Abstract
Objective:
Pericardial effusion is a frequent finding in patients who undergo cardiac surgery. There are currently limited data providing information regarding the factors that may contribute to postoperative pericarditis. The aim was to evaluate laboratory and echocardiographic features that may influence the presence of pericardial effusion 6 to 8 weeks following coronary artery bypass grafting (CABG).
Materials and Methods:
This was a prospective cross-sectional study that included 90 patients after CABG operation who were divided into two groups. A total of 32 (35.56%) patients with pericardial effusion on follow-up echocardiography formed the first group and 58 patients without pericardial effusion the second group, which were compared in respect to components that were taken prior to the operation.
Results:
The groups did not differ regarding sex (males 65.62% vs 63.79%, P = .86) or age (59.59 ± 9.29 vs 61.69 ± 10.71, P = .35). Platelet count (184.74 ± 58.79 vs 222.62 ± 88.97, P = .03) and left ventricular (LV) global longitudinal strain (GLS) (−14.64 ± 6.86 vs −16.96 ± 4.1, P = .04) demonstrated statistical significance.
Conclusion:
Prolonged postoperative pericardial effusion in small amounts may be found in patients, with preoperative lower thrombocyte count and LV GLS, which could be possible predisposing factors.
Keywords
Pericardial effusion is a frequent finding in patients who undergo cardiac surgery. Postoperative pericardial effusion may be found in as many as 85% of patients. 1 This form of pericarditis belongs to the group of inflammatory pericardial syndromes, termed postcardiac injury syndromes, which comprise postmyocardial infarction pericarditis, postpericardiotomy syndrome, and posttraumatic pericarditis. 2 Postoperative pericarditis is believed to have an autoimmune nature and it usually appears a few weeks after the pericardial injury during the cardiac surgery. 3 Pericardial effusions are more commonly found after coronary artery bypass grafting (CABG) than other cardiac surgery procedures. 4
There are currently limited data in the medical literature providing information regarding the factors that may contribute to postoperative pericarditis. Therefore, the study aim was to analyze the main laboratory and echocardiographic features that may influence the presence of pericardial effusion 6 to 8 weeks following CABG.
Materials and Methods
This was a prospective cross-sectional study that included 90 patients who underwent CABG procedure from January 2019 to July 2019 in the cardiosurgery department of a tertiary medical center. Exclusion criteria included patients who underwent other cardiac surgery interventions; patients with CABG who had postoperative bleeding that required reoperation; patients who had pericardial effusion prior to intervention; patients with history of chemotherapy, radiotherapy, hypothyroidism, and renal failure; and patients with poor echocardiography window. Demographic and history data, physical examination, laboratory tests, electrocardiography (ECG), and transthoracic echocardiography were obtained for each patient prior to entering the CABG intervention. Six to eight weeks after the surgery, patients were reevaluated by echocardiography, with the main aim of analyzing the pericardium. Patients in whom pericardial effusion was registered formed the first group, whereas those without pericardial effusion formed the second group. Data obtained before the surgery were compared between the two groups.
The study was approved by the Ethical Board of the institution and written informed consent was taken from every patient who entered the study.
Echocardiography
The echocardiography (Philips EPIQ 7C, X5-1 probe, Philips Medical Systems Nederland B.V., Bothell, WA.) examinations and measurements were performed according to the recommendations of the American Society of Echocardiography. 5 Pericardium was analyzed through several echocardiographic projections. Pericardial effusion was defined as the presence of echo-free space between the epicardium and pericardium. The echo-free space was then measured and was classified, according to size, into mild (less than 10 mm), moderate (10-20 mm), and severe (above 20 mm). Left ventricular end diastolic diameter (LVEDD), left ventricular end systolic diameter (LVESD), and septal wall and posterior wall thickness were measured from parasternal M-mode view according to standard criteria. Left ventricular ejection fraction (LVEF) and left ventricular end diastolic volume (LVEDV) were derived from left ventricular (LV) longitudinal strain rate measurements, as there are findings that consider these measurements reliable. 6 Left atrial (LA) diameter was measured in 2D projection at end ventricular systole in parasternal long axis view. Measurement of LA volume was done with area-length method using apical four-chamber and apical two-chamber views at ventricular end systole (maximum LA size), whereas L was measured from back wall to line across hinge points of mitral valve. Calculation of LA volume was made by the following formula: 8 / 3π ([A1][A2] / L). The LA volume index was calculated by dividing LA volume with body surface area. A wall motion score index (WMSI) was evaluated by the 17-segment model and the sum of all scores, acquired from wall kinetics, was divided by the number of segments. The pulsed wave Doppler was used to record trans-mitral flow from the apical four-chamber view. Peak velocity of early (E), late (A) atrial diastolic filling of the Doppler mitral flow, and E/A ratio were calculated as well as deceleration time of E wave. Tissue Doppler imaging was applied in the apical four-chamber view, where pulsed wave tissue Doppler imaging across septal and lateral mitral annulus was used. From the obtained negative deflection, maximal e′ and a′ velocities were measured. The average E/e′ was derived from the above measurements. For speckle tracking echocardiography, images from the apical four-chamber, three-chamber, and two-chamber views with ECG gating were attained. Gray-scale cine loops from three consecutive beats, while patient holding its breath, were obtained. Heart rate variation during acquisition was >10%, whereas frame rates were 30 to 100 Hz. Gain settings were adjusted to optimize the endocardial visualization. The endocardial border was manually adjusted at end systole. The software system then automatically generated the strain values for each segment and the average LV global longitudinal strain (GLS).
Statistical Analysis
Data were expressed as mean ± standard deviation (SD) and percentages. Comparison between parametric variables was performed using the two-tailed unpaired t test, and for categorical variables the χ2 test was used. For all tests, a P value of <.05 was considered significant. MedCalc Software Ltd. Version 20.007 and 20.008 were used to compare the two groups.
Results
From the 90 patients who fulfilled the criteria to be included in the study, 32 (35.56%) patients had pericardial effusion during follow-up echocardiography, 6 to 8 weeks after the CABG procedure. In all these patients, the pericardial effusion was categorized as a small amount, under 10 mm of echo-free space measured during diastole. The average amount of pericardial effusion was 5.11 ± 1.62 mm. None of the patients with pericarditis were symptomatic. According to the study design, patients with postoperative pericardial effusion formed the first group with 32 patients and patients without pericardial effusion on follow-up echocardiography formed the second group with 58 patients. The two groups were compared in respect to parameters that were taken prior to the operation. The groups did not differ in regard to sex (males 65.62% vs 63.79%, P = .86), age (59.59 ± 9.29 vs 61.69 ± 10.71, P = .35), body mass index (27.13 ± 4.05 vs 28.62 ± 4.11, P = .1), and heart rate at rest (83.16 ± 16.52 vs 80.21 ± 12.96, P = .35).
Comparison between the laboratory results is provided in Table 1. Platelet count was the only measured laboratory parameter that resulted with significant difference between groups (184.74 ± 58.79 vs 222.62 ± 88.97, P = .03), as lower platelet count at baseline tends to be associated with postoperative pericardial effusion. Nine patients (28.12%) from the first group had platelet count <150 × 109/L, whereas 10 (17.24%) from the second group, P = .2.
The Comparison of Laboratory Results Between the Two Groups of Study Patients.
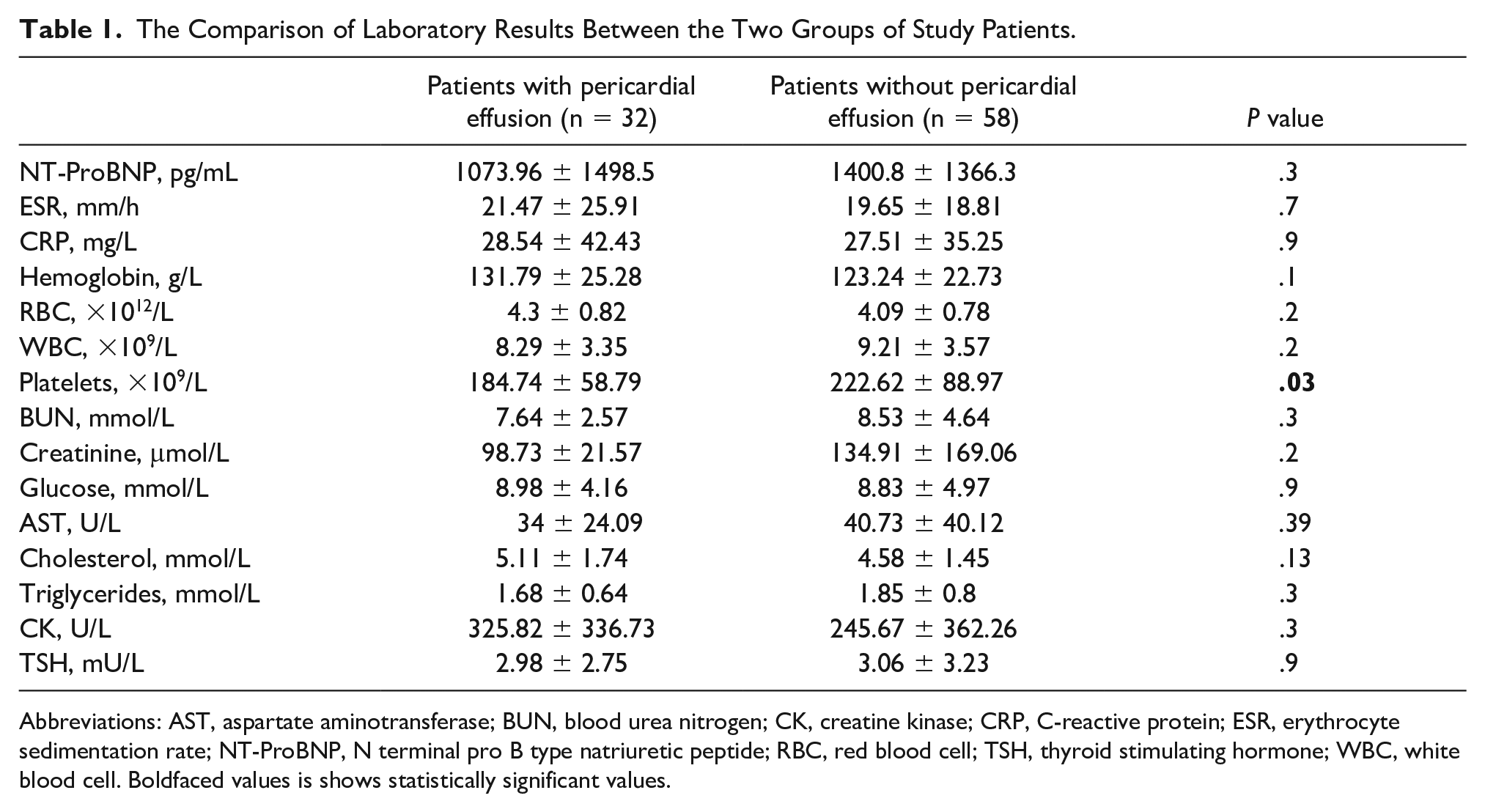
Abbreviations: AST, aspartate aminotransferase; BUN, blood urea nitrogen; CK, creatine kinase; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; NT-ProBNP, N terminal pro B type natriuretic peptide; RBC, red blood cell; TSH, thyroid stimulating hormone; WBC, white blood cell. Boldfaced values is shows statistically significant values.
Comparison between groups in regard to echocardiographic features is presented in Table 2. As seen in this table, LV GLS resulted the only echocardiographic feature analyzed with statistical significance (−14.64 ± 6.86 vs −16.96 ± 4.1, P = .04). Both groups had values below reference range, which can be explained by coronary artery disease in both groups; however, postoperation pericardial effusion group had significantly lower LV GLS values.
A Comparison of Echocardiographic Parameters Between the Two Groups of Study Patients.
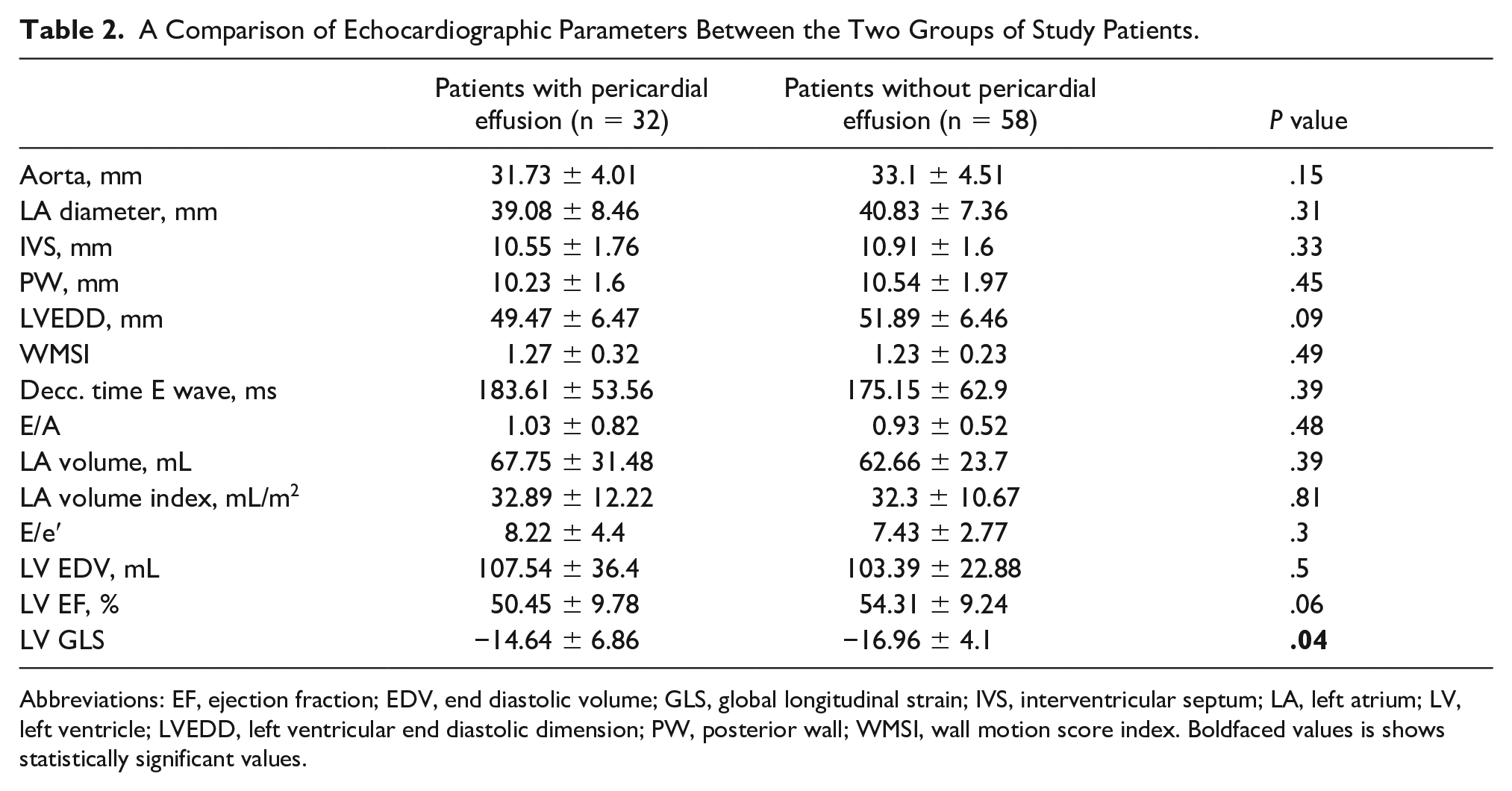
Abbreviations: EF, ejection fraction; EDV, end diastolic volume; GLS, global longitudinal strain; IVS, interventricular septum; LA, left atrium; LV, left ventricle; LVEDD, left ventricular end diastolic dimension; PW, posterior wall; WMSI, wall motion score index. Boldfaced values is shows statistically significant values.
Patients were recommended to take a nonsteroidal anti-inflammatory drug (NSAID) according to current guidelines; however, the definite choice of management of patients with postoperative pericardial effusion was left to the attending surgeon.
Discussion
Although postoperative pericardial effusion is a frequent finding following CABG, fortunately its course is usually benign and they randomly disappear within 7 to 10 days. However, in some instances, pericardial effusion may persist for longer and cause complications. 7 Pepi et al. found that 25 days after the surgery on follow-up echocardiography, of the 149 patients with moderate pericardial effusion, 40 patients had small effusion and 52 remained with moderate pericardial effusion, therefore demonstrating that resolution of moderate effusions may take time. It should be noted that they did not follow-up patients with initial small pericardial effusion and that no small effusions developed into cardiac tamponade or caused hemodynamic disturbances. 4 Weitzman et al. also concluded that pericardial effusion after cardiac surgery is very common, although complications are rare. Nevertheless, they also followed postoperatively patients for 30 days and did not provide data past this period. 8 In the present study, the pericardium was analyzed between sixth and eighth week after CABG and found that small pericardial effusion was present in around one-third of our patients. Although the prognosis of small postoperative pericardial effusions is good, it is believed that treatment is necessary to avoid development of chronic pericarditis.
Several laboratory results, as presented in Table 1, were analyzed prior to CABG with the aim of identifying risk factors for the occurrence of postoperative pericardial effusion. Lower thrombocyte count was the only laboratory parameter that resulted significantly lower in the group that developed pericardial effusion. As mentioned earlier, all pericardial effusions were of small amount, whereas patients who developed postoperative bleeding that required reoperation were excluded. Thrombocytopenia associated with pericarditis is described in a very small number of reports.9,10 However, aside from the well-known role of platelets in hemostasis, lately they have been linked to inflammation as well. For instance, studies have shown that platelets promote inflammatory response in atherosclerosis.11,12 Conversely, Goerge et al. in an experimental mice model found that inflammation plays a key role in inducing hemorrhage in thrombocytopenic patients. This study shows that platelets are necessary to protect smaller venules surrounded by little muscle cells and pericytes as these present the main site of inflammatory hemorrhage in the skin model. Furthermore, inflammatory hemorrhage in thrombocytopenia can occur at any inflamed site. 13 Therefore, it was assumed that the inflammation that was initiated by the surgical procedure lasted longer, than described in the literature, in our group of patients with postoperative pericardial effusion perhaps due to microcirculation hemorrhage that was induced by lower level of thrombocytes and inflammation. Moreover, as reported earlier, histologic findings of parietal pericardium of our patients who underwent CABG demonstrated early inflammatory process, with the presence of granulocytes and lymphocytes. 14
In regard to echocardiographic features, LV GLS was the only echocardiographic feature that demonstrated statistical significance between the two groups in the present study. The LV ejection fraction was also lower in the pericardial effusion group, although the mean value within normal reference range in both groups, however, without significant statistical difference. Nguyen et al. in their study identified LV end diastolic diameter as an independent predictor for postoperative pericardial effusion. They believed that the mismatch between the heart size and the pericardial cavity after surgical repair predisposes development of pericardial effusion. Among other risk factors associated with pericardial effusion in their study were also large body surface area, preoperative elevation of liver enzymes, and New York Heart Association (NYHA) class III/IV. 15 Conversely, Ashikhmina et al. found body surface area, immunosuppression, pulmonary thromboembolism, renal failure, urgency of operation, type of operation, and prolonged cardiopulmonary bypass as independent predictors for postoperative pericardial effusion. 16 In the present study, neither LV end diastolic diameter nor patient size influenced postoperative effusion; however, pericardial effusion in our patients was of small amount in contrast to the two afore mentioned studies.
Interestingly, a rather novel echocardiographic parameter, LV GLS obtained by speckle tracking echocardiography, resulted the only statistically significant preoperative factor in the group that developed pericardial effusion. Based on a review of the literature, currently there are no data that intertwine pericardial effusion and LV GLS. There is one report that states that the longitudinal deformation pattern of constrictive pericarditis includes preserved septal and reduced longitudinal strain values in LV free wall myocardial segments due to pericardial adhesions. 17 LV GLS is lately recognized as a more sensitive mean for detecting early LV systolic dysfunction than LV ejection fraction. Its role is also increasing in several cardiologic disorders, such as hypertrophic cardiomyopathy, coronary artery disease, and valvular heart disease. 18 The mean values of LV GLS were reduced in both our groups, which is understandable having in mind that all the current study patients had advanced coronary artery disease that required CABG. However, the group that ended up with postoperative pericardial effusion had significantly lower LV GLS values suggesting that this parameter might serve as an echocardiographic indicator for development of pericardial effusion. Further studies with more detailed study design and a larger number of patients should evaluate the importance of LV longitudinal strain in predicting postoperative pericardial effusion.
Conclusion
Postoperative pericardial effusion in small amount may be found in a considerable number of patients, as long as 2 months following CABG. This scenario may warrant medical treatment to avoid chronic pericarditis. Preoperative lower thrombocyte count may predispose prolonged postoperative pericardial effusion, perhaps due to microcirculation hemorrhage combined with inflammation. Lower LV GLS may be associated with the occurrence of pericardial effusion in patients following CABG.
Footnotes
Author Note
Rrezarta Alihajdaraj is affiliated to Faculty of Medicine, University of Prishtina, Pristina, Kosovo.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
