass also has a strong effect, both on the stability of ruthenates versus RuO2 and directly on the resistivity.464, 476 Alternative conductive phases for resistors can be based on IrO2 (properties similar to RuO2, more expensive,)477, 478 and compounds with CaO, SrO and BaO, which have the perovskite structure, are also possible.155 The synthesis and properties of RuO2, IrO2, their pyrochlore compounds and similar substances have been extensively studied.155, 203, 479 – 489 They belong to the class of electrically conductive oxides, which have been (very) extensively reviewed.490
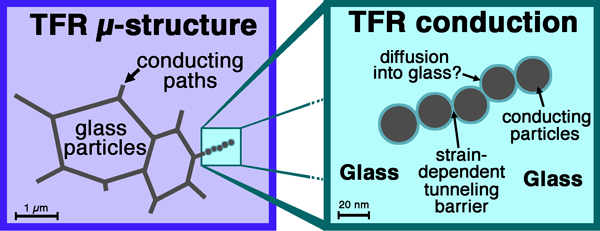
Microstructure and conduction mechanism of TFRs1
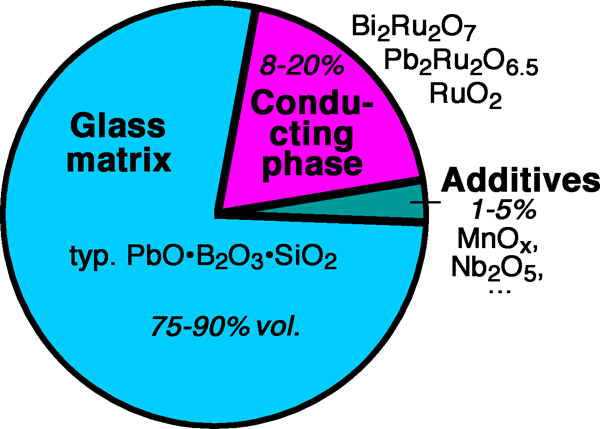
Typical composition (without temporary vehicle/binder) of TFRs with lead borosilicate glass matrix1
Such a complex resistor chemistry suggests possible particle glass chemical reactions, as Bi2O3 and PbO may be both in the glass and in the particles, and the configuration before firing may not be the equilibrium one. This is common in commercial systems, which frequently combine (Pb,Bi)2Ru2O7−y as the (initial) conductive phase dispersed in lead borosilicate glass, giving rise to exchange of Bi2O3 and PbO between the glass and the conducting phase as well as decomposition reactions.61,
62,
465,
491,
492 One may write the following main chemical equilibria
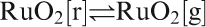
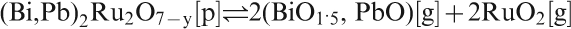
Equation (1) relates to the equilibrium between the pyrochlore ruthenate phase and the simple rutile oxide, and equation (2) describes the equilibrium composition of the pyrochlore phase relative to that of the glass; equations (3) and (4) relate to dissolution into/precipitation from the glass, which are quantitatively small due to the low solubility of RuO2 in the glass,493, 494 but may nevertheless play an important role in the electrical properties.495 Reactions (1) and (2) have indeed been observed experimentally,61, 62, 465, 491 and a detailed study, for PbO only, established lead ruthenate was stable/formed only at moderate temperatures in glasses with lead concentrations much higher than that used in commercial systems;464 this was confirmed in our studies on model RuO2 glass TFRs.85, 496 Therefore, most ruthenate based formulations are not at thermodynamic equilibrium during firing. However, even in model systems where the conducting phase is thermodynamically stable (in practice, those formulated using RuO2), the glass composition strongly influences the resistivity.476, 496
It must be additionally noted that the oxygen stoichiometry of Pb2Ru2O7−y is reported to be x = 0·5,483, 484 which implies an average valence of +2·5 for Pb (or +4·5 for Ru?) in this compound. This adds a further complication, as formation/decomposition of lead ruthenate may involve a change of the oxidation state of Pb, requiring exchange of oxygen with the firing atmosphere, an implication supported by results of TFR annealing studies in different atmospheres.497, 498 On the other hand, Bi and Ru essentially have the same valence in Bi2Ru2O7 as in the individual oxides Bi2O3 and RuO2; while Bi2Ru2O7 transforms at low temperatures to Bi3Ru3O11,460, 499 formally linked with a slight oxidation, this transformation is apparently not observed in TFRs, and does not involve a change of the Bi/Ru stoichiometry (Bi2Ru2O7 made by high temperature synthesis is actually reported to be slightly nonstoichiometric, Bi2Ru2O6·9 or Bi1·9Ru2O6·9 (see the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1), but this does not necessarily apply to ‘resistor grade’ nanopowders calcined at lower temperatures,483, 488 whose stoichiometry would require further study).
Therefore, it would seem logical to use a Bi based glass in order to enhance resistor stability to firing, given the otherwise similar properties to Pb based glasses. This would also lessen the property changes that classical resistors formulated with Pb based frits encounter when fired with Bi containing terminations.79, 500 Therefore, it is not surprising that several such compositions have been patented251 (Table 7, Hm). Nevertheless, making the switch is not trivial, given the intricacies of TFR chemistry. Recently, first encouraging results, some of which by our group, have been published on lead free TFRs with bismuth based glasses and RuO2 conducting particles,1, 70, 322, 323 for both standard 850°C251, 322 and relatively low (∼600°C) firing temperatures1, 70, 323 (see also Table 7, Hm/Hy/L-Bi12). The former is useful for application as standard TFRs, while the latter may be used for low firing compositions suitable for firing onto glass or high strength temperature sensitive metallic alloys to fabricate high performance force and pressure sensors.1, 68, 69, 71 An example of our results is given in Fig. 13, for resistors consisting of L-Bi12 glass with 11 mass-%RuO2 (∼9 vol. %) without additives. The results are very encouraging, featuring reproducible properties at firing temperature ⩾550°C and moderate, normal termination effects, i.e. without the increase of resistivity at the terminations77, 79 seen with the equivalent lead bearing systems.69
Sheet resistivity at 25°C and temperature coefficient (HTCR, 25–100°C), versus length and firing temperature, of experimental TFRs fired on Al2O3 with Ag terminations1
Several aspects must be studied carefully for Bi2O3 based glasses to become a standard matrix for TFRs. First, thermodynamic properties of RuO2, Bi2Ru2O7 and related compounds in regard to Bi based glasses must be known, especially the solubility of RuO2 in glass and the equilibrium between oxide and ruthenate, as was investigated in the lead based systems.
Additionally, as oxides of transition metals such as Ti, Zr, Nb, Mn, Fe, Cu and Zn are often added to control the temperature coefficient of resistance, it is important to know their possible interactions with Bi2O3 and RuO2. This has been the object of several studies, in pure oxide form, for ZnO,501 TiO2,502, CuO,503 and NiO.504 The Bi2Ru2O7 phase has been shown to be able to incorporate a significant amount of divalent transition metals (MO) as nominally (Bi2−xMx)Ru2O7−x/2 solid solutions, which seems to be a major difference with Pb2Ru2O7−y, where such solid solutions were not detected with CuO.505 For tetravalent TiO2, there is some disagreement as to the extent of Bi2(Ru,Ti)2O7 solid solutions.482, 502 Additionally, one must note that these studies only give clues to the observed behaviour in real TFR systems, because a) the activities of the oxides are different when dissolved in glass, and b) TFRs are usually non-equilibrium systems.
A more long term potential development would be the avoidance of costly precious metals, by using other metallic (or almost metallic) oxides and compounds as conductive phases, of which a very wide variety exists,490 or metal borides, silicides, etc. To circumvent the often observed processing issues with nitrogen firing TFRs,183 – 189, 190, 506 air firing systems should be developed. Several promising studies using molybdenum oxides have been carried out towards this goal,507 – 512 with the most recent embodiment512 having achieved transition to a bismuth glass frit, paving the way to low cost lead free resistors. These compositions are air fireable, because the conductive phase, MoO2 and/or other intermediate oxides (which are unstable in air) form in situ, after densification of the glass frit, by reduction of MoO3. This is brought about by a suitable reducing agent, usually boron. Another base metal conductive filler successfully investigated with bismuth glasses is SnO2/Sb,166 which seems, however, more suitable for higher ranges of surface resistance (>100 kΩ), in line with the semiconductive properties of this filler.
Other applications
Optics
Heavy metal ions such as Bi3+, Pb2+ and Tl+ feature a unique combination of properties: high mass and polarisability, together with low bond strength. For optical applications of HMO materials, be it in glassy, ceramic or single crystal form, this translates into high refractive indices n (Table 22), dispersion and optical non-linear susceptibility χ (3), as well as low phonon energies.36, 112, 124, 128, 329, 371 In addition, low levels of partly reduced Bi in both Bi based and other glasses have been found to display broad IR luminescence, with possible application to optical amplification353, 428 – 430 (see also discussion in the section on ‘Oxidation state of Bi species in glasses’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1). Glasses of HMO are a promising alternative to fluorides,37, 192, 193 due to their better chemical durability.
Optical properties of HMO glasses
*At wavelength (nm) indicated in index.
†Transmission (%) and thickness (mm) given in index.
Infrared cut-off wavelength is related to the highest frequency bond vibrations/maximum phonon energy. Therefore, HMO glasses for light amplification, and especially for far IR transmission, should contain no standard network formers (B2O3, SiO2, P2O5) or light intermediates with strong bonding such as Al2O3. Although GeO2 constitutes a good drop-in replacement for SiO2, much better IR performance can be obtained (Table 22) with non-standard formers such as Ga2O3 in the PbO–Bi2O3–Ga2O3 system36, 371 and TeO2 in Bi2O3–TeO2–WO3,123 and even systems entirely without glass formers such as Li2O–[BaO/PbO]–Bi2O3 381, 382, 388 and SrO–PbO–Bi2O3.117 Considerable care must be exercised when melting Bi containing glasses for optical applications, as they are quite susceptible to reduction by trace organics or excessive melting temperatures (the section on ‘Oxidoreduction issues’), and to contamination by crucibles.36, 387 Reduction or Pt pickup leads to darkening/staining of the glass, while even relatively low contamination by oxides such as SiO2 and Al2O3 will introduce high frequency vibrations into the network, degrading far IR transmission.
Obtaining a high refractive index and non-linear susceptibility, on the other hand, only involves the average glass properties, allowing limited use of standard network formers and thereby greatly facilitating vitrification. Synergistic vitrification through mixing of SiO2, B2O3 and Al2O3 and substituting some Bi2O3 by ZnO (the section on ‘Glass formation’) allows maximisation of HMO content; recent results on glasses optimised this way116 yield n = 2·31 at 633 nm and χ (3) = 4·9×10−11 esu at 748 nm. The glass composition was not specified exactly, but mentioned as Bi2O3–B2O3–SiO2, and is probably close to Smt-7 (Table 7). The high and very fast response of these glasses makes them very promising for high frequency fully optical switching.112
In the case of upconversion, recent measurements119, 371 indicate that average properties (phonon density) rather than maximal phonon energy dominates efficiency, which is supported by the good results achieved by others with doped/codoped HMO glasses containing a low amount of network formers,121, 124, 125, 325, 370 usually GeO2 given its lower impact on performance. An additional advantage of Bi2O3 based HMO glasses for this application is their good compatibility with lanthanide additions (the section on ‘Glass formation’).
Finally, the above considerations, though essentially focussed on glasses, are also applicable to similar crystalline (single crystal, ceramic or glass–ceramic) compounds, of which many exhibit promising optical properties.351
γ-ray protection glasses
Due to their ideal combination of high γ-ray absorption coefficient (imparted by their large atomic mass) and good glass forming ability as oxides, Pb and Bi are useful in γ-ray absorbing windows in the nuclear industry and high energy physics.129 – 135 Requirements for these glasses are in general similar to that for nonlinear optics: a HMO glass is needed for a good attenuation factor, but moderate concentrations of traditional network formers (B2O3 and SiO2) are allowed, facilitating fabrication of the thick parts needed for good shielding.
An early study of was carried out by Brekhovskikh in the PbO–Bi2O3–SiO2 system;129 ternary glasses down to ∼17%SiO2 and with density >8000 kg m−3 could be manufactured (Rad-1), but practical considerations (need for casting in thick sections requires ∼30%SiO2, Rad-2). An analogous borate glass (Rad-4, ∼21%BO1·5) was formulated130, but no indication was given on its castability in thick sections. The claim of a Bi2O3–SiO2 glass with ∼95 cation-%Bi131 is dubious, as the indicated density of 7284 kg m−3 corresponds to ∼70%Bi,248 In the light of the section on ‘Glass formation’, maximising HMO content in a Bi based Pb free glass would most likely best be achieved in a more complex system, such as SrO–BaO–ZnO–Bi2O3–Al2O3–B2O3–SiO2. Compared to PbO, γ-ray attenuation coefficients with Bi2O3 are similar129, 134 (the apparent observed increase when substituting PbO with Bi2O3 130 is simply due to the ‘molecular’ formulation, where Bi is counted twice versus Pb), yielding a progressive increase in HMO content from 69 to 79% on a cation basis.
This said, the main issue with γ-ray protection glasses lies with radiation induced darkening, for which results are relatively unclear.131 – 133, 135 Significant darkening in the short wavelength portion of the visible spectrum was observed after 2·5 kGy from 60Co, but no systematic trend versus composition was observed for Bi2O3–B2O3 glasses,133 which is also the case for Bi2O3–SiO2 glasses after 10, 20 and 40 kGy 60Co.131 This darkening on the other hand was absent or very low in the visible for Bi2O3–B2O3 glasses optionally doped with CuOy after up to 50 kGy 60Co.132 Although these latter results are very encouraging, systematic studies of the mechanisms of radiation induced damage and of the good resistance against it of some glasses, including doping/impurity effects, still need to be carried out to reliably achieve low darkening Bi based glasses.
Bi2O3 in (glass–)ceramics and crystals
A short discussion is given in this section of Bi2O3 in five types of crystalline materials:
piezoceramics
ionic and mixed conductors with high oxygen mobility
high temperature superconductors
scintillators
ferroelectric memories.
Besides these applications, Bi2O3 is a component of many electroceramics, in both the active crystalline phase or as a sintering aid,9 and compounds such as bismuth ruthenates (Bi2Ru2O7/Bi3Ru3O11), besides being a useful conductive phase for TFRs (see the section on ‘Glasses for TFRs’), are envisioned as chemical catalysts and electrodes.499, 513 Bismuth vanadate (BiVO4) is a relatively new yellow pigment that provides an alternative to toxic Pb and Cd based pigments,514 and crystalline bismuth borates have interesting optical properties,351 akin to the corresponding glasses (see the section on ‘Optics’). A quite extensive list of Bi2O3 based compounds and corresponding applications is given in recent work,403, 515 together with an extensive discussion on their structural features (some of which are also discussed in the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1, as they yield insight into the structure of the corresponding glasses). Finally, it is worth mentioning that derivatives of the analogous chalcogenide, Bi2Te3, are the dominant room temperature thermoelectric materials.516
Piezoceramics
Besides glass, the main application of PbO bearing materials is arguably PZT = Pb(Zr,Ti)O3 and similar ceramics, mainly used for their large piezoelectric or electrostrictive properties. As these ceramics are present in many small essentially disposable electronic devices, considerable effort has been devoted to finding lead free substitutes with at least acceptable performance. Recently, complex mixtures of Bi2O3 perovskite compounds such as Bi0·5(Na,K)0·5TiO3 and Bi(Fe,Sc)O3 with (Ba,Sr)TiO3 and (Na,K)NbO3 have given rise to promising materials achieving strains comparable to that of the classical PZT ceramics, although many issues remain.517, 518 Note that bismuth titanate (Bi4Ti3O12) is also a piezoceramic, albeit a specialised, high temperature and low activity one.
Ionic and mixed ionic electronic conductors with high oxygen mobility
The high temperature disordered defect fluorite cubic δ-Bi2O3 phase contains a very large amount of disordered oxygen vacancies, it may in fact be written as Bi(O3/4□1/4)2, i.e. with a quarter of the oxygen anion sublattice randomly empty, and therefore exhibits exceptionally high ionic conductivity. Although δ-Bi2O3 is only stable above 730°C, Takahashi et al. 519 established that this disordered phase may be stabilised at lower temperatures, much like cubic zirconia, by various dopants, thus giving birth to a new family of solid electrolyte materials519 – 522 with high ionic conductivity at moderate temperatures. For instance, Bi2O3 solid solutions stabilised by oxides such as Y2O3 or Er2O3 have ionic conductivities exceeding 1 S m−1 at 500°C, more than 20× that of cubic or tetragonal zirconia, and more recent ‘BIMEVOX’ materials with an oxygen deficient Aurivillius structure have even higher conductivity. However, as all Bi2O3 based materials, these ionic conductors have limited stability towards reduction, and their use is limited in solid oxide fuel cell (SOFC) applications. Nevertheless, they may still be used to this end, provided the reduction potential is limited or a thin protective layer of another electrolyte material is used on the reducing side. In addition, Bi2O3 ionic and mixed ionic electronic conductors may find other important applications such as air side electrodes for SOFCs, chemical electrodes, oxygen sensing and oxygen extraction from air.521, 523
High temperature superconductor materials
The discovery of high temperature (>100 K) superconductivity in the Bi–Sr–Ca–Cu oxide (‘BiSCCO’) family (with possible substitution of Bi by Pb)99, 524, 525 having low melting points opened up the possibility of greatly easier processing of these materials. In fact, these high temperature superconductor compounds have compositions (examples in Table 23) corresponding to the ‘heavy metal oxide’ glasses,36 and are therefore amenable to processing by the vitroceramic route, i.e. glass melting and quenching, followed by shaping and finally crystallisation and annealing to yield the superconducting phase39, 99, 101, 102, 104, 105 with zero resistance temperatures in excess of 100 K under optimal conditions.101 Low concentrations of suitable dopants do not significantly enhance the superconducting properties, but are added to facilitate processing and yield more consistent products. Reported melting temperatures are in the 1100–1300°C range, with long term annealing around 850°C to form the superconducting phase(s). These materials feature rather high resistivity in their glassy state,100 in contrast to amorphous Bi3Ru3O11.499
Compositions of selected BiSCCO superconductors, given in cation molar proportions [z = Pb/(Pb+Bi)]
*Oxides = PbO, BiO1·5, SrO, CaO, CuOy (y preparation dependent), AlO1·5 and MoO3.
Besides these materials, doping BaBiO3 (with Bi in mixed +3 and +5 valence) with K or Pb (Ba,K)BiO3 or Ba(Bi,Pb)O3 respectively, yields superconductors with transition temperatures up to 30 K that are of theoretical interest due to their simple cubic structure.526
Scintillators
In addition to heavy glasses potentially improving scintillation efficiency,36 crystals such as Bi4Ge3O12, Bi4Si3O12 and their solid solutions368, 527, 528 are useful as γ-ray scintillators, the active ion in this case being directly Bi3+ (i.e. ‘self-activated’). Potential preparation of Bi4Ge3O12 by the glass–ceramic route has been explored.368
Ferroelectric memories
Besides being useful as a piezoceramic, PZT in thin film form is also of interest for non-volatile ferroelectric memories. However, it was soon realised that its sensitivity to so called fatigue, i.e. degradation upon repeated switching, was an important, although alleviated by the use of oxide electrode contacts.529 This problem was largely solved by the introduction of Aurivillius type layered perovskite compounds derived from Bi4Ti3O12 such as Bi3·25La0·75Ti3O12 (simple substitution with La),530 SrBi2Ta2O9 531 or SrBi4Ti4O15,532 which feature vastly improved switching endurance.
Conclusions
The many examples discussed in this review show that replacement of lead by bismuth in TF and related glass frits seems the most feasible route in the short to middle term. In fact, this is already happening in several important industries such as electronics, displays and automotive/architectural glass, in spite of the still considerable technological issues. Moreover, promising new applications are currently undergoing development in the sensors and optics field.
The considerably higher price of bismuth, ∼10 times that of lead533 should not constitute a problem in many high added value applications, where a relatively low overall volume of material is used, such as TF electronics, sensor cells and specialised optics. Moreover, Bi is not expected to replace lead in high volume applications such as vehicle batteries, and only to a very modest extent in electronic solders, Bi–Sn and similar low melting eutectic solder having only limited use. The same applies for use of Pb–Bi eutectic (LBE) coolants.422
For TF electronics, the very strong increase in prices of all noble metals (Ag, Pd, Pt, Ru) in recent times533 is therefore a much more pressing issue for conductor and resistor materials. Development should be concentrated on new materials which reduce or altogether avoid their use, as has already been initiated for resistors.166, 512 On the other hand, the price of Bi can be a factor in high volume and very cost sensitive architectural and automotive applications,47 so further developments to reduce its use in these sectors are likely.
From a scientific point of view, more well controlled experimental data is necessary for bismuth based glasses, as the reported results, especially on glass stability and properties, are somewhat conflicting, even for the simplest systems. Detailed studies on devitrification, separately assessing the composition dependent dynamics of crystal nucleation and growth, are strongly needed as well, especially in the practically important low melting borosilicates and HMO gallate systems.
Also, the very disordered structure Bi3+ adopts within the glass network, and how it is influenced by glass composition and thermal history, requires further clarification, especially in the light of recent structural studies403 on the peculiar behaviour of ‘lone pair’ ions such as Bi3+, Sb3+, Pb2+ and Sn2+ (see also the section on ‘Coordination of bismuth in crystalline oxides’ in Supplementary Material 1 http://dx.doi.org/10.1179/1743280412Y.0000000010.S1). Apparently, PbO based glasses and two important envisioned substitutes, those based on Bi2O3 and on SnO, share fundamental structural features. Extending the detailed structural studies and computer modelling to glasses would lead to a clearer picture of how glass formation and structure are affected by the stereochemically active lone pair, the valence and the polarisability.
Finally, it must be noted that this review has only dealt with relatively standard glassmaking techniques of Bi based glasses; chemical methods such as solution coating of powders, nanopowder synthesis or sol–gel casting534 constitute promising alternative routes, potentially allowing the manufacture of novel glasses and glass matrix composites with a degree of control not achievable by standard methods.
Footnotes
Acknowledgements
The author gratefully acknowledges partial financial support from the Swiss National Science Foundation NCCR CO-ME programme, as well as the assistance and patience of present and former colleagues at EPFL, especially C. Jacq and M. Garcin for experimental work and help from N. Takano and I. Stolichnov with Japanese- and Russian-only sources. The coordination shell drawings were created with the Avogadro molecular editing software.
