Abstract
Objective:
To investigate the vagus nerve (VN) dimensional changes with Parkinson’s disease (PD), compared with healthy subjects. Additionally, it is important to investigate whether there is any relationships between these changes and patient’s motor and non-motor symptoms (NMS) of PD.
Materials and Methods:
A cohort of 43 patients with PD formed a group that was compared with 44 patients without PD, denoted as the healthy subject (HS) group. The diameter and areas of VN of study groups were measured using ultrasonography (US). The study groups were further divided into <65 and ≥65 subgroups, to evaluate the possible effect of age on the VN and evaluated relationships of VN dimensions, between subgroups. In the PD group, a correlational analysis was conducted between the diameter and area of the VN and the motor and NMS scores.
Results:
There was statistically significant difference in right (P = .002) and left VN diameters (P = .007) and in right (P = .001) and left VN areas (P = .007), between study groups. There was no significant difference in right and left VN diameters and the right and left VN areas, between subgroups. There was moderately negative correlation between gastrointestinal NMS scores and right VN area (r = −0.499, P = .002), left VN area (r = −0.499, P = .002), right VN diameter (r = −0.378, P = .023), left VN diameter (r = −0.385, P = .021), respectively.
Conclusion:
The US demonstrated that VN dimensions may possibly reduce in those patients affected by PD. In this cohort, it appears that an increase in gastrointestinal NMS scores may be explained by atrophy of the VN.
Parkinson’s disease (PD) is a common progressive neurodegenerative disease characterized by tremor, rigidity, bradykinesia, and postural instability. Although PD is considered a clinical condition with the mentioned motor symptoms according to the United Kingdom Brain Bank Criteria, 1 some non-motor symptoms (NMS), including olfactory disorders, gastrointestinal autonomic dysfunction, and sleep disorders, may precede motor symptoms. 2 Dysphagia, delay in gastric emptying, constipation, and decreased salivation are the most frequently reported gastrointestinal symptoms among PD patients. 3
The vagus nerve (VN) is the most important nerve and the neurological bridge of the brain-gut axis.4,5 It is known that pathologies occurring in the central nervous system affect the enteric nervous system; likewise, pathologies occurring in the enteric nervous system also affect the central nervous system. 6 Microbiota may also influence the central nervous system through the VN pathway and is associated with diseases such as PD. 7 Studies have shown that perikarion degeneration is accompanied by axonal destruction (Wallerian degeneration) in addition to α-synuclein deposition in PD. 8 It is known that PD also influences the VN’s dorsal motor nucleus. Degeneration of VN fibers (VN axons), along with the dorsal motor nucleus, can be associated with both non-motor and motor symptoms of PD. 9
The most common NMS of PD is constipation, with the literature reporting a prevalence between 28% and 80%.2,10 The gastrointestinal symptoms of PD are constipation, dysfunction of salivary glands, dysphagia, defecation disorder, and motility disorder of the upper gastrointestinal tract. PD mostly affects the upper gastrointestinal and lower gastrointestinal tract, and it has been shown that the main pathophysiological mechanisms of PD occur in the upper gastrointestinal tract due to striatal muscle dysfunction in the oropharynx and upper esophagus. It has been reported in the literature that PD delays gastric emptying and therefore causes gastroparesis during pathogenesis.11,12 The published literature indicates that gastrointestinal motility disorders are related to the axonal degeneration of the VN and degeneration of the myenteric plexus in PD. 13
The parasympathetic nervous system drives gastrointestinal motility, while the sympathetic nervous system acts as an antagonist to this motility. The VN system is a major component of the parasympathetic nervous system critical in regulating gastrointestinal motility. In PD patients, VN degeneration is common and has been associated with the development of PD. The area, diameter, and imaging features of the cervical segment of the VN can be evaluated using high-resolution ultrasonography (US). 14
In this study, the purpose was to evaluate the morphological changes in the VN’s area and diameter in PD patients using US. As the role of age-related atrophy had not previously been evaluated in the literature, the goal was to analyze age-related physiological VN atrophy in a group of healthy subjects. A secondary aim was to examine the relationship between morphological changes in the VN and non-motor and motor symptoms, caused by PD.
Materials and Methods
The study was accepted by local ethics committee and given the approval number 2018.147.10.12. Informed consent was obtained from all patients included in this study. A cohort of 43 patients with PD and 44 patients without PD were included, constituted a total of 87 patients. The PD group suffered from idiopathic PD as per the diagnostic criteria of the United Kingdom Parkinson’s Disease Society Brain Bank and had applied to the neurology outpatient clinic, at least 1 year between March 2018 and July 2019. These patients were provided with surveillance at intervals of 3–6 months. The patients’ demographic information and disease durations were obtained from the patient database registered in the computer system. The healthy subject (HS) group consisted of healthy individuals in the same age range as PD patients with normal neurological examinations.
Patients were excluded from the study if they had been who diagnosed with diabetes mellitus, followed up with or treated due to neurological diseases progressing with peripheral neuropathy, had other neurodegenerative diseases, had neck surgery for any reason, or had a history of neck trauma. The PD patients included in the study were evaluated using the Movement Disorder Society-Unified Parkinson’s Disease Rating Scale III (MDS-UPDRS III). An NMS questionnaire was used to evaluate the patients’ non-motor PD symptoms, which was validated for the Turkish population.15–17
Gastrointestinal symptoms of Parkinson’s disease include abnormal salivation, dysphagia, nausea, constipation, and defecatory dysfunction. Edwards et al 18 reported that except defecatory dysfunction, the other gastrointestinal symptoms are directly related to Parkinson’s disease instead of Parkinson’s medication. In this study, it was important to evaluate the gastrointestinal symptoms, except defecatory dysfunction, according to that study.
The VN areas and diameters of patients in both groups were evaluated individually in an anonymized manner by a radiologist, experienced in nerve US. To evaluate whether the VN’s area and diameter varied depending on age, patients in PD and HS groups were divided into subgroups of<65 and ≥65 years of age. The statistical differences between VN areas and diameters, MDS-UPDRS III scoring, disease duration, total NMS scores, and gastrointestinal NMS scores between the subgroups were examined.
Vagus Nerve Ultrasonography
Vagus nerve US was performed by an experienced radiologist, using a Toshiba Applio XG™ ultrasound equipment system. The right and left lower cervical VNs were evaluated separately at the level of the thyroid gland in the transverse plane with a 6–14 MHz linear transducer. The VN was found as a hypoechoic round structure, at the level of the thyroid gland, between the common carotid artery and the internal jugular vein. The continuity of the nerve, at this level, was detected with US. The axial area of the VN was measured in the axial imaging plane. Following this, the transducer was positioned at the longitudinal axis and the VN diameter was measured in longitudinal imaging plane (see Figure 1). 19
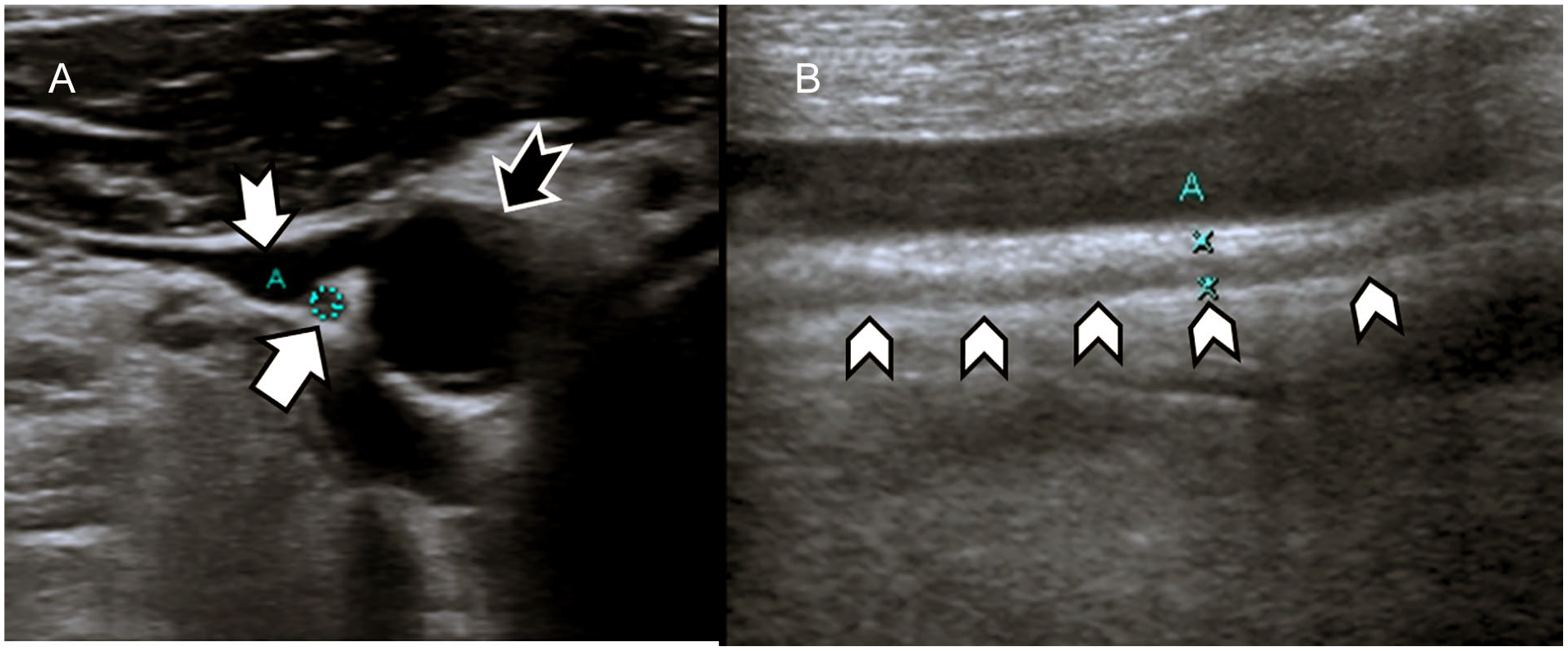
(A) In the axial plane, sonography demonstrates the vagus nerve (VN) (white arrow) between internal jugular vein (white-tailed arrow) and the common carotid artery (black-tailed arrow). (B) Ultrasonography shows the VN (arrow heads) in the longitudinal plane.
Statistical Analysis
Data were analyzed using IBM SPSS Statistics Version 24.0 (IBM Corporation, Armonk, NY, USA). The Kolmogorov–Smirnov test was used to determine whether the distribution of continuous numerical variables was close to normal. Descriptive statistics were expressed as the mean ± standard deviation and median (max–min) for numerical variables, while the number of cases and percentages (%) represented categorical variables.
The significance of the differences between two groups in terms of age was assessed with a Student’s t-test and gender-based differences were analyzed with a continuity-corrected chi-square test. Significant differences in continuous variables were evaluated using a Student’s t-test and the Mann–Whitney U test. Correlations between VN areas and diameters and MDS-UPDRS III scores, disease duration, and total NMS and gastrointestinal NMS scores, within the PD group, were completed using the Pearson and Spearman correlation analysis. The statistical significance was set a priori with P-values < .05.
Results
The mean age of the patients in the PD group was 63.30 ± 7.83 (range 42–73), and the mean age of those in the HS group was 62.36 ± 5.42 (range 54–76). There were 18 female participants in the PD group and 25 were male, while there were 31 female participants in the HS group and 13 were male. There was no statistically significant difference between ages of those in the PD and HS groups (P = .313). The mean MDS-UPDRS III of PD patients was 22.16 ± 15.65. Descriptive data and VN area and diameter US findings for the PD and HS groups (see Table 1).
The Descriptive Data and the Vagus Nerve Ultrasonographic Findings, in the Parkinson’s Disease and the Health Study Groups.
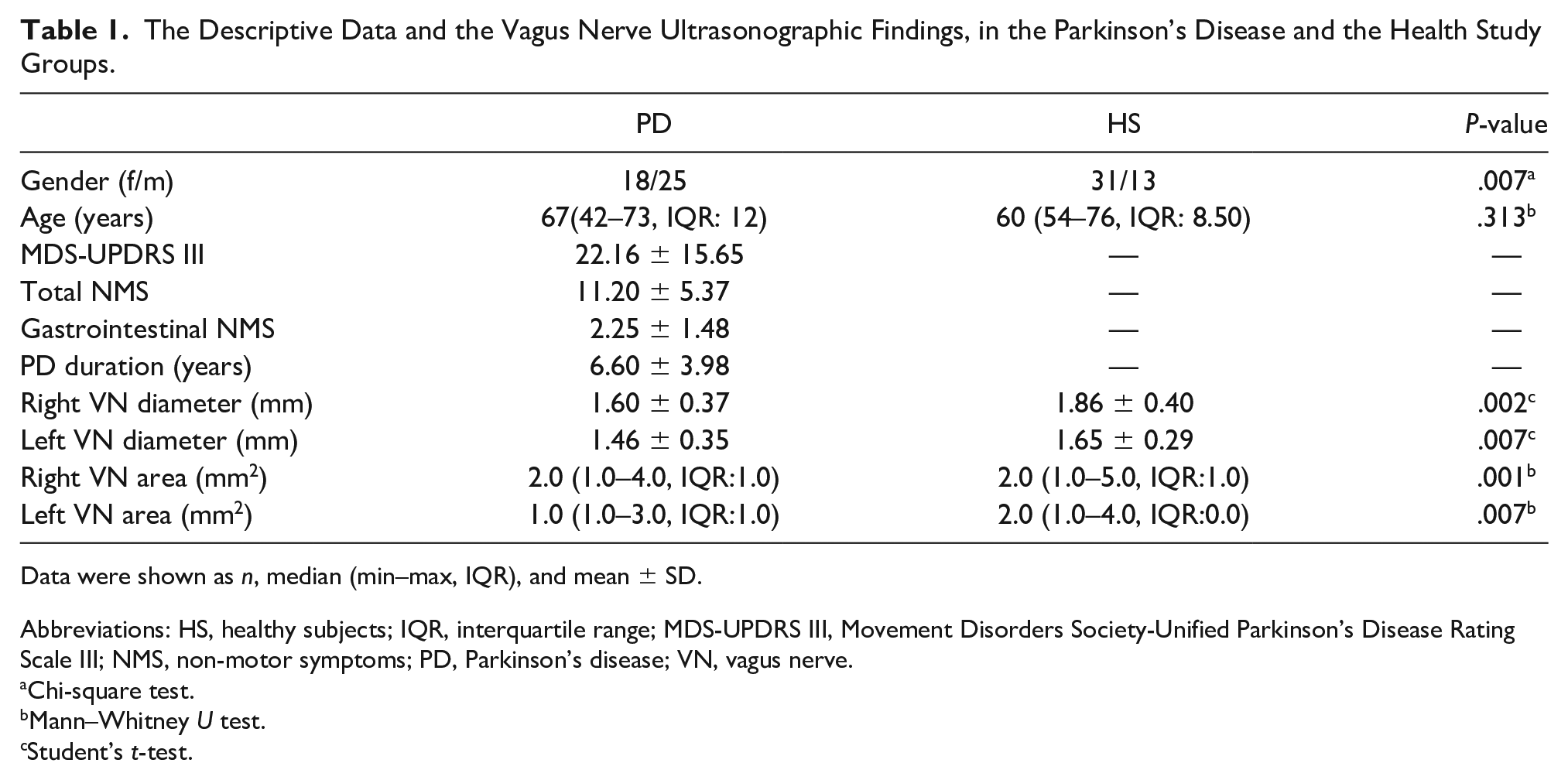
Data were shown as n, median (min–max, IQR), and mean ± SD.
Abbreviations: HS, healthy subjects; IQR, interquartile range; MDS-UPDRS III, Movement Disorders Society-Unified Parkinson’s Disease Rating Scale III; NMS, non-motor symptoms; PD, Parkinson’s disease; VN, vagus nerve.
Chi-square test.
Mann–Whitney U test.
Student’s t-test.
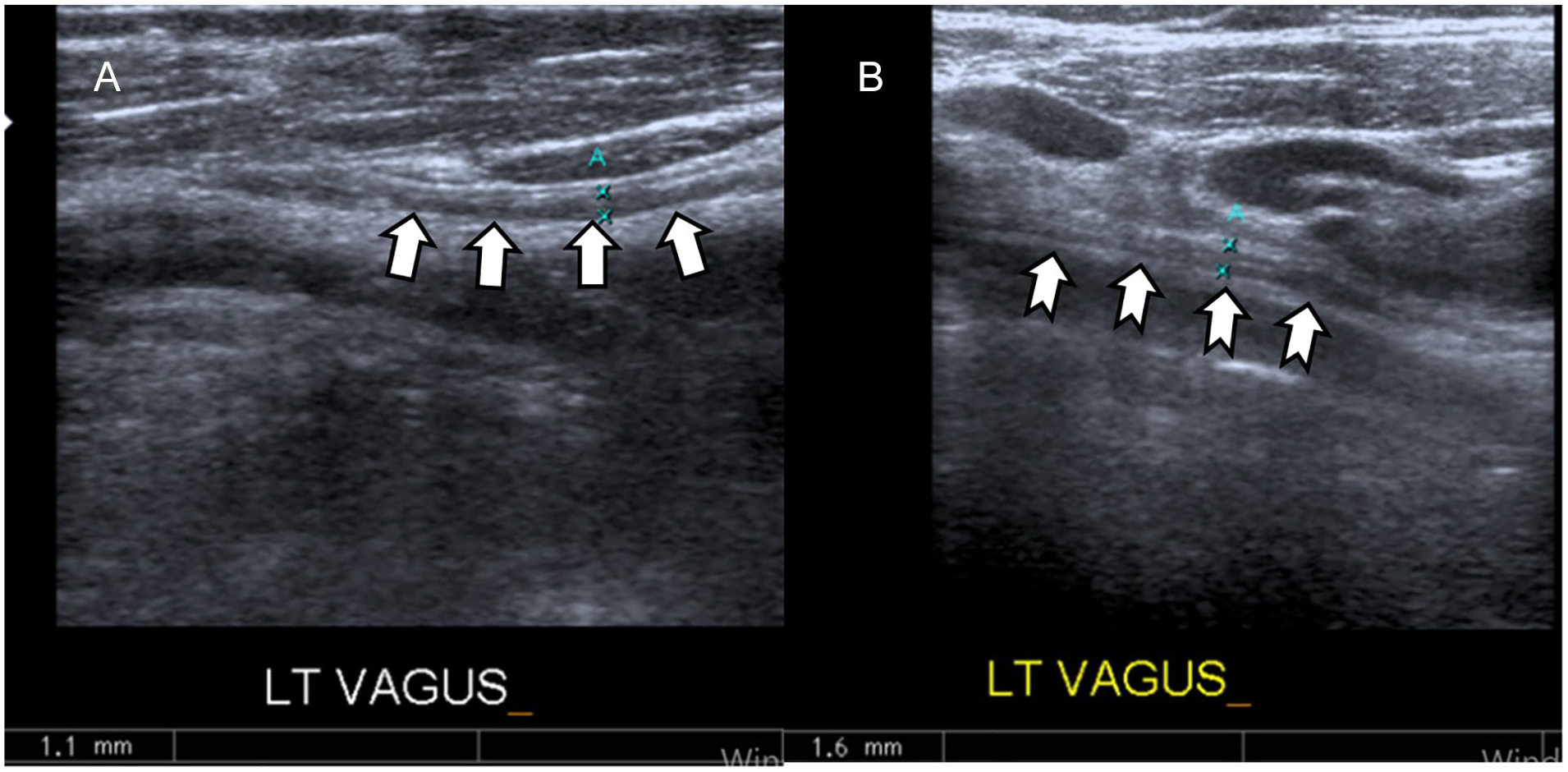
Between the PD and HS groups, the right VN area (P = .001) and the left VN area (P = .007) were significantly lower in the PD group when compared with HS group (see Figure 2). There was also a statistically significant difference between PD and HS groups between right VN diameter (P = .002) and left VN diameter (P = .007), respectively and right and left VN diameters were lower in PD group compared with HS group (see Figure 3 and Table 1).
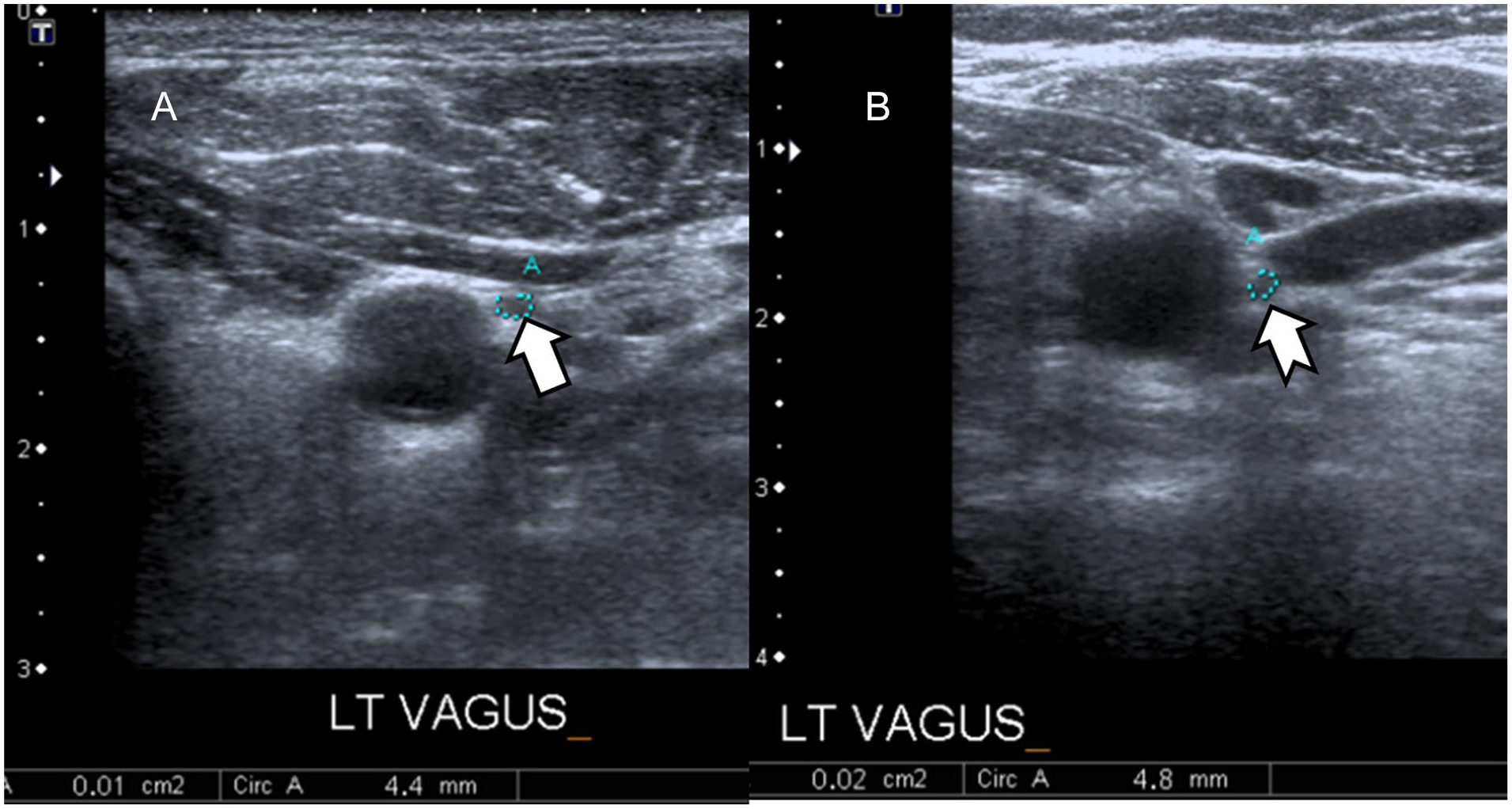
Sonography demonstrates the left vagus nerve (VN) (white arrow) in a Parkinson’s disease patient (A) and the left VN (white-tailed arrow) in a healthy subject (B), in the axial plane. The reduction of VN area can be clearly seen by the guidance of sonography (1 vs 2 mm2).
The mean MDS-UPDRS III score in PD subgroups above 65 years of age was 25.73 ± 17.90 and 18.05 ± 11.72 for those under 65 years of age. There was no statistically significant difference between MDS-UPDRS III findings, among PD subgroups (P = .109).
There was no significant difference between the right VN diameter (P = .598) and left VN diameter (P = .903) or right VN area (P = .587) and left VN area (P = .935), between the PD subgroups (see Table 2). Likewise there was no significant difference between right VN diameter (P = .358) and left VN diameter (P = .560) or right VN area (P = .221) and left VN area (P = .715), in the HS subgroups (see Table 3).
The Descriptive Data and the Vagus Nerve Ultrasonographic Findings, for the Parkinson’s Disease Subgroups.
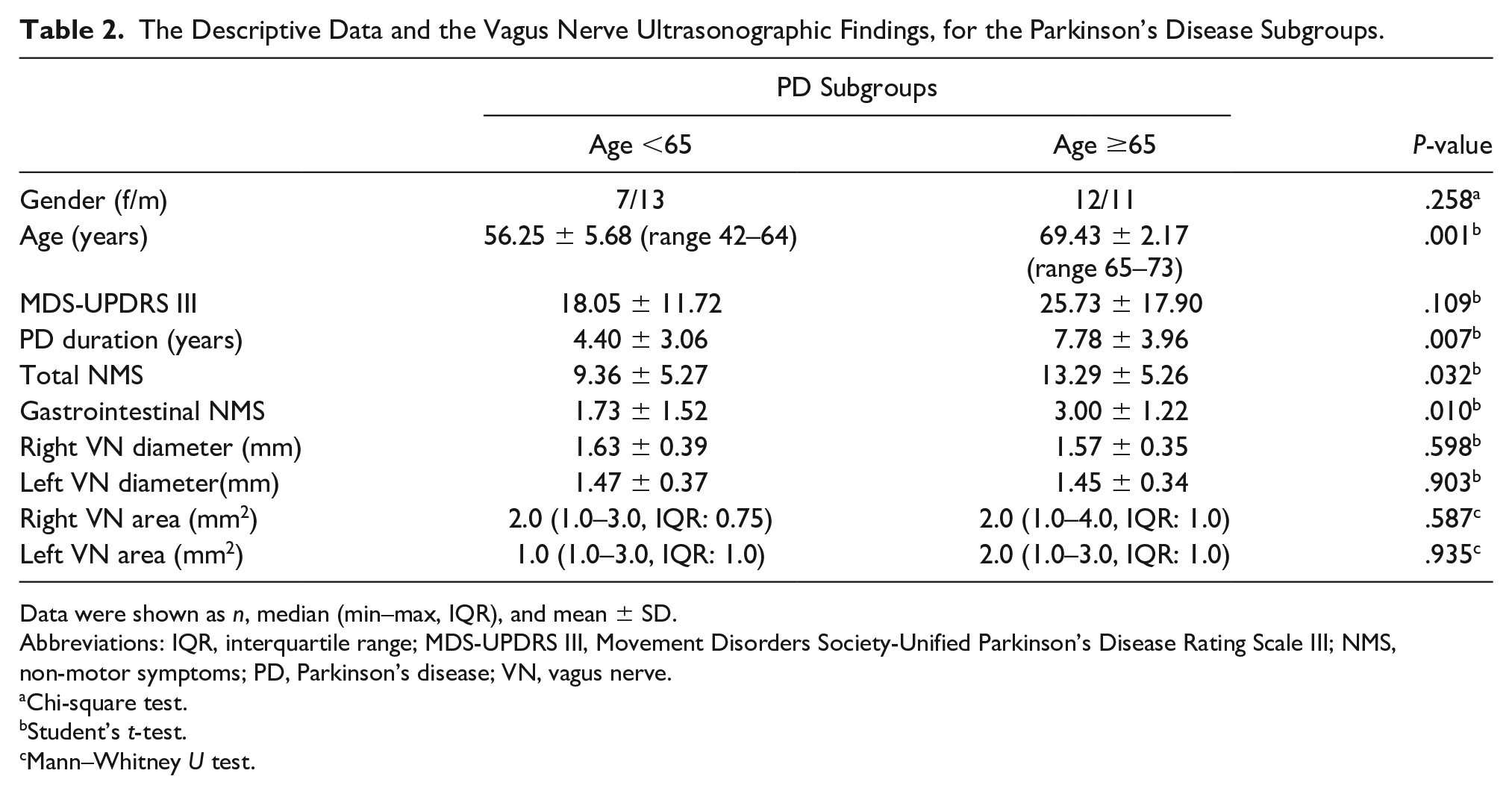
Data were shown as n, median (min–max, IQR), and mean ± SD.
Abbreviations: IQR, interquartile range; MDS-UPDRS III, Movement Disorders Society-Unified Parkinson’s Disease Rating Scale III; NMS, non-motor symptoms; PD, Parkinson’s disease; VN, vagus nerve.
Chi-square test.
Student’s t-test.
Mann–Whitney U test.
The Descriptive Data and the Vagus Nerve Ultrasonographic Findings, for Health Subject Subgroups.
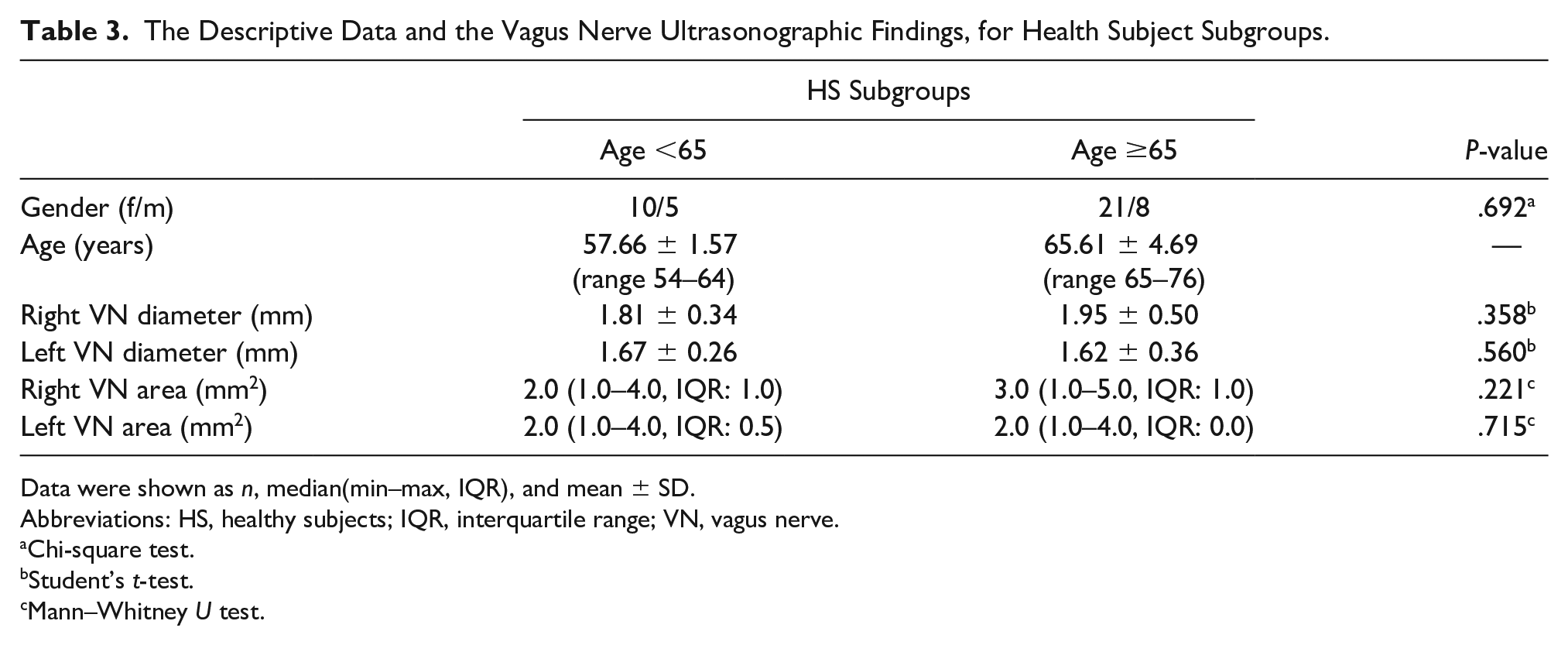
Data were shown as n, median(min–max, IQR), and mean ± SD.
Abbreviations: HS, healthy subjects; IQR, interquartile range; VN, vagus nerve.
Chi-square test.
Student’s t-test.
Mann–Whitney U test.
There was no significant correlation detected between disease duration and VN dimensions in the PD group. A moderately significant negative correlation was observed between MDS-UPDRS III scores and right VN diameter (r = –0.325, P = .044), as well as between total NMS scores and left VN area (r = –0.348, P = .038).
There was a significantly moderately negative correlation between gastrointestinal NMS scores and right VN area (r = –0.499, P = .002), left VN area (r = –0.499, P = .002), right VN diameter (r = –0.378, P = .023), left VN diameter (r = –0.385, P = .021), respectively (see Table 4).
The Correlational Analysis of the Vagus Nerve Dimensions and Parkinson’s Disease Scores.
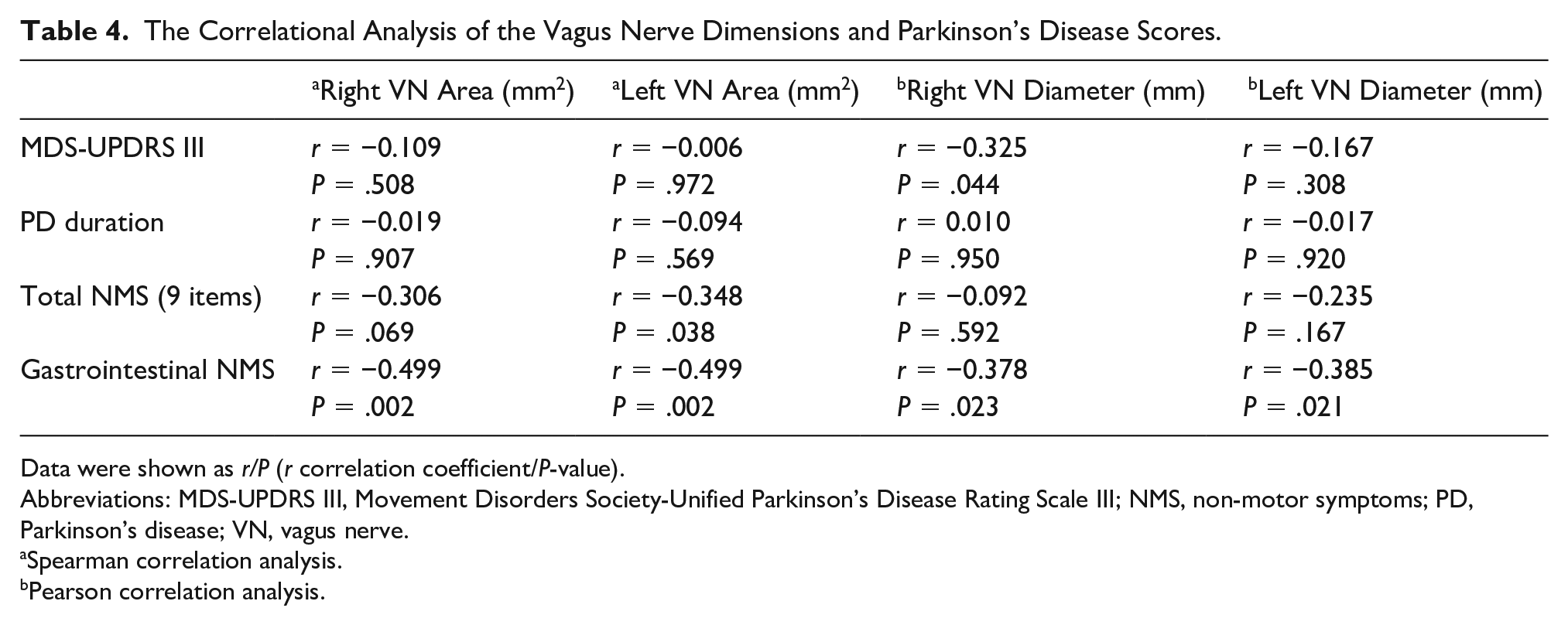
Data were shown as r/P (r correlation coefficient/P-value).
Abbreviations: MDS-UPDRS III, Movement Disorders Society-Unified Parkinson’s Disease Rating Scale III; NMS, non-motor symptoms; PD, Parkinson’s disease; VN, vagus nerve.
Spearman correlation analysis.
Pearson correlation analysis.
Discussion
Using US of the VN, this study demonstrated that these PD patients experienced a significant decrease in VN area and diameter, compared with the normal population. This could be explained by the decrease in overall VN size caused by the axonal degeneration associated with PD. This study also showed that there was no statistically significant difference between the area and diameter of the VN in PD and HS subgroups, and no significant age-related changes in VN dimensions in neither PD nor HS subgroups were observed. A statistically significant negative correlation was found between gastrointestinal NMS scores and the right and left VN area and diameter. These findings suggest that non-motor gastrointestinal symptoms seen in PD and may be related to axonal degeneration of VN.
Neuropathological studies have shown that the main pathophysiological mechanisms in PD affecting the gastrointestinal tract depend on the accumulation of α-synuclein inclusions (Lewy neuritis) in the enteric nervous system and the VN’s dorsal motor nucleus. 3 In their autopsy study, Del Treichi et al 20 showed that axonal degeneration also played a role in the development of NMS in PD, as well as dorsal motor nucleus degeneration.
US of the VN is an imaging technique that has been used effectively in the evaluation of the cervical VN. Some studies have employed US to demonstrate the effects of diseases that can influence VN.21–23 At the cervical level, high-resolution linear transducers can easily distinguish the VN from other anatomical structures, within the vagina caroticum. In their study with 35 PD and 35 control patients, Pelz et al 24 used US to examine the possible effects of PD on the VN. They reported significant atrophy in the VN area of PD patients thought to be related with the axonal degeneration of VN. The authors did not use the coincidental presence of diabetic neuropathy as an exclusion criterion. The evidence provided in the literature demonstrates that diabetic polyneuropathy can be one of the causes of vagal nerve atrophy. 21 In contrast, this study excluded patients with diabetes mellitus from the cohort to assess VN atrophy, strictly in PD patients. Walter et al 25 and Tsukita et al 26 have also reported significant statistical decreases in VN areas using US.
In addition to these studies mentioned above, this study evaluated the VN diameter and divided the study population into PD and HS subgroups, as under 65 and over 65 years. The right and left VN areas and diameters were evaluated, in both groups. The present findings showed no differences in VN dimensions between patients under 65 and over 65 years. This may suggest that there may be no age-related change in VN area and diameter, and that VN size changes in PD patients, are due to axonal degeneration.
In addition to the literature, it was important to underscore the relationship between VN dimensions to motor and non-motor symptoms of PD. In this study, there was no significant relationship between VN dimensional changes and total non-motor symptom scores of PD. It was intriguing to see a negative correlation between gastrointestinal NMS scores and decrease of both right and left VN areas and diameters. According to this finding, additionally to the literature, this study clearly depicted that gastrointestinal symptoms due to PD may be caused by degeneration and atrophy of VN.
Limitations
The limitations to this study are primarily due to the research design and the conveniently small sample sizes for both PD patients and control subgroups. There was also a lack of intra- and inter-observer reliability and no comparative evaluation with other imaging techniques. Conversely, there was not any reference value for atrophy of VN, found in the literature. For that reason, it was not possible to give a cut off value for atrophic VN dimensions. This situation should also be considered a limitation to the study.
Conclusion
This research study demonstrated that there is a significant decrease in VN area and diameter in accordance with disease pathophysiology in this cohort of PD patients, imaged with US. In this study, VN atrophy was depicted in PD patients, but was not related to age-related changes. This decrease may demonstrate the axonal degeneration of the VN in the development of PD and both non-motor and motor symptoms. Gastrointestinal symptoms associated with PD may be explained by axonal degeneration and VN size reduction.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
