Abstract
Breast implant ruptures are a recognized complication associated with both cosmetic augmentation and breast reconstruction. After a breast implant has been placed, a fibrous capsule forms around the shell of the implant. If the shell of the implant ruptures but the fibrous capsule remains intact, the rupture is known as intracapsular. If both the implant shell and fibrous capsule have ruptured, the implant content is freely extravasated into surrounding tissue, and the rupture is defined as extracapsular. This case study reviews a silicone implant rupture. Following an initial routine mammogram, bilateral extracapsular rupture was suspected and subsequently confirmed sonographically. Sonography was able to target focal areas of extracapsular silicone implant rupture as well as intracapsular rupture. The use of both diagnostic imaging modalities allowed for a quick, accurate diagnosis, and helped to establish the next step in appropriate treatment for the patient.
There are two types of silicone implant rupture. The more common intracapsular rupture transpires when the implant shell is disrupted without silicone escaping through the fibrous capsule. 1 Extracapsular implant rupture is described as silicone extending beyond the fibrous capsule of the implant, which occurs less often. Most implant ruptures occur 10-15 years after implant placement. 1 The likelihood of rupture increases with implant age. The average occurrence of rupture is approximately 2 implant ruptures per 100 implants. 1 The diagnosis of implant rupture can be done with an array of imaging modalities including magnetic resonance imaging (MRI), sonography, mammography, and computerized tomography (CT). 2 However, MRI is widely accepted as the imaging study of choice for diagnosing implant rupture. This case presents a patient with a well- visualized intracapsular and extracapsular rupture of a silicone breast implant. The rupture was initially detected with mammography and the diagnosis was confirmed by sonography.
Case Report
A 70-year-old female presented to the imaging center for her yearly mammogram, with complaints of bilateral breast pain and itching. The patient had subglandular silicone breast implants placed 40 years prior. A subglandular implant was placed in front of the chest wall muscle, whereas a submuscular implant was placed behind the chest wall muscle. One year ago, a routine screening mammogram was completed. The right breast implant was described as being unchanged, with a small area of calcified fat necrosis identified in the upper outer right breast (Figure 1). The left implant was also unchanged, with a superior lobulation noted (Figure 2). The patient reported feeling nodularity and tenderness around the border of the implants within the past year.
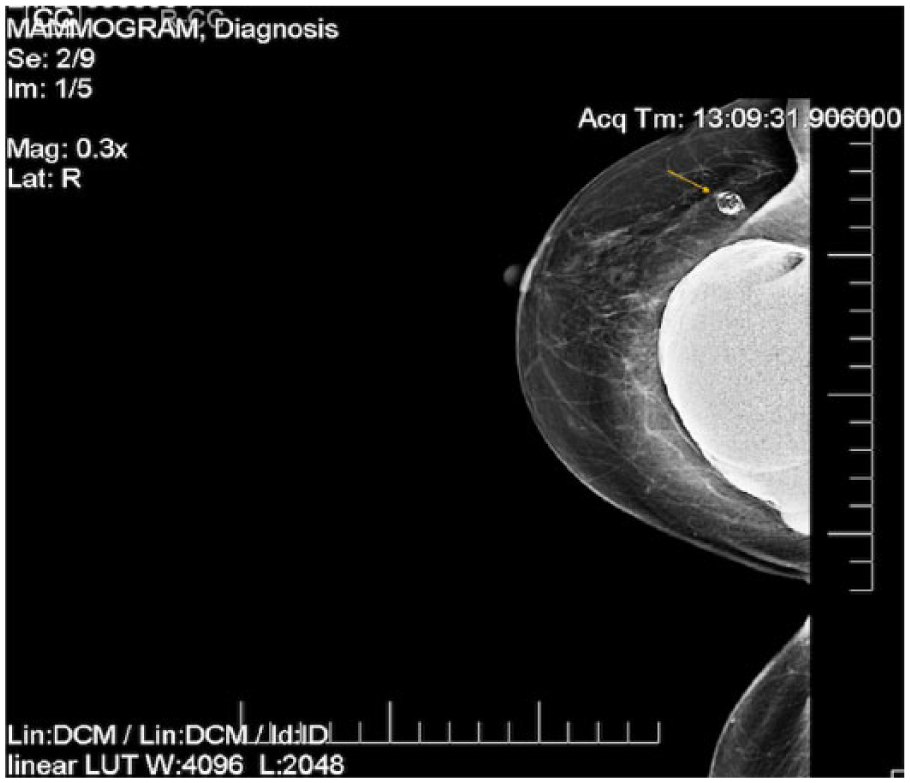
A mammographic craniocaudal projection of the right breast prior to implant rupture. An area of calcified fat necrosis is seen in the upper region of the breast (arrow).
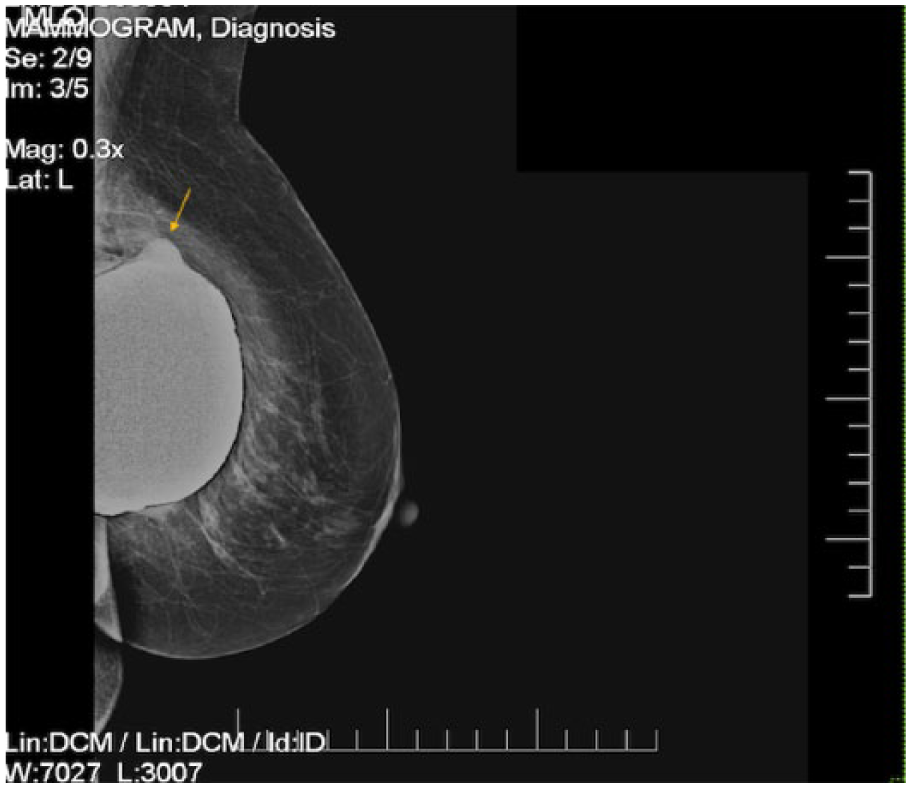
A mammographic mediolateral oblique projection of the left breast prior to rupture. A contour bulge is noted superiorly (arrow).
A mammogram was the first diagnostic test performed. Bilateral craniocaudal (CC) and mediolateral oblique (MLO) standard projections and tomosynthesis with 3D reformats were obtained. The mammogram showed that the breast tissue had scattered fibroglandular densities. Extracapsular silicone was documented bilaterally with a contour bulge along the superior margin of the left implant (Figures 3 and 4). Bilateral capsular calcifications were also noted on the images. A breast sonogram was ordered to further examine the focal areas of pain and itching.
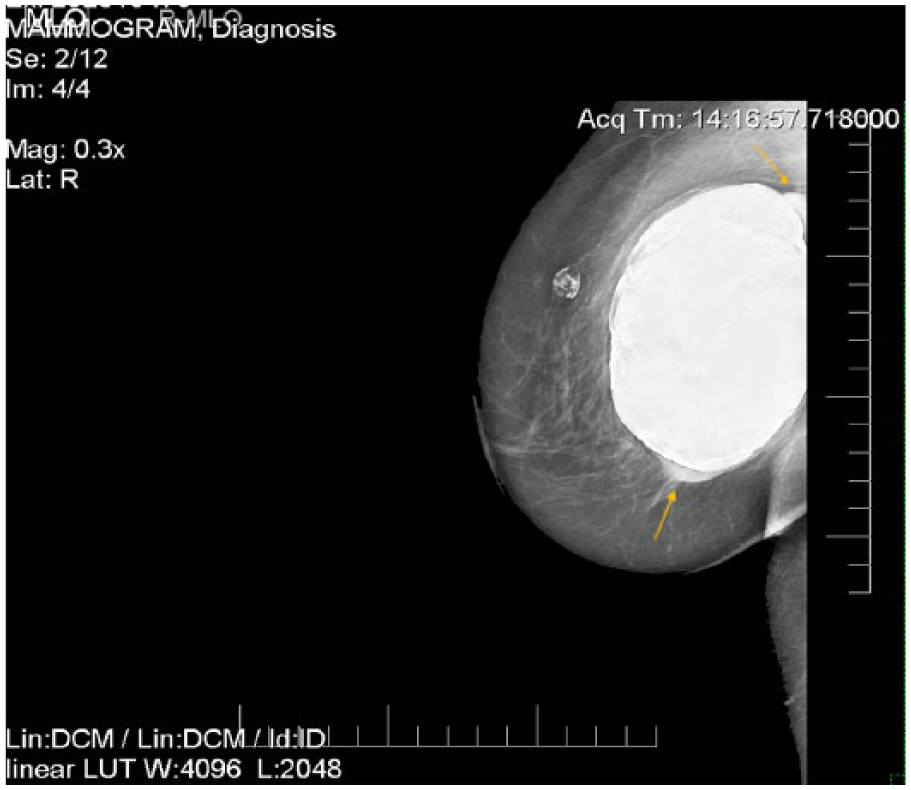
A mammographic mediolateral oblique projection of the right breast. The area of calcified fat necrosis is seen as well as areas of extracapsular rupture (arrows).
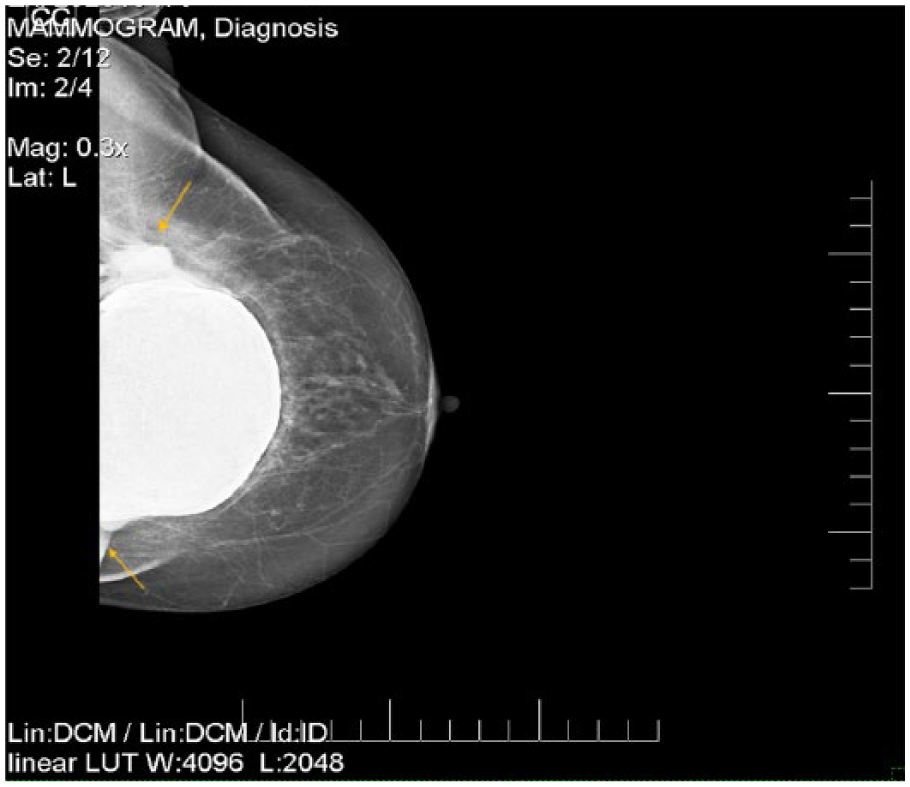
A mammographic craniocaudal projection of the left breast. Areas of extracapsular rupture are seen (arrows).
A limited bilateral breast sonogram was performed after the mammogram was completed. A Hitachi Arietta 70 ultrasound machine was used with a L55 linear-array transducer at a frequency of 13 MHz. The areas of the breast in which the patient was feeling pain and discomfort corresponded to focal areas of extracapsular silicone, with a snowstorm appearance demonstrated sonographically (Figures 5 and 6). Incidentally, intracapsular rupture was noted bilaterally, representing the “stepladder sign” (Figures 7 and 8). The sonography findings of intracapsular and extracapsular rupture confirmed the diagnosis.
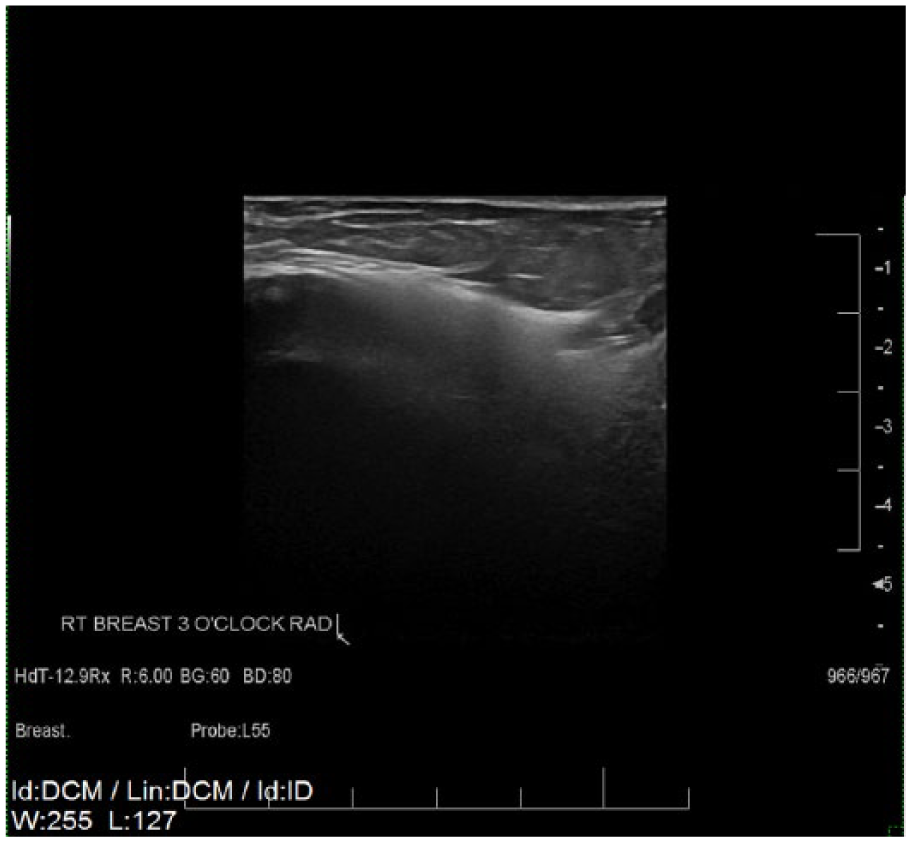
Radial grayscale sonogram of the right breast at 3 o’clock showing the “snowstorm sign” of extracapsular rupture.
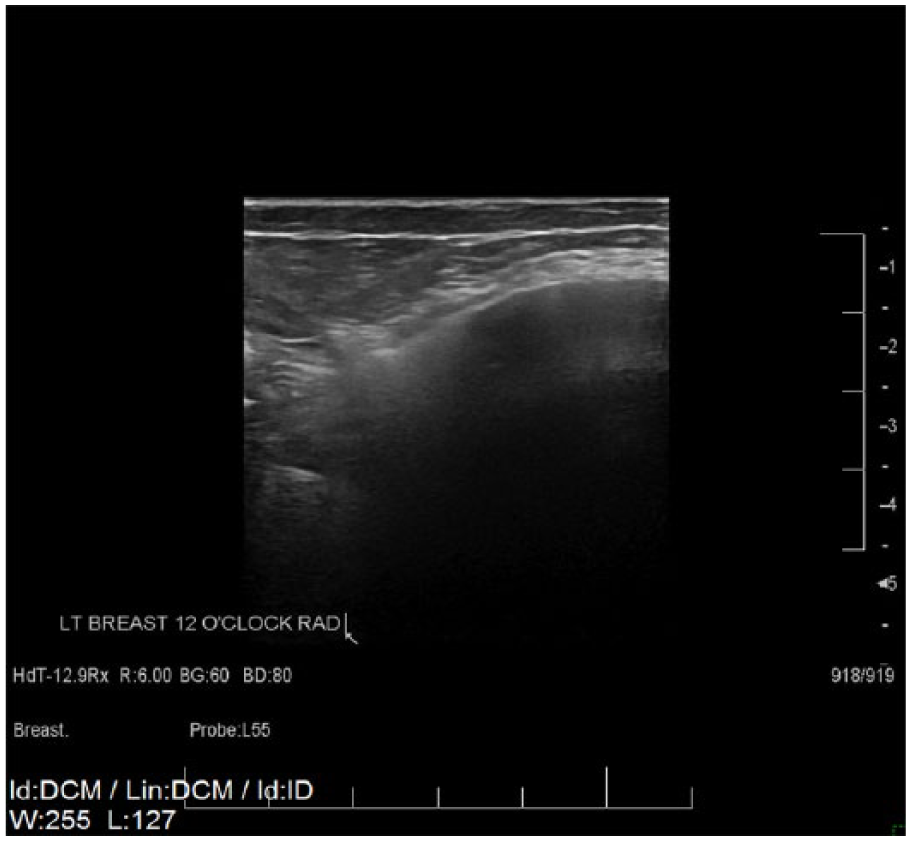
Radial gray-scale sonogram of the left breast at 12 o’clock showing the “snowstorm sign” of extracapsular rupture.
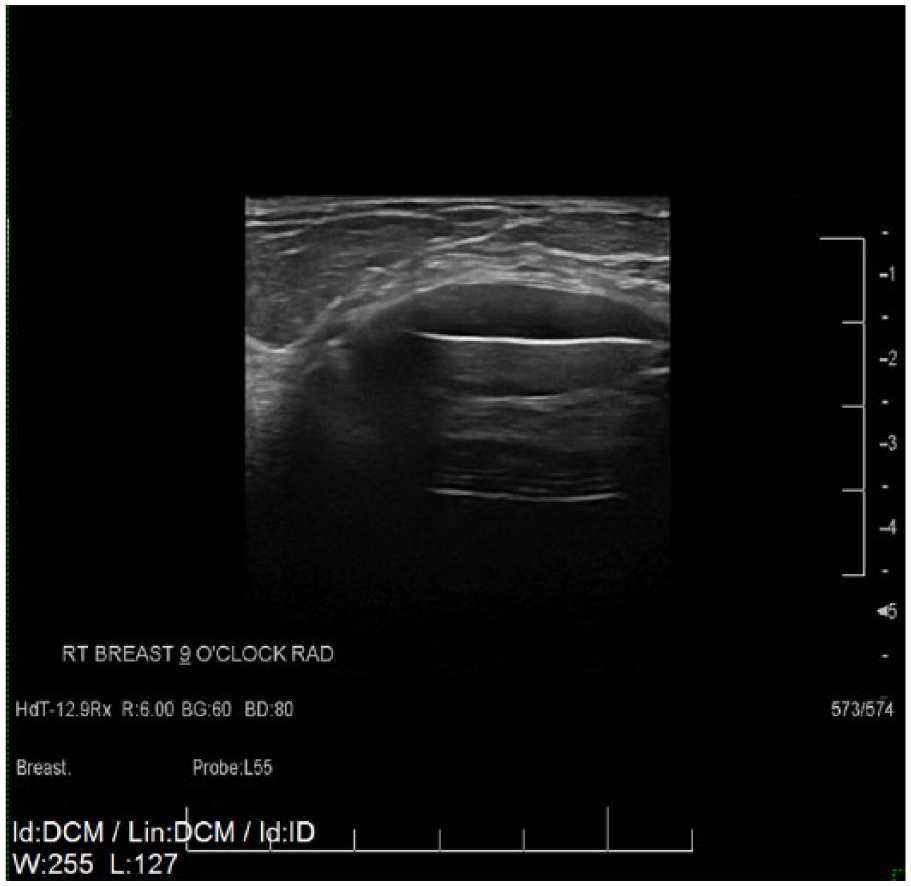
Radial gray-scale sonogram of the right breast at 9 o’clock demonstrating the “stepladder sign” of intracapsular rupture.
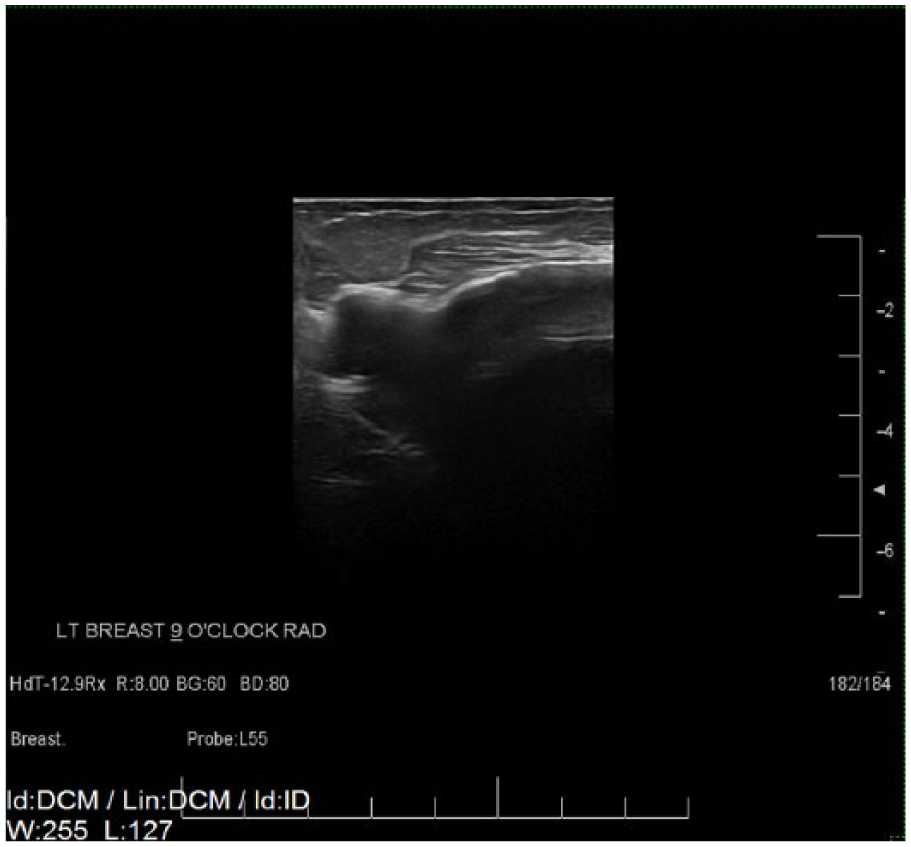
Radial grayscale sonogram of the left breast at 9 o’clock demonstrating the “stepladder sign” of intracapsular rupture and the “snowstorm sign” of extracapsular rupture.
The patient was advised to seek plastic surgery consultation for removal of the breast implants. Since there were no suspicious findings to suggest malignancy, a screening mammogram was recommended in one year.
Discussion
An implant rupture is described as a tear or hole in the outer shell of the breast implant. 3 When a silicone-filled breast implant is ruptured, the gel may remain within the fibrous shell of scar tissue that forms around the implant. 3 This is defined as an intracapsular rupture. When the silicone escapes outside of this fibrous capsule, it is known as an extracapsular rupture. By definition, an extracapsular rupture is indicative of an intracapsular rupture. The most common implant rupture is intracapsular. The longer an implant has been in the breast, the stronger the likelihood that the implant will rupture. Rupture most often occurs 10-15 years after implant placement. 2 Rupture rates vary significantly between manufacturers and method of patient evaluation. 2
Implant rupture can have various causes, although most have no traumatic origin. 2 Some possible causes of rupture include capsular contracture, compression during mammogram, damage by surgical instruments, normal aging of the implant, and physical stresses. 3 When a silicone breast implant ruptures, most patients do not experience any clinically significant signs or symptoms; these are known as “silent” ruptures. 2 Silicone implants, in particular, tend to leak slowly, making it difficult for a patient to recognize that a rupture has occurred. However, some patients may notice a change in size or shape of the breast. Individuals may also experience pain, tenderness, numbness, burning, or tingling of the breast. 4
Mammography and sonography are typically the standard first steps in the diagnostic workup of imaging breast implants, but MRI is thought to be the most useful imaging modality. 5 MRI produces high spatial resolution and contrast between the soft tissue of the breast and the implant, allowing for the highest sensitivity and specificity for assessing the integrity of the implant. 5 The usefulness of sonography for detecting implant ruptures varies. Although sonography does not use ionizing radiation, it is highly operator dependent. 5 With the aid of an experienced and well-trained sonographer, a negative sonogram can strongly support the integrity of the implant, which can limit the application of mammography or MRI to suspicious cases only. 5
Once a silicone gel implant has ruptured, the FDA recommends that it be removed as soon as possible. The longer the silicone is allowed to stay in the body, the more time it has to migrate to other parts of the body. 4 If the implant is not removed after it has ruptured, the silicone may migrate to other locations in the body and cannot be removed, causing other health problems. For example, a study conducted by the Office of Surveillance and Biometrics, Center for Devices and Radiological Health of the FDA, found women with ruptured silicone breast implants were more likely to have certain health issues than women who did not have ruptured implants. 5 The study established that women with ruptured silicone gel-filled breast implants were significantly more likely to be diagnosed with fibromyalgia, polymyositis, Hashimoto’s thyroiditis, pulmonary fibrosis, eosinophilic fasciitis, and polymyalgia than other women in the study. 5
Sonographically, an intact implant appears as a well-defined anechoic area with an echogenic shell. An intracapsular rupture is exhibited as the “stepladder sign” with sonography, representing a series of horizontal echogenic straight or curvilinear lines crossing the interior of the implant. 6 An extracapsular rupture appears as the “snowstorm sign.” This consists of a group of focal nodules with a widespread increase in echogenicity of the breast tissue and loss of normal parenchymal boundaries. 5 Other breast implant complications that may be differential diagnoses include a gel bleed, contracture, seroma, or infection. Unlike intracapsular and extracapsular rupture, a gel bleed is not detected with sonography since there is only a microscopic diffusion of silicone through an intact shell. A contracture on sonography will be confirmed by deformation of the implant, increased radial folds, and thickening of the fibrous capsule. 7 Seromas and infection are noted as fluid collections around the implant, which are easily differentiated from the signs of rupture.
Mammography, sonography, CT, and MRI have all been used to detect breast implant rupture, however MRI has long been accepted as the imaging modality of choice for detection. On MRI, the “linguine or teardrop sign” is a classic indication of an implant rupture. Limitations of MRI include the expense, the requirement for machines with specific breast coil technology, the time needed to complete the study, and patient limitations due to other medical device implants. 2 Sonography has recently become a popular alternative to MRI due to advantages such as decreased cost and time. The use of sonography for detecting breast implant rupture is limited by the fact that the modality is highly operator dependent. 2 Therefore, to optimize the detection of subtle signs of implant rupture it is critical that the sonographer be well trained in breast sonography.
When comparing mammography to the use of sonography, mammography has been considered a rather insensitive method for detecting breast implant rupture because it cannot consistently detect an implant that has ruptured within an intact fibrous capsule. 8 Conversely, sonography can reliably detect both intracapsular and extracapsular rupture. However, MRI is considered slightly more effective than sonography for detecting implant ruptures, particularly small intracapsular ruptures. 8 Sonography remains an operator-dependent imaging modality, unlike MRI. This could be the reason for the higher rate of detection of implant rupture with MRI. When using all sonographic signs of rupture, sonography has a sensitivity as high as 85% whereas the sensitivity of MRI is approximately 90%. 8 These statistics were prepared roughly 23 years ago, so they do not take into account major advancements in sonography that have occurred in more recent years. No current statistics for sensitivity of sonographic detection of breast implant rupture are available in peer-reviewed literature. Nonetheless, it is appropriate to assume that with modern-day ultrasound technology and equipment, sonography is better equipped to detect implant rupture than in previous years.
Conclusion
Silicone breast implant ruptures have been defined as intracapsular or extracapsular. With sonography, an intracapsular rupture is demonstrated as the “stepladder sign” and an extracapsular rupture is recognized as the “snowstorm sign.” MRI is thought to be the most accurate imaging modality for diagnosing breast implant rupture. However, this case demonstrates that sonography can play an important role in detecting and confirming breast implant rupture if the operator is well trained and knowledgeable. Sonography provides an examination that is lower in cost and quicker than MRI. The use of sonography to confirm implant rupture allows for a quick diagnosis in order to establish the appropriate course of treatment.
Footnotes
Acknowledgements
The authors thank Jennifer Culler, RDMS, for her image contribution, continuous encouragement, assistance, and guidance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
