Abstract
Surveillance of patients with congenital heart disease is difficult in some clinical settings. This case study follows the progress of a patient, born with prenatally missed diagnoses of tetralogy of Fallot (TOF) and total anomalous pulmonary venous return (TAPVR), for over two years. The patient acquired pulmonary vein stenosis following initial TOF/TAPVR repair, which made the case an interesting longitudinal study. Multiple cardiac catheterizations for stents and balloon angioplasties of the pulmonary veins were necessary. This case study illustrates this patient’s success with treatment options such as chemotherapy medications, as well as the failure of each treatment. It was also important to note that the interval of time between each interventional treatment weighed heavily upon results of frequent echocardiograms.
Keywords
Key Takeaways
Always include pertinent information such as color and pulsed spectral Doppler on every examination.
Patients may benefit from sutureless total anomalous pulmonary venous return repair to reduce scar tissue development and abnormal cell collection.
Rapamune treatment may be a resourceful way to prolong the pulmonary vein patency between cardiovascular procedures.
In normal cardiac circulation, the systemic veins drain deoxygenated blood from the body into the right atrium. This flow comes through the tricuspid valve, into the right ventricle, through the pulmonary valve, and into the pulmonary arteries, to be oxygenated. The pulmonary veins drain oxygenated blood from the lungs into the left atrium. This flow comes through the mitral valve, into the left ventricle, through the aortic valve, and out through the aorta. 1
Tetralogy of Fallot (TOF) is a cyanotic congenital heart defect (CHD) that requires surgical intervention. It consists of four abnormal components: a large perimembranous ventricular septal defect, pulmonary stenosis, right ventricular hypertrophy, and an overriding aorta. 2 TOF was first described in 1673, but in 1888, Étienne-Louis Arthur Fallot added the term tetralogy (from the Latin word tetra, which means “four”) given that the four abnormalities were found simultaneously.3,4 TOF is the most common cyanotic congenital heart defect. It is estimated that approximately 7% to 10% of patients with CHD have TOF. 5
Total anomalous pulmonary venous return (TAPVR) is a cyanotic defect in which the pulmonary veins drain abnormally into the right side of the heart. 6 This defect requires that prostaglandin be administered prior to surgical intervention, to stabilize the patient. 7 The first known case of TAPVR was diagnosed by Wilson in 1798. 8 Approximately 1 in every 7809 babies born in the United States has TAPVR, 9 with supracardiac being the most common type. The prevalence of TOF and TAPVR, in the same patient, is considered rare. 10 A number of patients post-TAPVR repair have presented with a difficult to manage pulmonary vein stenosis. This clinical situation has prompted surgeons to opt for a sutureless repair in some cases 11 and could prolong the time between planned interventions.
Case Report
An expecting mother came into the department for a fetal echocardiogram based on a “suspected cardiac defect,” detected sonographically at 35 weeks and three days. The process of diagnosing congenital heart defects, in an extremely active late-gestation fetus, can be challenging. The initial examination demonstrated a large perimembranous ventricular septal defect. However, due to the nature of the movement and dense fetal bone shadowing, additional fetal abnormalities went undetected. This case is devoted to these anomalies, including TOF and TAPVR, and the treatment options for recurring complications.
At birth, this Caucasian male was found to have TOF, with no significant pulmonary stenosis. A chest radiograph was completed and had diagnostic findings consistent with a mild cardiac enlargement and a hazy edema-like appearance (see Figure 1). The infant was exhibiting respiratory issues and was transported to another hospital. At that institution, the infant was diagnosed as having a supracardiac TAPVR to the superior vena cava (see Figure 2) and a dysplastic aortic valve. The patient was again transferred to another medical facility for surgical repair.
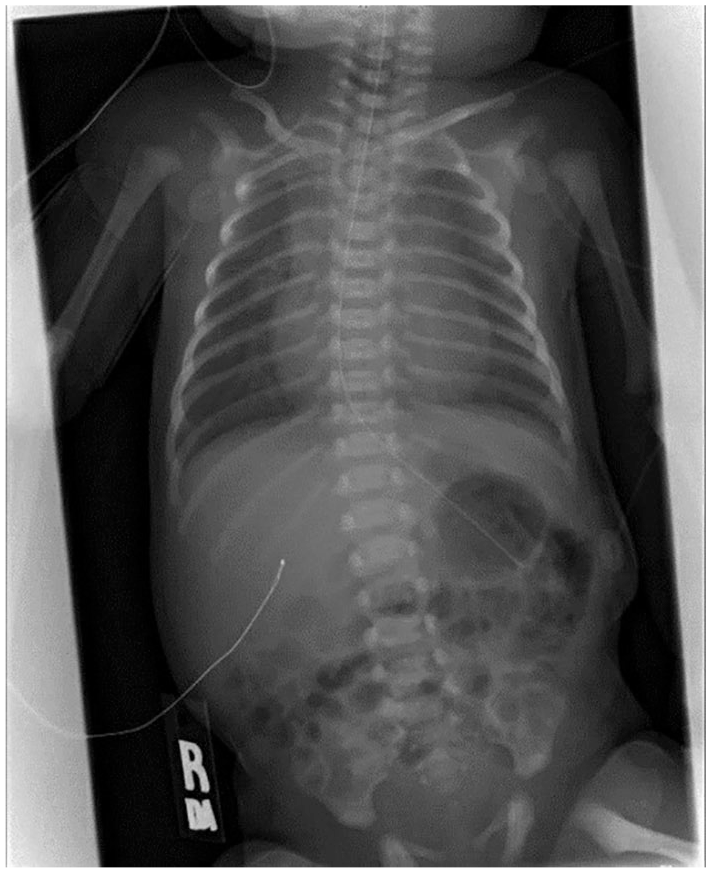
Initial chest radiograph revealing hazy edema-like appearance.
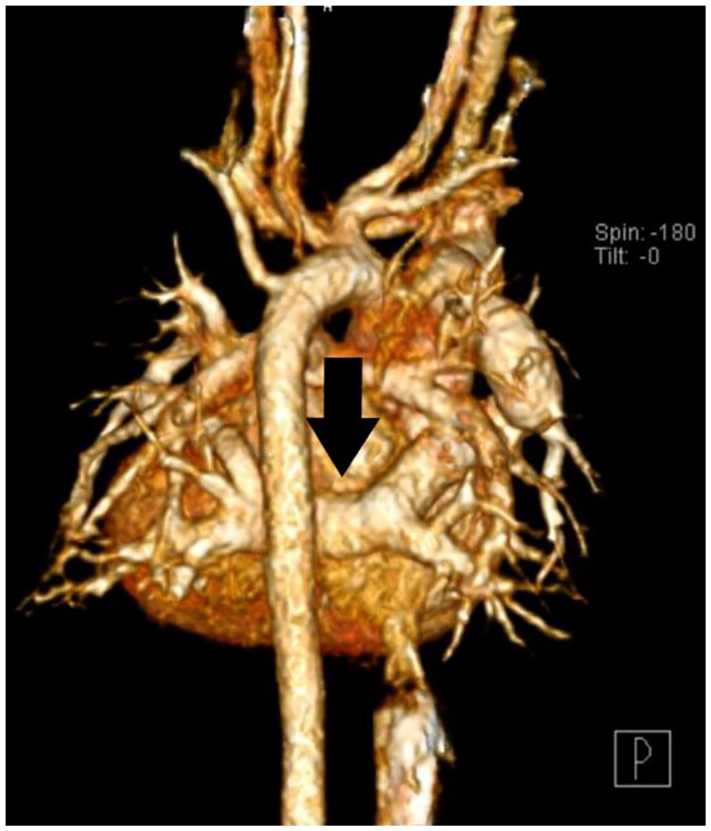
Three-dimensional image of a computed tomography scan demonstrating supracardiac total anomalous pulmonary venous return.
The patient’s first repair was completed when he was six days old, which included a valve-sparing complete repair of the TOF and supracardiac TAPVR. Echocardiograms were beneficial in assessing the development of complications following the open heart surgeries, in a noninvasive manner. The patient’s first postoperative echocardiogram showed a mild flow acceleration across the left pulmonary veins, via color flow and pulsed spectral Doppler. The mean flow gradients were approximately 2 to 3.0 mmHg. The right pulmonary veins appeared unobstructed.
A five-day postoperative echocardiogram revealed moderate flow acceleration across the left lower pulmonary vein. A mild to moderate flow acceleration was noted across the left upper pulmonary vein, with mean gradients of approximately 5 to 6.0 mmHg. The right pulmonary veins were determined to be unobstructed. The right ventricular function had become mildly depressed with a tricuspid valve regurgitation gradient of 36.0 mmHg. At this point, a computed tomogram (CT) was ordered, which revealed a high-grade stenosis at the confluence of the left pulmonary veins. It appeared that the connection of the left pulmonary veins was compressed due to being sandwiched between the heart and the descending aorta. There was stenosis present in the vertical vein as well as linear atelectasis in the right middle lobe. Upon reviewing the CT, performed prior to repair, the surgeon noted that a mild compression was present preoperatively (see Figure 3).
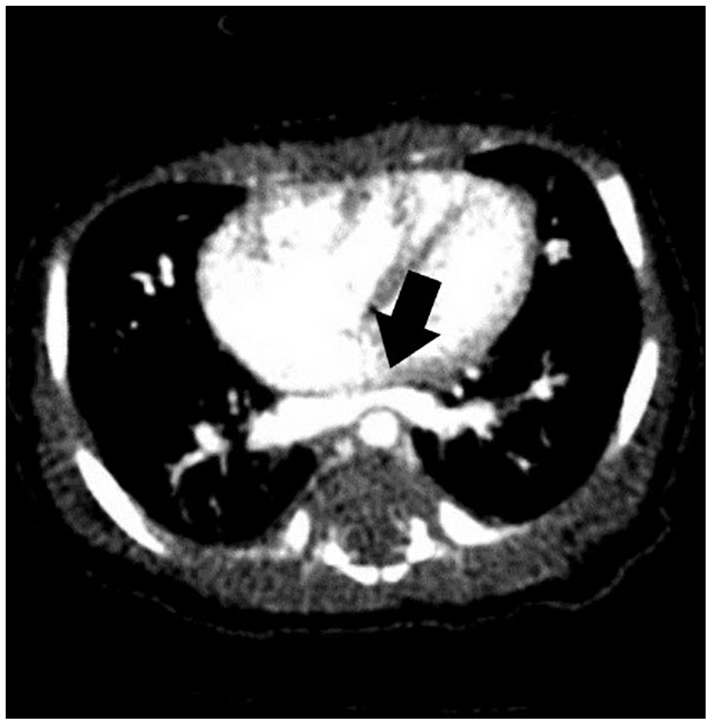
Preoperative computed tomogram demonstrating compression of the vein confluence.
Less than a month and a half after the patient’s first repair for pulmonary vein stenosis, the child returned with a superficial cellulitis of his sternal wound. Antibiotics were administered to help heal the infection. While hospitalized, an echocardiogram was performed, which revealed severe left pulmonary vein stenosis, requiring a return to the operating room (OR). In surgery, the patient’s veins were found to be stenotic and quite small, which was concerning and indicated that the stenosis was very aggressive. The surgeon completed a pericardial wall repair of the left pulmonary veins at that time. A postoperative echocardiogram indicated improvement in the left pulmonary veins, which now had a mean gradient of 2.0 mmHg.
Unfortunately, three weeks later, the patient began vomiting. An echocardiogram was performed, revealing that the left pulmonary vein stenosis was progressing. The stenosis was already exhibiting a mean gradient of 9 to 11.0 mmHg. Due to the worsening stenosis, it was determined that the patient would undergo a cardiac catherization for balloon dilation of the pulmonary veins. This procedure was deemed to be successful. An echocardiogram, less than a month later, revealed increased recurrent left pulmonary vein stenosis, with a mean gradient of 15.0 mmHg (see Figure 4). A moderate pulmonary stenosis of the right pulmonary veins was noted, with a mean gradient of 12.0 mmHg. The patient then underwent another cardiac catherization for angioplasty and stenting of the left pulmonary veins. This procedure produced significant improvement in the pulmonary vein patency (see Figure 5).
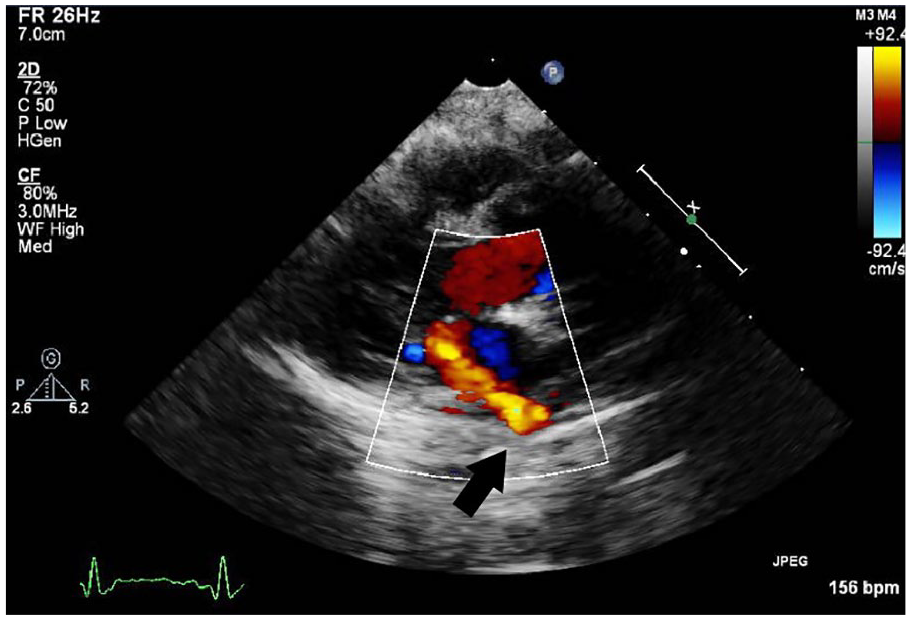
Left lower pulmonary vein stenosis on postoperative echocardiogram.
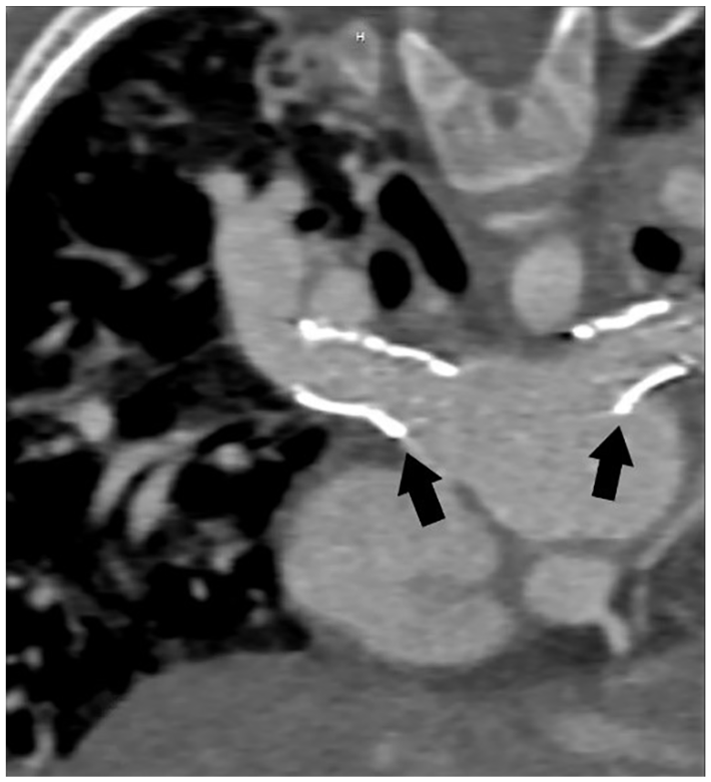
Postcatherization computed tomogram demonstrating pulmonary vein stenting.
At four months of age, the patient had experienced four interventional catheterizations and two open heart surgeries for pulmonary veins. Procedures such as echocardiograms were vital in assessing the progression of pulmonary vein stenosis. CT and cardiac catheterizations were also a common occurrence to the patient’s progress (see Figure 6). In fact, the patient had to have a cardiac catheterization once a month to dilate the pulmonary veins. This continued until the patient was started on a daily liquid chemotherapy medication, Rapamune (sirolimus). Clinical research from Boston Children’s Hospital showed Rapamune aids in decreasing abnormal blood cells that collect in the pulmonary veins, which cause stenosis. 12 The study indicated the need for interventions in patients who are using the Rapamune treatment, but the time between interventional catheterization is greatly increased. The Rapamune treatment surprisingly helped sustain the patency of this patient’s pulmonary veins and allowed for more time between interventions, but not without unintended side effects. This patient has had multiple upper respiratory infections, causing the need for hospital readmission, given that Rapamune is an immunosuppressant drug.
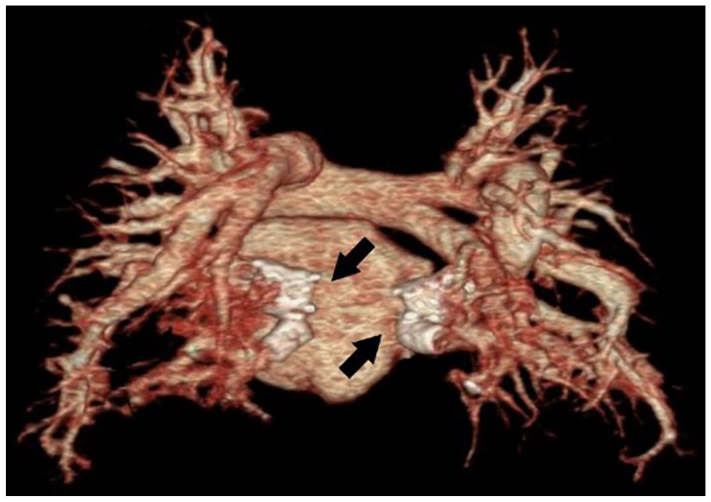
Three-dimensional image of a computed tomography scan demonstrating a total anomalous pulmonary venous return repair.
By two years of age, the patient had endured multiple stents and balloon dilations of his pulmonary veins, and the mean gradients were as high as 31.0 mmHg. The left upper pulmonary vein was difficult to assess via echocardiography as it had nearly regressed. The patient had a bidirectional atrial-level shunt and right ventricular hypertrophy. The patient has had operations for diaphragm plications, a G-tube placement, and a laparoscopic cholecystectomy. The patient also required home oxygen. Unfortunately, the child has also had seizures, head trauma, and a developmental delay, without evidence of a syndrome.
In addition to the aforementioned, the patient experienced Clostridium difficile colitis several times. The child continues to take multiple medications, including Lovenox injections.
Discussion
Research studies have shown that the management of recurrent pulmonary vein stenosis can be challenging. Recurrent pulmonary vein stenosis can occur in up to 18% of post-TAPVR repair patients. 13 Prognosis of patients with pulmonary vein stenosis is incredibly poor, 14 especially if all the veins are affected. 15 While there are necessary surgical interventions, there is only temporary limited success in achieving long-term pulmonary vein patency. Treatment options must be frequent with an aggressive approach to improve patient survival.
A sutureless repair is the best primary repair option for patients with TAPVR, with the hope of preventing a postoperative pulmonary vein stenosis. 16 Once stenosis occurs, a surgical revision is often required to relieve obstruction from the pulmonary venous confluence. 13 Transcatheter stent placements and balloon angioplasties are also warranted. The use of large stents can reduce the risk of restenosis significantly; however, a restenosis remains common after an intervention. 17 Currently, there is no known surgical intervention that can prevent progression of pulmonary vein stenosis. 18 For patients with terminal pulmonary vein stenosis, lung transplantation continues to be an option. 19 The number of patients who have recurrent pulmonary vein stenosis indicates a need for a nonsurgical intervention. Chemotherapeutic agents, such as Rapamune, may be a possible treatment for those with pulmonary vein stenosis to avoid terminal stage disease. 19
Echocardiography with color flow Doppler is a reliable imaging modality to accurately detect the presence and degree of obstruction in pulmonary veins. 8 Frequent echocardiograms allowed for documentation and ongoing information on this patient’s pulmonary vein stenosis. Color flow and pulsed spectral Doppler have been crucial in demonstrating a prolonged stabilization of the pulmonary vein stenosis since starting the Rapamune treatment.
Each intervention this patient needed improved the patency of the pulmonary veins, to some extent. However, prior to the Rapamune treatment, the restenosis of the patient’s veins frequently occurred with greater severity. The patient’s most recent cardiac catherization, at two years and nine months, revealed grossly patent pulmonary veins. This diagnostic finding precluded the need for balloon dilation after seven months, demonstrating the effectiveness of the Rapamune treatment.
Conclusion
As pulmonary vein stenosis increases, the pressure from the stenosis causes strain on the lungs. Without treatment, mortality rates are high. However, the remarkable difference observed in this patient’s pulmonary vein gradients was documented with serial echocardiograms. This was attributed to daily doses of chemotherapy treatment and underscored a method for prolonging the patency of pulmonary veins for recurrent pulmonary vein stenosis. This pediatric patient progressed from having multiple cardiovascular interventions to months of stability between each of these procedures.
