Abstract
A 53-year-old male was admitted to the hospital because of retrosternal and epigastric pain radiating to the back. Radiologic images showed a homogeneous soft tissue mass in the right upper lobe that was initially misdiagnosed as a lung cancer. Fortunately, a careful Doppler examination identified two vascular structures within the lesion. This diagnostic study suggested the execution of a more appropriate computed tomography angiography (CTA). The examination confirmed the diagnosis of a simple pulmonary arteriovenous malformation (AVM). The patient underwent embolization of the arterial side of the AVM and finally a posterolateral mini-thoracotomy to remove the lesion.
Keywords
Pulmonary arteriovenous malformations (AVMs) are abnormal communications between the arterial and venous circulation, resulting in right-to-left shunting. These malformations can be classified into simple and complex types. Simple pulmonary AVMs are supplied by a single feeding artery, while complex pulmonary AVMs comprise two or more feeding arteries. 1 Dyspnea, cyanosis, cerebrovascular events, and brain abscess are common clinical manifestations, although patients may be completely asymptomatic. On radiologic images, the dilated vessels may appear as a non-specific soft tissue mass in a relatively unusual orientation compared with adjacent vessels. 1 In such cases, this rare disorder may enter into the differential diagnosis with pulmonary nodules or masses. Computed tomography angiography (CTA) is the gold standard investigation for diagnosis, as it can imagine the abnormal vessels and further delineate the distribution and extent of the disease.1,2 Here, it is presented as an atypical case of an adult patient with a pulmonary AVM that has been initially misdiagnosed as a lung cancer. The aim of this case study is to warn clinicians to follow the complete diagnostic pathway with a careful observation of all the imaging findings, to avoid potentially fatal errors.
Case Report
A 53-year-old male was admitted to the hospital with complaint of retrosternal and epigastric pain radiating to back. He was virtually asymptotic, including dyspnea at rest. The patient’s clinical history revealed only a smoking habit and no past diseases to be deemed worthy of note. A chest radiograph of about 7 years ago showed no areas of consolidation, lobar collapse, or pulmonary masses/nodules. The physical examination yielded no significant cardiovascular, respiratory, or abdominal findings.
The patient had a new chest radiograph which showed a large opacity of 9–9.5 cm at the posterior segment of the upper lobe of the right lung (see Figure 1A and B). A chest computed tomogram (CT) confirmed the presence of a voluminous mass located in the right upper lobe and extending in the homolateral lower lobe (see Figure 1C). As the diagnosis of a lung cancer was highly suspicious, a positron emission tomography/CT (PET/CT) examination with fluoro-18-deoxy-glucose (18-FDG) and a whole-body contrast-enhanced computed tomogram (CECT) were scheduled, for staging purposes. The PET/CT examination was performed first, which documented an inhomogeneous accumulation of 18-FDG in the pulmonary mass with an extensive eccentric photopenic area (referred as a probable area of colliquation). No other areas of radiotracer uptake were highlighted on the examination (see Figure 1D).
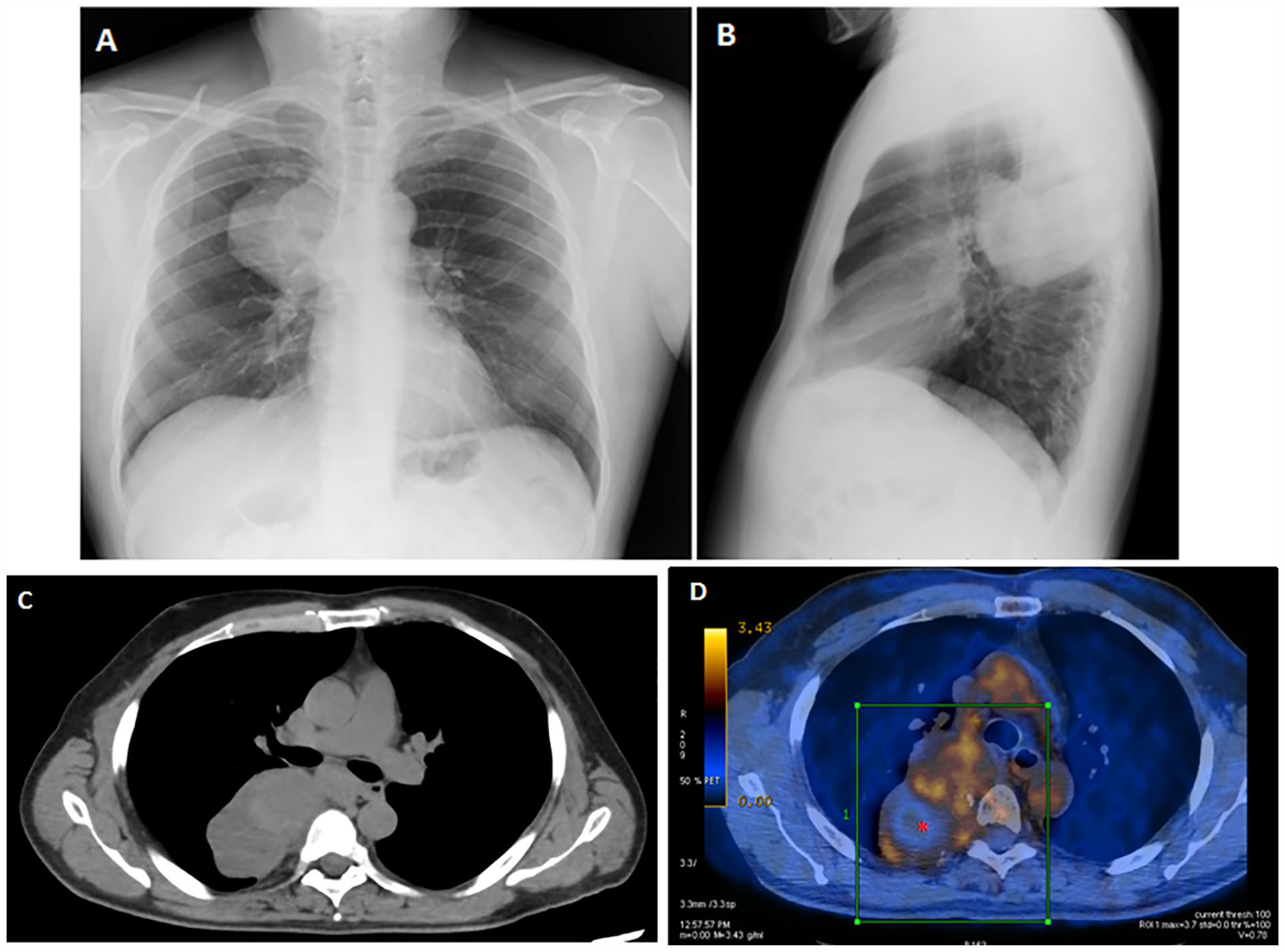
The posteroanterior (A) and lateral (B) chest radiographic views demonstrated a homogeneous soft tissue mass of 9–9.5 cm in the posterior segment of the upper lobe of the right lung. (C) The axial CT examination showed a homogeneous, well-circumscribed, non-calcified nodule of 9.5 × 5.5 cm in the right upper extending in the homolateral lower lobe. (D) A PET/CT examination showed an inhomogeneous accumulation of FDG in the pulmonary mass located between the right upper right lobe and the homolateral lower lobe (green square) and an extensive eccentric area of no radiotracer uptake (red asterisk). CT, computed tomography; FDG, fluor-18-deoxy-glucose; PET/CT, positron emission tomography/computed tomography.
While waiting for whole-body CECT examination, the patient was referred to the interventional sonography unit for a transthoracic ultrasonogram (TUS). The purpose of the study was to assess the presence of a satisfactory sonographic view to eventually plan a minimally invasive ultrasound-guided percutaneous needle biopsy (US-PNB). The examination was performed with an Esaote MyLab-9 scanner (Esaote-Biomedica, Genoa, Italy) using a convex multi-frequency (3–8 MHz) transducer. The ultrasound equipment system was properly set for the study of the adult thorax, as follows: depth varying between 70 and 140 mm, time gain compensation (TGC) of no more than 55%, focus pointed at the lesion, and activation of the tissue harmonic imaging. The patient was examined in a sitting position. The real-time grayscale image (TUS B-mode) demonstrated a hypo-anechoic subpleural lesion with corpuscular spots within, as well as a fluid-like pulsatile motion (see Figure 2A–C). The color Doppler interrogation clearly detected a mosaic-like vascular structure. Spectral wave analysis revealed a low resistive flow (see Figure 2D). Based on the TUS findings, a more appropriate CTA was performed (instead of the scheduled whole-body CECT). The CTA examination confirmed the diagnosis of a pulmonary AVM, which measured 9.5 × 5.5 cm. The lesion consisted of an abnormal connection between an aberrant bronchial arterial branch emerging from the aorta at the passage between D6 and D7, and the azygos vein. Markedly, ectatic vessels and an unenhanced area of probable thrombotic nature were clearly depicted within the mass (see Figure 2E). A cranial CT set of images showed no focal lesions or areas of pathological enhancement.
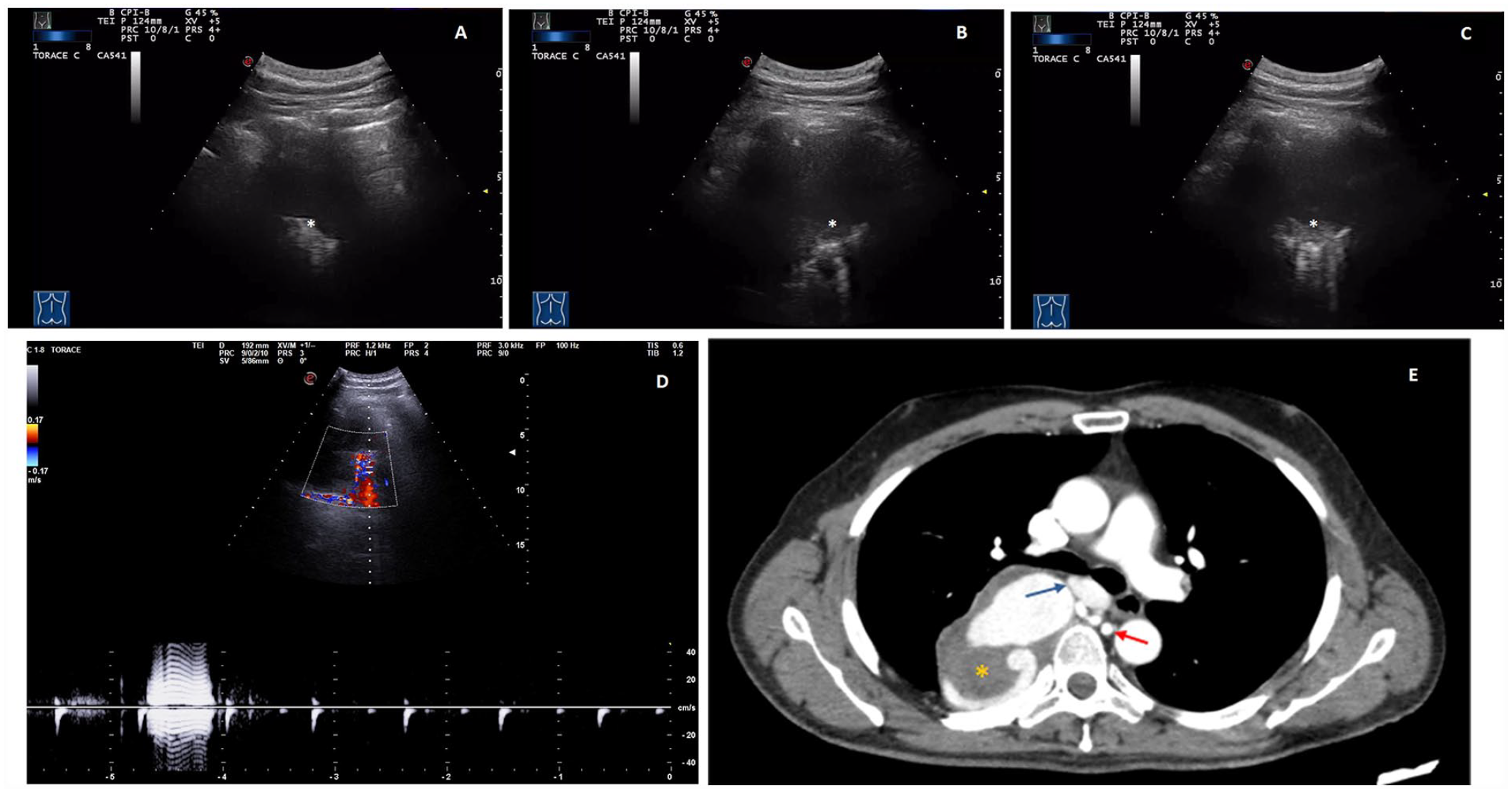
(A, B, C) A TUS showed a hypo-anechoic subpleural lesion with corpuscular spots within it (white asterisk) showing a fluid-like pulsatile motion. (D) The Doppler interrogation clearly demonstrated a mosaic-like vascular structure. The spectral wave analysis revealed a low resistive flow. (E) A CTA on early-phase sequences, demonstrated enhancement of the feeding artery arising from the descending aorta (red arrow), the aneurysmal part, the draining flow in the azygos vein (blue arrow), and an eccentric unenhanced area (yellow asterisk). CTA, computed tomography angiography; TUS, transthoracic ultrasonogram.
After a multidisciplinary consultation and agreement with the patient, a two-step treatment was planned, which consisted of an endovascular treatment for embolization of the arterial side of the malformation, followed by elective surgery for removal. Under general anesthesia and after heparinization, a vascular catheter was inserted through the right femoral vein and a pulmonary angiogram was obtained. The embolization of the arterial side of the AVM was performed through the introduction of two coil-type devices measuring 6 mm × 50 cm and 6 mm × 30 cm, respectively (Figure 3A–C). As the right single-access on video thoracoscopy showed that the mass extended to the mediastinum, the patient underwent a posterolateral mini-thoracotomy to remove the lesion. After isolation of the azygos vein, the mass was dissected from the chest wall and pedunculated on the afferent vessel already subjected to embolization. No patient complications were experienced following the procedure (see Figure 3D).
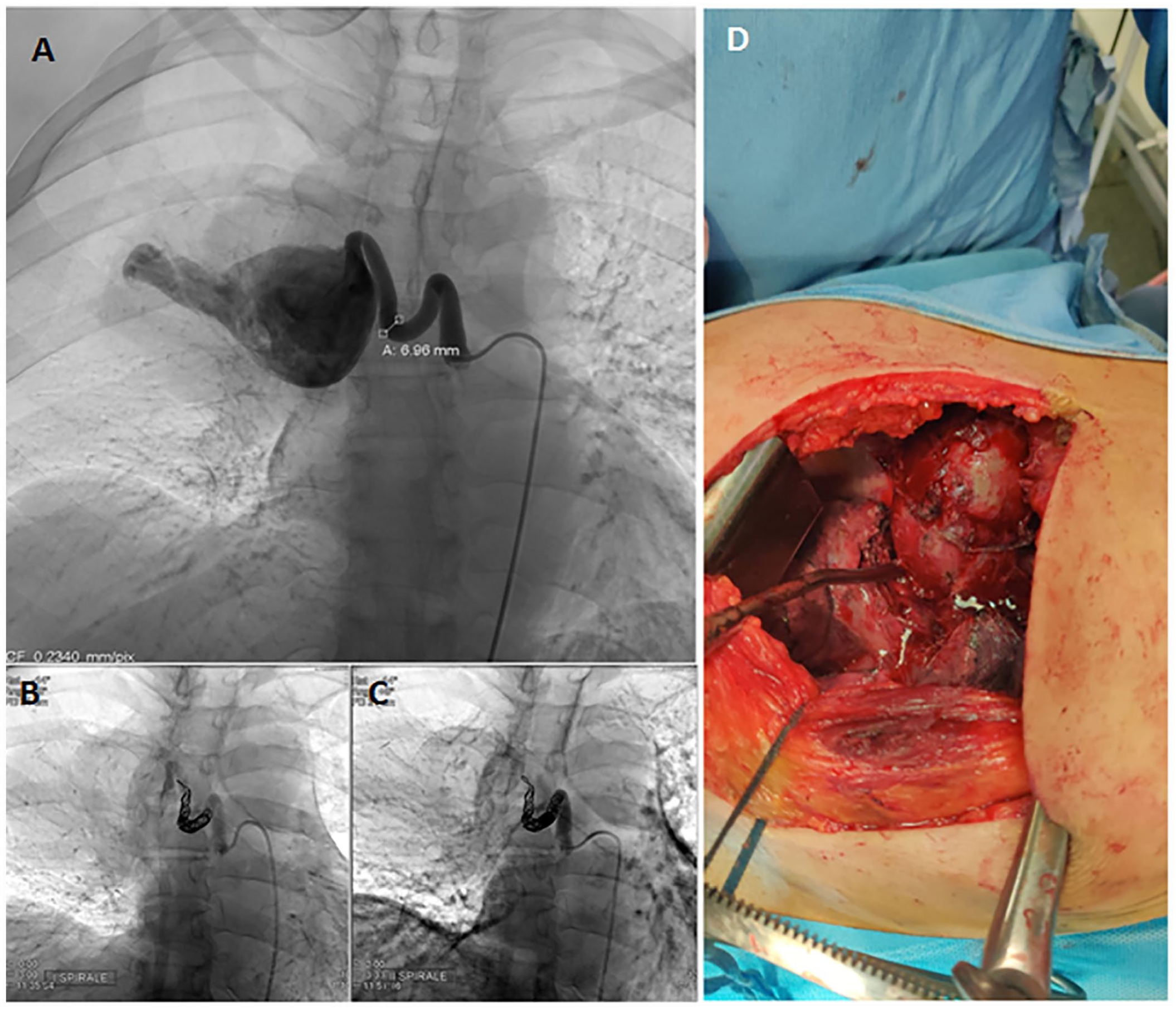
Image from a transcatheter pulmonary angiogram demonstrated pre-embolization images (A) and results after the placement of the first (B) and the second (C) coil-type devices. (D) A posterolateral mini-thoracotomy was completed with access through the fifth intercostal space, to remove the lesion.
Discussion
Pulmonary AVMs are rare vascular anomalies of the lung, with an estimated prevalence of approximately 38 per 100 000 individuals. 2 A female predilection has been recognized. Hereditary hemorrhagic telangiectasia (HHT), also known as Osler-Weber-Rendu syndrome, accounts for about 70% of pulmonary AVMs, while other cases are sporadic. 2 The pathological communication between the venous and arterial system can lead to hypoxemia and subsequent dyspnea (due to right-to-left shunting). Hemoptysis and hemothorax may also occur, but the most common complications are cerebrovascular events, including transient ischemia attacks, stroke, or brain abscess (due to paradoxical emboli). Although it is assumed that the vascular defects are present at birth, pulmonary AVMs may remain asymptomatic until adult life. A suggested explanation for symptomatic presentation in adult age is the gradual enlargement of the AVM as a response to increasing flow culminating in necrosis of the vessel wall and increased shunting fraction over aging. 3
The patient involved in this clinical case study did not present with any characteristic symptoms or signs (e.g., finger clubbing, cyanosis, and chest murmur), which would have allowed for the suspicion of a pulmonary AVM. Moreover, a diagnosis of HHT is clinically based on the presence of three of four of the Curaçao criteria, including the following: (1) recurrent spontaneous epistaxis, (2) multiple mucocutaneous telangiectasias, characteristically, including oral cavity, lips, fingers, and nose, (3) visceral AVMs, and (4) first degree relative with HHT. 4 Despite no genetic studies were done to rule out HHT, the patient did not clearly fulfill the Curaçao criteria.
Dilated vessel may appear as a non-specific soft tissue mass on standard chest radiograph and CT examination. 1 In a middle-aged smoker male who had a right upper lobe lung mass on recent radiologic images, but no pulmonary findings on prior chest radiographs, the most likely diagnosis was thought to be lung cancer. 5 The following PET/CT examination, showed a pathological radiotracer uptake in the lesion, further contributed to strengthen the erroneous hypothesis of malignancy.
TUS should be regarded as a useful non-invasive imaging method in the assessment and surveillance of several pleural and pulmonary conditions, providing complementary information to other radiologic imaging techniques. It allows the real-time examination of peripheral pulmonary lesions adhering to the parietal pleura, when not obscured by the bony structures of the rib cage. The use of color Doppler can simultaneously display blood flow information within the lesion. In addition, TUS is a safe and effective procedure for the guide of PNB of US-accessible peripheral lung consolidations.
A complete TUS should include the scanning of each hemithorax in the anterior, lateral, and posterior lung zones. Both transverse and longitudinal scans are important to avoid missing any alterations. A TUS should be ideally performed with the patient in a sitting position, as this allows all lung fields to be explored. In patients who are bedridden, the anterior chest wall may be examined in the supine position, while it will be necessary to modify the position into the lateral decubitus for the examination of the lateral and posterior chest wall. In the first instance, the examination should be conducted using convex probes with ranging frequencies from 3 to 5 MHz, as they allow for an adequate transducer depth which should range from 70 to 140 mm. High-frequency linear array transducers (8–12.5 MHz) may be employed to detail the pleural line, as they provide excellent resolution at short working distances. The TGC should not exceed 55% and the use of tissue harmonic imaging is preferable, to reduce artifacts.
Pulmonary AVMs can be imaged with TUS because they are mostly immediately subpleural or only partially embedded in the parenchyma. They may appear as nonspecific hypo-anechoic subpleural lesions on a B-mode grayscale image. Fortunately, close observation of the real-time TUS images allowed a skilled operator to locate corpuscular spots within the lesion that exhibited a fluid-like pulsatile motion. The subsequent color Doppler study clearly suggested a mosaic-like vascular structure. Based on these findings, the subsequent diagnostic workup was led toward the execution of a more appropriate CTA. 2 The intravenous injection of contrast agent on early-phase CT sequences allowed the diagnosis of a simple form of AVM consisting in a single feeding artery, an aneurismal part, and a draining vein. As pulmonary AVMs with a feeding artery diameter of > 3 mm are strongly associated with various neurologic manifestations, such as infarction, abscess, and seizure, a definitive treatment was highly indicated. 6 In general, the most radical and least invasive method of treatment should be preferred and embolization is recommended as first-line treatment. 7 However, the endovascular treatment alone is usually inadequate to achieve the complete closure in a large and centrally located pulmonary AVM. 8 As a result, the endovascular embolization of the arterial side of the AVM was performed as first step to stabilize the lesion and avoid complications. In addition, a minimally invasive video-assisted thoracic surgery (VATS) for removal of the lesion was planned. However, the extent of the lesion made it necessary to convert the VATS surgery into a lateral mini-thoracotomy. Peri- and post-operative courses were uneventful.
Conclusion
Pulmonary AVMs are relatively rare disorders that may enter in the differential diagnosis of pulmonary nodules. Despite epidemiologic data, including sex, age, smoking habits, signs and symptoms, and lesion location are frequently useful in orienting a specific diagnosis, this case demonstrated that the most probable diagnosis is not always correct. In fact, a biopsy of the pulmonary AVM could have resulted in a fatal error for the patient causing massive bleeding, if the correct diagnosis had not been made. A full investigation with careful observation of the findings is of immense importance for reaching a final definitive diagnosis and avoiding at the same time potentially fatal errors.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval was not sought for the present study because all data analysed were collected as part of routine diagnosis and treatment.
Informed Consent
The patient provided informed permission for publication of images and other clinical information relating to the case. Anyhow, all the images have been anonymized to protect patient’ privacy. As all the examinations and procedures described in this case report and for which the patient signed a written informed consent are part of our routine medical practice, the ethical approval by a Research Ethics Committee was deemed unnecessary.
