Abstract

Scope
The diagnostic medical sonography profession is comprised of specialties in abdominal, breast, cardiac, obstetrics, gynecology, musculoskeletal, pediatric, phlebology, vascular, and other emerging clinical areas. These diverse areas of sonography all use ultrasound as the primary technology in their daily work.
Sonographers have a responsibility to ensure that the ultrasound transducer (“transducer”) and other equipment used are properly reprocessed. These guidelines specifically address the use, handling, cleaning, disinfection or sterilization, transport, and storage of transducers (hereinafter referred to as “reprocessing”) used in diagnostic medical sonography, but not those used for endoscopic, intracardiac, intravascular, or laparoscopic purposes.
Because of the sensitivity of the transducer’s materials and electronics to some reprocessing techniques, gaps may exist in the ability to perform the desired method of reprocessing for the transducer. Therefore, while these guidelines address current best practices, users of these guidelines should recognize that best practices will evolve over time based on technological advances and new research.
To ensure the facility has the capability and capacity to adequately clean and reprocess the transducer, sonographers should obtain and review the manufacturer’s Instructions for Use (IFU) or other guidance on use, handling, and reprocessing of the manufacturer’s equipment before purchasing. Manufacturers and suppliers should ensure guidance on use, handling, and reprocessing specific to each machine, transducer, or other equipment is easily accessible on their website. Manufacturers and companies that refurbish transducers should take the steps necessary to prevent contamination during or after the manufacturing or refurbishment process. However, regardless of the source, a newly acquired, non-sterile transducer should be processed as if it was used previously.
While these guidelines focus on the reprocessing of the transducer, the importance of infection prevention and control principles apply equally to the ultrasound machine and any ancillary equipment used during the procedure (e.g., cables, keyboards, beds, chairs, IV poles, oxygen systems, cords). Cross-contamination of the ultrasound machine, ancillary equipment, and other surfaces (e.g., gel bottles, light switches, door knobs/handles) presents significant infection prevention and control risks and challenges. Where applicable, refer to the manufacturer’s IFU or the facility’s policies for the proper reprocessing (as applicable) for ultrasound machines, transducers, and any ancillary equipment. New technologies (e.g., transducers connected to smartphones or tablets) will add new risks and challenges that the manufacturer, facilities, and sonographers must address in the future.
Development Process
At the end of 2017, the Society of Diagnostic Medical Sonography’s (SDMS) Sonography Disinfection & Infection Control Task Force developed draft guidelines consistent with existing regulations, standards, and current best practices. In early 2018, the Task Force shared the draft with SDMS members, and sonography and infection prevention and control-related organizations and manufacturers for input. The Task Force met to develop a second draft in early 2019 and shared the second draft with SDMS members, and sonography and infection control-related organizations and manufacturers for input. The Task Force distributed the final draft guidelines for organizational endorsement or support in December 2019 and the guidelines were finalized in May 2020.
Acknowledgements
Jennifer Bagley, MPH, RDMS, RVT, FAIUM, FSDMS (Chair)
Roy Boukidjian, MSN, PHN, CIC, NE-BC
Mary Whitsett, BS, RDMS, RVT, RT(R)(T)
Talisha Hunt, BSRT, RDMS, RDCS, RVT (Board Liaison)
Don Kerns, JD, CAE (Staff Liaison)
Mary Rodriguez, BS, CAE (Staff Liaison)
1. Introduction
Evidence-based and reproducible infection prevention and control practices are essential to ensure that sonography procedures are safe for patients and sonographers. Infection prevention and control guidelines and standards covering sonography have been in place for many years in the United States, but pre-date the rapid expansion of the use of sonography in many medical specialties and procedures.
In particular, those guidelines and standards incorporate the Centers for Disease Control and Prevention (CDC) Guidelines for Disinfection and Sterilization in Healthcare Facilities (published in 2008, reviewed in 2017), the Association for the Advancement of Medical Instrumentation (AAMI) standard ST58:2013: Chemical Sterilization and High-Level Disinfection in Healthcare Facilities, the Food and Drug Administration (FDA), and the AAMI standard ST34:2014: Water for the Reprocessing of Medical Devices, and the Spaulding Classification.1-5 Other transducer reprocessing guidelines have also been released, both in the United States and internationally, including from the following organizations: American Institute for Ultrasound in Medicine, World Federation for Ultrasound in Medicine and Biology, European Society of Radiology, European Committee for Medical Ultrasound Safety, Australasian Society for Ultrasound in Medicine/Australasian College for Infection Prevention and Control, Health Service Executive Ireland and Health Facilities Scotland.6-12
This document builds upon earlier guidelines and provides a comprehensive set of recommendations that any sonographer can use to answer disinfection and transducer reprocessing questions or to implement the recommendations. Sonographers should consult the manufacturer’s IFU for use of appropriate cleaners, disinfectants, and sterilants that have undergone both material compatibility and reprocessing validation and efficacy testing.
2. Infection Prevention and Control Risks and Challenges
Proper infection prevention and control practices are necessary to protect patients and sonographers. Contaminated transducers and ultrasound gel or coupling agent (hereinafter referred to as “gel”) have been associated with infection outbreaks, including those in the bloodstream, urinary tract, respiratory tract, biopsy sites, and wounds. There are also documented cases of hepatitis B and C with some resulting in patient death.13-19 These infections were attributable to a failure to follow evidence-based and reproducible infection prevention and control practices (e.g., proper transducer cleaning and disinfection, appropriate transducer cover, sterile gel). As a result, regulatory, accrediting, and public health agencies, including the FDA, The Joint Commission, and the CDC have issued several alerts regarding transducer use and reprocessing.20-22
Ongoing infection control challenges include changes in microbial resistance and its impact on disinfectants and disinfection practices. The COVID-19 (SARS-CoV-2) pandemic in 2020 underscores the importance of proper infection control and disinfection of all non-disposable equipment used by sonographers. Use of a properly reprocessed transducer with an appropriate transducer cover and gel can reduce the risk of transmitting bacteria and other pathogens during sonographic procedures and promote patient safety.
3. Understanding How the Spaulding Classification Affects Transducer Reprocessing
The Spaulding Classification determines the disinfection and sterilization requirements for medical devices based on the level of infection risk associated with their use. 5 Sterilization is defined as the destruction or inactivation of microorganisms, which minimizes infection transmission risk. Some healthcare professionals and literature refer to “disinfection” as “sterilization”. Medical device reprocessing standards, regulations, and guidelines widely use this classification system.1,2,24-26 For example, the CDC guidelines categorize medical devices reprocessing requirements based on risk using the Spaulding Classification, which has different disinfection and sterilization requirements (Table 1).
How the Spaulding Classification Affects Transducer Reprocessing (adapted from the CDC guidelines 1 )
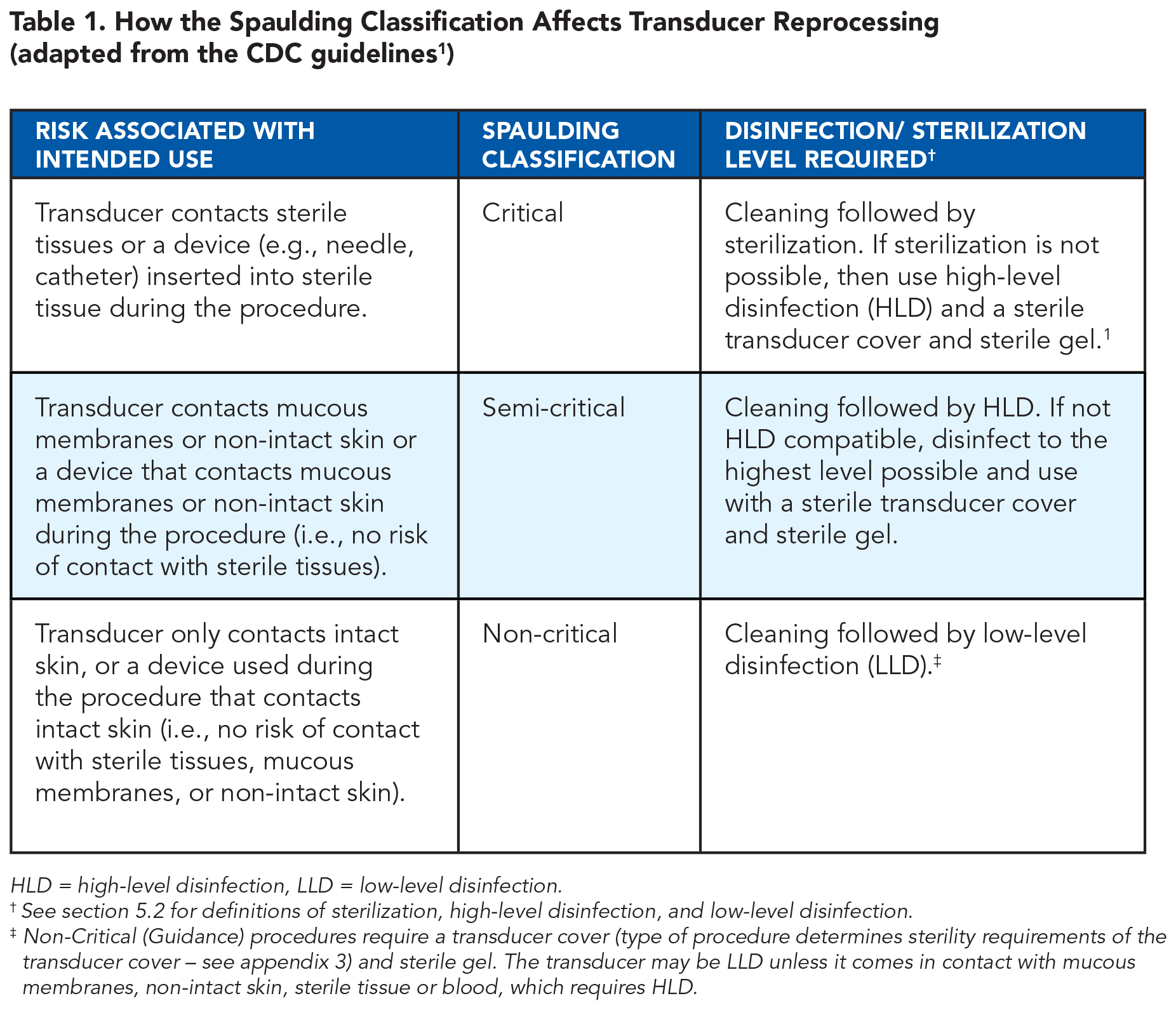
HLD = high-level disinfection, LLD = low-level disinfection.
See section 5.2 for definitions of sterilization, high-level disinfection, and low-level disinfection.
Non-Critical (Guidance) procedures require a transducer cover (type of procedure determines sterility requirements of the transducer cover – see appendix 3) and sterile gel. The transducer may be LLD unless it comes in contact with mucous membranes, non-intact skin, sterile tissue or blood, which requires HLD.
The intended use of the transducer and its contact risk in the procedure determines its expected classification into one of three groups:
are endogenously free from all living organisms. This includes, but is not limited to the vascular system, joints and joint spaces, other internal body fluids (e.g., blood, synovial fluid), the vasculature, internal body organs, peritoneum, and retroperitoneum.
NOTE: Use of a transducer cover does not change the Spaulding Classification or disinfection process as transducer covers have been shown to have micro-perforations and they can break, open, or tear.1,26
The expected classification could change during or after the procedure if the transducer comes in contact with mucous membranes or sterile tissues. The sonographer may classify the same transducer differently when it is used in another procedure. Sonographers should work closely with their facility’s infection control and risk management personnel to determine and document their infection control and disinfection policies and procedures. Any deviations from these policies and procedures (e.g., due to a transducer manufacturer’s IFU limiting the type of approved disinfectant) should be documented and reviewed in accordance with the facility’s policies and procedures.
3.1. Assigning the Expected Spaulding Classification Based on Sonographic Procedure
Make the expected Spaulding Classification determination before the procedure commences. It is sometimes difficult to anticipate what tissues or devices the transducer might contact (e.g., catheter, sterile tissue, mucous membranes, intact, or non-intact skin). Where some risk of contact exists, it may be prudent to apply the higher classification.
4. Sonographic Procedure
Refer to the steps below to prevent infection transmission before, during, and after performing the procedure.
4.1. Before the Procedure
Ensure the procedure area is clean and surfaces have been appropriately disinfected, the transducer has been appropriately reprocessed, and any necessary ancillary equipment is clean for the upcoming procedure.
4.1.1. Select the Correct Transducer
Determine the correct transducer for the procedure, visually inspect the transducer (e.g., damage, visible soil, bioburden), and prepare the transducer based on the Spaulding Classification guidance regarding the procedure. If the patient’s circumstances unexpectedly change before or during the procedure, ensure that the selected transducer is still appropriate based on the reassessment of the procedure and its alignment with the Spaulding Classification.
4.1.2. Select the Correct Transducer Cover
The choice of transducer cover will depend on the procedure to be performed. For critical procedures, a transducer cover is optional with a sterilized transducer, but a sterile transducer cover should be used with a high-level disinfected transducer. 1 At the minimum, use a non- sterile transducer cover for all semi-critical procedures (e.g., endocavitary); however, a sterile cover is preferred. Transducer covers are optional for non-critical procedures, although a transducer cover and sterile gel are required for Guidance procedures (See Appendix 3). Do not reuse transducer covers.
Transducer Cover Selection Based on the Spaulding Classification
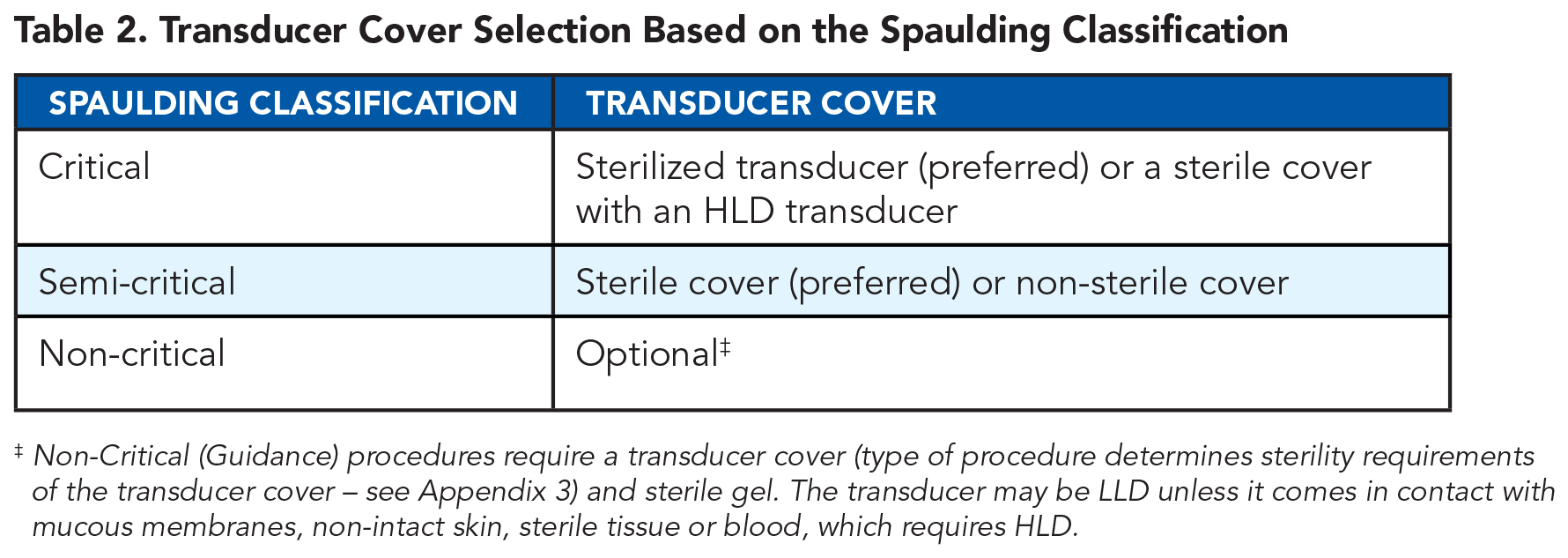
Non-Critical (Guidance) procedures require a transducer cover (type of procedure determines sterility requirements of the transducer cover – see Appendix 3) and sterile gel. The transducer may be LLD unless it comes in contact with mucous membranes, non-intact skin, sterile tissue or blood, which requires HLD.
4.1.3. Select the Correct Ultrasound Gel
A single-use, sterile gel packet is recommended for all critical procedures and is preferable for semi-critical procedures.6,12 A single use, non-sterile gel packet is recommended for all semi-critical procedures and is preferred for non-critical procedures. Do not use a multi-use gel bottle for critical and semi-critical procedures. A multi-use gel bottle is the least preferred option for non-critical procedures due to the potential for patient cross-contamination and gel contamination.
Ultrasound Gel Selection Based on the Spaulding Classification

Non-Critical (Guidance) procedures require a transducer cover (type of procedure determines sterility requirements of the transducer cover – see Appendix 3) and sterile gel. The transducer may be LLD unless it comes in contact with mucous membranes, non-intact skin, sterile tissue or blood, which requires HLD.
Discard unused portions of a single-use gel packet; do not use on other patients. If using a multi-use gel bottle, ensure the tip of a multi-use gel bottle does not come in contact with the patient, transducer, and any ancillary equipment. If the multi-use gel bottle tip comes in contact, discard the multi-use gel bottle. If a multi-use gel bottle is used with an isolation patient or in an isolation precaution setting, discard the multi-use gel bottle. The use of a refilled gel bottle as well as heating of gel is discouraged, as this can lead to microorganism growth, pathogen transmission, cross-contamination, and possible outbreaks.
27
4.2. During the Procedure
During the procedure, use caution to prevent the transfer of pathogens from potential contaminated surfaces (e.g., clothing, non-sterile gloves, other equipment, and environment) to the reprocessed transducer, transducer cover, or gel.
If during the procedure the transducer cover is damaged (e.g., tear, puncture), immediately discard the damaged transducer cover, and replace the damaged transducer cover with a new transducer cover. In such case, the transducer’s expected Spaulding Classification may need to be altered if the transducer came in contact with sterile tissue or mucous membrane because of the damaged cover and may require different reprocessing procedures. If feasible, replace the contaminated transducer with an appropriately reprocessed transducer.
4.3. After the Procedure
After the procedure, inspect and remove the transducer cover (if used) and discard in a designated, approved receptacle, and inspect the contaminated transducer for possible damage. Immediately clean (e.g., remove any remaining gel, visible soil, or bioburden) and disinfect the transducer if classified as non-critical based on the procedure performed. If the transducer is classified as critical or semi-critical, place it in a transport container (e.g., container with lid, impermeable bag), label container as dirty with a biohazard symbol, and deliver it to the reprocessing area. Clean and disinfect all high-touch surfaces in the procedure area including, but not limited to bed railings and the ultrasound machine’s console and controls (see the manufacturer’s IFU or other guidance). Discard waste (e.g., used gloves, wipes, drapes) in a designated, approved receptacle.
5. Transducer Reprocessing
According to the CDC Guidelines for Disinfection and Sterilization in Healthcare Facilities, there are various factors that affect the efficacy of reprocessing including: prior cleaning, removal of bioburden, the organic and inorganic load that is present, type and degree of microbial contamination, concentration and exposure time of the germicide, and the physical features of the transducer or equipment (bevels, crevices, lumens, etc). Additionally, temperature and pH of the process and relative humidity may affect the sterilization process. 1
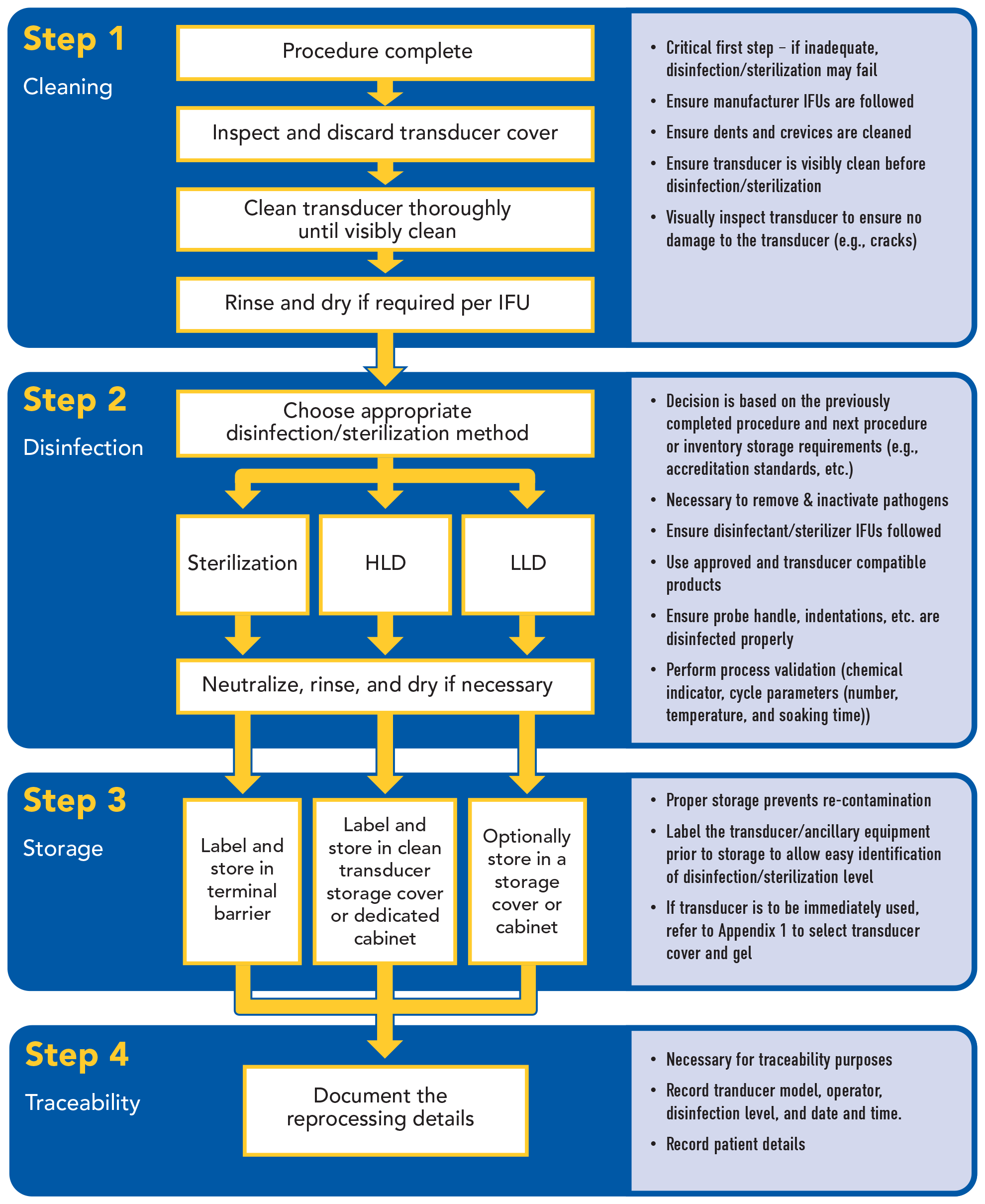
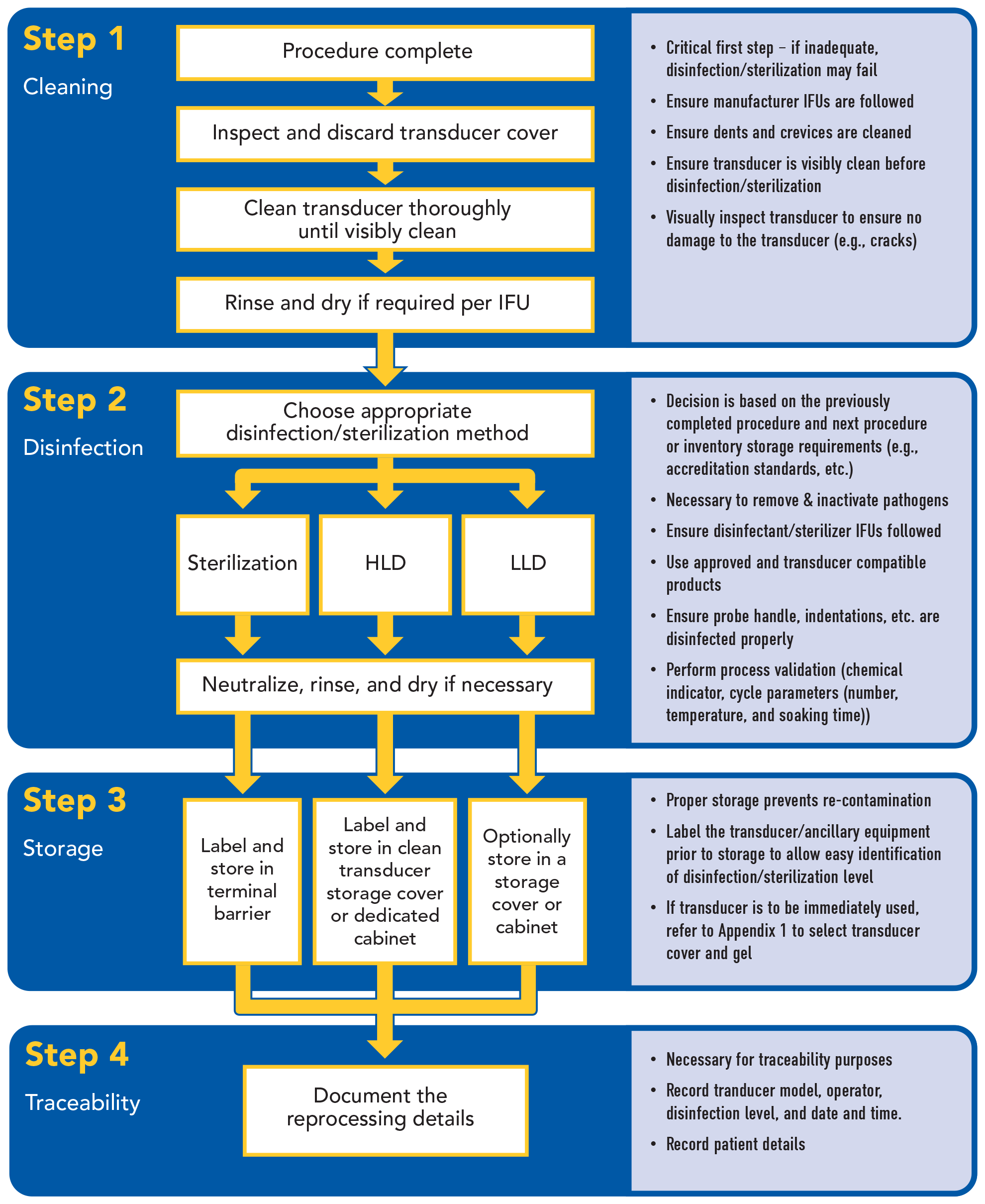
Reprocess the transducer and disinfect any ancillary equipment used between each patient use. Reprocessing refers to the procedure undertaken to prepare an instrument for reuse. Reprocessing includes the cleaning, disinfection or sterilization, transport, and storage. Additional considerations such as traceability are also important. See details below.
5.1. Cleaning
Effective disinfection or sterilization requires adequate cleaning. Cleaning should remove all visible gel, soil, and bioburden on all surfaces of the transducer or any ancillary equipment including any indentations or complex surfaces. Potential cleaning agents for the transducer include neutral pH cleaner, approved wipes, soap and running water, and enzyme soaks. Selection of a cleaning process should factor in manufacturer’s IFU, cleaning efficacy, cost, time, complexity, safety, and designated location for reprocessing.
5.2. Disinfection and Sterilization
Disinfection or sterilization refers to the destruction or inactivation of microorganisms, which in turn minimizes infection transmission risk. Choice of disinfection or sterilization level depends on the Spaulding Classification of the transducer or other ancillary equipment, which is based on use in the prior completed procedure and the intended use in the next procedure. Use of a transducer cover does not change the Spaulding Classification since transducer covers have been shown to leak or tear. 1 The transducer manufacturer’s IFU should be consulted for the recommended disinfection or sterilization methods. Other factors to consider include, but are not limited to disinfection or sterilization efficacy, cost, cycle time, complexity, safety, designated location, and simultaneous transducer reprocessing capability. See below for further detail on sterilization, high-level disinfection, and low-level disinfection.
5.2.1. Sterilization
The FDA defines sterilization as the complete removal of all viable microorganisms, including bacterial endospores, to the extent of achieving a sterility assurance level (SAL) of at least 10-6. 3 Ideally, all critical transducers and devices that are to be inserted into sterile tissue (e.g., needles, catheters) would be sterilized. However, if the transducer cannot be sterilized (see manufacturer’s IFU), HLD is acceptable if the transducer is used with a sterile transducer cover and sterile gel.1,12 Common sterilization options for the transducer include ethylene oxide gas, hydrogen peroxide gas plasma, and liquid chemicals with extended contact times (e.g., glutaraldehyde, ortho-phthalaldehyde). 2 It is critical to ensure that any sterilization process is compatible with the transducer and will not cause damage (see manufacturer’s IFU).
5.2.2. High-Level Disinfection (HLD)
HLD is the removal of all microorganisms except bacterial endospores, of which small numbers are permitted to remain.1,6-8,12 HLD is required for a semi-critical transducer that contacts mucous membranes or non-intact skin. HLD is also suitable for a critical transducer in the event they cannot be sterilized if the transducer is used with a sterile transducer cover.1,12
According to the CDC Guidelines for Disinfection and Sterilization in Healthcare Facilities, unlike sterilization, disinfection is not sporicidal. A few disinfectants, known as chemical sterilants, will kill spores with prolonged exposure times (3–12 hours). At similar concentrations but with shorter exposure periods (e.g., 20 minutes for 2% glutaraldehyde), these same disinfectants are called high-level disinfectants and will kill all microorganisms except large numbers of bacterial spores. 1
Automated HLD methods include hydrogen peroxide mist devices and liquid soak devices using approved liquid chemicals such as glutaraldehyde, ortho-phthalaldehyde, and accelerated hydrogen peroxide. Manual HLD can safely occur using HLD vapor control soaking stations with approved liquid chemicals such as glutaraldehyde, ortho-phthalaldehyde, peracetic acid, hydrogen peroxide, and accelerated hydrogen peroxide. Automated processes are preferable due to the reduced risk of operator error.7,12,28,29
Select HLD methods that are efficient, effective, reliable, reproducible, and safe for the transducer, sonographer, and environment. A process that high-level disinfects the transducer (head and handle) with low level disinfection of cables and connectors is preferred, as these components can harbor clinically relevant pathogens.23,30 Refer to the manufacturer’s IFU to ensure that the HLD process selected is compatible with the transducer and for other suitable HLD options. Refer to the FDA’s website (
5.2.3. Low-Level Disinfection (LLD)
LLD is the inactivation of vegetative bacteria, enveloped viruses, some non-enveloped viruses, and most fungi in a practical period of time (≤10 minutes). 1 LLD is used for a non-critical transducer that only contacts intact skin. 1 Common low-level disinfecting agents include quaternary ammonium compounds, alcohols, and phenols available as sprays and disinfectant wipes. Ensure that the chosen LLD disinfection method is compatible with the transducer. Alcohols are often contraindicated due to material incompatibility.
5.3. Rinsing and Drying
Perform rinsing and drying after disinfection or sterilization if necessary, based on the reprocessing method and required by the manufacturer’s IFU. Thoroughly dry the transducer before storing. These steps may be necessary to ensure that no chemical residues remain on the transducer after reprocessing.
Some automated processes may not require a rinsing step. Follow the disinfectant manufacturer’s IFU for rinse requirements, rinse water quality and sterility, the number of rinses, and drying method. Following HLD, perform the rinses in a separate clean container using critical water (treated water). 4 Sterile water is required for immunocompromised patients.1,2,4,31
5.4. Documentation and Traceability
The facility should develop a written policy outlining the process for documentation and traceability. Documentation and traceability measures are needed for critical and semi-critical transducers requiring sterilization and HLD but may not be necessary for non-critical transducers requiring LLD. 2
Document all reprocessing details including, but not limited to, the transducer model, serial number, or other unique identifier, reprocessing personnel identifier, validation result (e.g., chemical indicator/ test strip, temperature, soaking time), date/time, and patient identifier. 2
To ensure traceability, reprocessing records must be linked to the patient on whom the transducer is used. Traceability is ensured through manual (e.g., logbooks, stickers) or automated processes (e.g., electronic health record, tracking systems). In the event of a reprocessing failure or an outbreak, complete documentation for each transducer reprocessing cycle will aid in traceability for patient trace-back.
5.5. Storage
Proper storage reduces the risk of re-contamination of the transducer from environmental contaminants or accidental contamination. Storage practices, including maximum storage duration should be consistent with the transducer’s intended use, manufacturer’s IFU, and facility’s policies. Suitable options for transducer storage include storage covers, boxes, or cabinets (e.g., HEPA- filtered, ventilated or non-ventilated). Clearly label the container holding the transducer with disinfection level, storage date, and maximum storage duration. Maintain distinct separation of clean and dirty transducers. Dirty transducers should not be placed in the same cabinet as clean transducers.
6. Infection Prevention and Control Considerations for Transducer Reprocessing Workflow
There are many things to consider when establishing an infection prevention and control workflow for transducer reprocessing. Perform reprocessing with a dirty-to-clean workflow. See below for some considerations to assist in streamlining this process.
6.1. Types of Procedures
Consider the type of procedure, location, and reprocessing requirements. In some clinical settings, the transducer may be used for the same type of procedure throughout the day or may be used for a variety of procedures. Therefore, the sonographer should select the transducer based on the intended use. The facility’s policies should address whether to have appropriately disinfected transducers available in storage or to reprocess transducers as needed.
6.2. Reprocessing Time
Consider the length of each patient procedure and the reprocessing time. If the reprocessing time is more than the amount of time available between scans, the facility may need additional transducer inventory. It may be helpful to map out the patient-to-patient workflow, including reprocessing, to identify potential efficiency gains. For example, it may be possible to conduct reprocessing in parallel to the patient change-over process.
6.3. Reprocessing Location and Transportation
After the procedure, immediately remove the transducer cover, excess gel, and other contaminated items (e.g., gowns, sheets) and dispose of them in a designated, approved receptacle. The transducer can potentially be reprocessed in the exam room, if the cleaning and disinfection processes are suitable for use in that environment and a dirty-to-clean workflow is established. 12
If the transducer cannot be safely reprocessed in the exam room, transport the transducer in a designated, approved container to a separate room for reprocessing and then return the transducer in a designated, approved clean or sterile transport container, as appropriate. Handle and transport the transport containers in a manner to prevent contamination of the transducer, other equipment, and the facility. Transport containers used to transport contaminated items by hand should be maintained in a position parallel to the floor. 2 Transport containers and any transport carts should be disinfected. Clearly mark designated, approved containers as clean or dirty with a biohazard symbol.
6.4. Compatibility
The facility must have the capability and capacity to adequately clean and reprocess the transducer. Consult the manufacturer’s IFU or other guidance on use, handling, and reprocessing of the manufacturer’s equipment before purchasing. If an entity other than the manufacturer has repaired the transducer, ensure the manufacturer’s IFU for cleaning and reprocessing is still valid.
6.5. Establishing Policies and Standard Operating Procedures (SOPs)
Before adopting a reprocessing workflow, consider the total cost of reprocessing including equipment, consumables, personnel, time, personal protective equipment, and turnaround time. It is also important to look at how safe, reliable, and reproducible the process is for the patient, sonographer, and transducer.1,2 Additionally, consider how reprocessing time might impact patient throughput.
The facility should have written reprocessing policies and standard operating procedures (SOPs) that are specific to the types of procedures, transducers, ancillary equipment, clinical settings, and reprocessing requirements. Align the policies and SOPs with current manufacturer’s IFU, regulations, guidelines, and standards. Conduct ongoing risk assessments and review of policies and SOPs and update accordingly.
6.6. Personnel
Personnel assigned to perform transducer reprocessing (typically the sonographer) should undergo documented competency-based training and assessment on transducer reprocessing at least annually. Policies should define the roles and responsibilities of the personnel assigned to manage and ensure compliance with procedures and reprocessing requirements.1,2
6.7. Evaluation and Quality Improvement
A process that monitors infection prevention and control practices related to ultrasound use and reprocessing workflow should be in place for each clinical setting. The process should reflect an assessment of risks with the procedure to both patients and sonographers. The process should include methods for observation, performance measurement, intervention, feedback, and documentation to identify practice gaps. Report process results as part of a continuous quality and performance improvement approach.
Maintenance of the transducer and ancillary equipment should follow the same process of risk assessment, performance measurement, intervention, feedback, and documentation. Damaged equipment may harbor pathogens, tear transducer covers, or impact the ability of the sonographer to safely and accurately perform the procedure. If equipment damage is identified, such as a transducer with a crack, abrasion, or tear, there should be processes in place to promptly identify the damage, remove the equipment from use, and document the damage and repair.
7. Process Overview: Identification of Factors Impacting Spaulding Classification
Determine the prior disinfection level of transducer to be used. Determine if there are any factors that can or will affect the transducer, cover, or gel needed and the procedure to be performed (e.g., open wound, unsanitary location, a high-risk immunocompromised patient). Determine the expected Spaulding Classification and post-procedure disinfection requirements (based collectively on the known factors above).
Determine if any additional factors are identified during the procedure (e.g., transducer came in contact with a mucous membrane) that may affect the actual Spaulding Classification and post-procedure disinfection requirements.
Determine if there are any additional factors identified after the procedure (e.g., transducer came in contact with mucous membranes or sterile tissues) that may affect the actual Spaulding Classification and post-procedure disinfection requirements.
Determine the actual Spaulding Classification and post-procedure disinfection requirements (based collectively on the factors identified in Steps 1, 2, and 3 above).
8. Sonography Procedures: Transducer Disinfection and Infection Control Worksheet
The
Footnotes
Appendix 1: Preparing for a Sonography Procedure
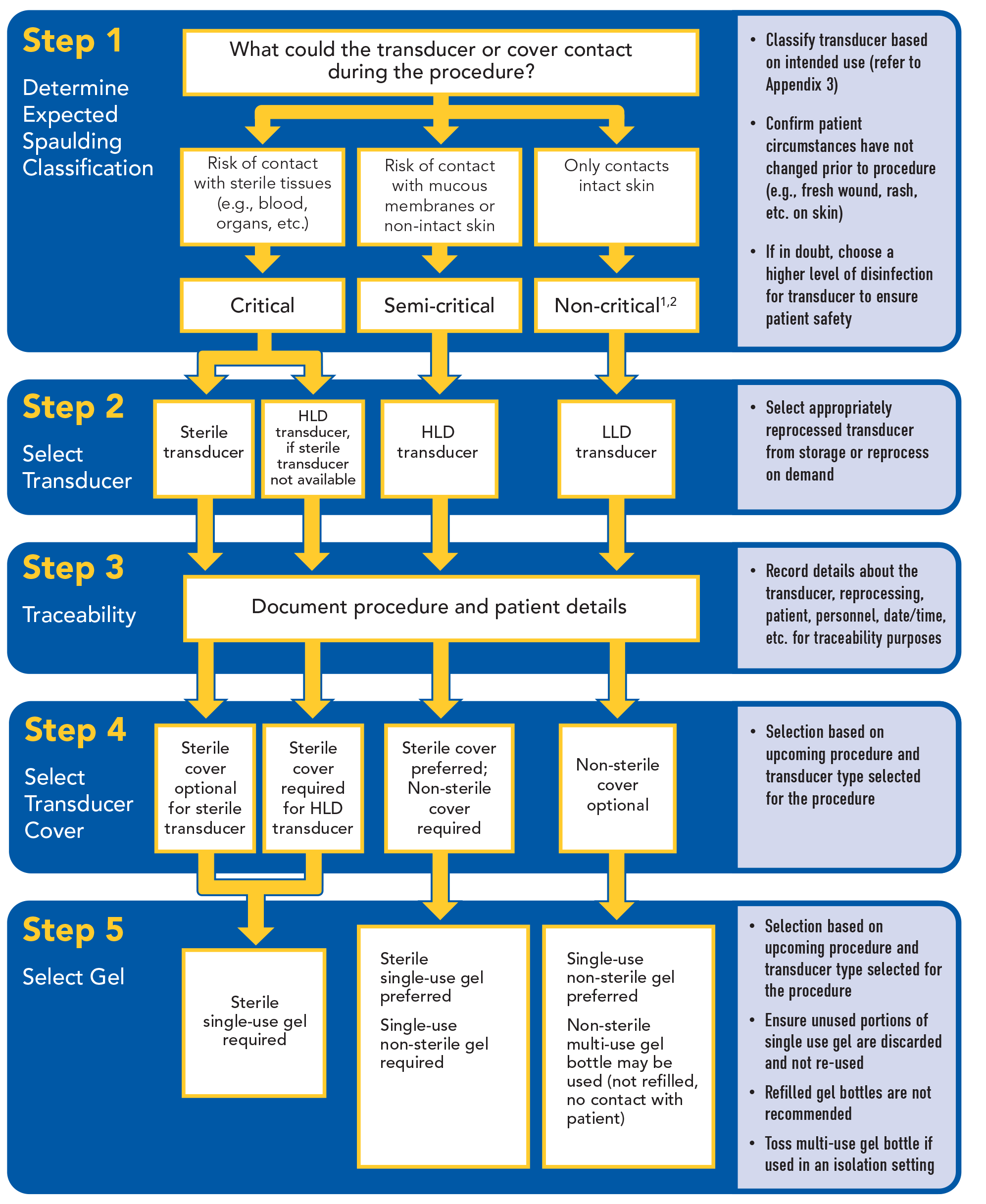
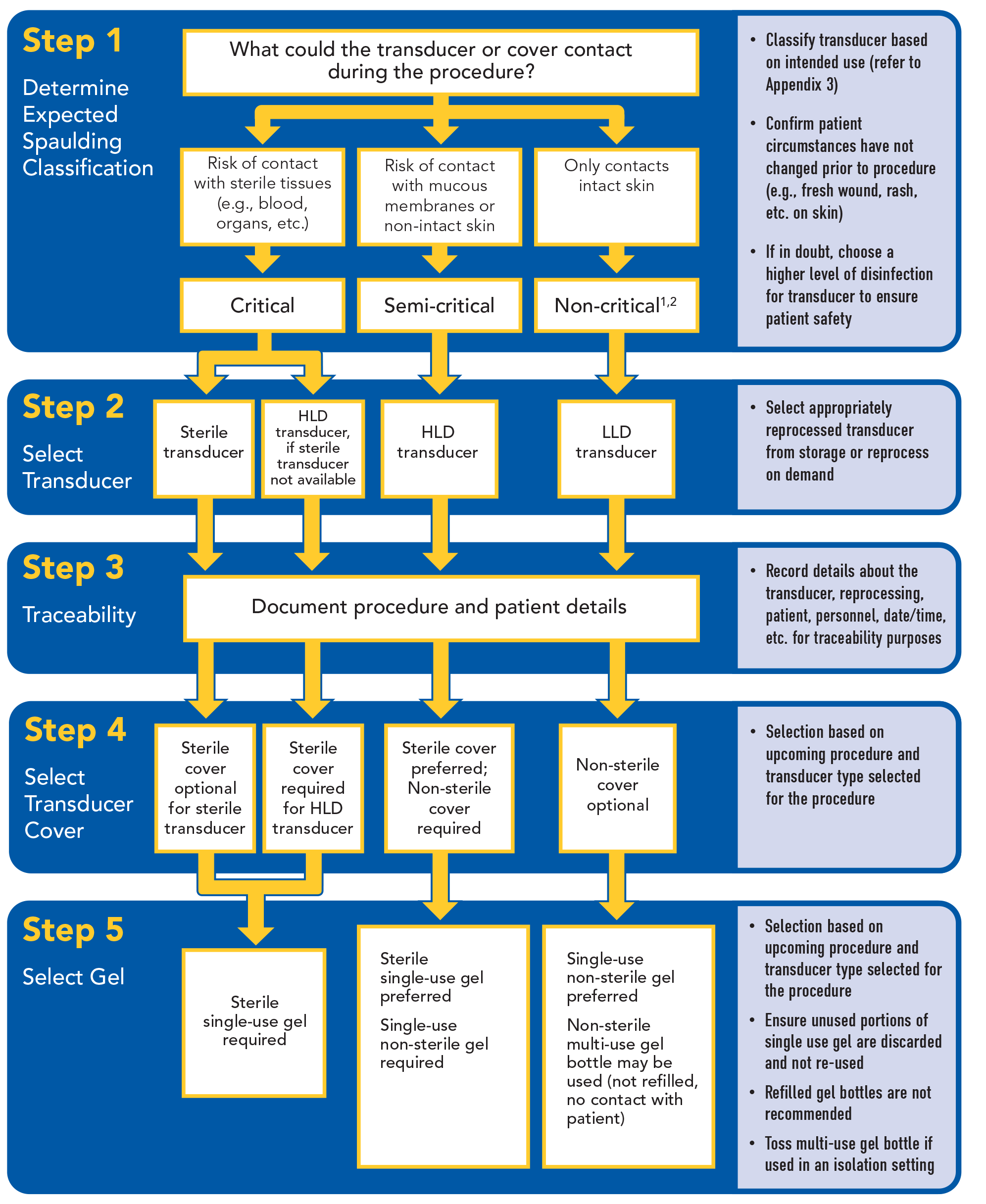
|
Appendix 2: Reprocessing the Ultrasound Transducer and Ancillary Equipment
|
Appendix 3: Sonography Procedures: Transducer Disinfection and Infection Control
The list of sonography procedures below describes the Spaulding Classification and best practices. Note that the expected Spaulding Classification for a sonography procedure could change upon patient condition (e.g., has open wound or infection in the scan area) or during the procedure (e.g., blood is present). Follow the same assessment of risk and Spaulding Classification for sonography procedures not listed.
10. Glossary of Terms and Abbreviations/Acronyms
organic materials that remain on the surfaces of instruments interfere with the effectiveness of these processes.
