Abstract
Objective:
The use of a hand-held ultrasound device provides a portable, cost-effective solution for renal assessments in rural settings, but their accuracy and reliability in measuring kidney metrics remain uncertain. The aim of this study was to evaluate the validity and reliability of the Lumify hand-held ultrasound device (LHUS) in capturing basic kidney metrics in healthy adults.
Materials and Methods:
Twenty individuals underwent renal sonography measurements, which included length, height, width, cortical thickness, parenchymal echogenicity, and renal resistive index (RRI). These measurements were completed using LHUS and the Epiq Elite ultrasound equipment system. LHUS measurements were repeated for test-retest reliability (TRR). Validity and TRR were assessed using intraclass correlation coefficient (ICC), Bland-Altman plots, coefficient of variation (COV), and mean absolute percent error (MAPE).
Results:
There was poor agreement (0.414, p = .048) between equipment systems for RRI. All other measures demonstrated moderate to excellent agreement (ICC = 0.75–0.94, p < .001) between systems. The left kidney showed poor agreement for RRI (0.06, p = .45). MAPE for all measurements between devices was <5%, indicating acceptable accuracy. All repeated measurements showed moderate to excellent reliability (ICC = 0.71–0.96, p < .001), with low mean COV for each measurement (0.02–0.08).
Conclusion:
Based on this study, LHUS appears to be a valid and reliable tool for basic kidney metrics in healthy adults, but caution should be used when obtaining an RRI, especially in the left kidney.
Sonography plays an important role in the assessment of kidney structure and function, offering insights into renal health through measurements, such as length, width, height, volume, cortical thickness (CT), parenchymal echogenicity (PE), and renal resistive index (RRI).1 –3 In traditional clinical settings, these measurements are typically obtained using high-end ultrasound equipment systems, which, while accurate and reliable, are often expensive, bulky, and require a stable environment to operate effectively. This makes standard ultrasound systems less practical in rural and low-resource settings where access to advanced medical equipment is limited.4,5
In many rural or remote areas, health care providers face significant challenges in delivering quality care due to the lack of medical infrastructure and the high cost of standard diagnostic tools. 6 Hand-held ultrasound devices have emerged as a promising solution to these challenges. These devices are portable, cost-effective, and user-friendly, making them suitable for use in low-resource environments where traditional ultrasound equipment are not feasible. 7 The portability of hand-held ultrasound devices allows health care providers to bring diagnostic capabilities directly to the patient, even in the most inaccessible locations. 8
While a hand-held ultrasound device offers advantages, its broader adoption hinges on its proven accuracy and reliability in measuring critical kidney metrics. Despite their growing popularity, there is limited research on the validity and reliability of hand-held ultrasound devices in measuring basic kidney metrics. Previous studies have primarily focused on the use of these devices for diagnosing specific kidney pathologies (hydronephrosis, kidney cysts, and nephrolithiasis),9 –12 without thoroughly evaluating their accuracy and consistency in measuring key kidney metrics, such as length, height, width, volume, CT, PE, and RRI.
Given the increasing reliance on hand-held ultrasound devices in rural and low-resource settings, establishing their validity and reliability for kidney measurements is essential. This study aimed to assess the construct validity and test-retest reliability (TRR) of the Lumify hand-held ultrasound device (LHUS) (Philips Healthcare, Andover, Mass.) in measuring basic renal metrics in healthy adults. The measurements obtained with the LHUS were compared with those from the Epiq Elite (Philips Healthcare, Andover, Mass.) ultrasound system (ESUS), to determine the accuracy and reliability of these ultrasound devices.
Materials and Methods
Twenty healthy individuals (10 females and 10 males), above the age of 18 (18–29 years) with no known medical issues, were recruited for this study. Participants were recruited through flyers and personal referrals targeting university students and community members in Laramie, Wyoming. Using G*Power (v 3.1.9.7, Heinrich-Heine-Universität Düsseldorf), it was determined that 20 individual’s organ measurements would provide enough statistical power to complete this data analysis. An effect size (q = 0.66) was calculated using correlations from two similar studies (r = 0.97 and r = 0.51).13,14 These correlations were used to reflect the relationship between the measurement outcomes from the LHUS and the standard ESUS. The effect size was calculated specifically to investigate the intraclass correlation (ICC) between the LHUS and ESUS. By inputting the effect size, an alpha level of 0.05, and a power level of 0.80, the power analysis determined that 40 measurements were needed for sufficient statistical power. While 20 participants were recruited, each provided measurements for both the right and left kidneys, resulting in a total of 40 measurements for the analysis.
To validate this method, a Pearson correlation analysis was performed between the measurements from the right and left kidneys of each participant. The analysis revealed no significant correlation between the two sets of measurements (r = 0.18, p = .64), indicating that measurements from the right and left kidneys were independent of each other. While including both kidneys per participant was intended to increase data density, this finding suggests that the measurements of the two kidneys can be considered independent for the purposes of this study.
Participants below the age of 18, those with a history of renal agenesis, kidney dysplasia, nephrectomy, dialysis, medical conditions like hypertension, renal and cardiovascular disease, type I/type II diabetes, as well as specific kidney diseases, such as kidney stones, glomerulonephritis, polycystic kidney disease, and those who had two or more urinary tract infections in the past year, were excluded from the study. All exclusion criteria previously mentioned were identified using a health history questionnaire. This study was approved by the Institutional Review Board (IRB #20220916KM03389) and the study was performed in accordance with the ethical standards as stipulated in the 1964 Declaration of Helsinki. Informed written consent was obtained from all participants before their involvement with the study.
Research Design
The construct validity of the LHUS was investigated by comparing kidney measurements to a contemporary standard ESUS. Comparisons were done separately on the left and right kidneys for each of the 20 study participants. Participants attended two separate sessions. During the first session, anthropometric measurements were recorded. Less than 14 days later, participants returned for a second session, during which sonography assessments of renal length, width, height, CT, PE, and RRI were taken for both the right and left kidneys, using both the LHUS and ESUS. This separation of sessions ensured that anthropometric and sonographic measurements were conducted independently. Each assessment was performed once for each kidney using both the LHUS and ESUS, with the order of ultrasound devices was randomized. To evaluate the TRR of the LHUS, measurements with this device were repeated once more after a 5-minute break. While multiple measurements are generally recommended to enhance accuracy, the decision to take a single measurement in this study was based on previous published research.9 –11,13,14 The randomized order of ultrasound devices further minimize bias, allowing for a direct comparison between the LHUS and ESUS. Future research may benefit from multiple measurements to strengthen the findings, but the methodology was appropriate for the aim of this study. To protect the internal validity of this study, measurements taken on each ultrasound device by the same sonographer, that is, Registered Diagnostic Medical Sonographer (Abdomen and OB/Gyn) and Registered Vascular Technologist (RVT) with 5 years of clinical experience.
Measurements
Participant weight was measured by a calibrated digital scale (SECA Medical; Hamburg, DE), and height was measured by a calibrated wall mounted stadiometer (Health O Meter; McCook, IL, USA). Body mass index (BMI) was calculated by dividing the weight in kilograms (kg) by the height in meters squared (m2). Body fat percentage and lean muscle mass were measured using dual energy X-ray absorptiometry (GE Lunar Prodigy Advance; Madison, WI, USA). To obtain the sonographic measurements, the participants laid on a padded table in the lateral decubitus position. The images and measurements taken with the LHUS used a curvilinear transducer, which was compatible with a smartphone or tablet. 13 This transducer is a curved array type with a 5–2 MHz frequency range, has a 67.5º field of view, high-resolution imaging for abdominal applications, and tissue Doppler imaging. 15 The ESUS used the same transducer type (curved array with a 5–2 MHz frequency range and has a 67.5º field of view). Renal length, height, and CT were measured using the image based on the maximum longitudinal dimension, 16 and the width was measured in the section perpendicular to the longitudinal axis of the kidney. 17 Kidney volume was calculated manually based on the formula, 0.50 × kidney length × kidney width × kidney height. 17 CT was measured as the shortest distance from the renal sinus fat to the renal capsule, 16 and PE was determined by comparing hepatic echogenicity for the right kidney and splenic echogenicity for the left kidney. 18 Increased PE was defined when the renal parenchyma was more echogenic than the liver or spleen. 18 The sonographer rated the PE as normal, abnormal, or severe abnormality. 19 The sonographer assessed PE using the following criteria: 19 PE was rated as normal (Rating 1) when it was homogenous and had echogenicity similar to or slightly lower than the liver or spleen. It was classified as abnormal (Rating 2) if there was increased echogenicity, suggesting early renal pathology, but without significant structural abnormalities. Severe abnormality (Rating 3) was assigned when there was marked hyperechogenicity, often accompanied by cortical thinning, loss of corticomedullary differentiation, or other structural changes. The LHUS and ESUS use Doppler to measure blood velocity in the arteries, including peak systolic and end-diastolic velocities. To measure RRI, peak systolic and end-diastolic velocities of the distal renal artery were measured with the transducer set at a 60-degree angle. 20 RRI was then calculated manually using the formula, [(peak systolic velocity − end − diastolic velocity) / peak systolic velocity)]. 20
Data Analysis
Data were reported as the mean ± standard deviation with range (minimum–maximum) for anthropometric data and kidney measurements. A one-way analysis of variance (ANOVA) was performed to investigate potential gender differences in anthropometric measurements between male and female participants. Spearman’s rank correlation was used to assess the relationship of PE measurements between the LHUS and ESUS. Consistency between LHUS and ESUS measurements for each metric of interest was assessed via ICC using two-way mixed-effects regression models. 21 Because of the differences in morphology and position next to other organs, separate ICC analyses were done for the left and right kidneys of each participant. The sample size for each of these analyses was 20. ICC values were reported according to the guideline of Koo and Li 21 in which a coefficient < 0.50 is indicated as poor agreement, values between 0.50 and 0.75 as moderate agreement, values between 0.75 and 0.90 as good agreement, and values > 0.90 as excellent agreement. Mean difference and coefficient of variation (COV) between the LHUS and ESUS of each measurement for the left and right kidneys were also calculated. In addition, Bland Altman plots were used to further assess the agreement and potential bias between the LHUS and ESUS. Finally, the mean absolute percent error (MAPE) between the LHUS and ESUS was calculated from the formula ([LHUS measurement–ESUS measurement]/ESUS measurement) × 100. The criteria for validity of the LHUS were determined a priori and were considered valid if both of the following criteria were met. (1) The average of the ICC values for all measurements was good to excellent (ICC between 0.7 and 0.99) 21 and (2) The Bland Altman limits of agreement for each measurement are found to be clinically insignificant. 22 The Bland Altman limits of agreement were determined to be clinically significant, a priori, if they were greater than 1 cm for length, width, height, and CT, greater than 50 cm3 for volume, and greater than 0.2 for RRI. Clinical significance was based on what the sonographer determined to be a meaningful difference.
To assess the TRR of the LHUS, an ICC using a two-way mixed-effects model based on mean rating and absolute agreement was used. 21 To further examine the TRR, the COV between the repeated LHUS measurements was found, and the mean COV for each renal measurement was calculated. COV values were reported according to the guideline of Ospina and Marmolejo-Ramos 23 in which a coefficient < 0.05 is indicated as low variation, 0.06–0.09 as moderate variation, and > 0.10 as high variation. The LHUS was reliable if both of the following criteria were met. (1) The average of the ICC values for all repeated measurements was good to excellent (ICC between 0.7 and 0.99) 21 and (2) the average COV for all repeated measurements was below 0.05. p-values were set at a significance level of < .05 and all statistical analyses were performed using IBM SPSS statistics (v 26 IBM Corp., Armonk, NY, USA). All figures were created using GraphPad Prism (v 9.5.1 La Jolla, CA, USA).
Results
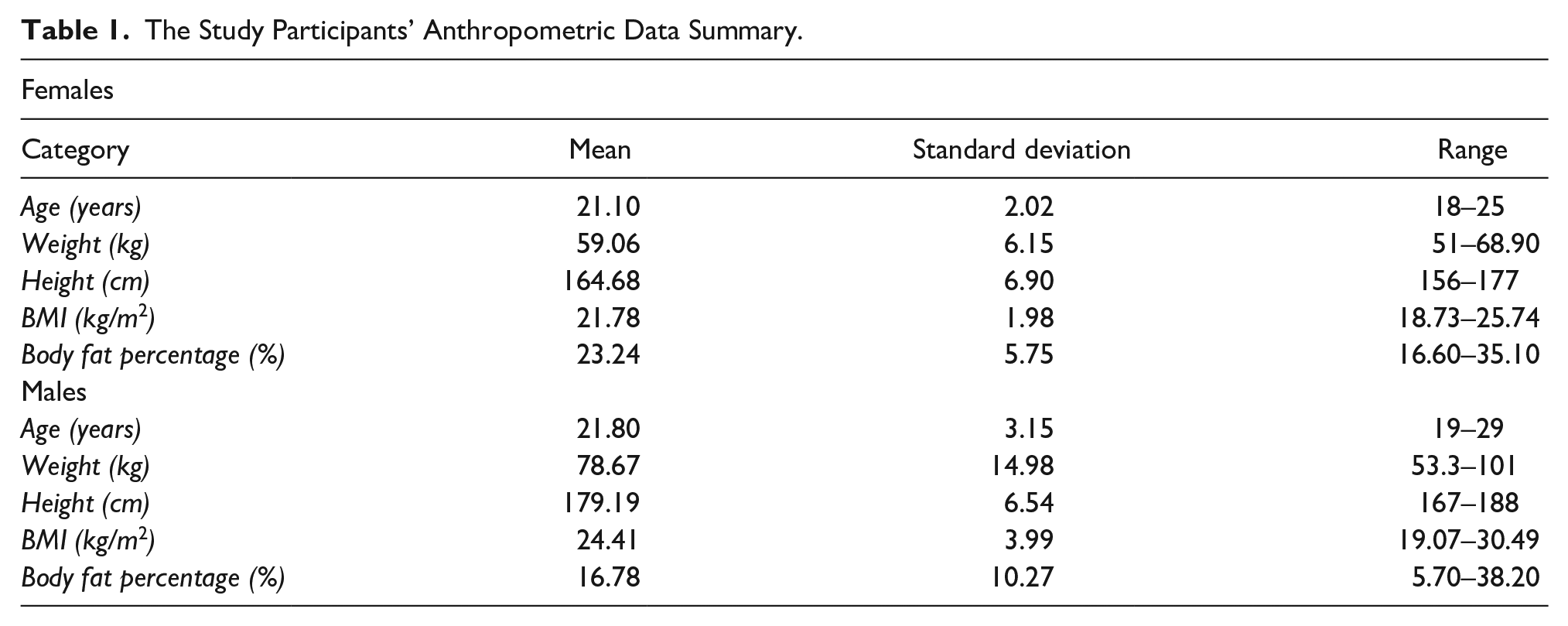
There were no data excluded from any of the participants who underwent sonographic measurements. Anthropometric data of the participants are provided in Table 1. All means, standard deviations, and ranges of each measurement from both ultrasound devices are provided in Table 2. Height (p < .001) and weight (p = .001) were significantly different between male and female participants, with females being shorter and lighter. However, no significant differences were observed in BMI, body fat percentage, and age (all p > .05). Example images taken from the LHUS of the right and left kidneys are provided in Figure 1.
The Study Participants’ Anthropometric Data Summary.
The Measurement Mean, Standard Deviation, and Range Measures Taken With the Hand-held (LHUS) and Standard Ultrasound Devices (ESUS).
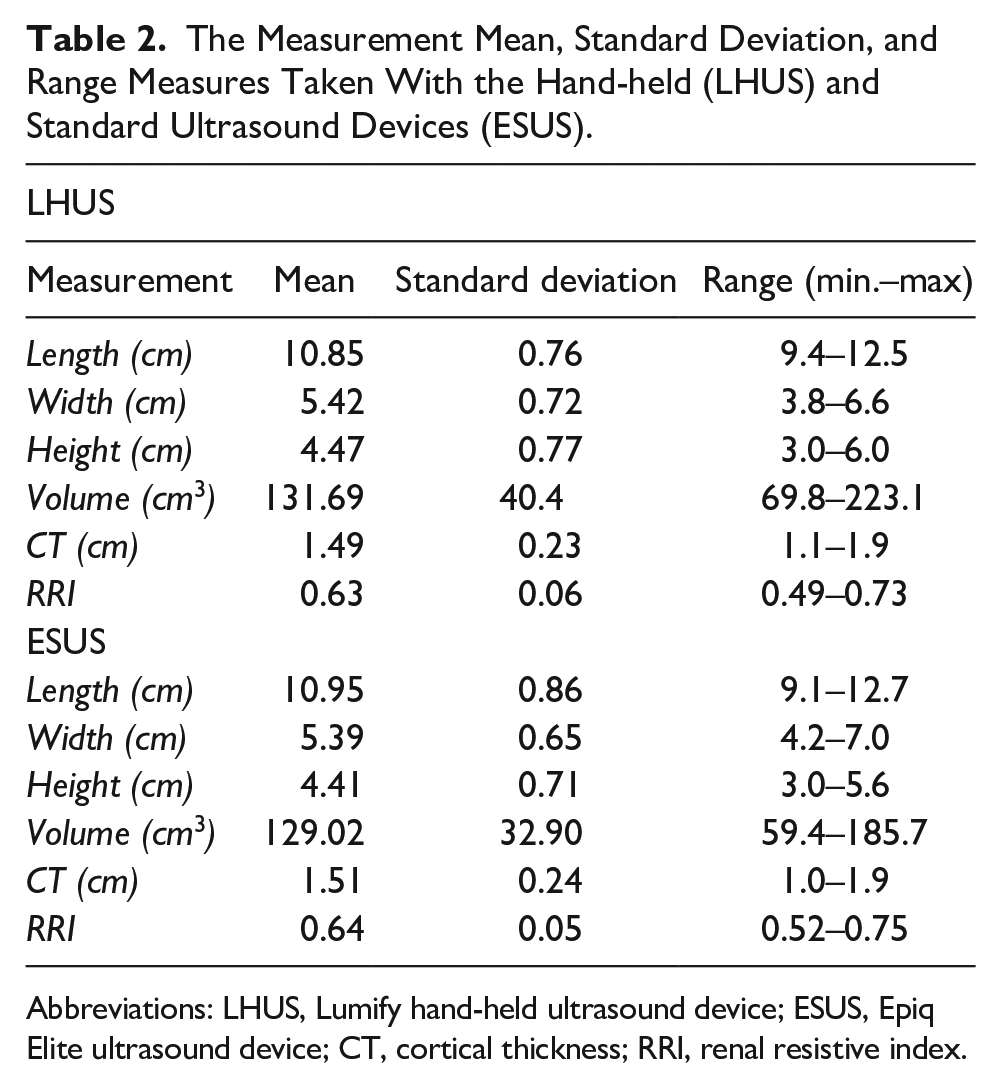
Abbreviations: LHUS, Lumify hand-held ultrasound device; ESUS, Epiq Elite ultrasound device; CT, cortical thickness; RRI, renal resistive index.
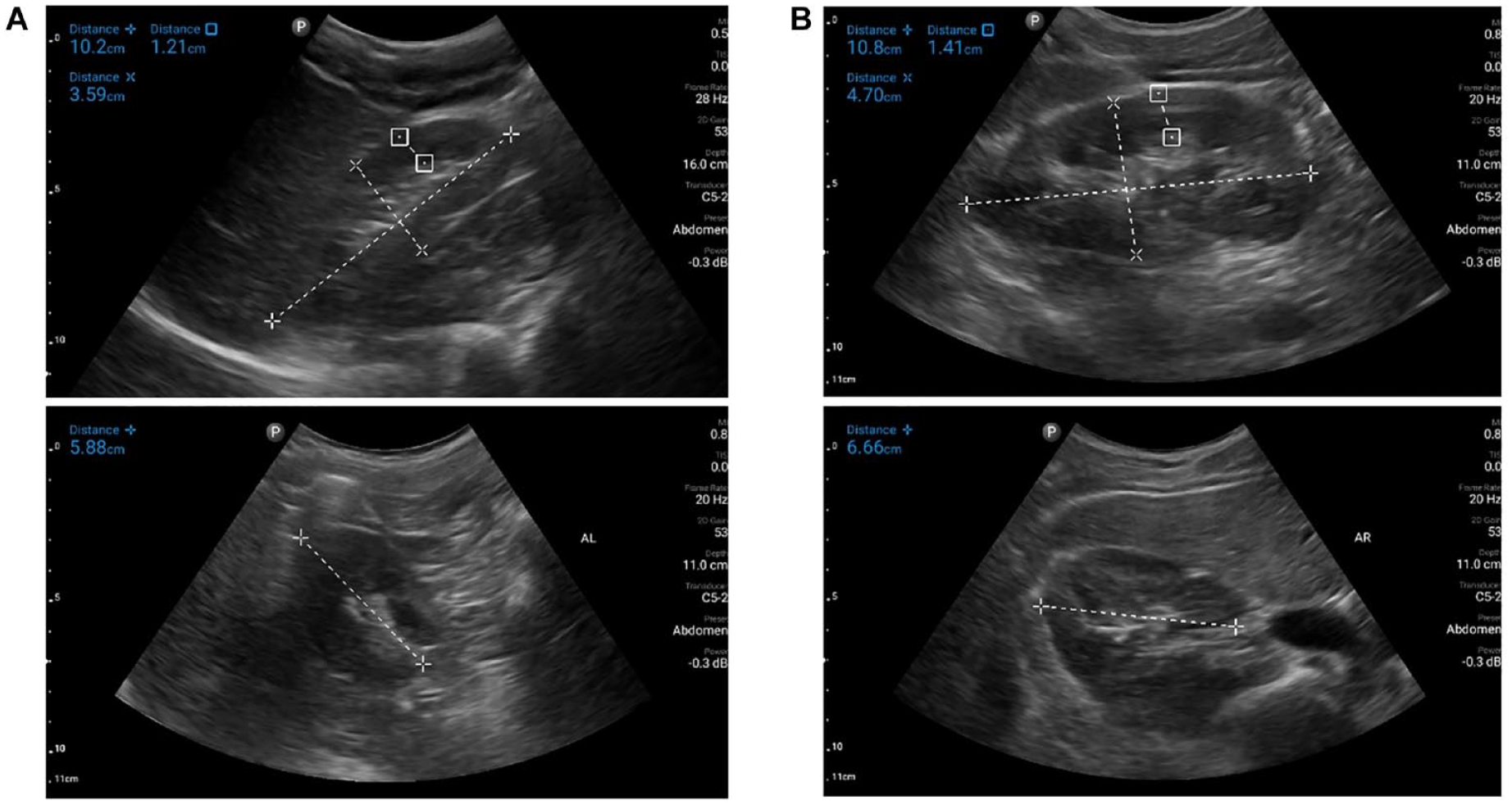
Sonographic images captured by the LHUS of the right (A.) and the left (B.) kidneys. The measurements displayed in these images are in the sagittal and transverse plane and include length, width, and CT. LHUS, Lumify hand-held ultrasound device; CT, cortical thickness.
Validity
Data for the ICC, separate left and right ICCs, and MAPE analyses are included in Table 3. All PE grades from the LHUS and ESUS were ranked as 1 (normal), and thus Spearman’s rank correlation could not be calculated. Bland Altman plots for RRI revealed that 95% of the differences between the LHUS RRI and ESUS RRI fell within two standard deviations of the mean difference (limits of agreement) (See Figure 2), thus, indicating agreement between the two devices when performing RRI measurements. A Bland Altman plot showed proportional bias between LHUS and ESUS when measuring kidney volume. As mean kidney volume increased, the variability between the two ultrasound units increased, which was attributable to the larger measurements acquired by the LHUS in comparison with the ESUS. Despite the increased variability, the larger differences remained within the limits of agreement. The ICC analyses comparing the LHUS and ESUS revealed excellent agreement for length, and volume (0.94 and 0.92, respectively), good agreement for width and height (both 0.86), moderate agreement for CT (0.74), and poor agreement for RRI (0.41) (all p < .05). Separate ICC analyses revealed no considerable differences in ICC values between the left and right kidneys for length, width, height, and volume. However, there were substantial differences in ICC values between left and right kidneys for CT and RRI, where the left kidney had very poor agreement and the right kidney showed moderate to good agreement (See Table 3). The mean difference and COV of each measurement for separate right and left kidneys were found to be larger in the left kidney for all measurements except for height (See Table 4). MAPE for all measurements between the LHUS and ESUS was found to be acceptably accurate (all < 5%). 24 The LHUS was found to be valid based upon an average ICC value of 0.78, and the Bland Altman limits of agreement for each measurement were clinically insignificant.
ICC, Separate Left and Right ICCs, and MAPE Based on the Study Data.
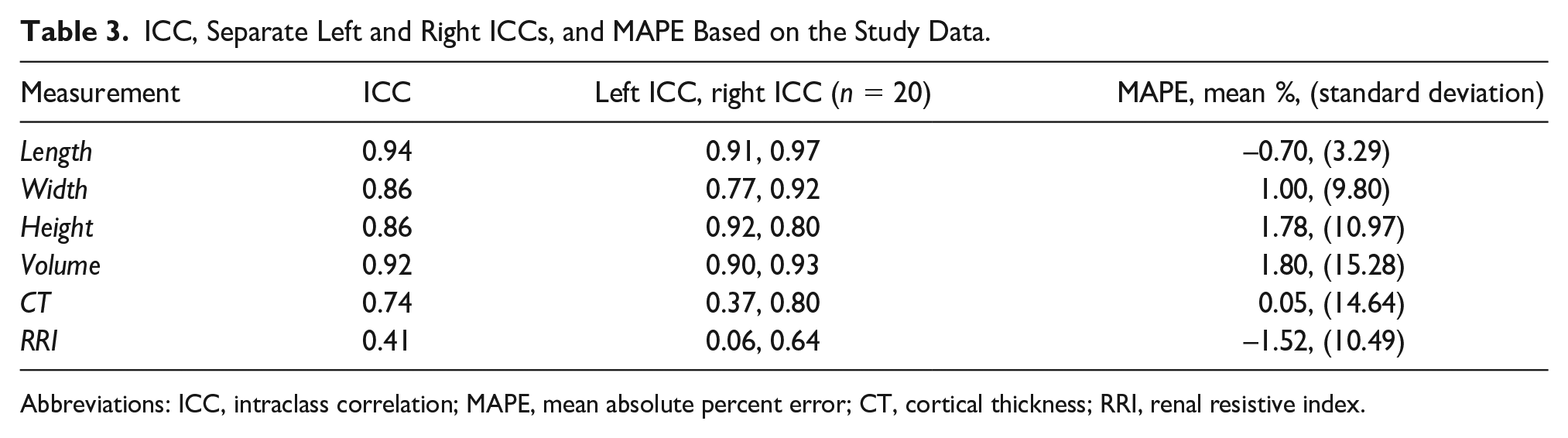
Abbreviations: ICC, intraclass correlation; MAPE, mean absolute percent error; CT, cortical thickness; RRI, renal resistive index.
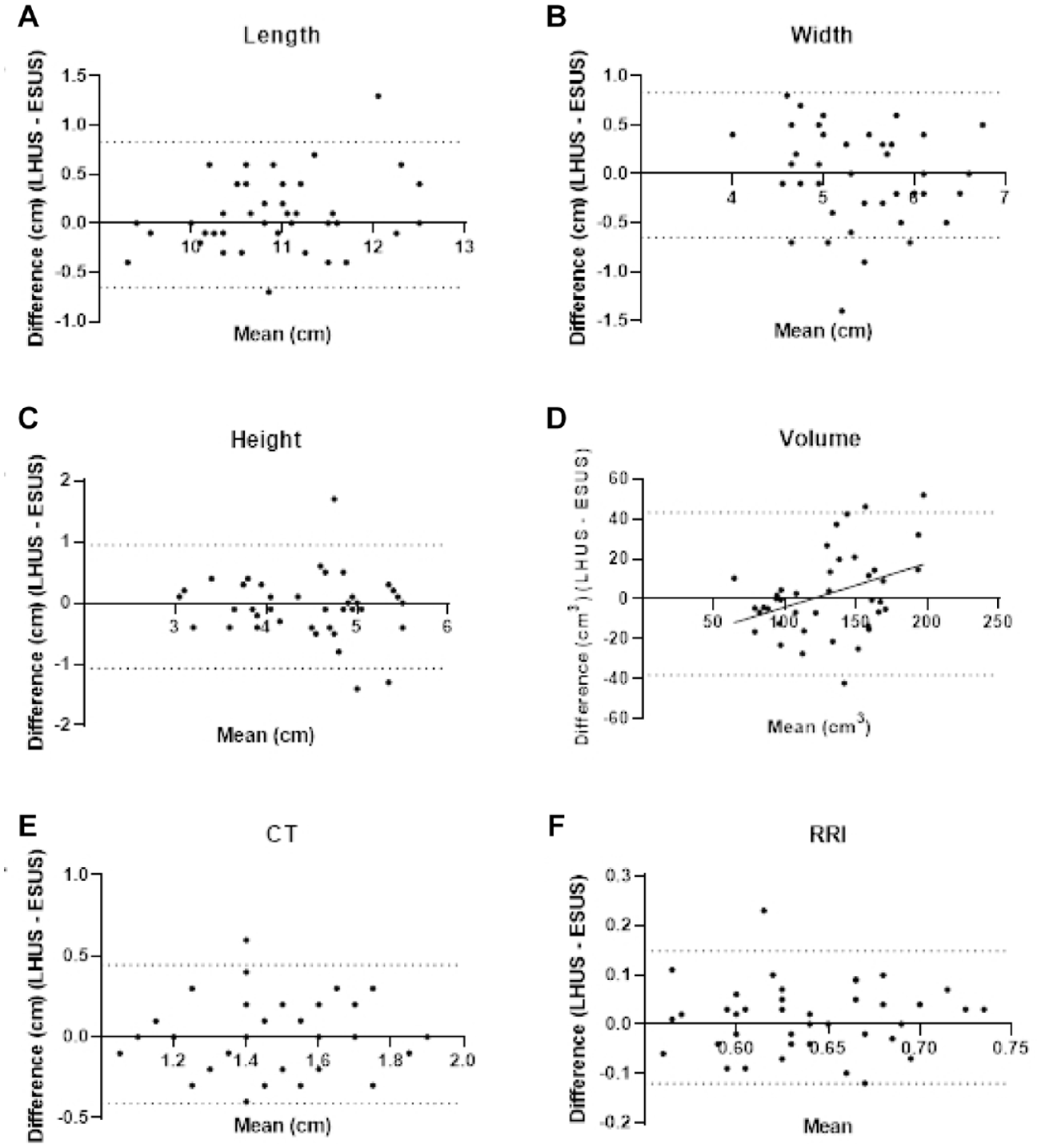
Bland Altman plots that compare assessment of (A) length, (B) width, (C) height, (D) volume, (E) CT, and (F) RRI between the LHUS and ESUS where the solid black line represents the average difference and the dotted black lines represent the limits of agreement per comparison. LHUS, Lumify hand-held ultrasound device, ESUS, Epiq Elite ultrasound device, CT, cortical thickness, RRI, renal resistive index.
Mean Difference and COV Between LHUS and ESUS for the Participants’ Left and Right Kidneys.

Abbreviations: COV, coefficient of variation; LHUS, Lumify hand-held ultrasound device; ESUS, Epiq Elite ultrasound device; CT, cortical thickness; RRI, renal resistive index.
Test-Retest Reliability
ICC revealed significant excellent agreement for length, volume, and CT (0.96, 0.93, and 0.90, respectively), good agreement for width and height (0.87 and 0.88, respectively) and moderate agreement for RRI (0.72) (all, p < .05). Mean COV for each measurement was low, ranging from 0.02 to 0.08. The LHUS was found to be reliable based upon an average ICC value of 0.87, and an average COV of 0.04.
Discussion
This study was designed to determine whether the LHUS is a valid and reliable tool to measure dimensions of the kidney, specifically, kidney length, height, width, volume, PE, and CT, in young healthy adults. However, practitioners should exercise caution when measuring RRI on the left kidney in young healthy adults due to the observed poor agreement that was documented with this ESUS.
The LHUS demonstrated moderate to strong agreement to the standard ESUS and was reliable in measuring basic dimensions and echogenicity of the kidney in a sample of young healthy adults. These findings are consistent with studies by Lavi et al, 11 and Barreiros et al, 12 which showed that their hand-held ultrasound device was accurate in its ability to measure basic dimensions of the kidney. Particularly, kidney length measured by their hand-held ultrasound was not significantly different and was strongly correlated with their standard ultrasound equipment system.11,12 However, one study showed contradictory results, where measurements of kidney’s length and width were significantly different from their standard ultrasound equipment system. 9 One possible reason for these conflicting results is that in the study by Stock et al, 9 their hand-held ultrasound device employed a 2–4 MHz, phased array transducer, whereas their standard ultrasound equipment system used a 2–6 MHz, curved array transducer. Curved transducers provide a larger field of view and have a low-frequency rate, 25 which allows these transducers to display full images of organs deep within the abdominal region. Conversely, phased array transducers have a narrower field of view with a high-frequency/frame rate, which allows it to view fast moving structures, such as the heart. 25 A previous study by Maar et al, 26 found significantly different measurements of renal cortical blood flow between different transducer types. Other previous studies have also demonstrated significant differences in measurements performed by different transducers in skeletal muscle 27 and the pneumothorax. 28 These previous studies demonstrate the importance of using the correct transducer for different tissue and organ types and the measurement type of interest. Lavi et al, 11 do not report which type of transducers were used in their study. However, Barreiros et al, 12 report that both their hand-held ultrasound and standard ultrasound equipment systems used the same type of transducers.
In the current study, the level of agreement for RRI between the LHUS and the ESUS was poor, based on the sample of young healthy adults. Separate ICC analyses for RRI of the left and right kidneys revealed that there was nonsignificant agreement in the left kidney, whereas the right kidney showed significant moderate agreement. In addition, the mean difference and COV between the LHUS and the ESUS was greater on the left kidney than the right. Such a discrepancy between the left and right ICC values led to a generally poor level of agreement for RRI. Previous studies have shown that the sonographic measurements on the left kidney can be less accurate than the right. A study by Ulusan et al, 29 found that the diagnostic accuracy of sonography to identify kidney stones was significantly lower in the left kidney than the right. Another study by Sasipattarapong et al, 30 reported that most diagnostic false positives for identifying hydronephrosis occurred in the left kidney. These findings suggest that diagnosing conditions in the left kidney may be more challenging due to its more superior position, which often requires a sonographer to use an intercostal window to capture an image. Moreover, the lack of an acoustic window and presence of bowel gas in the stomach near the left kidney can reflect the acoustic pressure waves, leading to difficulties in obtaining the best image quality. These challenges may have led to inaccurate or more variable RRI measurements of the left kidney, which in turn may have influenced the poor level of agreement between the LHUS and the ESUS. In fact, evaluation of morphological ICCs shows consistently lower agreement in the left kidney for four of the five variables. The left kidney is often a source of measurement error due to its shape, which differs from that of the right kidney. This issue is commonly linked to the dromedary hump, a noticeable bulge on the lateral border of the left kidney occurring in about 0.5% of cases. 31 This normal variation in the kidney’s contour is caused by the splenic impression on the superolateral part of the left kidney.31,32 Because of this unique anatomical feature, measuring the length and CT of the left kidney can be particularly challenging.
The LHUS uses Doppler to measure peak systolic and end-diastolic velocities of the renal artery and uses these values to calculate RRI. Previous studies investigating the use of hand-held ultrasound devices to measure RRI have produced mixed results. A study by Jin et al, 33 found excellent agreement between hand-held ultrasound devices and standard ultrasound equipment systems, when measuring carotid artery resistive index. Whereas a study by Trinquart et al, 34 demonstrated that hand-held ultrasound device had poor agreement compared with a reference standard ultrasound equipment system, when quantifying hepatic artery resistive index. In the same study, Trinquart et al, 34 also found no significant difference in the diagnostic accuracy of identifying normal and abnormal resistive index. So, although the objective measurements may have differed, the interpretation of the values was similar. The participants in the current study were all healthy with no kidney abnormalities. Thus, it cannot be concluded whether the LHUS has the same diagnostic ability to identify normal and abnormal RRI, as observed in the study by Trinquart et al. 34 A study by Lui et al, 35 identified two subjective human factors when measuring blood flow that might explain contradictory results in the literature. They found that inaccurate angle and placement of the transducer increased the variability of peak velocity measurements from 1%–2% to 4%–6%. In addition, both measurement error and variability were strongly dependent on the choice of Doppler gain level, where a more saturated gain showed 20% greater measurement error than the low and ideal gain chosen by the sonographers. It is possible that in the present study, the choices regarding angle of placement and gain level by the sonographer could have influenced the agreement between the LHUS and the ESUS.
In addition to subjective error, it is possible that the current study observed poor agreement for RRI because of the frequent trunk rotation of the participants during the data collection. The study design measured both the right and left kidneys, which prompted participants to rotate three times to lie down on both right and left sides. Previous literature has shown that posture change can influence renal blood flow. A study by Lin et al, 36 observed a significant influence of posture change on RRI when individuals rotated from the lateral decubitus position to the supine position. Changes in renal blood flow during posture change from the supine to upright position are also well documented.37 –39 In the current study, participants rotated left and right in the lateral decubitus position, but the effects of posture change while remaining in a lateral decubitus position have not previously been quantified.
Limitations
Major limitations due to the research design and the participant sample of convenience, result in threats to internal and external validity. In addition, the participants were young (mean = 21.5 years, range = 18–29 years) and healthy adults, which further effects the generalizability of these results to other populations. Aging can affect the morphology of the kidney,17,40 however, sonography seems to be unaffected, as it can be used to aid in the diagnosis of chronic kidney disease in older adults.41,42 It is unclear whether the validity of a hand-held ultrasound device, to take abdominal images in older adults, has been evaluated and warrants future investigation. Although the sample of participants included a wide range of BMI (18.7–30.3 kg/m2) and body fat percentage (5.7–38.2), most participants fell within healthy limits. The current study was not powered to investigate the effect of BMI and body composition on the accuracy and TRR of hand-held ultrasound. Previous studies have shown BMI and body fat percentage to be related to decreased sonographic quality.43 –45 After performing a post hoc analysis, no differences were found in agreement across BMI and body fat percentage. Future studies should investigate the TRR and validity of a hand-held ultrasound device to take abdominal images in individuals with high BMI and body fat percentage, as the image quality could potentially affect objective measurements.
Only one sonographer performed the sonographic measurements, and only one hand-held ultrasound device was evaluated. While using a single sonographer could introduce a risk of bias due to personal technique or preference, this approach was chosen to allow for a controlled evaluation of the specific ultrasound system, by minimizing variability introduced by differences in technique, experience, and preferences. This approach helped to isolate and specifically assess the performance characteristics of this LHUS. However, the absence of an interrater agreement evaluation means that the findings may not be broadly applicable to other systems. High interrater agreement and system comparison are crucial for ensuring quality in research and assessment. Therefore, future studies should involve multiple examiners and hand-held systems to analyze interrater agreement and compare different systems. Certainly, quality control evaluation of all transducers is critical to assure proper maintenance and performance.
Conclusion
In conclusion, this LHUS was found to be valid and reliable in measuring basic dimensions of the kidney in a sample of young healthy adults. Clinicians should exercise caution when performing RRI with an LHUS in young healthy adults, especially in the left kidney as the current study demonstrated nonsignificant agreement with a standard ESUS. Continued use of an LHUS and future studies should consider controlling for which side of the body, subjective errors, and trunk rotation when taking kidney hemodynamic measures.
This study’s implications for rural and remote settings are significant, as the validated use of the LHUS may enhance health care delivery in areas with limited medical infrastructure. By providing accurate kidney assessments, an LHUS may enable health care providers to identify renal issues early and initiate timely interventions. This is crucial in underserved populations, where traditional ultrasound equipment systems may be unavailable. Integrating an LHUS into clinical practice may help to bridge the health care gap, ensuring individuals receive the necessary diagnostic attention, regardless of their location.
Footnotes
Acknowledgements
Special thanks are extended to Ivinson Memorial Hospital for granting access to essential ultrasound equipment and dedicated space. The authors are also appreciative of the participants whose voluntary contributions played a crucial role in the successful execution of this study.
Ethics Approval
Ethical approval for this study was obtained from the Institutional Review Board (IRB #20220916KM03389).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Rocky Mountain Regional Chapter of the American College of Sports Medicine provided funding for this study.
