Abstract
Given the significant morbidity and mortality associated with tubo-ovarian abscess (TOA), prompt diagnosis and initiation of treatment is critical. If a diagnosis is made early, medical management remains an option thereby improving the chance of conservation of reproductive organs. TOA is a sequalae of pelvic inflammatory disease (PID) and the diagnosis remains difficult due to the wide inclusion criteria for this condition, which should exclude other causes of pelvic pain. This case study presents two atypical cases of TOA in which neither patient met the diagnostic criteria for PID, on initial presentation and delayed diagnosis. Sonographic findings of enlarging, complex hyper-vascular masses and markedly elevated CA-125 levels instead raised concerns for malignancy influencing management decisions. In retrospect, utilizing these two predictive diagnostic tools may facilitate an earlier diagnosis of TOA therefore even without overt PID. This becomes more important when the clinical signs support a potential diagnosis of malignancy.
Keywords
Tubo-ovarian abscess (TOA) is a potentially life-threatening condition often resulting from complications of pelvic inflammatory disease (PID). As this condition frequently occurs in women of reproductive age, medical management is preferable to surgical intervention and when surgical treatment is deemed necessary, a conservative approach is desired. 1 Acute PID remains an elusive diagnosis in the clinical setting given the wide variation in presenting signs and symptoms associated with this condition. The Centers for Disease Control and Prevention suggests that treatment be initiated in sexually active young women if they are experiencing pelvic or lower abdominal pain. Treatment is recommended given that PID is the likely cause so long as one or more of the following minimum clinical criteria are present on pelvic examination: cervical motion tenderness or uterine tenderness or adnexal tenderness. 2 Even with this broad inclusion criteria, women may escape diagnosis given they have subtle signs or are entirely asymptomatic. A delay in diagnosis and therefore treatment can lead to significant complications. Known sequalae of PID include infertility, ectopic pregnancy, chronic pelvic pain, recurrent infection, and abscess formation. A TOA occurs in 5% to 15% of women with PID, and rupture of this abscess can lead to sepsis and mortality. 3 Broad spectrum antibiotics are considered as a first-line treatment and have success rates of up to 75% to 85%. 4 Treatment failures result in a need for surgical intervention which can lead to removal of vital organs for reproduction. Therefore, to increase specificity, additional diagnostic criteria have been included in some definitions of PID including fever, mucopurulent cervical or vaginal discharge, elevated erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP), and presence of cervical N gonorrhea or C trachomatis. 3 Despite these additions, the diagnosis remains non-specific so additional tools that raise the suspicion for TOA could ultimately lead to early identification and intervention.
In TOA, the infection generally spreads from the tubes to the ovaries and into the pelvis, resulting in a breakdown of the adnexal structures. Sonographically, this will appear as a complex, multiloculated mass where tubes and ovary are indistinguishable with a poorly delineated mass involving one or both adnexae. 5 There may or may not be free fluid present in the pelvis. Increased color Doppler flow in the multilocular, thick cystic walls is not uncommon, given the inflammatory nature of PID/TOA. However, the increased vascularity can make it hard to distinguish these lesions from a malignant condition. The sonographic features of malignancy are not unlike TOA and include a large mass (>5 cm) with poorly delineated margins, indicating invasion of surrounding tissues. All malignancies require neovascularization, so the presence of increased, high velocity and erratic areas of flow contribute to the concern of potential malignancy. 5 The sonographic differential diagnosis when these types of masses are present can also include hemorrhagic cysts, teratomas, serous or mucinous cystadenomas, or endometriomas, although these tend to display less vascular characteristics and more clearly defined margins. 6
A few studies in the past have suggested a relationship between PID/TOA and CA-125 levels. CA-125 is currently considered the most useful tumor marker in monitoring epithelial ovarian cancer. 7 Elevations in CA-125 invariably put malignant conditions on the differential when an adnexal mass is identified. However, it is well known that CA-125 levels can also be elevated in a variety of benign and inflammatory conditions. 4 This is because CA-125 has been found in the peritoneum and epithelial tissues of Mullerian origin such as the fallopian tubes, with irritation raising these levels.8,9 Despite this, severe elevations in CA-125 >500 to 1000 U/mL are still rare in benign conditions. One report stated that serum CA-125 levels >65 U/mL are associated with benign conditions in approximately only 13% of patients. Furthermore, if levels exceeded 1000 U/mL, only 3% of cases were found to be benign. 7 This rare association may lead to a higher suspicion of malignancy, particularly when a complex adnexal mass is found with sonography. The treatment for malignancy is often surgical and irreversible, while for TOA, it is medical and reversible. Any prognostic indicators facilitating the differentiation is thus significant. This case series provides the clinical and sonographic findings of TOA, associated with significantly elevated CA-125 levels. In addition, these cases demonstrate complex adnexal masses that preceded overt signs or symptoms of typical PID.
Case Series Report
Case 1
Patient was a 23 yo G1P0 with history of elective termination of pregnancy 6 months prior and presented for an annual examination and birth control counseling. Gonorrhea and chlamydia cultures were negative at the time of pregnancy and were also negative at time of annual pelvic examination. After counseling with her primary gynecologist, a decision was made to have a hormonal intrauterine device (IUD) inserted and insertion was uncomplicated. A month later, the patient presented to office for routine IUD follow-up. At that time, she complained of daily cramping and bleeding since insertion. A physical examination was documented as normal including no cervical motion tenderness, non-tender uterus and adnexa upon palpation, and IUD strings visualized. Approximately 2 weeks later, patient called the emergency service line and stated she was continuing to have cramping. She was advised to go to the emergency room for evaluation, where she was told the IUD was in the lower uterine segment based on sonography. The IUD was removed without difficulty in the office that same day. Incidentally, the same sonogram completed in the ER showed a normal right ovary and a complex left ovarian cyst measuring 6 cm. The radiologist interpreted the complex mass as a likely hemorrhagic cyst. Pain was initially thought to be related to improper positioning of the IUD; however, given the complex nature of the ovarian cyst, a CA-125 level was drawn. In addition, the patient was now complaining of non-specific vaginal discharge, therefore cultures were also taken. The CA-125 results were found to be elevated at 471.4 U/mL (normal lab range 0–38.1 U/mL). A repeat sonogram documented a larger 8.3 cm complex, hyper-vascular, multiseptated cystic mass with layering and low-level echoes. Furthermore, the right ovary was now showing a solid amorphous lesion with internal flow measuring 2.8 cm (Figures 1 and 2).
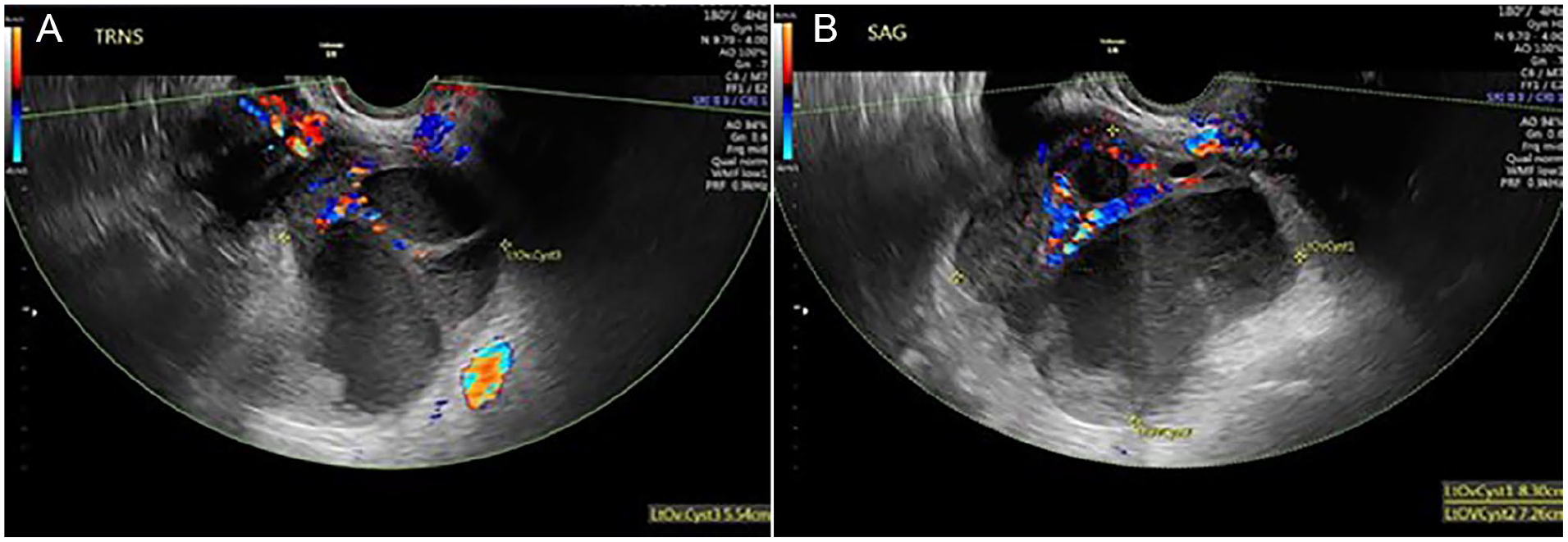
Transverse (A) and longitudinal (B) sonographic images of left adnexal mass.
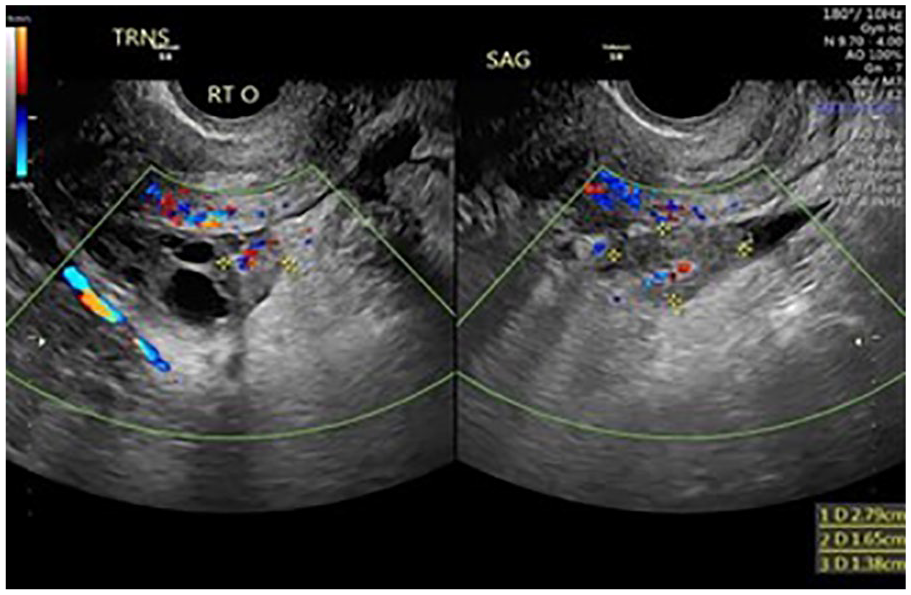
A sonographic image of an echogenic lesion with flow at medial edge of the right ovary.
Due to her cramping in context of pelvic masses and significantly elevated CA-125, surgical consultation was requested with a gynecologic oncologist. To further stratify risk, an OVA-1 test was also obtained and showed an elevated risk of 10 (premenopausal risk cut off is 5) and specified a 70% risk of malignancy. Approximately 1 week after this referral, vaginal cultures resulted positive for chlamydia trachomatis. The gynecologic oncologist conducted an examination that documented a continued absence of cervical motion or adnexal tenderness. It was also noted that the patient had some fullness in the cul-de-sac, without nodularity. In addition, some new tenderness in the patient’s abdomen was documented. Due to the fact that the mass was large and she was now experiencing chronic pelvic pain, surgical intervention was recommended. She was prepared for a laparoscopic ovarian cystectomy and possible oophorectomy. Intra-operative findings, found a TOA and an intraperitoneal drain was placed, and the patient was started on a a course of intravenous antibiotics. A repeat sonogram was completed 1 month after surgery and showed a complete resolution of the bilateral masses, a normal uterus and ovaries.
Case 2
This patient was an 18 yo G0 who presented to our office with a month of intermittent sharp pains associated with bloating. The patient reported that her symptoms were worse with certain foods and therefore she thought that she had appendicitis. In the emergency room, her work up was negative for appendicitis. She was told that she had a simple 2 cm follicular cyst and to follow-up with a gynecologist. The patient had no other pertinent medical or surgical history. There was no tenderness or abnormalities on her bimanual examination. A follow-up sonogram was scheduled with findings remarkable for bilateral complex, multiseptated and vascular cystic masses which contained internal echoes, solid portions and layering. The right adnexal mass measured 5.6 cm with an additional 3 cm simple component and the left adnexal mass measured 5.2 cm (Figures 3 and 4).
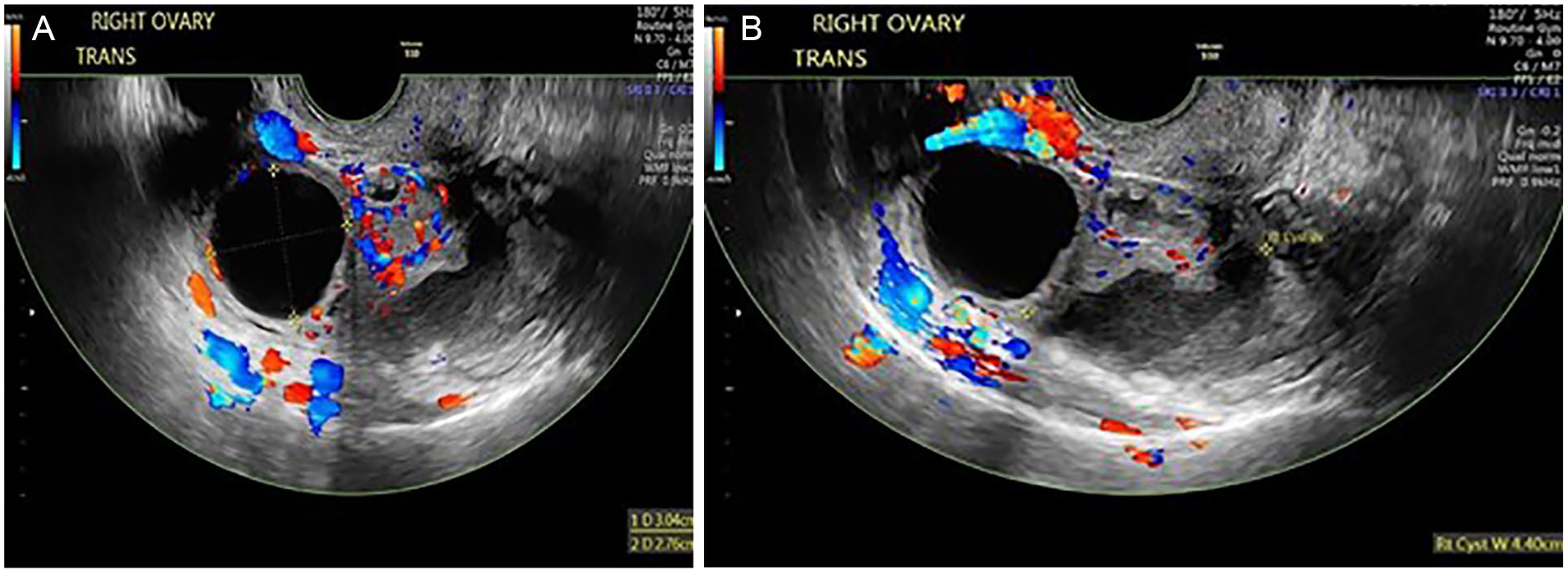
Sonographic views of the right adnexal mass: (A) Transverse view showing hypervascularity. (B) Transverse view, noting the poorly delineated margins of the mass.
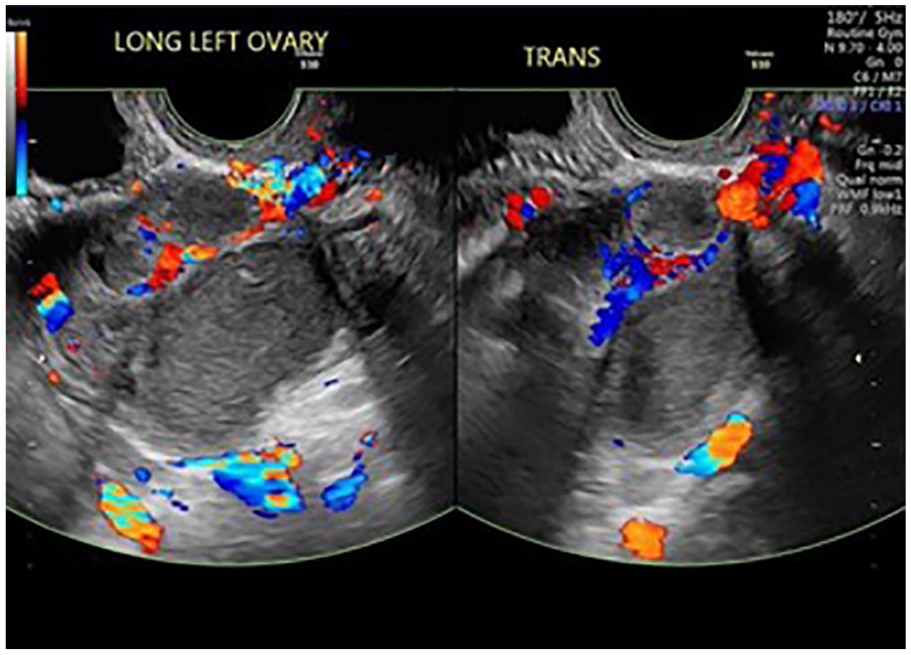
Sonographic views of the left adnexal mass: Longitudinal and transverse images, with color Doppler of the left adnexa mass.
Given the concern for these rapidly enlarging masses, a CA-125 was sent and noted to be markedly elevated at 1057 U/mL (normal value <35 U/mL). A repeat sonogram was done 1 week later and again, there were bilateral complex hyper-vascular adnexal masses with amorphous borders. The right adnexal mass was now measuring 7.2 cm and left adnexal mass was 4.7 cm. Tumor markers of carcinoembryonic antigen (CEA), CA 19–9, alpha-fetoprotein (AFP) were sent for analysis and the results were normal; however, the CA-125 had risen to 1129 U/mL. Neuron-specific enolase and lactate dehydrogenase (LDH) were sent as a result of discussion with gynecologic oncologist, and these biomarkers were elevated. A pelvic magnetic resonance imaging (MRI) examination was ordered for further elucidation of the masses and to assist in surgical planning. The MRI report noted a right normal right ovary with follicles and a 4.5 cm tubular mass which was a suspected hydrosalpinx. In the left adnexa, there was a 4.7 cm mass with 3.4 cm cystic component and nodularity. This lesion was also a suspected hydrosalpinx; however, the report indicated that a tumor or cystadenoma could not be distinguished given the elevated CA-125. The report also documented no enlarged lymph nodes were noted in the pelvis. A white blood cell (WBC) count was repeated and was again noted to be within normal limits. A cervical swab was then done and noted to be positive for chlamydia. Patient was treated with 1 gram of azithromycin orally and follow-up sonogram in 1 month demonstrated significant regression in size and decreased complexity of the bilateral masses, which both now measured approximately 2 cm. The CA-125 had also decreased to a level of 133 U/mL and 2 weeks later decreased to a normal level of 18 U/mL. Six weeks later, a pelvic sonogram was done and showed complete resolution of the adnexal masses and normal ovarian architecture.
Discussion
PID is typically diagnosed if a patient presents with abdominopelvic pain with tenderness on cervical motion and/or adnexa on examination, without any other explanatory diagnosis. Aside from acute lower abdominal pain, a high diagnostic suspicion of PID occurs when a reproductive-aged woman presents with fevers, chills, and a vaginal discharge. When unilateral or bilateral adnexal masses are visualized with ultrasound, a diagnosis of TOA can be made quickly and an appropriate intervention can be initiated. The sonographic features of TOA are typically complex, unilateral or bilateral cystic masses with internal debris, layering, thick wall, tubular or tortuous shape, septations, and free fluid. The ovaries tend to be indistinguishable from the fallopian tubes. 5 Often, there are areas of hypervascularity within these masses seen with color Doppler. This is not surprising given the inflammatory nature of TOA; however, these Doppler findings can also raise the suspicion for a potential malignancy.
This case series illustrates the non-classic presentation of TOA devoid of fever or pelvic examination tenderness, with overlapping features concerning for malignancy. Therefore, it is important to have a high index of suspicion in patients with chronic non-specific abdominal pain and an associated complex adnexal mass. While a CA-125 biomarker is typically drawn in these cases, it would be beneficial to obtain lab values such as white blood count, ESR, and a CRP. The lab results can be paired with cervical testing for gonococcus and chlamydia, as noted in previous studies.4,7–9 However, the results of gonorrhea and chlamydia testing are not readily available in an emergency room setting. Therefore, an elevated CA-125 with a complex hyper-vascular adnexal mass could prompt initiation of antibiotic treatment for presumed PID/TOA. It is also important that a further outpatient diagnostic work-up be pursued to rule out a possible malignancy. As noted in these two patient cases, despite unusually high CA-125 values, a malignancy was not found.
Prior studies have suggested that an abscess diameter greater than 7 cm along with age and bilateral findings may predict need for surgical intervention. 10 These two case reports suggest that despite the presence of these typical predictors associated with need for surgery, conservative medical management can be effective if initiated early. In addition, trending the CA-125 levels and surveillance with sonography can be useful in determining clinical improvement in those patients.
Conclusion
Rapidly enlarging vascular adnexal masses were documented by close surveillance and continuous sonographic examination to determine whether a pelvic malignancy existed. However, it is important to consider that an untreated TOA could behave in a similar manner. Elevated CA-125 values might signal a malignant process, but it can also be noted with many benign diseases. As in malignancy, reduction in CA-125 and decrease in size and complexity of adnexal masses can occur simultaneously with disease resolution, based on this case series. However, unlike an ovarian carcinoma, disease resolution of TOA can often occur without surgical intervention when caught quickly. Therefore, early use of CA-125 in young patients with pelvic pain and a complex adnexal mass may facilitate the diagnosis. Future research could help determine if extremely high CA-125 values and certain sonographic features such as septations and vascularity could add to the diagnostic criteria for TOA. This would be helpful for determining diagnostic options for non-specific pelvic pain in an otherwise healthy young female, especially in an emergency room setting.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
