Abstract
Objective:
Myofascial trigger points (MTrPs) are pathognomonic of myofascial pain syndrome. The detection ability of MTrPs via ultrasonography is underreported and the characteristics of MTrPs are not sufficiently standardized. The objective was to summarize the characteristics and diagnostic abilities of ultrasonography for MTrP investigations.
Materials and Methods:
A multi-database, and bibliography hand-search was implemented. Studies of ≥10 patients, published after 1980, appraising ultrasonography as a diagnostic aid for myofascial pain syndrome were included. The Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) tool was used to evaluate the diagnostic accuracy of the included studies.
Results:
Six cross-sectional studies met the inclusion criteria. The back and trapezius muscles were the most studied locations. The diagnostic studies were of low-medium risk of bias. The studies reported a large range of diagnostic metrics (accuracy 58%–100%, sensitivity 33%–91%, specificity 75%–100%, positive predictive value 91%–100%, negative predictive value 47%–97%, positive likelihood ratio 3.6, and negative likelihood ratio 0.12–0.67).
Conclusion:
This review found low-medium risk of bias evidence in support of ultrasonography for MTrP investigations. The clinical studies identified in the scoping review used gray-scale ultrasound equipment systems with a 5 to 14 MHz transducer to diagnose MTrPs and the local twitch response, and MTrPs were visualized mostly as hypoechoic nodules.
Myofascial pain syndrome is a localized condition affecting up to 85% of the population1,2 and is characterized by pain in specific muscle fibers or muscle fiber groups.3–5 Myofascial pain can hinder quality of life due to associated comorbidities such as sleep disturbances and depression.6–9 In addition, the chronic nature of myofascial pain syndrome predisposes afflicted patients to maladaptive emotional responses such as catastrophizing and rumination of existing pain and fear of physical movement. 10 The aforementioned muscle fiber pain can be attributed mainly to localized contractions called myofascial trigger points (MTrPs), which are pathognomonic of myofascial pain syndrome.11–13
MTrPs are thought to develop from etiological factors and muscle overload, causing a localized self-sustaining muscular fiber contraction. 10 Clinically, MTrPs are 2 to 5 mm hyper-irritated hard nodules in subjectively palpable stretched muscle fibers, known as taut bands.12,14 The local twitch response is a fleeting contraction or “flinching” of the muscle fiber that occurs with the release of pressure on an MTrP.12,14 MTrPs can cause notable referred or localized pain.15–18 They can present in two forms: the active form, which can cause un-induced referred and/or familiar pain, and the latent state, which is generally asymptomatic unless palpated.3,4,16
There is no “gold standard” for the detection of MTrPs.19,20 Although multiple examination guidelines (e.g., the Diagnostic Criteria for Temporo-Mandibular Disorders) and consensus documents (e.g., International Consensus on Diagnostic Criteria and Clinical Considerations of Myofascial Trigger Points) do exist,11,15,20,21 but their capability to diagnose MTrPs varies greatly.11,15,21 Typically, these protocols use a process of diagnosis by elimination that follows a comprehensive patient history and a clinical examination focused on compression of muscle tissue.11,12,15,21,22 This compression is intended to either identify a textural abnormality in the muscular tissue, or more commonly, elicit a combination of clinical findings (e.g., referred pain) that are agreed upon as diagnostic criteria.19,20 Currently, the most widely accepted signs for MTrP diagnosis are “spot tenderness,” “referred pain,” and “the local twitch response.” 20 The clinical acceptance and significance of these features, as defining signs of MTrPs, evolves and changes with time (e.g., a combination of two signs is accepted by some while others require a minimum of six signs). 20
Clinical palpation has been used as the primary modality for diagnostic compression of muscular tissue; however, it is controversial due to poor study quality and variable reliability.23–26 The complexity of integrating various diagnostic criteria into palpation is likely to produce inconsistent findings and hence reduce palpation’s reliability for MTrP detection. 27 In addition, clinical palpation can only provide subjective reporting, such as the general location and estimated pain level in contrast to measurements of the exact location, size, depth, or shape of MTrPs within the muscle. These findings may be further confounded by inconsistent pressure applied between clinicians; as an example, it has been reported that almost 60% of clinicians do not use palpation pressure within the recommended range of 0.8 to 1.2 kg. 28 Hence, other diagnostic tools such as electromyography, 29 histochemistry, 29 thermography, 30 pressure algometry, 30 and magnetic resonance imaging elastography30,31 have been studied. However, their high cost and limited availability and clinical value hinder their use.29,30 As a result, there is a clear need for research on an easily accessible chair-side diagnostic tool that can be used to aid in the identification of MTrPs and the diagnosis of myofascial pain. One such diagnostic tool is ultrasonography.
Ultrasonography is a real-time, relatively inexpensive, risk-free, and portable diagnostic tool that uses 2 to 20 MHz acoustic pressure waves.32–34 Diagnostic ultrasonography started in obstetrics, and its uses have since expanded to include visualization of musculoskeletal structures. 35 This has since led to studies evaluating ultrasonography in the diagnosis of MTrPs.36–39 In most investigations on this topic, ultrasonography is combined with other tools (e.g., elastography, computational modeling, or Vector Machine Classifier) to diagnose or guide interventions.40–44 These investigations may be premature, as the literature is yet to describe a consensus regarding ultrasonography’s capabilities, parameters, or the sonographic characteristics of MTrPs. For example, studies demonstrate a wide variety of target descriptions: a hyperechoic band, 36 hypoechoic elliptical region, 38 or simply a different echo-architecture than the surrounding muscle tissue. 36 As is the case with clinical palpation, 20 the standardization of sonography and a consensus of sonographic traits in the diagnosis of MTrPs will aid in the use of ultrasonography across clinical specialties and provide a more robust baseline for future studies. Therefore, this scoping review was aimed at presenting the diagnostic capabilities and parameters of ultrasonography, in MTrP investigations. Furthermore, the objective was to summarize the available descriptive characteristics of MTrPs, as seen via ultrasonography.
Materials and Methods
An a priori protocol was designed following the Systematic Reviews and Meta-Analyses-Extension for Scoping Reviews (PRISMA-ScR) checklist. 45 The Preferred Reporting Items for Systematic Reviews and Meta-Analyses checklist 46 was followed in the reporting of this review. With the assistance of an institutional librarian, a search strategy was developed (see Supplement 1) for Ovid Medline using the following MeSH phrases and keywords: “myofascial pain syndrome,” “Trigger Points,” “local twitch response,” “ultrasonography,” and “echography.” These terms were then combined with the Boolean formula “OR”/”AND” when appropriate. 47 The search strategy was then adapted to Embase Classic + Embase, and Cochrane CENTRAL®, as well as gray literature search (Google Scholar [first 200 hits]). All databases were searched from inception until January 2021. A hand-search of references was also conducted from the included studies and related papers,15,48–51 books, 52 and reviews.36–39
Eligibility Criteria
Data sources had to: (1) be a primary study published fully in a peer-reviewed journal; (2) be investigating patients suspected of having myofascial pain syndrome in any muscle; (3) be aimed to diagnose MTrPs or the local twitch response; (4) have used stand-alone ultrasonography as the index test; (5) have used “Palpation” as a reference test for detecting MTrPs or “Visual Inspection” as a reference test for the local twitch response; and (6) have calculable or reported diagnostic metrics (e.g., diagnostic percentages, concordance, accuracy, sensitivity, specificity, or positive/negative predictive values). Studies published before 1980 were excluded, as study methodologies and technological development were limited before that time (i.e., color Doppler development occurred in 1985). 53 Studies were also excluded with sample sizes of <10 patients as well as case reports and conference proceedings, as diagnostic accuracy measurements from such studies are of limited generalizability. Finally, studies were excluded that measured the diagnostic or therapeutic capabilities of a combination of ultrasonography and other tools (i.e., vibration elastography) as they can improve or lower the diagnostic ability of ultrasonography as a stand-alone modality.
References were uploaded to EndNote X9, 54 and duplicates were removed following Bramer et al. 55 All titles and abstracts were uploaded into Covidence®56 and were independently screened by two reviewers (ME, AS). Disagreements were resolved by consensus or arbitration by a senior reviewer (AA). This process was repeated during the full-text review.
To ensure the efficiency and accuracy of data abstraction, data-charting was independently undertaken by two experienced reviewers (ME and AS). A senior reviewer (AA) acted as an arbitrator in case of disagreement. Eleven variables were captured: muscle studied, sample size, study location, age range, sex distribution, patient inclusion criteria, target lesion, reference test description, index test description, index test target definition, and diagnostic accuracy results.
Critical Appraisal of Individual Sources of Evidence
As recommended by the
The risk of bias across studies was assessed based on methods of patient selection and enrollment, study design, and patient exclusion. It was also important to assess the methods of index test and reference standard application and interpretation, blinding, threshold (index test), and ability to correctly diagnose the target lesion (reference standard). Finally, the authors assessed the conformity of patients to the tests, the standardization of the tests, the time interval between the tests, and the analysis of the patients.
Applicability was assessed based on patient selection in terms of study setting, intended use of index test, presentation of the target lesion, and presence of prior testing. The index and reference test(s) applicability was also assessed based on the alignment, their conduct, and interpretation.
Results
There were 273 unique studies identified after de-duplication of the initial 357 titles. From these, 51 full texts were evaluated. Six studies,49,60–63 with a total sample size of 354 patients, were finally identified as sources of reliable evidence (see Figure 1). The reasons for the exclusion of the studies in the full-text review stage are presented in Supplement 2.
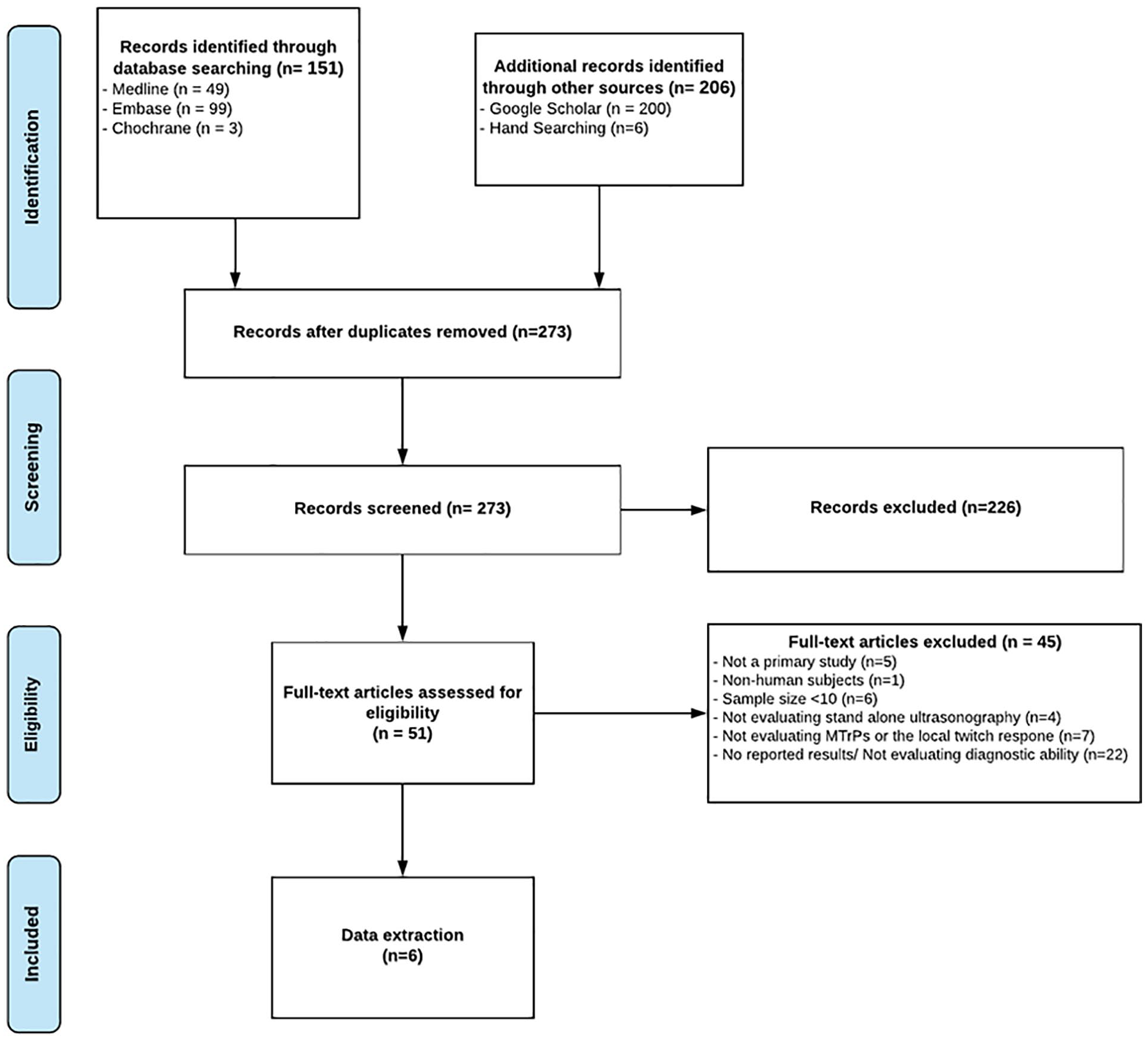
The study flow chart associated with this literature review.
Study Descriptions
The included studies (see Table 1) are cross-sectional clinical diagnostic research conducted in the United States, 60 Iran, 61 South Korea, 62 Egypt, 61 Malaysia, 49 and Australia. 63 One study was published in 1999, 63 and the remaining studies were published from 2011 to 2016. Only two studies49,62 had a sample of more than 100 patients, and the remaining studies had 50 patients or less. Two studies did not report on the sex distribution of the patients.1,49 The remaining studies had a prevalent female patient base (ranging from 56% 60 to 73%).1,49 The average age range of participants was 18 60 to 86 years old. 61
The Primary Studies Samples and Test Descriptions.
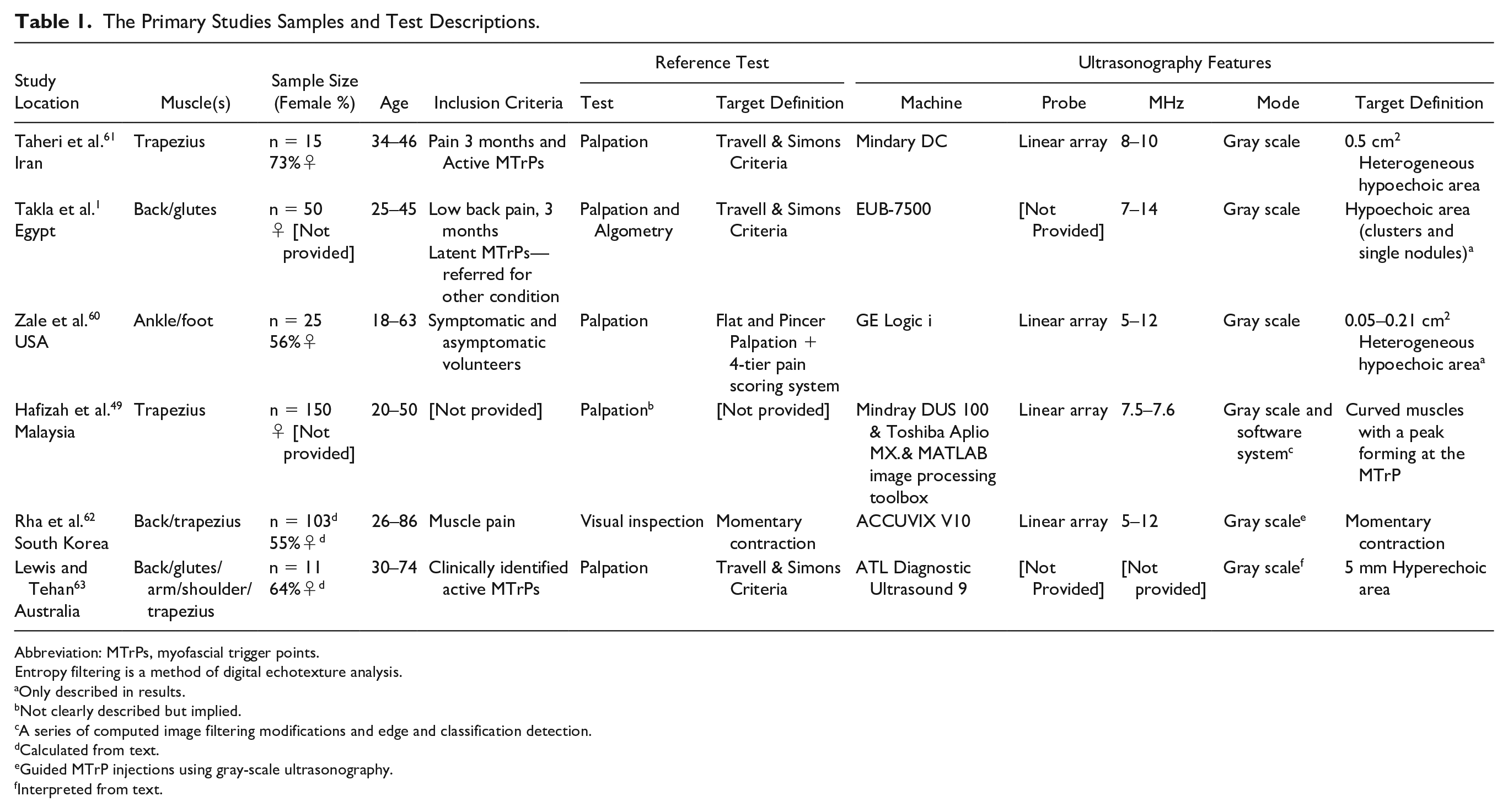
Abbreviation: MTrPs, myofascial trigger points.
Entropy filtering is a method of digital echotexture analysis.
Only described in results.
Not clearly described but implied.
A series of computed image filtering modifications and edge and classification detection.
Calculated from text.
Guided MTrP injections using gray-scale ultrasonography.
Interpreted from text.
Descriptions of Reference Tests
The reference tests (Table 1) included visual inspection of the local twitch response during needle insertion into MTrPs 62 and standardized palpation. In the majority of the studies, palpation was conducted via MTrP palpation alone following the Travell and Simons criteria,1,61,63 described as the presence of tender nodule in a tense band. One study used flat and pincer palpation with a 0- to 3-tier pain scoring system 60 where 0 represented no pain and no trigger point; 1 represented no trigger point nodule found but pain felt; 2 represented both pain and physical nodule presence; and 3 represented pain felt and nodule presence in both palpation and ultrasonography.
The reference standards were conducted by physiotherapists,61,63 a physical therapist, 1 a medical sonographer, 60 and a medical doctor. 62 One study 49 did not describe the palpation technique or administrator.
Descriptions of Index Tests and Target Conditions
The index tests (see Table 1) were conducted by a diagnostic medical sonographer, 60 radiologist,1,61,63 or medical doctor. 62 One study 49 did not describe the ultrasonography administrator. All utilized linear-array probes for the gray-scale ultrasonography, in frequencies of 5 to 14 MHz, except for one study, 63 which did not report the frequency. The target conditions were MTrPs or the twitch response. MTrPs were defined as 0.05 to 0.5 cm2 heterogeneous,60,61 hypoechoic clusters 1 or single nodules.1,60,61 Two exceptions were Lewis and Tehan, 63 who described MTrPs as 5 mm hyperechoic nodules; and Hafiza et al., 49 who described the MTrPs as the peak of a curved muscle structure. The twitch response was defined as a momentary contraction.62,63
Descriptions of the Diagnostic Capabilities of Ultrasonography
Descriptions of the diagnostic metrics are available in Table 2. One study 62 described the local twitch response and included only target positive cases. In other words, only MTrP-positive subjects (diagnosed by the reference test) were subjected to an index test, and subjects with “negative” results did not undergo comparison. As such, the rates of false or true negatives could not be calculated. This study reported that ultrasonography identified an equal amount of twitch responses as visual inspection in the trapezius muscle and 30% more twitch responses in the lower back during MTrP injections. 62
The Diagnostic Metrics Utilized.
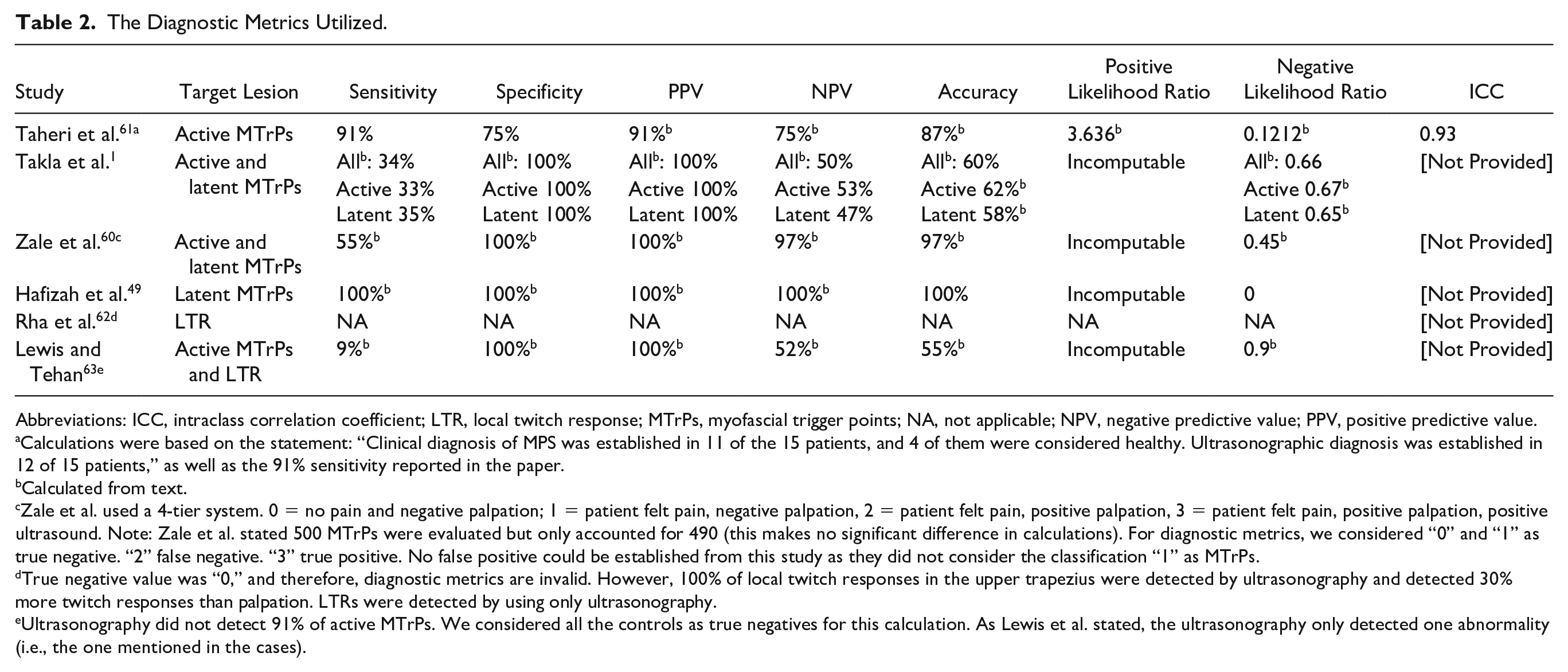
Abbreviations: ICC, intraclass correlation coefficient; LTR, local twitch response; MTrPs, myofascial trigger points; NA, not applicable; NPV, negative predictive value; PPV, positive predictive value.
Calculations were based on the statement: “Clinical diagnosis of MPS was established in 11 of the 15 patients, and 4 of them were considered healthy. Ultrasonographic diagnosis was established in 12 of 15 patients,” as well as the 91% sensitivity reported in the paper.
Calculated from text.
Zale et al. used a 4-tier system. 0 = no pain and negative palpation; 1 = patient felt pain, negative palpation, 2 = patient felt pain, positive palpation, 3 = patient felt pain, positive palpation, positive ultrasound. Note: Zale et al. stated 500 MTrPs were evaluated but only accounted for 490 (this makes no significant difference in calculations). For diagnostic metrics, we considered “0” and “1” as true negative. “2” false negative. “3” true positive. No false positive could be established from this study as they did not consider the classification “1” as MTrPs.
True negative value was “0,” and therefore, diagnostic metrics are invalid. However, 100% of local twitch responses in the upper trapezius were detected by ultrasonography and detected 30% more twitch responses than palpation. LTRs were detected by using only ultrasonography.
Ultrasonography did not detect 91% of active MTrPs. We considered all the controls as true negatives for this calculation. As Lewis et al. stated, the ultrasonography only detected one abnormality (i.e., the one mentioned in the cases).
The diagnostic metrics (see Table 2 and Figures 2 and 3) of the remaining studies are summarized as follows: accuracy of 55% 1 to 97%, 60 sensitivity of 33% 1 to 100%, 49 negative predictive value of 47% 1 to 100%, 49 specificity of 75% 1 to 100%,1,49,60 positive predictive value of 91% 61 to 100%,1,60 positive likelihood ratio of 3.64, 61 and negative likelihood ratio of 0.1261 to 0.67. 1 We excluded Lewis and Tehan 63 from the ranges above due to a likely inaccurate MTrP definition (hyperechoic instead of a hypoechoic lesion). Only Taheri et al. 61 reported inter-rater reliability (intraclass correlation coefficient of 0.83 and 0.93 for the longitudinal echogenicity and area of MTrPs, respectively).
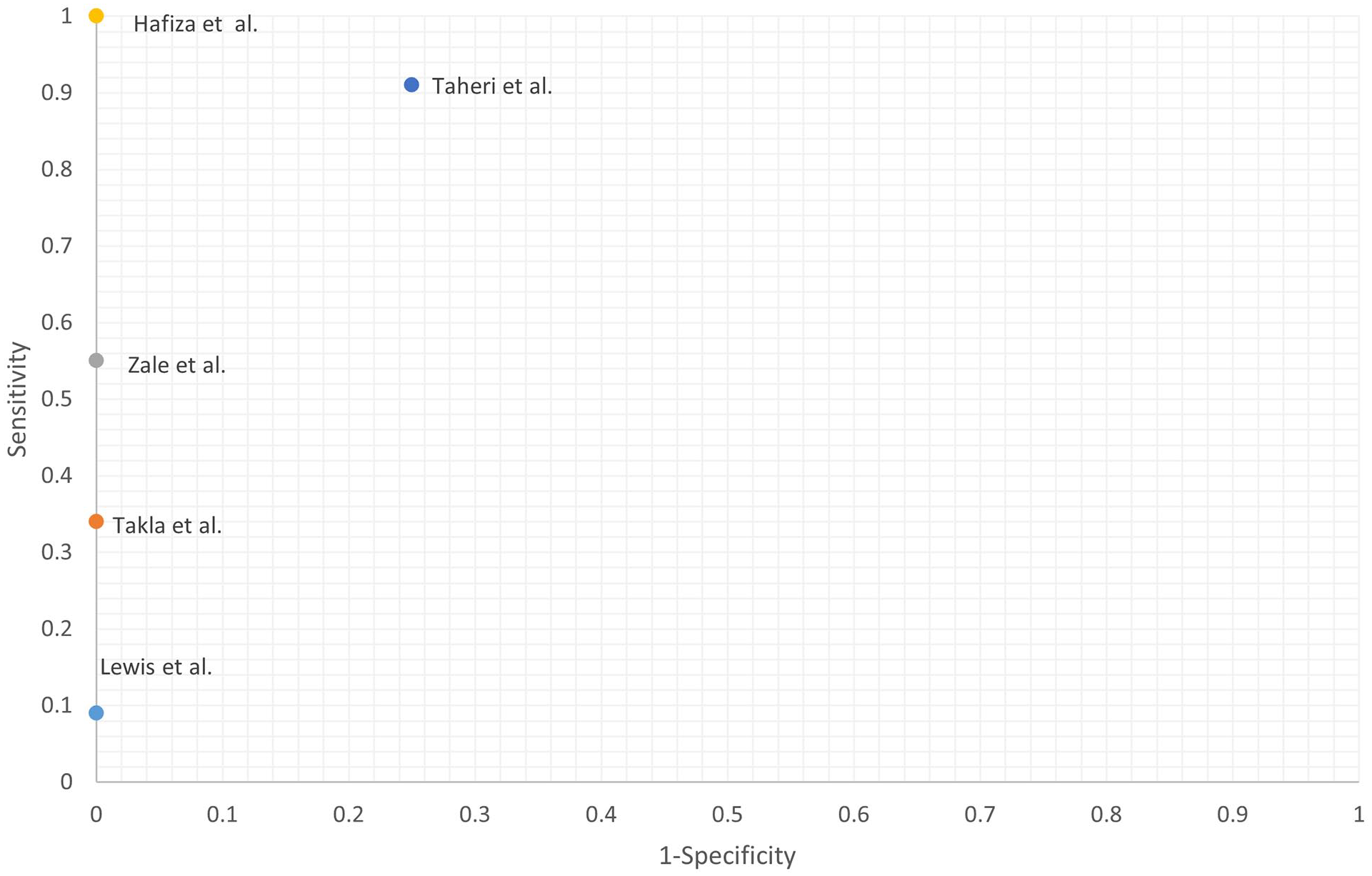
The receiver operating characteristic (RoC) plot Of studies included in this literature review.
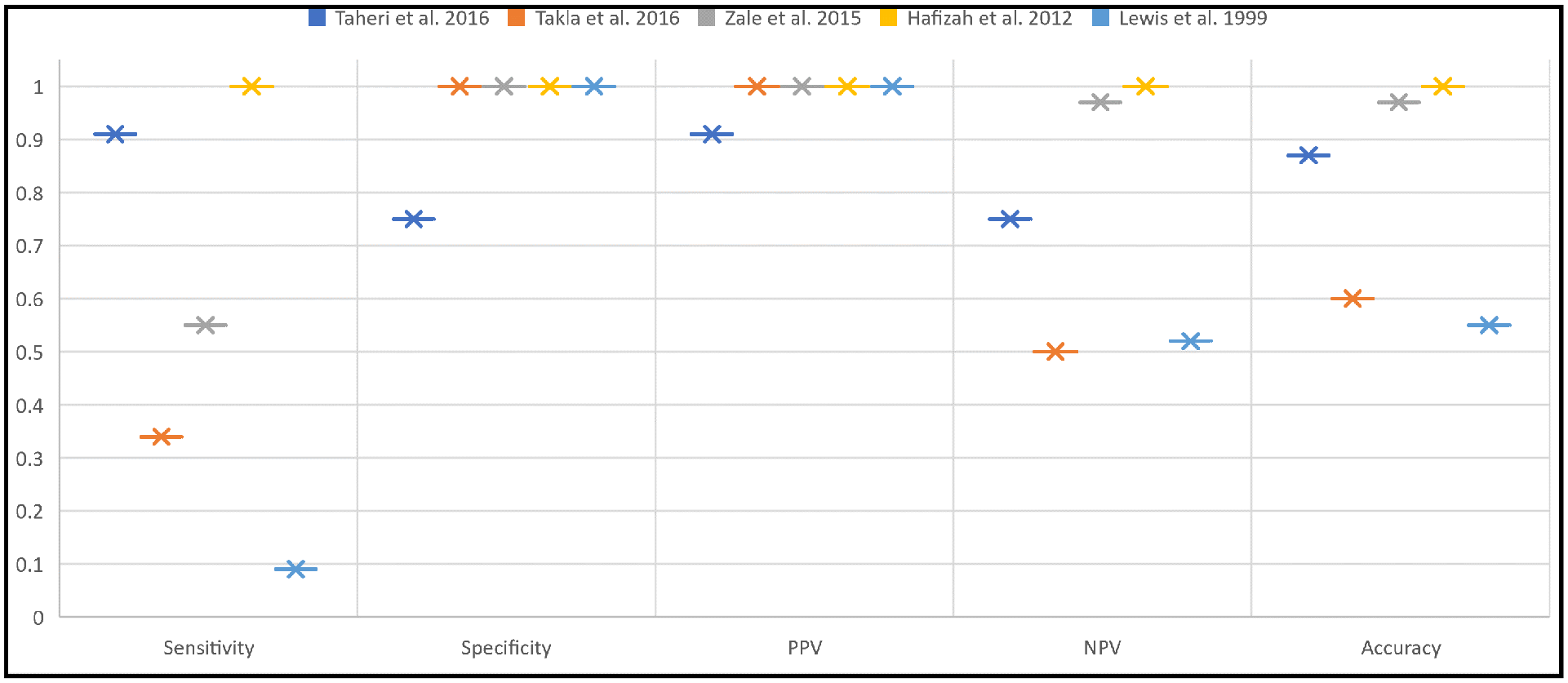
The Diagnostic Capabilities Reported In The Literature Review. NPV, Negative Predictive Value; PPV, Positive Predictive Value.
Methodological Quality of Included Studies
A full Modified QUADAS-2 evaluation58,59 is presented in Table 3 and Figure 4. For transparency, excerpts and rationales for the QUADAS-2 evaluations are presented in Supplement 3. Despite having a low sample size, the study performed by Taheri et al. 61 had the lowest risk of bias. The remaining studies showed a low to medium risk of bias, with each study showing one to two items suggestive of high or unclear risk of bias. The items of concern were the following.
The Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) Risk of Bias Assessment.
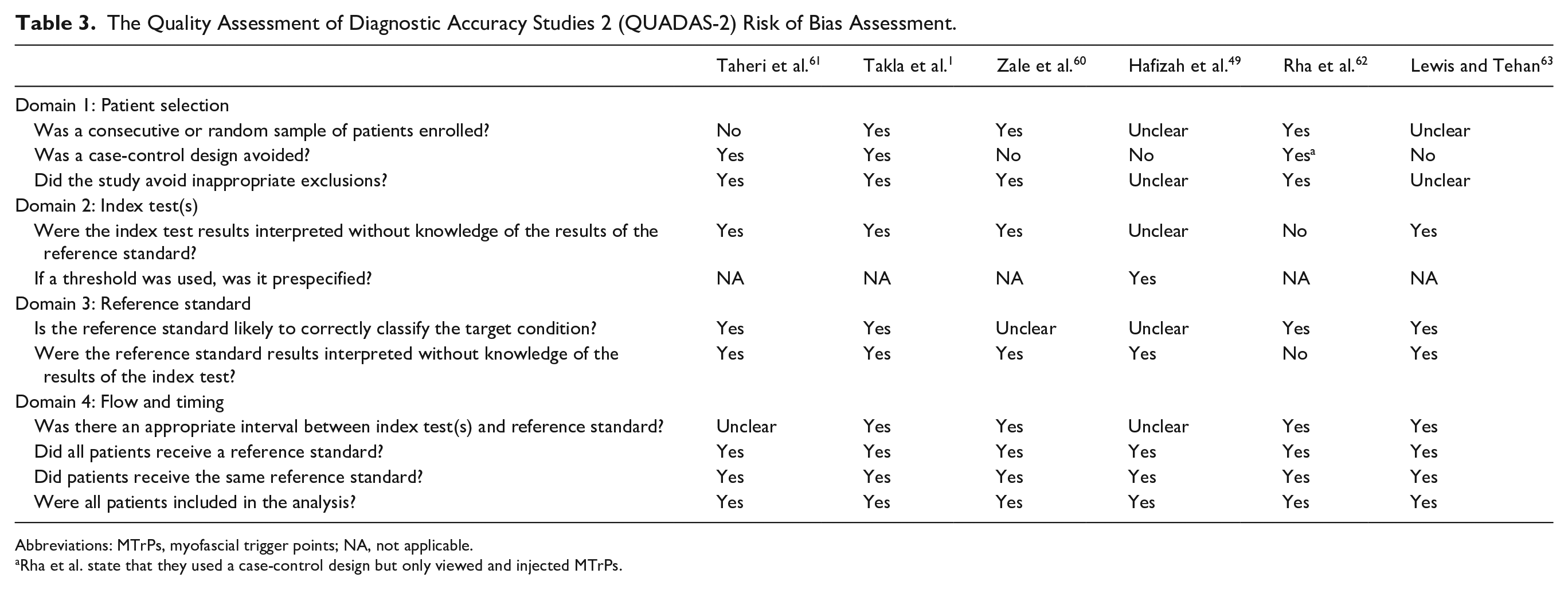
Abbreviations: MTrPs, myofascial trigger points; NA, not applicable.
Rha et al. state that they used a case-control design but only viewed and injected MTrPs.
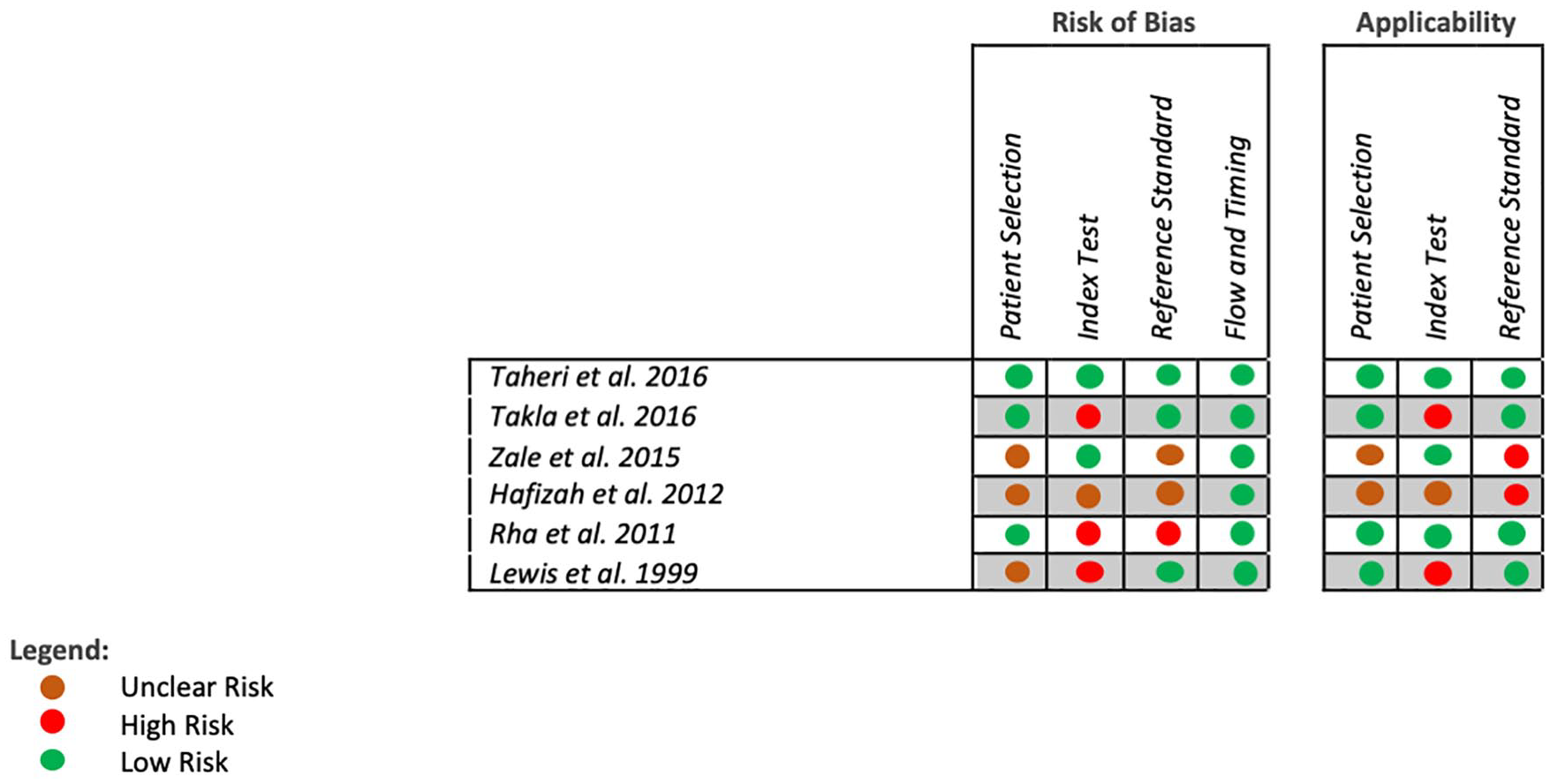
The Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) RIsk of Bias illustration.
In the QUADAS-2 Risk of Bias Assessment
In the QUADAS-2 Risk of Applicability Assessment
Discussion
In this review, the characteristics of the available ultrasonographic MTrP investigation were summarized and the attributes related to study population, ultrasonography, palpation parameters, and the reported diagnostic abilities and critical appraisal of the primary studies were presented.
A consensus on the parameters was found for diagnostic characteristics and the presentation of MTrPs, in ultrasonography. The trigger points appeared to be 0.05 to 0.5 cm2 nodules of varying hypo-echogenicity (Figure 5). However, the consensus was not absolute as there are notable deviations in described presentation, specifically: clusters of hypoechoic structures, 1 and a curved muscle form with MTrP, 49 as well as the hyperechoic presentation. 63 This indicates that further investigation is needed to clarify if these presentations (namely hypoechoic clusters) are a variation of the standard hypoechoic nodule and how frequently such manifestations occur. Overall, a significant imprecision in the ranges of diagnostic ability was noted. Some contributing factors to this wide range may be the limited subjective nature of diagnostic interpretation, small sample sizes, the methodological variability, the investigator’s profession, and the varying target definitions.
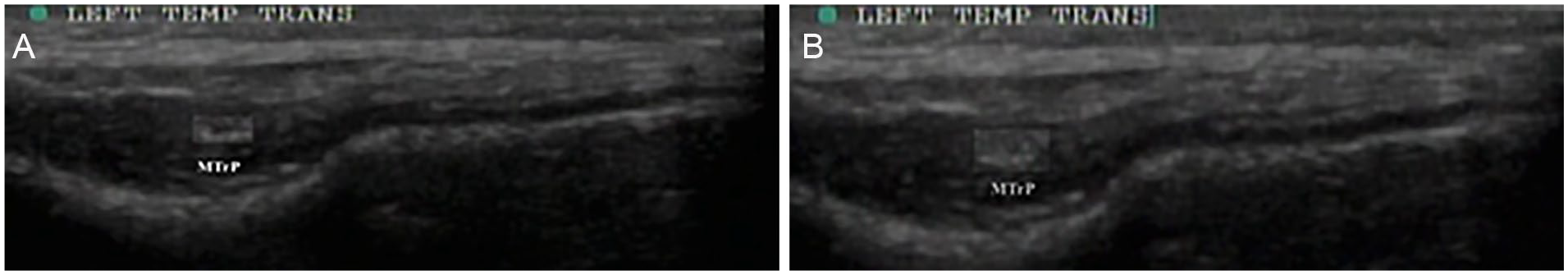
Myofascial trigger point (MTrP) size in both the relaxed and contracted states.
These results indicate that the value of ultrasonography is multifaceted. The high values for specificity (Figures 2 and 3) and positive predictive values indicate ultrasonography’s significance in “ruling in” the diagnosis of MTrPs. This need for increased diagnostic confidence for MTrPs is evident in cases where myofascial pain syndrome contributes to a combination of ailments. As such, ultrasonography would provide the clinician with an objective confirmatory diagnosis of MTrPs that would otherwise be lost in the “general symptomatology.” Examples of such conditions are temporomandibular disorders, as they are often multifactorial in origin with overlapping symptomatology. 64
Ultrasonography may aid in reducing the overestimation of the presence of MTrPs. Myofascial pain is a common syndrome that presents with a variety of manifestations. These manifestations often overlap with symptoms of other conditions. 65 To make things worse, these conditions are often comorbid with myofascial pain. Examples of common comorbidities can be connective tissue diseases, neuropathic pain disorders, migraine, tension-type headaches, gastrointestinal disorders, and hypothyroidism. 66 Another example of particular importance is fibromyalgia.64,65 It can be approximated that 75% of fibromyalgia patients are suspected of also having myofascial pain syndrome. 64 The overlap of the conditions and their symptomatic similarity have often caused them to be confused for one another or even thought of as the same condition. 66 As such, sonographic results may change the diagnostic management strategy of numerous patients by increasing the diagnostic confidence and, in turn, changing the approach to treatment.
Ultrasonography may also aid in reducing underdiagnosed MTrP conditions. Palpation is useful in diagnosing textural abnormality and eliciting a painful sensation but would be limited if the area in question is already painful due to another condition (e.g., inflammatory disease, regional infection, and active rheumatoid arthritis) or is anatomically inaccessible (e.g., in subjects with high body mass index). Myofascial pain is commonly resultant of, or at least correlated with, joint problems or parafunctional abnormalities, 10 which are common manifestations of many of the study exclusions (e.g., high body mass index or musculoskeletal fracture). It should be noted that although such conditions can be characteristic of a significant portion of the target population, making them susceptible to MTrPs, they were listed as part of the exclusion criteria of the included studies to perhaps make the diagnosis goals attainable by palpation. When considering that ultrasonography’s diagnosis is primarily image-based, excluding these conditions in the included clinical studies might have simultaneously contributed to the overestimation of diagnostic metrics for clinical palpations and the underestimation of such metrics for ultrasonography.
Some overarching limitations in the included studies were missing/unreported data, noted inconsistencies, and significant heterogeneity of reference test and index test target definitions and interpretations. This caused the review team a considerable challenge of translating each study’s result into a form adaptable to an overview. The choices in data presentation in the included studies did not assess diagnostic ability as the primary outcome. Although invaluable as descriptive and foundational studies, this reporting tendency limits the diagnostic applicability and generalizability of the studied topic. To mitigate this limitation, we conducted diagnostic metric calculations whenever feasible. The inconsistencies in data outcome standardization and heterogeneity in the definitions and interpretations of the reported outcomes are also a limiting factor. For example, the area of the MTrPs is an objective measurement that has clinical treatment and follow-up implications but was only reported in two of our included studies. To mitigate this, our data collection was as inclusive as possible and accommodated for heterogeneous data (e.g., target description instead of MTrP area). Also, none of our included studies reported on pain grade of the studied MTrPs and instead limited their description to active or latent MTrPs. This limits the possible data synthesis regarding the correlative or causative relationship between the size, appearance, and depth of the MTrP as well as the pain severity. Finally, we only identified 6 studies with 354 included patients, which may be due to the novelty of the topic. Muscle variety was also a limitation, as four of the studies investigated, at least in part, the back and neck areas. To mitigate this, we conducted a comprehensive search of four major databases in all languages and gray literature and reference searching of reviews and related studies.
Only one study 61 reported intrarater reliability of the ultrasonographic parameters by re-evaluating patients within an hour. Although invaluable in determining the consistency of the result, this does little to establish the applicability of ultrasonography across multiple investigators, limiting the information regarding the reproducibility of the results in a different clinical setting.
This review has some unique strengths. To our knowledge, it is the first scoping review and critical summary of the literature on the use and diagnostic capabilities of ultrasonography for the diagnosis of MTrPs and the twitch response. In addition, it provides a baseline for consensus documents and further diagnostic studies by defining the reported characteristics of MTrPs in ultrasonography. This review has showcased that the diagnostic capability ranges are imprecise (e.g., sensitivity 33%–100%) and that most of the included studies focused on identifying MTrPs and the local twitch response in the trapezius and quadratus lumborum muscles. Hence, there is a gap of knowledge in the diagnostic capabilities of ultrasonography on other muscles of the body. Of special note are the muscles of mastication, as it is reported that 50% of patients with temporomandibular disorder (an ailment that afflicts 15% of adults) 67 suffer masticatory myofascial pain syndrome.4,64,68–70
This review also showed that the ultrasonography examiners were radiologists, medical sonographers, or medical doctors. The included studies used variable methods of establishing the clinical adequacy of their ultrasonography examiners (e.g., clinician experience, specialty clinical training, preinvestigation briefing). The lack of optimal training or experience in applying the test or interpreting its results is of significant impact in diagnosing MTrPs. It is unclear how much modular training is needed for an acceptable level of adequacy in MTrP identification via ultrasonography. This choice of experts also indicates another opportunity for training other allied health care professionals, such as dentists and physiotherapists. Training these specialists to use ultrasonography as a chair-side evaluation can be beneficial, as they routinely see myofascial pain patients.
Considering the review findings, this investigation also highlighted some implications for future research and some clinical notes. There have been recent updates regarding the use of the palpation reference standard, its standardization, and its service across clinical specialties. 20 It stands to reason that this standardization should also be attempted for the diagnostic tools used to identify MTrPs. This scoping review is a first step in the standardization and interdisciplinary utilization of this technology. For this to be achieved, future studies must describe nodule sizes relative to examined muscles. This knowledge can further hone sonographic investigation and allow us to further understand MTrP characteristics in deep and superficial muscles of all sizes.
When calculating traditional metrics such as sensitivity and specificity, it can be assumed that the reference standard is an error-free “gold standard.” However, the reference test of clinical palpation is uniquely prone to misdiagnosis errors, which can overestimate or underestimate the accuracy of ultrasonography. Future studies need to investigate the
It is suggested that future clinical studies of ultrasonography in MTrP diagnosis follow the
The primary study authors did not provide any specific methods or protocol for MTrP ultrasonography. Nevertheless, some clinically important takeaways can be taken from the included studies. Clinicians may find bimonthly quality control testing of ultrasonography machines important to maintain diagnostic accuracy and consistency. 60 It is also important to define and maintain appropriate imaging settings (i.e., time gain compensation, depth, and sector size) 73 as appropriate to the muscle/area being scanned. Using musculoskeletal presetting defaults that optimizes the sonographic technology parameters (e.g., spatial compounding and upshifted frequencies) 60 may be an appropriate pragmatic method of achieving optimization and consistency for the sonographers. In terms of patient infection control, it is advisable to use a sterile coupling gel and latex-free transducer cover especially during ultrasonography-guided injection therapy. 62 It may be appropriate that clinicians adapt predefined scanning protocols in their relative fields such as the American Institute of Ultrasound in Medicine protocol.60,74
The authors recommend that while imaging MTrPs via ultrasound, patients should be positioned in a comfortable position that allows the clinicians to applying the transducer perpendicularly above the MTrP sites 1 (i.e., upright sitting position for trapezius) 61 and the imaging procedure should be limited to 30 minutes for patient comfort. This should be done while maintaining the following key points:
The diagnostic ultrasonography equipment system should be used with a transducer that provides a transmit frequency of 5 to 10 MHz, and the pathological appearance of MTrPs should be identified as 0.05 to 0.21 cm2 heterogeneous hypoechoic areas during clinical investigations.
The ultrasound transducer should be coated with a layer of ultrasound gel and positioned on the overlying the examined muscles. Muscles are to be scanned for MTrPs by maneuvering the probe perpendicular to the muscle fibers. Once MTrPs have been located, the transducer position should be tilted as needed to obtain an adequate view. Each muscle should be imaged in the same position 1 for a better quantification of the imaged lesion features (see Figure 5) in both longitudinal and cross-sectional views. 61
The investigator should then perform both contracted and non-contracted examinations (to view the trigger points while the muscle is tensed and not).
Color Doppler of the MTrPs should be performed both contracted and non-contracted to illustrate blood flow of the MTrPs (blood vessel presence, systolic/diastolic flow).
Size and characteristics and depth of the MTrPs recorded in the non-contracted state are to take precedence over contracted muscle image measurements to allow for maximum size approximation. The digital images should be stored for record keeping. However, diagnosis from printed images is discouraged as a static image does not fully relay the MTrPs reaction under the compressive force and regular muscular movement.
Conclusion
This scoping review of low-to-medium risk of bias studies has found that the clinical studies used 5 to 14 MHz gray-scale ultrasonography to diagnose MTrPs and the local twitch response and visualized MTrPs as heterogeneous hypoechoic nodules. It is important to note that there was a wide range of ultrasonography diagnostic metrics across the included studies; nevertheless, the high specificity and positive likelihood ratio show strong promise in ultrasonography’s use as a confirmatory diagnostic tool when implemented independently or in conjunction with other diagnostic aids.
In conclusion, the available literature regarding ultrasonography’s diagnostic potential in myofascial pain is scarce and spread over multiple medical disciplines and specialties. This is due to the study’s novelty and the nature of myofascial pain and in all skeletal muscles. Identifying MTrPs through ultrasonography needs additional primary and replication studies to be established as a reliable diagnostic tool.
Supplemental Material
sj-docx-1-jdm-10.1177_87564793221102593 – Supplemental material for Diagnostic Applications of Ultrasonography in Myofascial Trigger Points: A Scoping Review and Critical Appraisal of Literature
Supplemental material, sj-docx-1-jdm-10.1177_87564793221102593 for Diagnostic Applications of Ultrasonography in Myofascial Trigger Points: A Scoping Review and Critical Appraisal of Literature by Mohamed Elbarbary, Adam Sgro, Michael Goldberg, Howard Tenenbaum and Amir Azarpazhooh in Journal of Diagnostic Medical Sonography
Supplemental Material
sj-docx-2-jdm-10.1177_87564793221102593 – Supplemental material for Diagnostic Applications of Ultrasonography in Myofascial Trigger Points: A Scoping Review and Critical Appraisal of Literature
Supplemental material, sj-docx-2-jdm-10.1177_87564793221102593 for Diagnostic Applications of Ultrasonography in Myofascial Trigger Points: A Scoping Review and Critical Appraisal of Literature by Mohamed Elbarbary, Adam Sgro, Michael Goldberg, Howard Tenenbaum and Amir Azarpazhooh in Journal of Diagnostic Medical Sonography
Supplemental Material
sj-docx-3-jdm-10.1177_87564793221102593 – Supplemental material for Diagnostic Applications of Ultrasonography in Myofascial Trigger Points: A Scoping Review and Critical Appraisal of Literature
Supplemental material, sj-docx-3-jdm-10.1177_87564793221102593 for Diagnostic Applications of Ultrasonography in Myofascial Trigger Points: A Scoping Review and Critical Appraisal of Literature by Mohamed Elbarbary, Adam Sgro, Michael Goldberg, Howard Tenenbaum and Amir Azarpazhooh in Journal of Diagnostic Medical Sonography
Footnotes
Acknowledgements
We would like to thank Maria Zych for her assistance in developing the search strategy. We would also like to thank Ariel Oren and Alac Alkhateeb for their assistance in writing this paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
