Abstract
Vasa previa is a life-threatening complication of a velamentous cord insertion. Understanding variations of this condition is vital to ensuring optimal patient outcomes. The case presented is of a 27-year-old primigravida woman whose condition was diagnosed during her second-trimester obstetric sonographic examination. The case demonstrated an unusual variation of vasa previa, in which a single fetal vessel separated from the membranous insertion located near the fundal placenta and traversed posteriorly through the chorionic membrane to within 0.83 cm of the internal os. The case was followed with the most up-to-date standards recommended by the American Institute of Ultrasound in Medicine.
Vasa previa is a life-threatening condition defined as unprotected fetal blood vessels within the membranes overlying or inserting within 2 cm of the internal cervical os.1–3 Rupture of the unprotected fetal vessels during late pregnancy or labor results in a 54% fetal mortality rate due to exsanguination in undiagnosed cases.1–4 This condition is uncommon, occurring in 1 per 2500 births,1–3 and can only be present with either a velamentous cord insertion (VCI) or a succenturiate lobe.1–3,5 A VCI, also called a membranous cord insertion, is when the umbilical cord inserts into the membranes rather than directly into the placental mass and the umbilicus vessels course through the membranes unprotected by the umbilical cord or Wharton jelly.6,7 This condition is present in 1.5% of singleton pregnancies and 6% of multiple-gestation pregnancies. 6 The more common variation of type 1 vasa previa is when the placental mass is in the lower uterine segment and the VCI is over or close to the internal cervical os.1–3,5 The presented case is an uncommon variation of type 1 vasa previa, in which a single fetal vessel separates from the membranous insertion near the fundal placenta and traverses inferiorly through the chorionic membrane to within 2 cm of the internal os. A succenturiate lobe, or bilobed placenta, is defined by an accessory lobe connected to the main placental mass by unprotected fetal vessels coursing through the membranes and is present in <1% of pregnancies. 2 Type 2 vasa previa can be present with a succenturiate lobe with the unsupported fetal vessels between the placental bodies crossing near or over the cervical os.1–3,5
By using gray-scale and color Doppler as well as pulsed wave sonography, it is possible to diagnose VCI and vasa previa prenatally.1,5 Sonographically, the unprotected vessels of a VCI and vasa previa and the vessels connecting succenturiate lobes all have a similar appearance. In gray-scale, they appear as immobile linear structures that adhere to the uterine wall; with color Doppler, the umbilical cord insertion point shows vessel separation outside the placental mass; and pulsed wave Doppler can be used to demonstrate whether vessels are of maternal or fetal origin.
An atypical variation of VCI is presented in this case study, but having an understanding of all the possible variations of this condition and recognizing the sonographic appearance is vital to ensuring optimal patient outcomes.
Case Study
The case presented is of a 27-year-old primigravida woman with a gestational age of 22 weeks based on the last menstrual period, who presented for sonographic evaluation with an indication of small fetal size according to the expected due date. All images were acquired with an Acuson S2000 (Siemens Healthcare) with a 6C2 curvilinear-array transducer. Additional images were taken with an EC9-4 endocavity transducer. At the start of the initial examination, the lower uterine segment was interrogated with a transabdominal approach in gray-scale (Figure 1) and color Doppler (Figure 2). The gray-scale image showed a linear structure indicative of a possible vessel in the area of the internal os. Use of color Doppler confirmed that the structure was a vessel; however, the origin of the vessel was still unknown. Spectral Doppler was then used to confirm that the vessel was of fetal origin (Figure 3), and further inspection of the area showed that the placenta was not implanted in the lower uterine segment. An additional image taken in transverse with an endocavity transducer (Figure 4) indicated that the vessel lay within 0.83 cm of the internal os of the cervix.
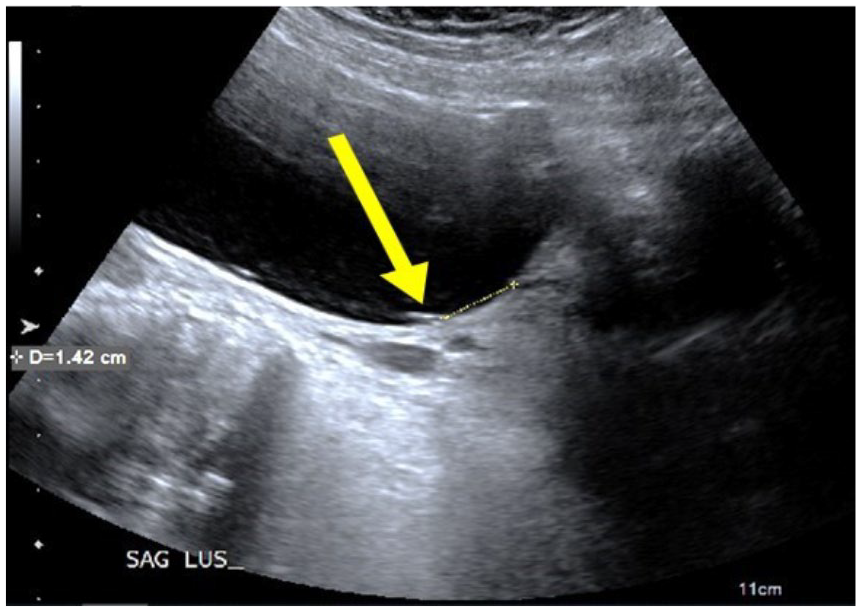
Yellow arrow shows nonmobile linear structure adhered to uterine wall near the internal cervical os.
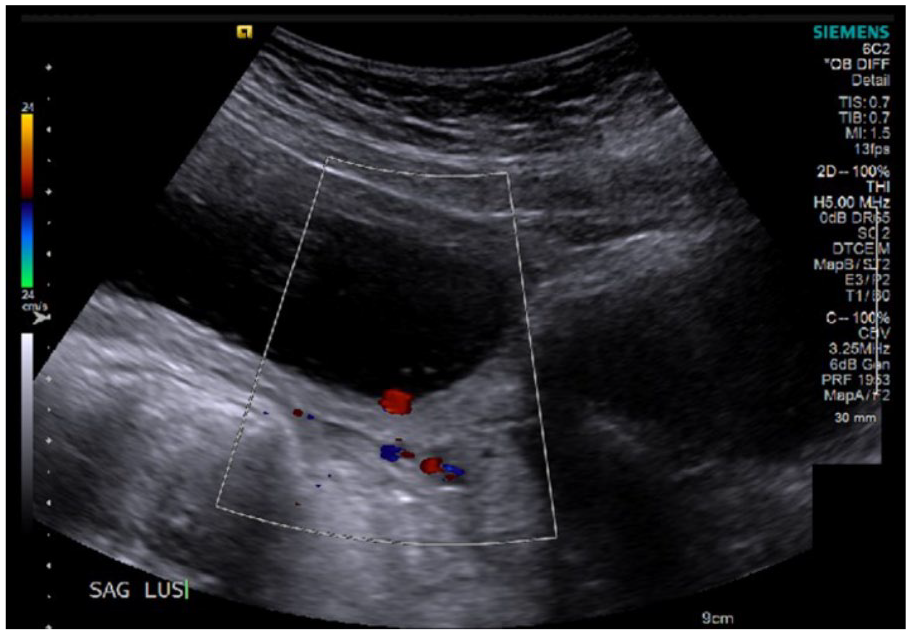
Color Doppler of linear structure confirms that the structure is a vessel with blood flow.
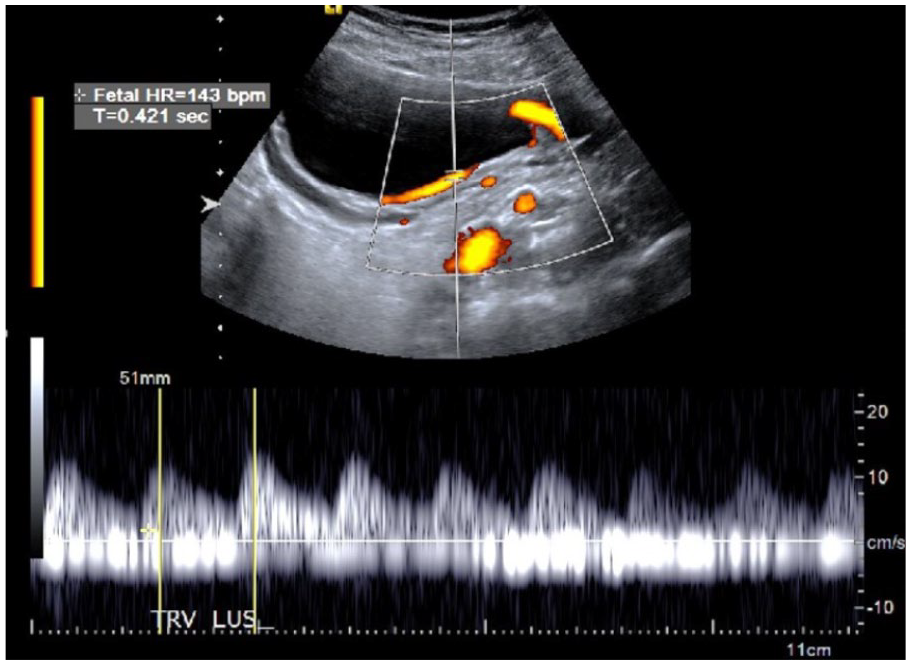
Spectral Doppler confirms that vessel is of fetal origin.
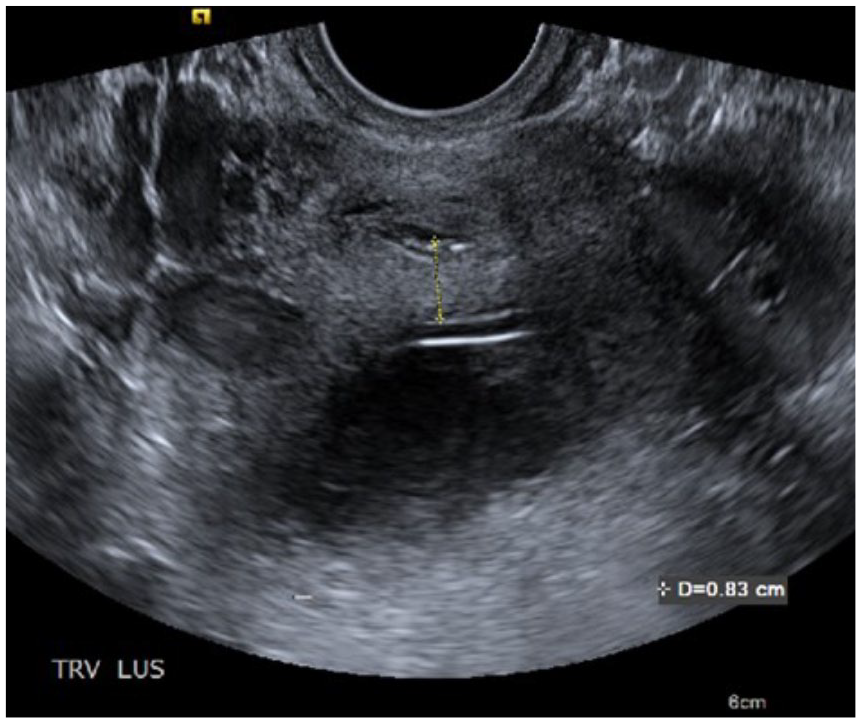
Endovaginal image in transverse shows that fetal vessels are 0.83 cm from internal os.
To determine the source of the fetal vessel, the uterus was carefully interrogated, and a placental mass was noted in the fundus. No additional placental tissue was identified, ruling out the possibility of a succenturiate lobe placenta. A sagittal image of the placenta appeared to show a normal placental cord insertion (Figure 5); however, a right coronal plane showed what could be either a battledore or marginal cord insertion (Figure 6). Because the examination had already identified the vasa previa and ruled out a bilobed placenta, an attempt was made to visualize a “mangrove sign.” 8 This sign was proposed in 2012, as a VCI can sometimes appear to look like a mangrove tree with a trunk splitting into multiple root stems well above the surface of the ground. In the case of a VCI, the umbilicus splits into multiple unprotected fetal vessels outside the placental surface (Figure 7). 8 The VCI (Figures 8 and 9) and mangrove sign (Figure 10) were seen by slowly sweeping through the placental cord insertion area in sagittal and transverse. Additionally, a cine clip was taken documenting the insertion site and another of the single fetal vessel traversing through the membranes along the posterior uterine wall to the lower uterine segment. These two videos are available in the supplemental material for this article online.
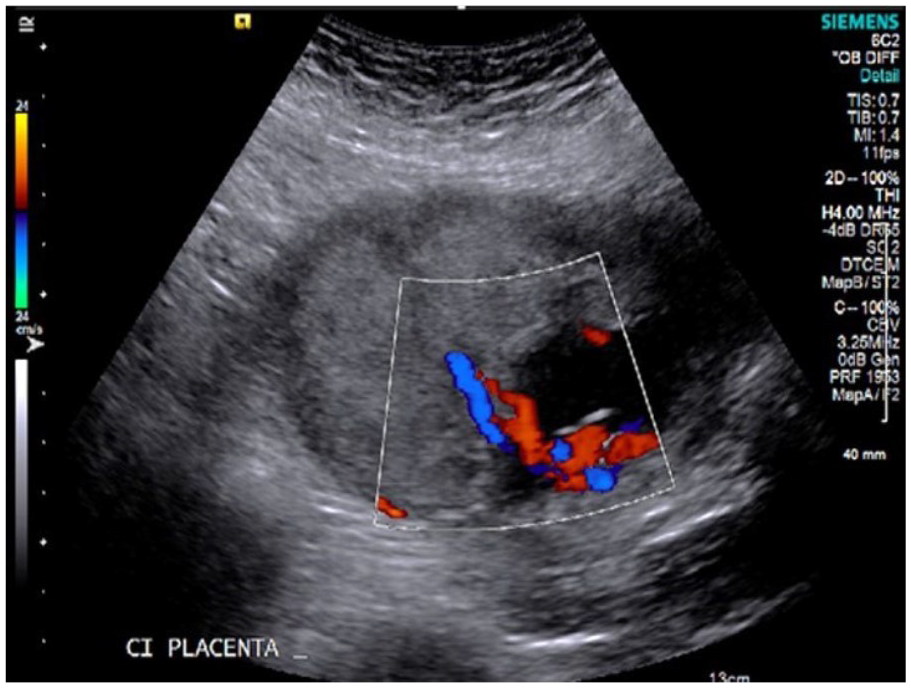
Placental cord insertion in the sagittal plane appears to show a normal cord insertion
Right coronal image shows a possible marginal or battledore insertion, as indicated by the yellow arrow.
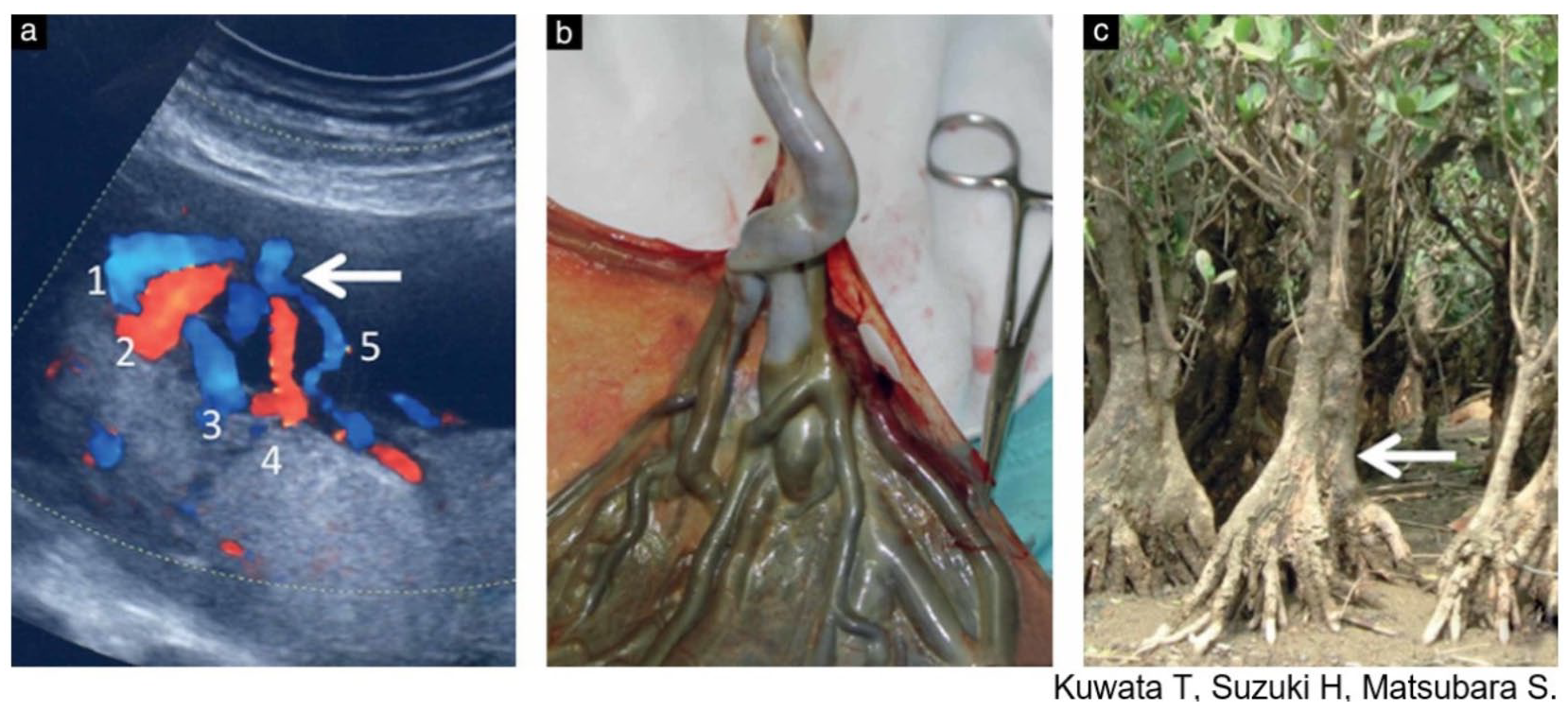
“(a) Color Doppler image showing the ‘mangrove sign’ for velamentous cord insertion. Although the cord itself is not shown, at least five large vessels (numbered 1–5) could be seen branching from the point marked by the arrow. These ran between the fetal membranes to the placental surface. (b) Macroscopic findings of the cord and the placenta from this patient following delivery. A velamentous cord insertion was identified. (c) Photograph of a mangrove tree in subtropical Amami Island, Japan. The tree trunk represents the cord and the arrow is analogous to the point marked by the arrow in (a), i.e. the point from which the trunk/cord branches to the several large roots/velamentous vessels.” Figure and caption reprinted with permission. 8
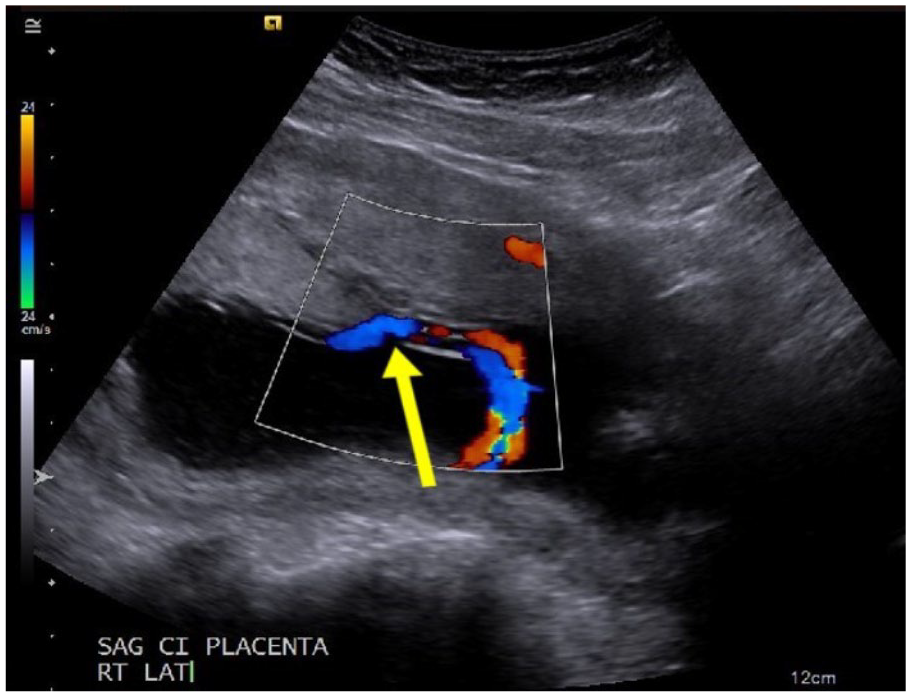
The sagittal right lateral image shows aberrant velamentous cord insertion. The yellow arrow points to insertion into membranes. The green arrow points to a single fetal vessel that travels through membranes inferiorly along the posterior uterine wall to the lower uterine segment.
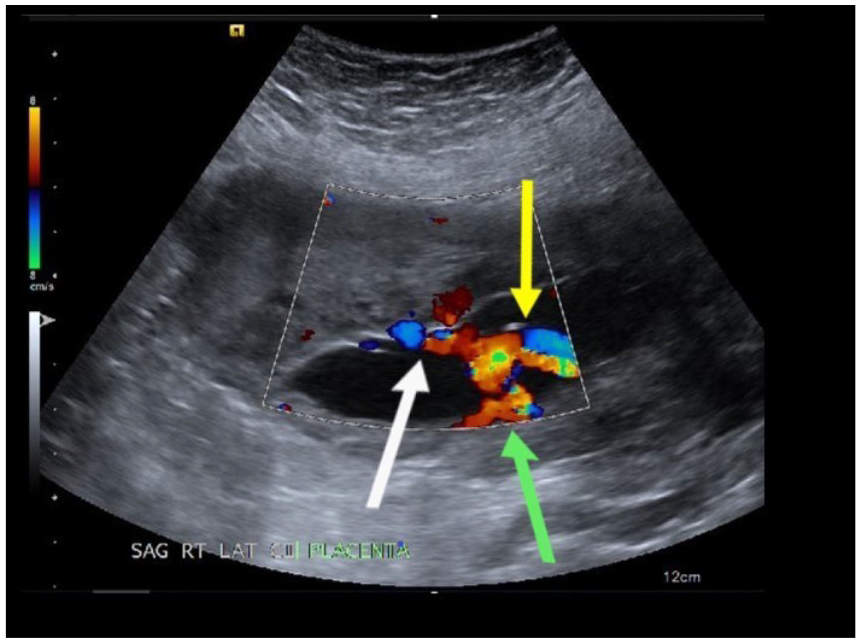
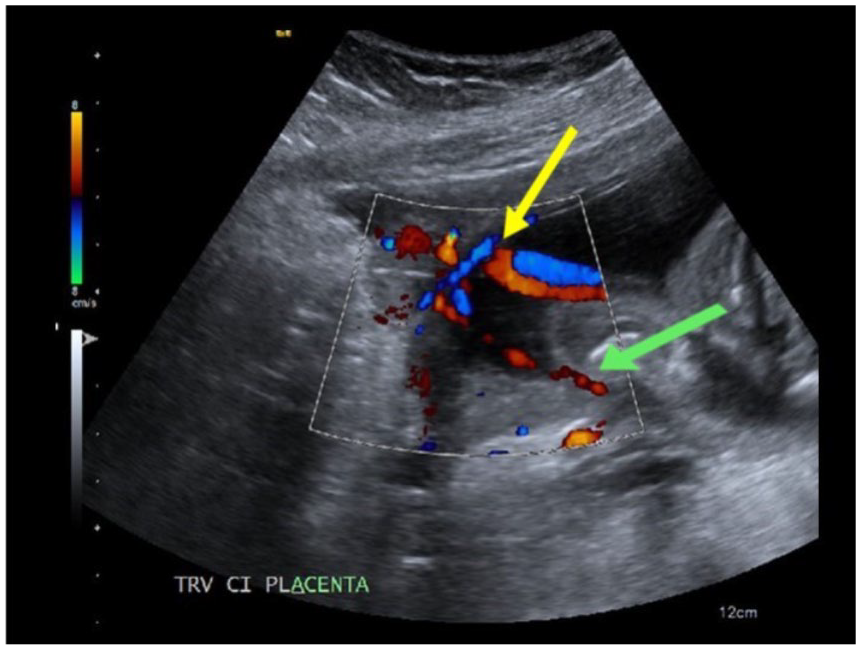
The cord insertion is noted in the transverse oblique view. The yellow arrow points to insertion into membranes. The green arrow points to a single fetal vessel that travels through membranes inferiorly along the posterior uterine wall to the lower uterine segment. The white arrow points to vessels that travel through membranes from insertion to placental mass.
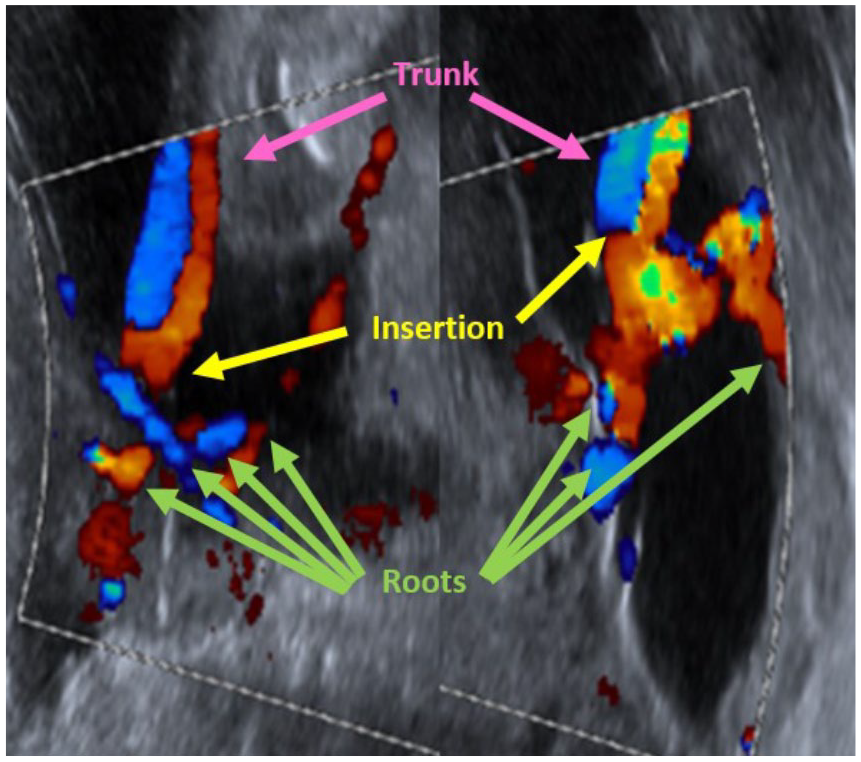
Cord insertion images have been rotated 90° to better appreciate the mangrove sign. The pink arrows point to the “trunk” (umbilical cord). The yellow arrows point to insertion into membranes. The green arrows point to the “roots,” which are the separated fetal vessels.
Biometric calculations by sonographic evaluation indicated a gestational age of 17 weeks. This was a 5-week difference from the size that would be expected based on the last mensural period date. However, the fetal age was not adjusted at this time, as it was unknown if the fetus was small due to incorrect dates or intrauterine growth restriction. Placentas with abnormal insertions have a lower density of vessels, and when those vessels are further elongated by velamentous insertion, there is even greater restriction on the transfer of nutrients to the fetus. 6 No other anomalies were seen at that time, and fetal heart rate was within normal limits at 143 bpm. Due to the abnormal cord insertion and vasa previa, the pregnancy was deemed high risk, and a 4-week follow-up was scheduled. At the follow-up appointment, biometry measurements indicated that fetal growth since last examination was consistent with time elapsed, and another 4-week follow-up was scheduled. The third examination showed continued appropriate growth and no change in diagnosis, and the due date was corrected to match the estimate by sonographic evaluation to a gestational age of 26 weeks.
In this case, monitoring the patient for signs of imminent delivery was paramount, as the highest risk for rupture of the unprotected fetal vessels is when the membranes break.1,2 The treatment plan for this patient included hospitalization at 33 weeks, administration of antenatal corticosteroids to assist in fetal lung development, and scheduled cesarean delivery on or prior to 35 weeks. Unfortunately, the patient was noncompliant and did not return to the facility for corticosteroid treatment or planned hospitalization. The patient did present to the facility’s emergency department at 34 weeks with an unrelated health issue. The baby was delivered by cesarean the next morning with a birthweight of 2150 g and Apgar scores of 6 and 7 at the 1- and 5-minute marks, respectively. Postsurgical inspection of the placenta did confirm VCI with a thickened cord diameter indicating separated umbilical vessels. Both mother and baby were cleared for release 4 days later.
Discussion
Anomalous insertion of the umbilical cord occurs in 7.8% of singleton pregnancies and 16.9% of twin pregnancies. 6 Abnormal cord insertions do not have any associated maternal signs or symptoms and can go undiagnosed until delivery. 2 There are 2 types of abnormal cord insertions. Marginal cord insertion, also called battledore placenta, is when the cord inserts within 3 cm of the edge of the placental mass. VCI, also known as membranous cord insertion, is defined as “insertion of the umbilical cord into membranes before it enters the placenta.” 7 There are two theories on the pathogenesis of VCI. The first states that the cord implants into the trophoblast of the decidua capsularis rather than the trophoblast that forms the placental mass. 9 The second, trophotropism, is when placental tissue in the lower uterine segment migrates to the fundal area, where there is more optimal vasculature2,3,9–11 and the initial centrally located cord vessels elongate.
VCI is the more severe abnormal cord insertion type and has triple the risk of perinatal death as any other type of abnormal cord insertion. 6 In this case of VCI, the umbilical cord vessels separated at the membrane insertion site, with all of the vessels except one traveling through the membranes toward the placenta. The exception was a single vessel that diverged to travel inferiorly along the posterior uterine wall to the internal os. Only one reference to this atypical variation, called “aberrant velamentous,” could be located in a literature search. 2 This variation added a second high-risk complication of vasa previa to this case. Prognosis is good if the patient is monitored for fetal risk factors, such as low birth weight, abnormal intrapartum fetal heart rate pattern, and low Apgar score. Maternal risk factors, including placental abruption and preterm delivery 12 remain in place. Due to the high-risk nature of this pregnancy, the most conservative approach of commonly accepted protocols in treatment was proposed, which includes hospitalization starting at 33 weeks to monitor for signs of imminent preterm birth, as well as heart rate pattern and general health of the fetus, and to reduce the possibility of placental abruption. Additionally, the administration of antenatal corticosteroids is recommended to assist in fetal lung development in the advent of an emergency caesarean, and a scheduled delivery by caesarean on or prior to 35 weeks is recommended to lower the risk of vessel sheering, rupture, or compression, any of which could result in perinatal death.
Although it is unknown why the patient did not follow the suggested treatment plan, the sonographic techniques used and documentation recorded were all based on currently accepted guidelines for a high-risk patient presenting with this condition. These guidelines have progressed over the last 20 years as ultrasound machines have improved, and various studies showed that the inclusion of specific information greatly increased fetal outcomes while adding minimal time and effort by the sonographer. 2 For example, shortly after 2000, most case reports on VCIs included the suggestion for cord insertion sites to be regularly inspected,11,13,14 as they were not often included at that time. By 2007, routine inspection of the cord insertion point was an accepted practice, and a new area of interrogation was being called for: the inclusion of lower uterine segment to inspect for vasa previa. In 2008, a literature review on the importance of checking for vasa previa with gray-scale, color, and spectral Doppler, if needed, included a plea to incorporate this protocol into common practice in obstetrical sonography to prevent the deaths of otherwise healthy babies. 1 Inspection of the lower uterine segment is now common practice, and there was a study released in 2014 arguing that this screening examination should be performed on all pregnancies. 2 In 2010 a data-based study was released that discussed the use of cervical length measurements as an indicator for risk of spontaneous preterm birth in high-risk pregnancies. 9 An additional study on the management of pregnancies with a complication of vasa previa released in 2017 showed that the rate of cervical length shortening had a high correlative value indicative of an increased risk of imminent preterm birth. 15
At the time of the case presented, the obstetric facility followed the American Institute of Ultrasound in Medicine’s (AIUM’s) performance of standard diagnostic obstetric second- and third-trimester ultrasound examinations to assess for vasa previa, which included documentation of the internal os in gray-scale and color Doppler, as well as color Doppler of the cord insertion site. However, it did not routinely monitor cervical length as part of the sonographic examination protocol. It should be noted that the AIUM standards 16 just released in October 2018 now include criteria for cervical length measurements and routine inclusion of this documentation into second- and third-trimester examinations. With this change, medical professionals are able to use cervical length shortening criteria as a diagnostic indicator when assessing the possibility of imminent preterm birth. It is conceivable that this new diagnostic information can assist future patients with high-risk pregnancy to avoid prolonged hospital stays by monitoring the rate of cervical length shortening as a predictive value for imminent preterm labor; it could also be used as an alternative to inpatient admittance for monitoring.
Conclusion
Use of gray-scale, color Doppler, and spectral Doppler on the cervix is essential when interrogating the lower uterine segment. It is also important to perform a thorough interrogation of the umbilical cord insertion to rule out VCI and other cord anomalies. The use of cine clips and differing angles of insonation can ensure comprehensive documentation of any unusual findings. Last, obstetric facilities as well as individual sonographers should consider incorporating the new AIUM guidelines by including cervical length monitoring to assist in determining optimal delivery time. In conclusion, there is often a gap created when new innovations, techniques, or methodologies allow for the improvement in the diagnostic value of examinations. Exploring these new techniques and methods is the basis for improved patient outcomes, which should be the goal of every medical professional.
Footnotes
Acknowledgements
I would like to thank and recognize the Diagnostic Medical Sonography Program at El Centro College and program coordinator Lynn Schluns, BS, RDMS, RVT, for providing the impetus needed to stretch my comfort zone. I would also like to express my sincere gratitude to my mentor, Rymma Dudar, RDMS, for her encouragement and for pointing me in the right direction. Last, I must acknowledge Renee Ricketts Hansen for her extensive assistance with writing revisions.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
