Abstract
Objective:
The objective of this study was to evaluate the association between reduced fetal thymus size and intrauterine growth restriction (IUGR). This study was devised to determine the association between thymus size and any abnormal Doppler indices within the fetal umbilical artery (UA), as well as the middle cerebral artery (MCA).
Materials and Methods:
Forty-six pregnancies between 20 and 38 weeks of gestation with IUGR and 46 normal pregnancies within similar gestational age (GA) range were included. The transverse diameter of fetal thymus was measured. In the IUGR group, the fetal umbilical artery (UA) and middle cerebral artery (MCA) Doppler flow velocities were recorded.
Results:
The mean GA of fetuses with IUGR (33.5 weeks) was higher than control group (30.3 weeks). To adjust for the effect of GA, analysis of covariance (ANCOVA) was performed. The adjusted mean thymus diameters were 19.02 mm in IUGR and 21.25 within the control group (mean difference = 2.23 mm; P = .02). The mean (±SD) thymus size in 16 fetuses, with abnormal Doppler findings, was significantly lower than in the group with normal Doppler findings, 17.45 (±2.50) vs 22.02 (±5.39) mm; P < .001.
Conclusion:
IUGR may be associated with reduced fetal thymus size, especially when coupled with abnormal Doppler findings. The thymus size in a group of IUGR fetuses, with abnormal Doppler findings, was smaller than IUGR fetuses, with normal Doppler findings.
The thymus is a primary lymphoid organ with immune functions that has an important role in fetal T cell lymphocytes development. 1 Under normal conditions, the maximal diameters and perimeters of the thymus in fetuses measured during obstetric sonography usually increases as gestational age (GA) advances. 2 Tangshewinsirikul and Panburana reported that 50th percentile of thymus maximal diameters at 20th, 30th, and 38th weeks of gestation were 15.8 mm, 30.9 mm, and 40.1 mm, respectively. 2
Some studies have shown a possible association between abnormal fetal thymus size with adverse pre- and postnatal outcomes. For instance, Aksakal et al reported that small fetal thymuses (<5th percentile), measured between 24 and 37 weeks of GA, had a sensitivity of 91% and specificity of 81%, in predicting histological chorioamnionitis, as well as women with preterm premature rupture of membranes (PROM). Of the 27 fetuses whose thymus transverse diameter was below the 5th percentile, 22 patients had histological chorioamnionitis. Conversely, only 2 out of 23 patients, whose thymus diameter was above the 5th percentile, developed chorioamnionitis. 3 Reduced thymus size has also been reported to have an association with preeclampsia. 4 Eviston et al, in their case-control study of 53 preeclamptic and 120 healthy pregnancies, reported that the fetal thymus was smaller in preeclamptic pregnancies (16.2 mm) compared with control group (18.2 mm) with a mean difference of 2.1 mm. 4 This difference was independent of other measurements including GA, maternal body mass index (BMI), and anthropometry indices of the fetuses, such as the head and abdominal circumference (HC, AC) and femur length (FL). 4
Some studies have also evaluated intrauterine growth restriction (IUGR) with fetal thymus size.5,6 For example, Cromi et al described the ratios of fetal thymus perimeter to biometric parameters, such as the biparietal diameter (BPD) and FL, in fetuses with IUGR and showed that this ratio is significantly lower in comparison to control fetuses with similar GA. 5 Likewise, Olearo et al and Ekin et al found that thymus size in IUGR fetuses was significantly smaller than a control group of healthy uncomplicated pregnancies.6,7 In addition, they reported that thymus size in IUGR fetuses with abnormal umbilical artery (UA) and uterine artery Doppler flow was smaller compared with those with normal flow.
Neuroendocrine adaptation in response to malnutrition caused by placental insufficiency or changes in enzyme expression of syncytiotrophoblasts have been proposed, as the possible mechanisms of thymic involution. 7 Moreover, an intrauterine infection with accompanying inflammatory response can lead to thymocyte depletion and reduction in the thymus size. 6 There are some inconsistencies between the results of the primary studies. To underscore these inconsistencies, Cromi et al 5 and Ekin et al 7 showed that thymus size was smaller in IUGR fetuses compared with a control with normal growth; however, in a recent meta-analysis, this was disputed. Caissutti et al found that when IUGR was described as a birthweight <5th or in the 10th percentiles, no significant association was noted between thymus size and restricted fetal growth. 8
To some degree, the differences in the limited number of publications on this topic may be justified by differences in the definitions, size or volume measurement of the thymus, and sonographic methods. Hence, it seems that more studies are still required to add to the evidence about fetal thymus size and IUGR. Therefore, this study was executed to evaluate the association between IUGR and reduced fetal thymus size measured during obstetric sonography. In addition, the aim was to determine whether an association existed between decreased thymus size and abnormal Doppler flow velocity waveforms, in fetuses with IUGR.
Materials and Methods
Design
The study was performed in a university hospital and lasted 10 months. The study protocol was approved by the Medical Ethical Committee of the university. Written informed consent was obtained from pregnant women.
Forty-six pregnancies with IUGR and 46 normal pregnancies were included. The study subjects were selected consecutively from women referred to the department for routine or clinically indicated obstetric sonography. Inclusion criteria were singleton pregnancy, a GA between 20 and 38 weeks, intact membranes, absence of known maternal or fetal infection, absence of known structural malformation or chromosomal abnormality, and a lack of active labor during sonography.
Sonographic Examination
All participants underwent a 2D prenatal sonographic examination by a radiology resident using a GE Voluson E6 (GE Medical Systems, Zipf, Austria) equipment system. Fetal biometric parameters including AC and amniotic fluid index (AFI) were measured. A sonographic estimated fetal weight (EFW), in grams, was calculated on the equipment system. IUGR was defined as a sonographic measurement of fetal AC< 10th percentile for GA. 9 GA was calculated based on the last menstrual period (LMP) and crown-rump length (CRL) measurement in the first trimester (11–14 weeks of gestation), during the sonogram. In the case of a disagreement between the two methods, serial sonograms were performed in the first and second trimesters and were evaluated to determine GA.
In pregnancies with IUGR, fetal UA and middle cerebral artery (MCA) Doppler flow velocity waveforms were studied. UA resistance index > 95th percentile, MCA RI < 5th percentile, and UA RI to MCA RI ratio > 95th percentile for GA were considered abnormal. During the sonogram, the transverse diameter of fetal thymus was measured for all participants. The attending radiologist, experienced in obstetric imaging, made thymus measurements, in the transverse section of the fetal thorax, at the level of great vessels (three-vessel view). In this view, the thymus appeared as a homogeneous structure between the sternum and the great vessels of the heart. Measurement of the maximum transverse diameter of thymus was in axial plane perpendicular to the line connecting the fetal sternum and the spine. The measurement was repeated three times and the mean of these measurements was considered as the final thymus transverse diameter. If obtaining the optimal view of the thymus, for measuring its maximum transverse diameter, was impossible due to oligohydramnios, fetal position, or maternal obesity, the case was excluded.
Statistical Analyses
The statistical analyses were performed using SPSS software version 22.0 (IBM Corp., NY, USA). Descriptive indices including frequency, percentage, mean, median, and standard deviation (SD) were used to express data. To determine the normality of the continuous variables, for the IUGR and control groups, the Kolmogorov-Smirnov test, visual inspection of histograms, and comparison of mean and median values were completed. Since maternal age and GA had nonnormal distributions, the Mann-Whitney U test was used, for comparison between groups. Likewise, thymus transverse diameter had nonnormal distributions in the two groups (especially in control group), as well as in the groups with normal and abnormal Doppler findings. Hence, the Mann-Whitney U test was used to compare thymus transverse diameters between the two groups. Conversely, the EFW and AFI had normal distributions, within the groups; therefore, the Student t test was used for comparisons. The statistical significance was set at P < .05 a priori. In addition, a Pearson correlation coefficient was calculated to determine the strength of association between the transverse diameter of the thymus and GA. Since GA is related directly to thymus size and GA of the IUGR group was significantly higher than in control group, comparison of mean thymus diameters could lead to a false interpretation. Therefore, it was decided to conduct analysis of covariance (ANCOVA), an advanced form of analysis of variance (ANOVA), which combines both ANOVA and regression analysis. This was done to take off the covariate effect of GA and to investigate the adjusted mean values of thymus diameters.
Results
A total of 92 fetuses were included (46 fetuses diagnosed to have IUGR and 46 normal fetuses). Mean (±SD) maternal age was 30.06 (± 6.33) years (range, 17–43 years). Gestational diabetes and preeclampsia were not diagnosed in any of the mothers. Table 1 shows comparison of continuous variables between the two study groups. As observed, GA was higher in the IUGR group than in the control group. However, there were no significant differences between the two groups in terms of AFI and estimated fetal weight.
The Comparison of Maternal Age, AFI, Estimated Fetal Weight, and Gestational Age Between IUGR and Control Groups.
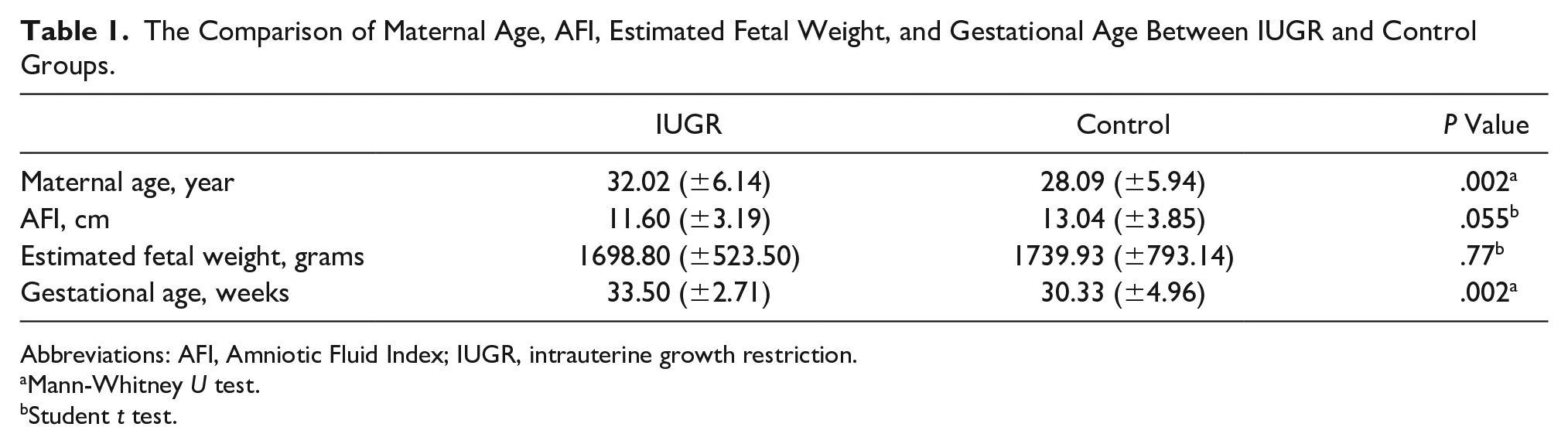
Abbreviations: AFI, Amniotic Fluid Index; IUGR, intrauterine growth restriction.
Mann-Whitney U test.
Student t test.
Mean (±SD) thymus transverse diameter was 20.14 (±5.47) mm in the whole sample (range, 10.50–37.99 mm). The mean and ±SD for thymus size in IUGR and normal fetuses groups were 20.43 (±5.06) and 19.84 (±5.89) mm, respectively. See Figures 1 and 2 for examples of a selected fetal thymus, captured with sonography.
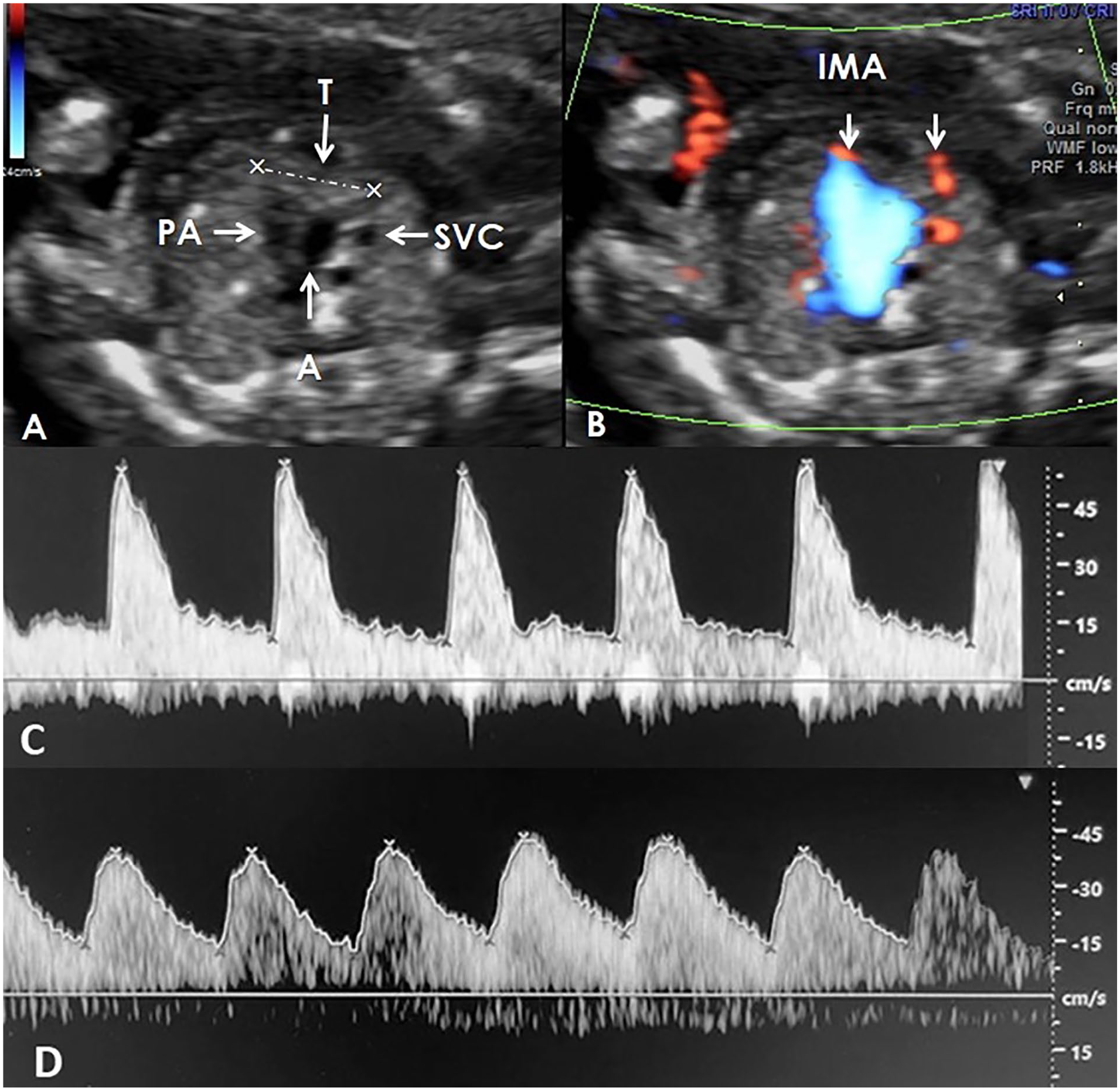
(A) and (B) Measurement of the transverse diameter of the thymus, in a normal fetus with the gestational age of 24w + 2d, according to the first trimester sonogram. (C) The middle cerebral artery Doppler waveform. It provided a pulsatility index (PI) of 1.89, and a resistive index (RI) of 0.80 (about the 50th percentile for gestational age). (D) The umbilical artery Doppler waveform. It provided a PI of 0.96 and an RI of 0.64 (between the 5th and the 50th percentile for gestational age). A, aorta; IMA, internal mammary arteries; PA, pulmonary artery; SVC, superior vena cava; T, thymus.
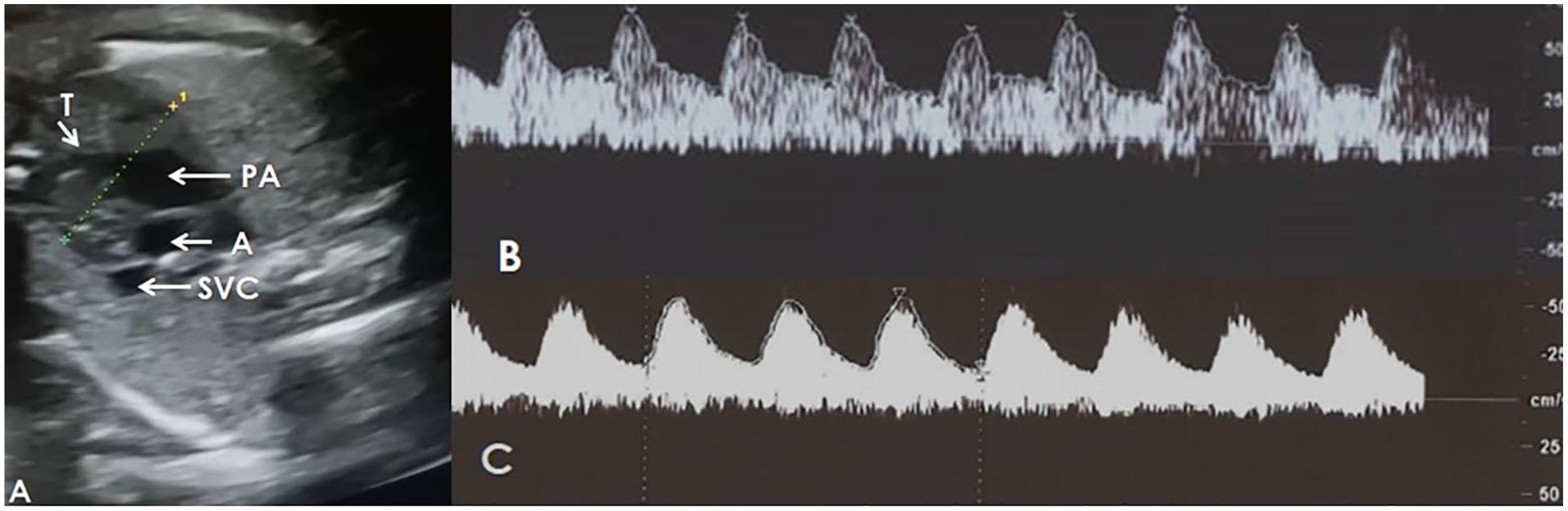
(A) The measurement of the transverse diameter of the thymus in a fetus with the gestational age of 30w + 3d, according to the first trimester ultrasound. The abdominal circumference was measured at 224.8 mm (below the 3rd percentage for gestational age). (B) The middle cerebral artery Doppler waveform. It provided a pulsatility index (PI) of 0.82 and a resistive Index (RI) of 0.52 (below the 5th percentile for gestational age). (C) The umbilical artery Doppler waveform. It provided a PI of 1.31 and an RI of 0.76 (about the 95th percentile for gestational age). A, aorta; PA, pulmonary artery; SVC, superior vena cava; T, thymus.
A correlation was observed between GA and thymus diameter (r = 0.62; P < .001). Since GA had a significant correlation with thymus size and in the bivariate comparisons (see Table 1), GA of IUGR group was higher than in control group, it was necessary to perform ANCOVA to account for the covariate GA. The analysis (See Table 2) showed that thymus size had significant association with IUGR. Adjusted mean value of the thymus size was 19.02 mm in IUGR group. This was significantly lower than the adjusted mean of thymus diameter in normal group (21.25 mm); P = .02 (see Table 3).
The Analysis of Covariance Model for the Covariate Effect of Gestational Age on the Dependent Variable (Thymus Diameter).

Abbreviation: IUGR, intrauterine growth restriction.
The Adjusted Mean Thymus Size and Mean Difference in IUGR and Normal Fetuses.
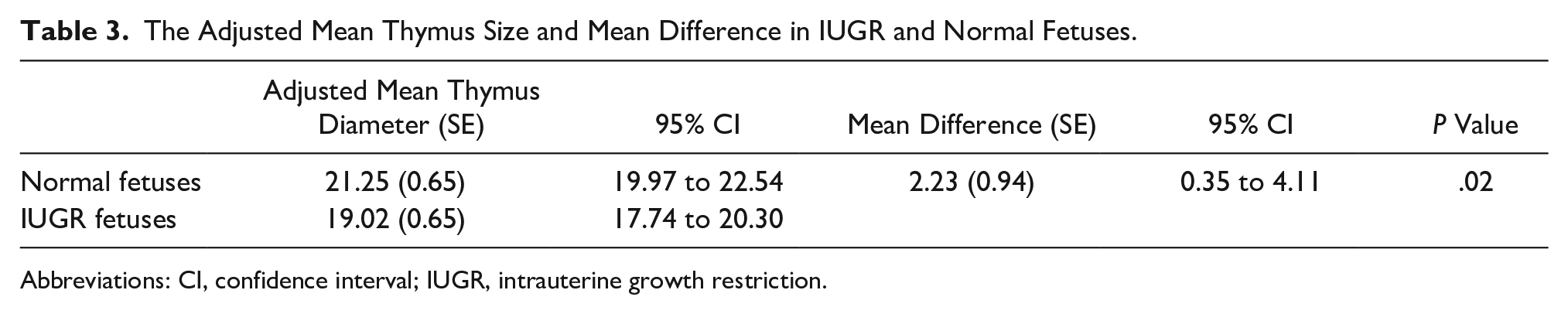
Abbreviations: CI, confidence interval; IUGR, intrauterine growth restriction.
Doppler Sonography
Of 46 IUGR fetuses, the Doppler demonstrated an abnormal finding in 16 fetuses (34.78%). The GA in the abnormal Doppler group was 32.75 weeks, and it was 33.90 weeks in fetuses with normal Doppler findings (P = .17). The mean (±SD) thymus transverse diameter in 16 fetuses, with an abnormal Doppler, was significantly lower than in the group with normal Doppler findings; 17.45 (2.50) vs 22.02 (5.39) mm; P < .001. The median thymus size in the abnormal Doppler group was 17.41 mm, and it was 20.85 mm in fetuses with normal Doppler results.
These results demonstrated that although no statistically significant difference existed between normal and IUGR groups, regarding unadjusted mean fetal thymus transverse diameter, this could be attributed to difference in GA between normal (mean GA of 30 weeks) and IUGR (mean GA of 33 weeks) groups. By adjusting the values for GA, it was evident that fetuses with IUGR had in fact a smaller fetal thymus, than the normal group.
Discussion
Measuring thymus size, to define its normal range, has been done, and the evidence published by Asghar et al showed that fetal thymus diameters and volume increase with gestational weeks. 10 Based on a limited literature search, a few studies have directly evaluated the association between IUGR and thymic involution. Confirming the present study, Cromi et al found that the ratio of thymus perimeter to BPD, FL, and EFW, in IUGR fetuses (>20th gestational week), were significantly lower than their control group. 5 Olearo et al demonstrated that fetal thymic volume/AC ratio, in a control group, was higher than IUGR and SGA (small for gestational age) fetuses, measured at 20 to 37 weeks of gestation. 6 Similarly, Ekin et al showed that transverse diameter of the fetal thymus was significantly lower in IUGR group compared with control fetuses at 24 to 40 weeks’ gestation. 7 Brandt et al evaluated the association between second trimester fetal thymus measurements and adverse obstetric outcomes. 11 Their results are inconsistent with the findings of the current study and those of afore mentioned studies. These investigators found no association between thymus measurements and SGA or pregnancy-related hypertension. This discordant result could be the consequence of using a different set of inclusion criteria. Brandt et al included pregnant women at GA of 18 to 23 weeks, but in the present study, pregnant women were recruited with a GA between 20 and 38 weeks. 11 It is likely that the fetal thymus size in the third trimester is more associated with IUGR and SGA.
Various underlying mechanisms have been proposed for this association. Olearo et al. has suggested that the same trigger, like an early subclinical infection, could affect trophoblastic invasion, resulting in placental insufficiency, IUGR, and thymic involution, due to thymocytes depletion. 6 The association between intrauterine infections and preeclampsia with infections and thymic involution, may support this theory. Another proposed mechanism is the neuroendocrine adaptation due to placental insufficiency. In a physiological state, a balance between leptin and glucocorticoids manages the apoptosis of thymocytes. Leptin inhibits thymocyte apoptosis with two mechanisms: direct inhibition of thymocyte apoptosis and suppression of glucocorticoids, via the hypothalamic-pituitary-adrenal gland axis. Placental insufficiency results in chronic malnutrition and consequently reduction in leptin level. Reduced leptin level and increased level of glucocorticoids result in more thymocyte apoptosis and thymus involution. 7
Another mechanism that has been proposed for association between IUGR and thymus involution is enzymatic changes. Kosicka et al reported that 11β-hydroxysteroid dehydrogenase 2 (11β-HSD2) is an enzyme that is highly expressed in placenta and catalyzes the conversion of biologically active cortisol to a less active cortisone. 12 They added that reduced activity of placental 11β-HSD2 that occurs due to placental insufficiency and fetal malnutrition in IUGR results in more exposure to maternal cortisol. 12 It has been suggested that overexposure to maternal cortisol can lead to more thymocyte apoptosis as previously explained. 7
Doppler of the uterine artery has been studied more often than UA and MCA in prediction of adverse pregnancy outcomes. 13 Comparison of thymus size between IUGR fetuses with normal and abnormal UA and MCA findings showed that fetuses with abnormal Doppler values had a smaller thymus size. This observation has been shown to agree with other previous studies.6,7 Importance of this finding is that abnormal UA and MCA Doppler findings, in IUGR fetuses, is associated with poorer perinatal outcomes. These fetuses have a higher risk for the unfavorable outcomes, like neonatal intensive care unit admission, perinatal mortality, and neonatal complications. 14 One of the proposed underlying mechanisms for abnormal UA Doppler waveforms, in IUGR, is vasoconstriction. This occurs at the placental bed, due to reduction in oxygen delivery to fetus, caused by placental insufficiency.15,16 Deletions of genes located on chromosome 22q11.2 can lead to disruptions in the development of the third and fourth pharyngeal pouches, hypoplasia (75% of cases), or total aplasia (<1% of cases), of the thymus. These deletions can lead to a variable phenotype, including DiGeorge syndrome (e.g., immunodeficiency, dysmorphic features, and cardiac anomalies). Children with this syndrome may have growth or developmental retardation. In a study on 10 fetuses, diagnosed with DiGeorge syndrome prenatally, IUGR was detected in one fetus. 17 Another study on 42 cases with confirmed 22q11.2 deletion syndromes did not find any indication of IUGR. 18
Limitations
There were some limitations to the present study, due to the nature of using a convenient sample. For the purposes of this study, IUGR was defined as the AC< 10th percentile; therefore, it did not allow for the exclusion of constitutionally small fetuses. In this study, the normal group was not matched to IUGR group, regarding the GA of the pregnancies. However, an attempt was made to overcome this limitation by performing ANCOVA to adjust for the effect of GA. In addition, the transverse diameter was chosen, as the index of the thymus size, but due to the lobulated shape of the thymus, measurement the sonographic 3D volume may be a more accurate index. Finally, it was not possible to follow the pregnancies to assess the outcomes and any complications of the pregnancies during the third trimester or post-partum.
Conclusion
In conclusion, IUGR may be associated with reduced fetal thymus size. In addition, the thymus size in this group of IUGR fetuses, with abnormal Doppler findings, was smaller than IUGR fetuses, with normal Doppler values. It will be important to stage future studies to evaluate the association of reduced thymus size and the different etiologies for IUGR.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
