Abstract
Pelvic lymphoceles are collections of leaked lymphatic fluid which can be postsurgical complications. These are most prevalent following a radical prostatectomy with simultaneous pelvic node dissection to address prostate cancer. Although a common occurrence after this type of procedure, a small percentage may cause significant complications, including deep vein thrombosis. This case study demonstrated an obstructive pelvic lymphocele that was incidentally detected with sonography, during a lower extremity venous Doppler examination.
A pelvic lymphocele is a collection of lymphatic fluid that leaks from afferent lymphatic vessels. They can stay patent, after the surgical removal of pelvic lymph nodes, otherwise known as pelvic lymph node dissection (PLND), or pelvic lymphadenectomy. Pelvic lymph node dissection is a technique used in both urologic and gynecologic oncology procedures and is used to prevent and/or stage metastatic disease.1,2 This is critical, given that pelvic lymph nodes, connected to afferent lymphatic vessels, can be the first site for metastatic cells to migrate (sentinel lymph nodes). Lymphoceles, in the pelvis, predominantly collect adjacent to the external iliac vein and artery. 3 Lymphoceles can form in any anatomical location whenever lymph nodes or lymphatic tissue is resected and/or damaged. They can be found in the pelvis and retroperitoneum, as these are common areas for them to collect. 4 Although lymphoceles are the most frequent postsurgical complication, for a radical prostatectomy with PLND, only a small percentage of lymphoceles become symptomatic.1,3,5 The case report provided is of a symptomatic, obstructive pelvic lymphocele, which was discovered as the cause of the patient’s edematous right lower extremity, during a venous Doppler examination.
Case Report
A 71-year-old male presented for a bilateral lower extremity venous Doppler examination. This was based on a referral by his oncologic urologist for suspected deep venous thrombosis, as evidenced by moderate edema in the right lower extremity. The patient stated discomfort emanating from his right pelvic region. He had a robotically assisted radical prostatectomy for adenocarcinoma with concurrent bilateral PLND of the hypogastric, external iliac, and obturator fossa lymph nodes. The surgery had occured 17 days prior. The PLND yielded 12 right and 18 left pelvic lymph nodes, all of which were negative for metastatic disease. The Doppler study of the right common femoral vein (CFV) was performed with a 2-9 MHz linear-array transducer using a GE LOGIQ E10 ultrasound system (GE Healthcare Ultrasound, Waukesha, Wisconsin, USA). Coaptation throughout the entire right lower extremity was successful, with more force than usual being needed to collapse the deep veins. Normal phasic spectral Doppler waveforms and spontaneous color flow Doppler were unable to be obtained in the right lower extremity venous system. The Valsalva maneuver did not affect venous flow. Manual augmentation was the only way to fill the veins with color Doppler and demonstrate any spectral Doppler waveforms (see Figure 1). Since normal phasic venous flow with respiratory variations on spectral Doppler could not be obtained, especially in the right CFV, the sonographer suspected a proximal obstruction. Utilizing a 1-6 MHz curved-array transducer, the right CFV was followed superiorly to reveal a 5.9 × 7.9 × 6.7 cm avascular, septated cystic structure (see Figure 2A and B). This mass was compressing the right external iliac vein and obstructing venous return (see Figure 3). The cystic structure was diagnosed as a right pelvic lymphocele, a potential complication of a radical prostatectomy with PLND. The radiologist noted that the findings were indicative for impending deep venous thrombosis and the patient was immediately sent for a computed tomogram (CT) of the abdomen and pelvis, to confirm the findings on sonogram (see Figures 4A, B, and 5). Besides confirmation of the right pelvic lymphocele, an additional 3.4 × 1.8 cm left pelvic lymphocele was discovered on the CT examination but was deemed subclinical (see Figure 5). The patient was referred to interventional radiology for CT-guided percutaneous drainage of the right pelvic lymphocele. A subsequent lower extremity venous Doppler confirmed the return of normal phasic venous flow in the right lower extremity, with no evidence of deep venous thrombosis (DVT; see Figure 6). Owing to continued substantial lymphatic leakage, an intranodal lymphangiography, followed by lymphatic embolization, was performed 30 days after the prostatectomy with PLND.
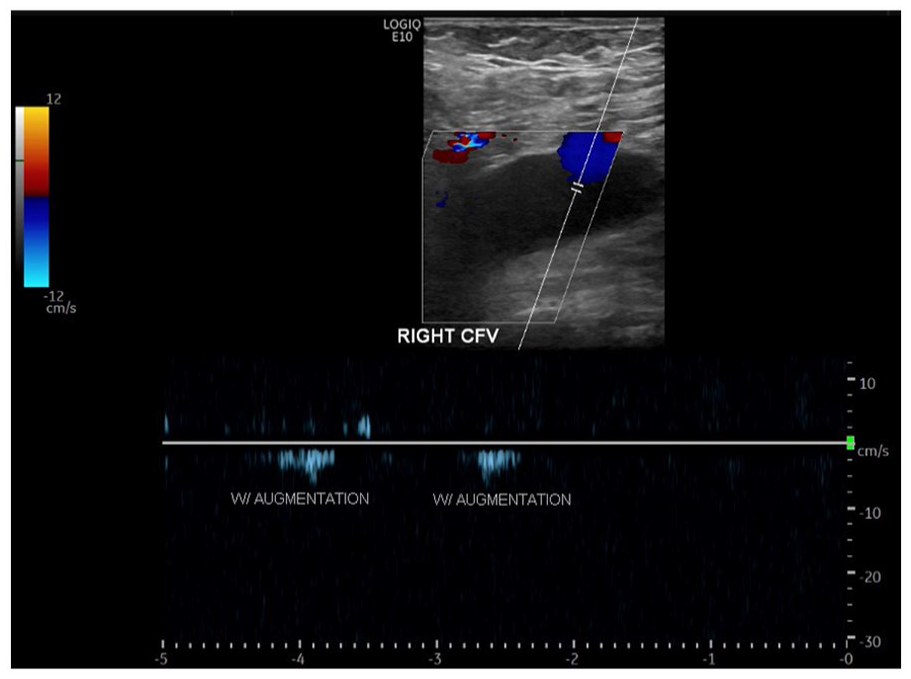
Spectral Doppler waveform of the right CFV demonstrates no spontaneous phasic flow, in addition to no color Doppler (except for greater saphenous vein inflow). The waveform shows minimal flow with consecutive augmentations. CFV, common femoral vein.
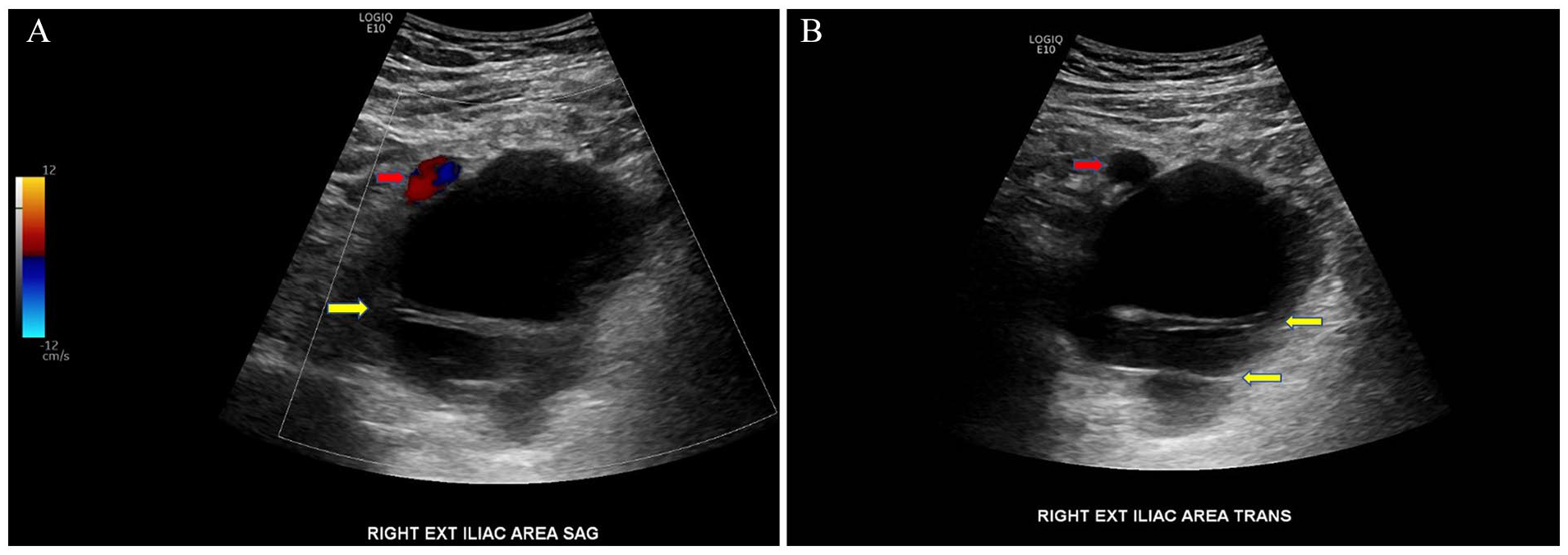
Sagittal (A) and transverse (B) sonographic views of an avascular, mostly anechoic, cystic pelvic lymphocele containing septations (yellow arrows) adjacent to the right external iliac artery (red arrows).
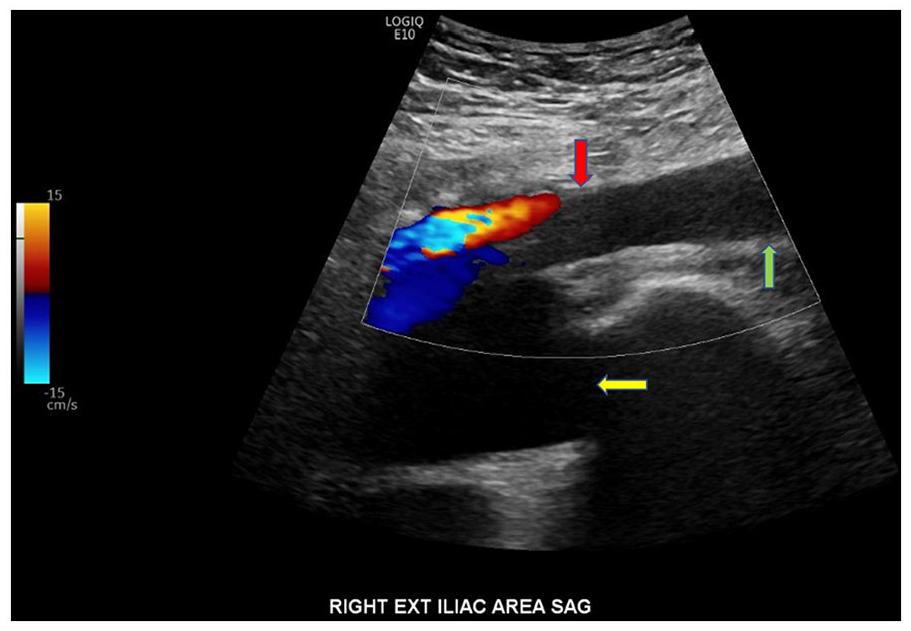
A sagittal sonographic view of the pelvic lymphocele (yellow arrow) narrowing the right external iliac vein, obstructing venous return. Notice the lack of color Doppler in the right external iliac vein (red arrow) and superior portion of the right CFV (green arrow) inferior to the lymphocele. CFV, common femoral vein.
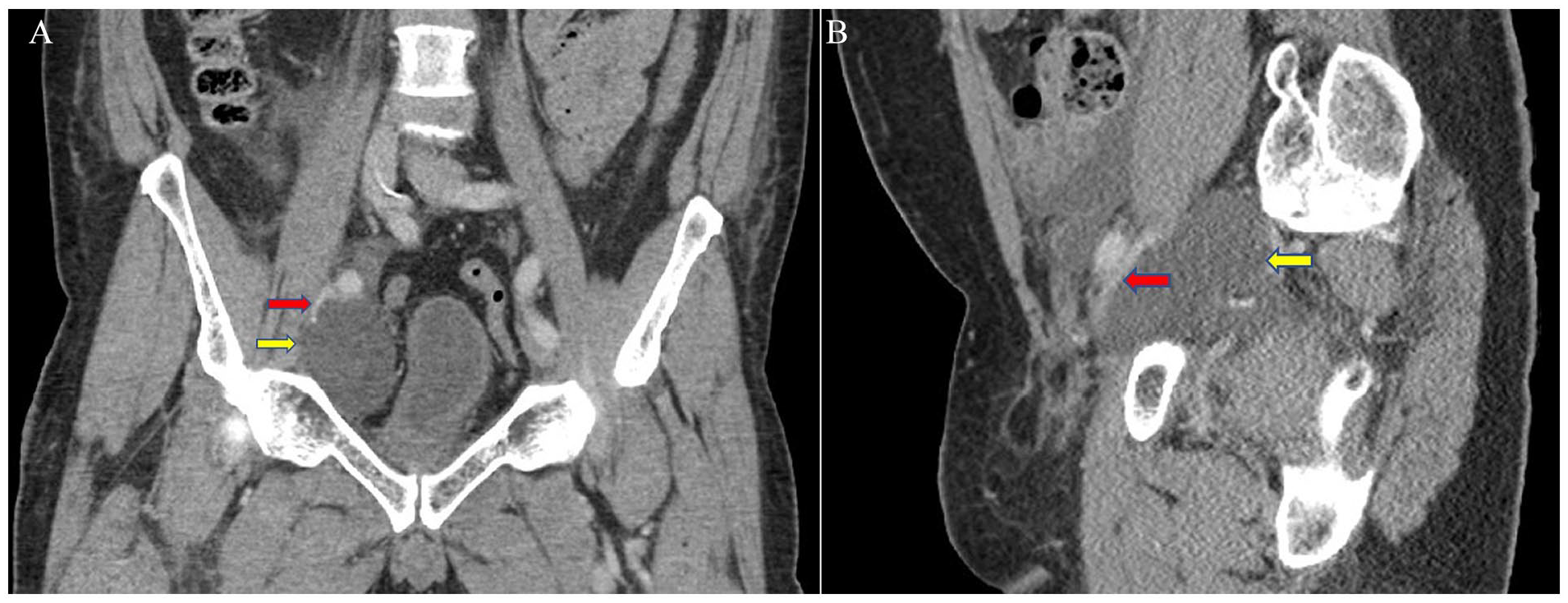
Coronal (A) and sagittal (B) CT images demonstrates the right pelvic lymphocele (yellow arrows) causing significant narrowing of the right external iliac vein (red arrows) CT, computed tomogram.
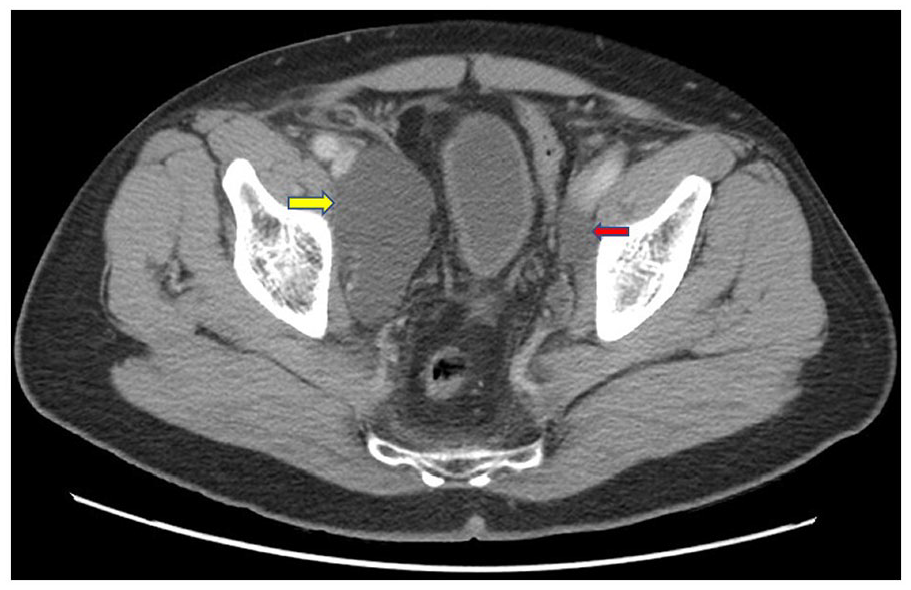
Axial CT image demonstrates the large right pelvic lymphocele (yellow arrow) adjacent to the right external iliac vessels. A small left pelvic lymphocele (red arrow) is partially imaged. CT, computed tomogram.
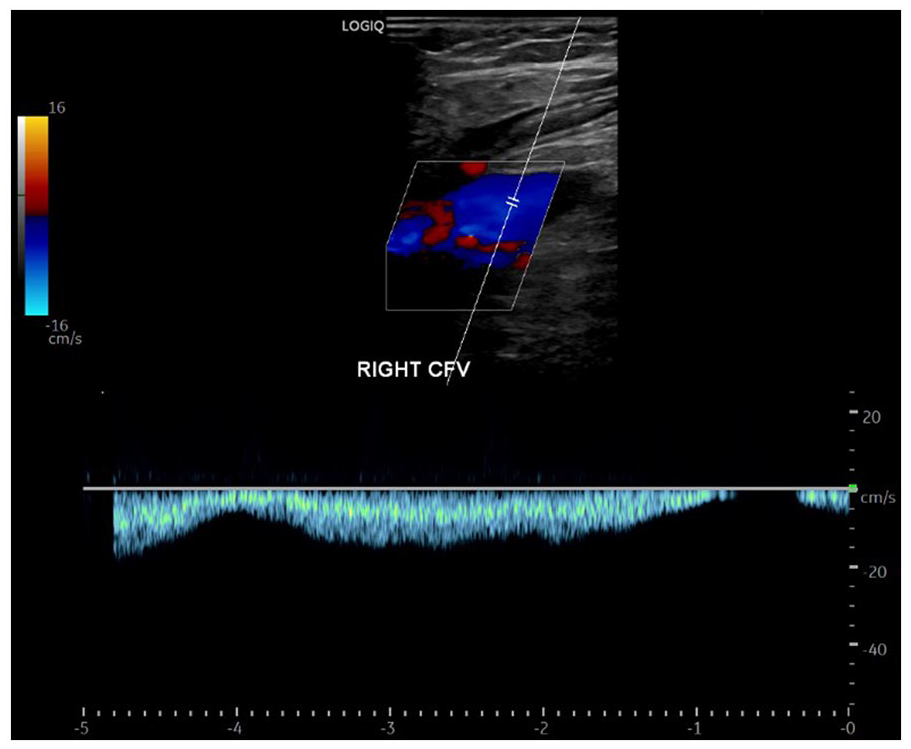
Triplex Doppler image that was part of the lower extremity venous Doppler examination performed after the right pelvic lymphocele percutaneous drainage. The spectral Doppler waveform demonstrates return of normal phasic respiratory variations in the right CFV. CFV, common femoral vein.
Discussion
Pelvic lymphoceles are the most common postoperative complication after radical prostatectomies with PLND, but only a small percentage lead to symptoms and complications. They have even been found in cases of radical prostatectomies without PLND, 6 as well as renal transplantation.4,7 Radical hysterectomies, along with PLND, are performed to mitigate and/or stage metastatic disease from endometrial and cervical cancers and similar rates of pelvic lymphoceles can occur, including those that are symptomatic. 7 A precise occurrence of pelvic lymphoceles, after PLND, cannot be determined since most occur without symptoms, subsequently go undiagnosed, and eventually regress without intervention.8,9 Moreover, pelvic lymphoceles often go undetected because post-surgical imaging is not routinely done 8 unless warranted by signs and symptoms of possible complications. This has resulted in a wide discrepancy in the reported incidence of pelvic lymphoceles after prostatectomies, with PLND, with research evidence ranging from 9% 5 to 61%. 10 Similarly, there is also a discrepancy in reported symptomatic pelvic lymphoceles: ranging from 2.3% 10 to 7.4% 3 based on the total number of post-operative patients, or 4.2% to 59.1% of patients with lymphoceles, respectively. Complications from symptomatic pelvic lymphoceles, which includes DVT, have been reported as soon as 3 weeks 11 post procedure and up to 6 months 5 after prostatectomy with PLND.
Symptoms occur from extrinsic compression by the lymphocele to nearby anatomy. Lower extremity edema occurs due to a proximal venous obstruction and congestion, which may lead to DVT from stagnant blood flow (stasis is one component of Virchow’s triad). Venous congestion is caused by increased pressure in the lower extremity venous system due to the inability of blood to drain proximally from the leg. This is what makes coaptation more difficult than usual during a venous Doppler examination.
Beyer et al. 11 found that 16.8% of patients in their cohort developed a DVT (majority were calf muscle thrombosis) and/or a pulmonary embolism (PE), within 3 weeks following radical prostatectomies with lymphadectomies. Every case with diminished pelvic venous flow was found to have a concurrent ipsilateral pelvic lymphocele, with 34.2% of these cases developing DVT and/or PE. This demonstrates the importance of sonographers paying keen attention to the phasicity of the CFV spectral Doppler waveform. This can likely be the first indication that there may be a more proximal venous obstruction. Minimal flow or a continuous, flattened waveform without respiratory variations in the CFV is indicative of a proximal venous obstruction.
Pelvic and/or lower extremity pain may occur due to compressed pelvic nerves. Unresolved symptomatic pelvic lymphoceles can also cause nerve injury. Urinary frequency, hydronephrosis, and potential renal failure 12 can result from compression of the urinary bladder and/or ureters. Constipation6,9 and/or bloating 13 can be caused by compression of the rectosigmoid colon. 9 Pelvic lymphoceles may also become infected, potentially leading to sepsis, a serious and potentially life-threatening postoperative complication.
Weinberger et al. 14 identified specific sonographic features of pelvic and retroperitoneal lymphoceles that were found to favor either symptomatic or asymptomatic lymphoceles after pelvic and/or paraaortic lymph node dissections for gynecological cancers. An oval/ovoid shape (found 47.2% of the time), anechoic or low-level echoes (72.7% and 59.2% respectively), septations (80.0%) and debris (79.2%) were all features more often found in symptomatic lymphoceles. Conversely, a round shape (seen 82.7% of the time) and hyperechoic (what Weinberger et al. described as a ground-glass appearance) (90.8%) were overwhelmingly found in asymptomatic lymphoceles. This case study supports the results of Weinberger et al., as the symptomatic lymphocele was oval, predominantly anechoic, and contained septations (see Figure 2A and B). Interestingly, Weinberger et al. believed that the potential reason oval lymphoceles tended to become symptomatic, as opposed to round ones, was because they took up more surface area in the body, potentially causing more compression on the surrounding anatomy. 14
The differential diagnoses for lymphoceles are other types of post-surgical collections of fluid, such as urinomas, seromas, hematomas, and abscesses. Urinomas are postoperative pockets of urine and have a similar sonographic appearance to lymphoceles, as they are avascular and either simple or septated. Anatomically, they can be found in both the pelvis and retroperitoneum, as are lymphoceles. A CT with a delayed postcontrast phase, which shows urine leakage, is needed for proper diagnosis. A seroma is a complex accumulation of serous fluid that may contain solid-appearing avascular echoes, which are, in fact, hematomas. They often resolve without incident. Abscesses are infected postsurgical fluid collections with a mixed echogenicity. The distinguishing characteristic of an abscess on sonography is the considerable amount of color Doppler, in and around the capsule. 14
Prevention or mitigation of pelvic lymphoceles is key to reducing the risk of complications. This can be accomplished in several ways. The first is to possibly avoid the need for PLND altogether. Pelvic lymph node dissection may not be necessary in patients who are in a low-risk category based upon set preoperative criteria such as prostate-specific antigen (PSA) level, Gleason score, and tumor node metastasis (TNM) stage. In these patients, the risk for lymph node metastasis is no greater than 1%. 15 Ultimately, both surgeon and patient need to weigh the benefits and risks of this decision. Surgeons can also minimize postoperative pelvic lymphoceles and their respective complications by carefully clipping/cauterizing all lymphatic vessels in or around the PLND site. This may minimize the number of patent lymphatic vessels and decrease the potential for leakage and lymphocele development in the pelvic cavity. 9
Interventional techniques used to treat symptomatic pelvic lymphoceles include percutaneous catheter drainage with or without sclerotherapy, lymphatic embolization, and laparoscopic or open-drainage/marsupialization. There is a significant incidence of recurrent lymphoceles after percutaneous drainage.2,3 Therefore, sclerotherapy can be utilized, which is the injection of a sealing agent (sclerosant) through the indwelling catheter to close the lymphocele pocket. However, sclerotherapy may require more than one attempt and does not always seal the entire lymphatic leak. An alternative, two-step initial intervention has been used with more success: intranodal lymphangiography followed by lymphatic embolization. This procedure is indicated if the daily drainage from the percutaneous catheter is greater than 50 mL. Lymphangiography involves the localization of the lymphatic leak, using fluoroscopy. Subsequently, the adjacent upstream lymph node/lymphatic vessel, causing the leak, is embolized with an injection of surgical glue. 16
There is conflicting research as to whether pelvic lymphoceles occur at the same rate during open and laparoscopic/robotic PLND, with concurrent prostatectomies. Zorn et al. 17 found that pelvic lymphoceles occurred at the same rate, regardless of surgical method, while Solberg et al. 10 found a higher incidence (61%) in the open surgical approach versus laparoscopic PLND (37%). Orvieto et al., 1 Keskin et al., 5 Heers et al., 8 and Gust et al. 12 investigated potential links between patient factors such as age, body mass index, prostate volume/weight, number of lymph nodes excised, and length of time in surgery as increased risk factors for lymphocele formation after prostatectomy with PLND. Unfortunately, they were not able to find a connection. However, Naselli et al. 3 found that the number of lymph nodes removed had a high correlation with symptomatic lymphocele formation postoperatively. Similarly, Capitanio et al. 18 found that males above age 65, as well as patients who had more than 20 lymph nodes removed during PLND had an increased risk for symptomatic pelvic lymphoceles. In addition, the authors found that the risk for symptomatic pelvic lymphoceles increased 5% with every additional year of age and for every additional lymph node removed. Although Orvieto et al. 1 were unable to predict which patients would develop a lymphocele preoperatively, they found postoperatively that those patients who had metastatic lymph nodes, a higher volume of cancer in the prostate gland, and extracapsular extension in the specimen, were more likely to develop pelvic lymphoceles. Since there are conflicting data, it is not known which patients are more at risk for developing pelvic lymphoceles.
There is some discrepancy between research in overall pelvic lymphocele incidence. This may be due to research studies using different imaging techniques, for pelvic lymphocele detection. 1 Naselli et al., 3 Keskin et al., 5 Heers et al., 8 and Gust et al. 12 used sonography, whereas Orvieto et al. 1 and Solberg et al. 10 performed only CT, postoperatively. Keskin et al. 5 expressed their advocacy for using sonography in the detection of asymptomatic pelvic lymphoceles but performed CT and/or lower extremity venous Doppler on symptomatic patients, as did Naselli et al. 3 Although radiation free and cost-effective, as opposed to CT, sonography has its limitations. The quality of sonography is heavily dependent on the skills of the operator. In addition, sonography may yield falsely low sensitivity in research studies if the patient has a challenging body habitus due to obesity or bowel gas.
Conclusion
Although a small percentage of pelvic lymphoceles become symptomatic, it is imperative that both patients and sonographers are aware of the potential serious complications. Upon discharge from the hospital or surgical facility, patients need to be educated on potential complications resulting from pelvic lymphoceles, such as DVT, PE, and infection. More specifically, patients need to be vigilant regarding the various signs and symptoms of DVT (lower extremity edema and/or leg pain) and PE (difficulty breathing and/or chest pain). Medical attention should be sought immediately, as most cases of DVT and PE have been reported to occur within 8 to 21 days after radical prostatectomies, with PLND. 11 It is also imperative that the sonographer is aware of the patient’s surgical history so that an ipsilateral pelvic lymphocele, or any pelvic pathology that can significantly obstruct the deep venous system, is not overlooked. This can be a potential cause for the lack of spontaneous phasic venous flow and/or unusual difficulty coapting the lower extremity deep veins, as demonstrated in this case study.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval was not sought for the present study since only anonymized patient information was utilized, and the presented information was researched and developed to be shared for medical and/or educational purposes.
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
