Abstract

In 2016 the Food and Drug Administration (FDA) approved contrast-enhanced ultrasound (CEUS) to image and characterize liver lesions as benign or malignant using gray-scale sonography. The technique includes the intravenous (IV) administration of a gas microbubble that the vascular system disperses to the liver, thus enabling liver lesion characterization. With the inclusion of CEUS to liver imaging protocols, interpreter accuracy has been shown to improve to 88% over a baseline of 35% with gray-scale sonography alone. 1 Sensitivity and specificity can improve to 83% and 84% from 40% and 63% with gray-scale sonography alone. 2
How Contrast-Enhanced Ultrasound Works:
The microbubble contrast agent is a high molecular weight, low-soluble gas encased in an elastic shell. 3 (Figure 1) It is approximately 3-4 microns, which is smaller than red blood cells, and passes easily through the pulmonary circulation. The microbubble remains in the vascular system throughout the examination. 3
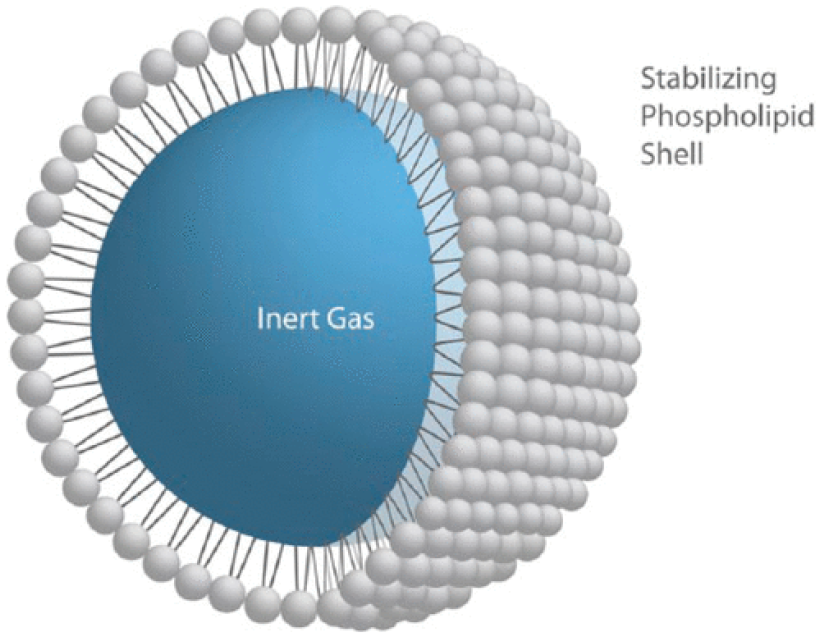
Schematic of a phospholipid-encased ultrasound contrast microbubble. 3 Image used with permission from Jim Baun, BS, RDMS, RVT, FSDMS.
The microbubble shell is highly compliant, responsive to the compressions and rarefactions of the ultrasound pressure wave, and oscillates in the ultrasound field. There are three possible bubble-ultrasound wave interactions depending on the acoustic output power. Figure 2 illustrates how the bubble interacts with ultrasound exposure. 3 When the power is very low, the bubble will oscillate linearly in tandem with the compressions and rarefactions of the ultrasound wave resulting in signal enhancement. When there is moderate to low power, the bubble will oscillate non-linearly, creating a harmonic frequency response that is two times the send frequency. The ultrasound system displays the harmonic response by using pulse cancellation techniques that eliminate the signal from tissues and only display the bubble harmonic responses. At high power, the sound waves burst the bubble, leaving behind a strong back-scattering echo. Because the objective of contrast-enhanced ultrasound is to image the microbubble within the vascular spaces, the sonographer should use moderate to low pressure or low-mechanical index (MI) technique to elicit a bubble harmonic response.
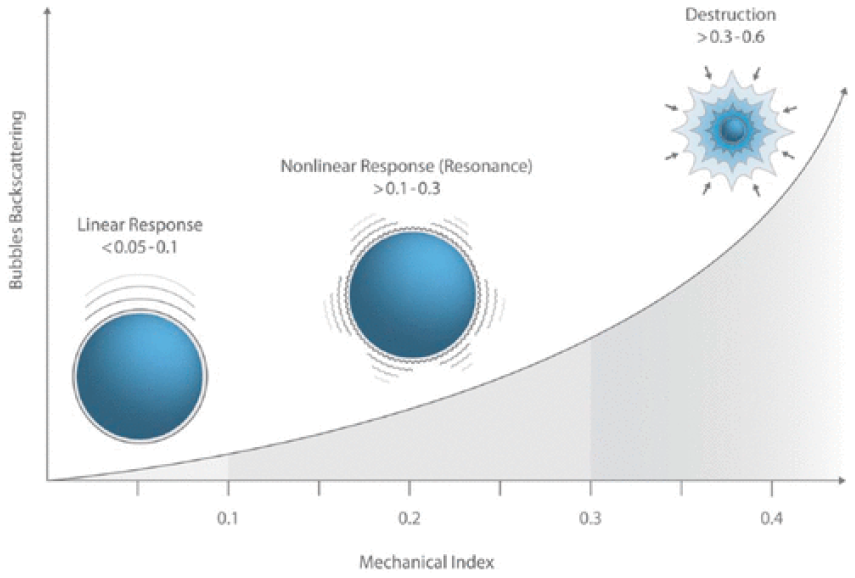
Schematic representation of microbubble response to increasing levels of acoustic power (mechanical index). 3 Image used with permission from Jim Baun, BS, RDMS, RVT, FSDMS.
Safety:
Because ultrasound contrast agents are vascular agents, they are not nephrotoxic and do not collect in the renal spaces. They do not carry the same risks as iodized contrast agents used in computed tomography (CT) or gadolinium used in magnetic resonance imaging (MRI). The main concern with ultrasound contrast agents is anaphylactic reactions due to a sensitivity to the sulfur hexafluoride gas. 4 Adverse events from liver contrast scanning are low, 0.008% in over 23,000 patients without any patient deaths reported. 5
Planning:
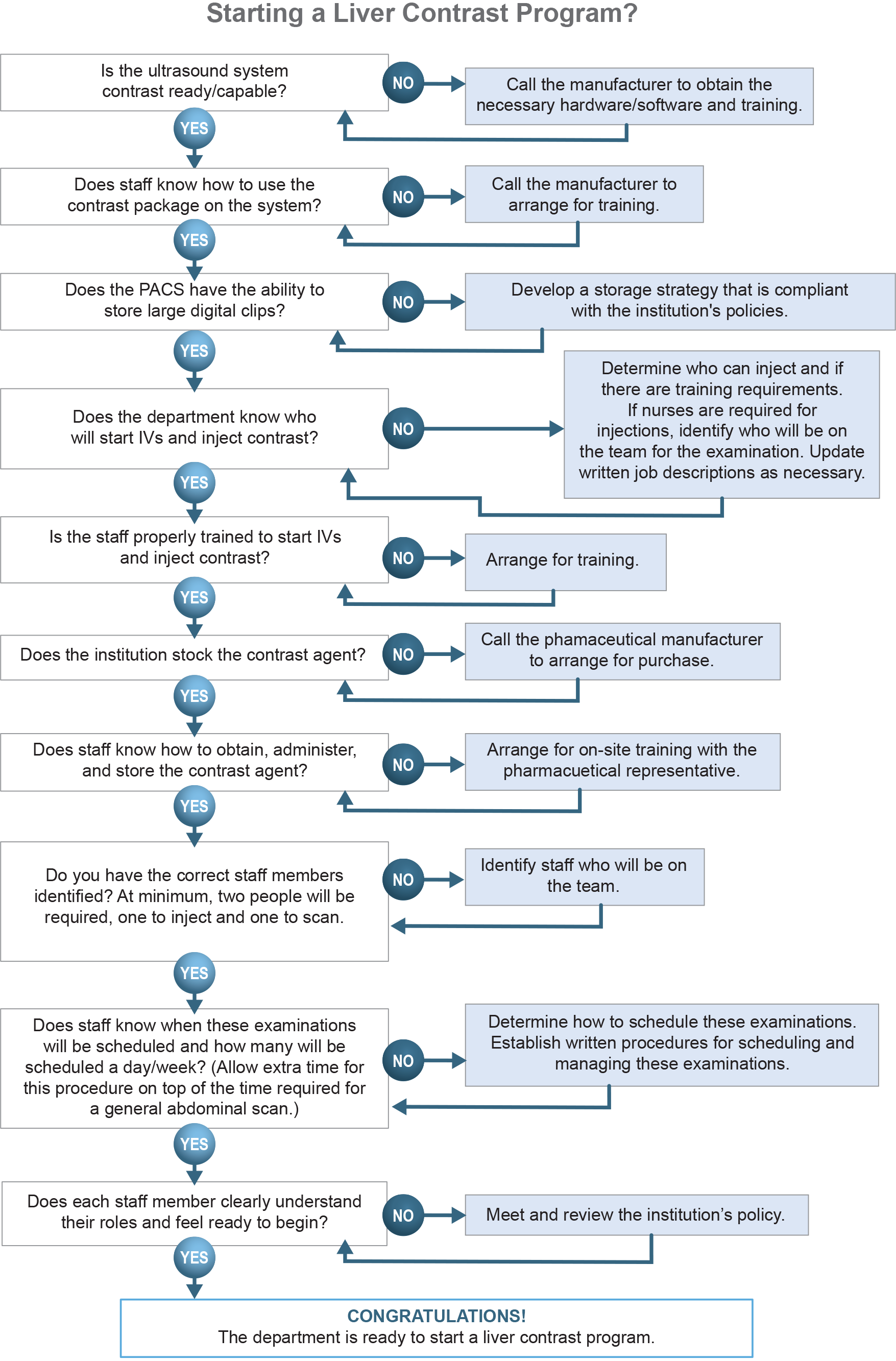
Prior to implementing a contrast program, the staff must conduct an evaluation to ensure proper preparation.
A contrast-enhanced ultrasound requires ultrasound machines to have contrast software and/or hardware. The staff should consult the equipment manufacturer to determine if the department’s ultrasound machines have the proper software and/or hardware or if they need additional equipment.
Staff may require additional training with the manufacturer’s clinical specialist on how to operate the software and hardware, including the specific controls involved in contrast-enhanced ultrasound. Once the predetermined staff member injects the contrast, there is not time to attempt different optimization techniques.
With each new software and hardware update, the staff should determine how the updates affect contrast-enhanced ultrasound.
Some of the required equipment features include:
Live dual or side-by-side imaging of gray-scale/contrast-enhanced images
Dual measurement/caliper placement to help identify poorly visualized lesions in contrast mode
A stop-watch/timer function
Extended clip capabilities of up to five minutes
Optimized contrast pre-sets
The department should maintain a continuous quality-monitoring program for equipment and transducers used for contrast-enhanced ultrasound. Damaged transducers and equipment used for contrast-enhanced ultrasound will quickly show flaws that may not be visible in gray-scale ultrasound.
The department may find it cost-effective to use a preventative maintenance contract with the manufacturer(s) to maintain the equipment. When in-house biomedical engineering performs equipment maintenance, it is essential they receive proper training.
If the department sends out transducers for repair, they should inspect them and perform quality assurance testing prior to patient use to ensure the repairs meet performance expectations.
As of June 2019, Bracco’s LUMASON®, a non-refrigerated kit, is the only pharmacologic agent the FDA has approved for use in contrast-enhanced liver ultrasound. The staff will need to meet with the pharmaceutical representative to learn more about the contrast agent.
Specific information needed includes:
How the properties of the contrast agent affect storage and shelf-life
Mixing and activating the contrast agent
Injection needle size
Required dose
Rate of injection
Number of injections available per vial
As new and existing contrast agents gain FDA approval for liver imaging and come to market in the United States, it is important that staff meet with those pharmacological representatives to learn the specifics of the agents as each may have its own requirements.
Prior to the first contrast-enhanced ultrasound, the department must decide who is responsible for the IV insertion and contrast administration. First, document the state laws, if any, that govern healthcare personnel starting and administering IVs. Additionally, review the institution’s policies on which personnel can insert IVs and administer contrast agents. Given the high level of expertise required to learn how to start an IV as well as maintain competency, some institutions may decide to use designated nursing staff to insert IVs and/or administer contrast. If the lab wants to propose authorizing sonographers to perform IV insertion and contrast administration, the following should be considered:
With the proper education, and training, it is within the scope of practice for sonographers to start IVs and administer contrast agents. 6
If the institution authorizes sonographers to start IVs, a written policy should detail this process and outline guidelines and requirements for training and competency. Written sonographer job descriptions should also reflect IV insertion duties. A training program and an ongoing competency evaluation system is also necessary to ensure patient safety and comfort.
Regardless of who is administering the contrast, they will need training on how to handle the contrast agent.
The examination will always require two individuals – one person scanning and one person administering the contrast agent. Due to the timing of arterial phase after injection, it is impossible for the same person to scan and administer the contrast agent.
Since this examination requires two people, the scheduling staff must consider the extra staffing and time requirements when scheduling patients.
Before performing the initial contrast-enhanced ultrasound, the department should determine if the PACS can store large digital clips which may be up to several minutes long. If there is no PACS, the department should develop an archival strategy.
As of January 2019, there are new approved Current Procedural Terminology (CPT) billing codes for contrast-enhanced ultrasounds. 7 Hospitals, offices, and outpatient facilities will use CPT Code 76978 for US targeted dynamic microbubble sonographic contrast characterization (non-cardiac) initial lesion, and the add on code 76979 for additional lesions with separate injections. 7 Use the code Q9950 to report the use of the LUMASON® microbubble contrast agent. 7 There are other cardiac contrast agents that have their own CPT codes and billing staff should not confuse them with the liver ultrasound contrast CPT codes. Staff should consult with their billing departments to ensure proper use of the currently available CPT codes. 8
Performing the Contrast-Enhanced Liver Ultrasound
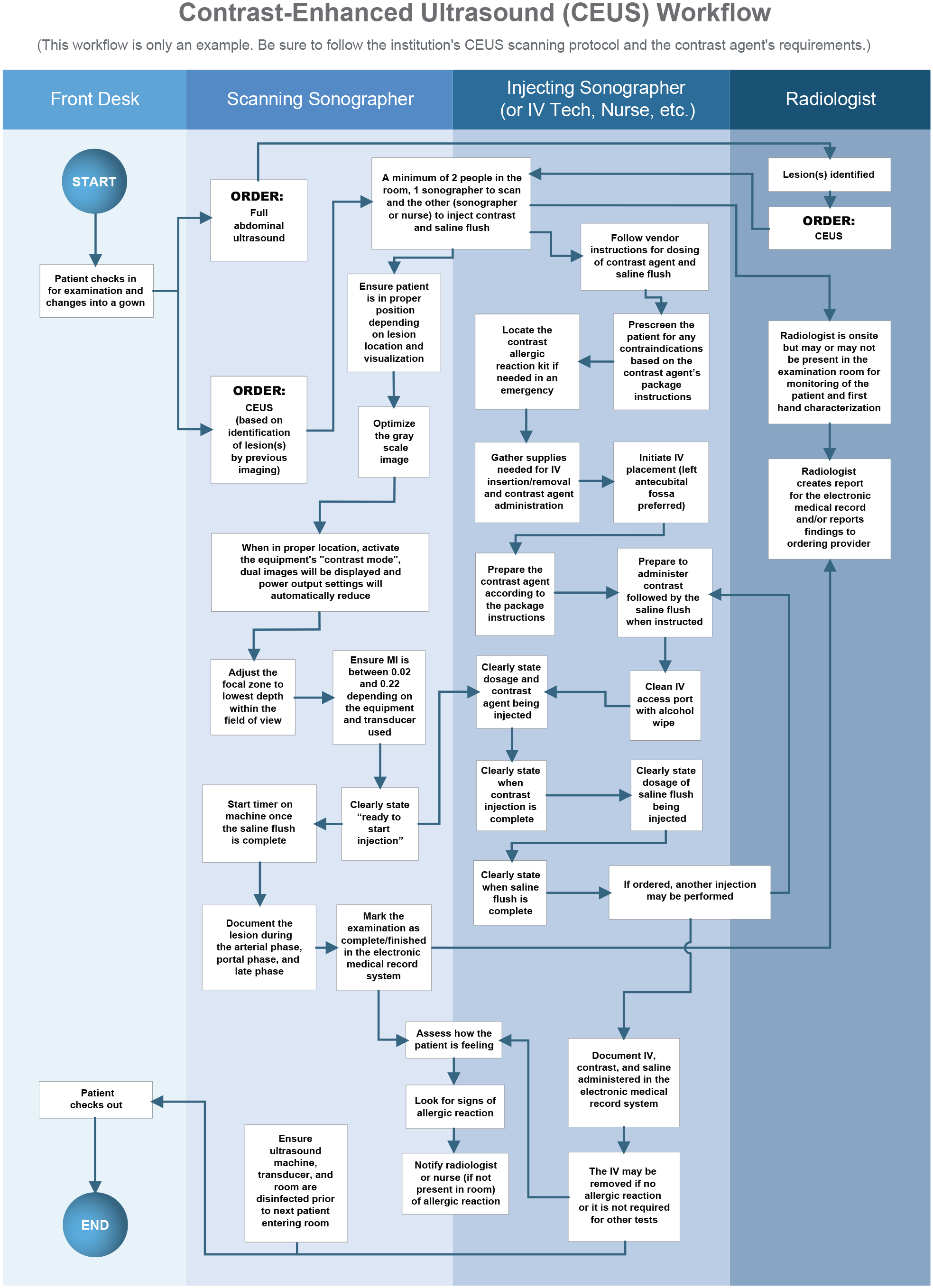
The examination begins with a standard gray-scale evaluation of the liver to locate the lesion in question. This part of the examination is known as baseline scanning or pre-injection scanning. During this time, the sonographer should identify the patient position that best depicts the lesion with normal respiration. Because the sonographer must image the lesion over several minutes, a breath hold is not practical for the patient. The best patient position is the one that moves the lesion closest to the transducer, ideally less than 10 cm in depth. Lesion visibility during the contrast portion of the examination is more important than maintaining specific imaging planes.
Once the scanning sonographer has identified the optimal patient position for lesion visualization, the examination moves into the injection phase.
The patient should be pre-screened for contraindications based on the pharmaceutical package insert instructions.
A staff member will place an IV, preferably in the patient’s left antecubital vein. This location is optimal because the sonographer typically scans from the patient’s right side, thus rendering the right arm inaccessible
After the scanning sonographer determines the best patient position in the pre-injection scan, they will activate the contrast mode on the machine, which will bring up the live-dual or side-by-side imaging mode as well as reduce the acoustic output power settings.
The displayed MI will be between 0.02 and 0.22 depending on the equipment and transducer used.
The staff member responsible for the IV will slowly inject the appropriate dose of the contrast agent over approximately 2 seconds (a fast bolus will destroy the microbubbles) using a 20-22 gauge needle and then follow with a 5-10 ml saline flush.
The staff member administering the contrast agent should clearly communicate when the injection starts and the saline flush is complete.
The staff should observe the patient for any adverse events in accordance with departmental protocol and pharmaceutical requirements.
Once the staff member injects the contrast, the examination moves into the post-injection phase.
Consistent adherence to the protocol is necessary so the scanning sonographer can accurately document enhancement patterns and not miss specific phases.
Once the injection starts, the scanning sonographer activates the clip store function and the contrast timer. For some manufacturers’ equipment, these steps may be reversed.
The scanning sonographer will maintain the transducer position and observe the contrast arrival to the lesion.
The scanning sonographer should image and store the enhancement patterns and the radiologist should document them in the examination report.
The sonography department should develop a protocol on how to acquire the clip and the length of the initial clip. Clips of 60 seconds or less are ideal since they capture the initial arterial phase while meeting the storage limitations of some PACS systems or imaging equipment.
The sonographer(s) and/or the radiologist present in the examination room must observe the lesion for at least five minutes post-injection to capture the portal venous and late phases, as the information obtained in these phases is also relevant to the uptake pattern and overall lesion characterization.
The staff should create a protocol outlining the intervals at which the scanning sonographer should obtain clips and static images during this 5-minute duration.
Do not image the lesion continually for 5 minutes, as even at low power, continual scanning may result in bubble destruction.
At the end of the examination, staff should archive the data for review and comparison with prior or upcoming studies or other imaging examinations.
If the pharmaceutical contrast agent requires post-examination monitoring, it should occur according to the pharmaceutical safety instructions.
Staff should document the pharmaceutical contrast agent administration in the patient’s electronic medical record including but not limited to:
□ Date and time; □ Contrast agent name, dose, and lot number; □ Saline flush; □ Initiation and removal of the IV; and □ Any adverse events
Conclusion:
The contrast-enhanced liver ultrasound requires additional ultrasound equipment, protocols, and personnel. Consistent adherence to the institution’s protocol provides the best results. Pre-planning should include meeting with the machine manufacturer’s clinical specialist to ensure proper equipment is available and that the sonography department knows how to operate the contrast functions on the machine. Additionally, the sonography department should meet with the pharmacologic representative to ensure that the staff knows how to administer and store the contrast agent. With proper planning and preparation, institutions can successfully implement a liver contrast-enhanced ultrasound program.