Abstract
Dilated cardiomyopathy (DCM) is a disease of the myocardium characterized by changes in cardiac chamber dimension and initial deterioration in systolic function accompanied by eventual diastolic function impairment with possible progression to cardiac failure. Early identification of ventricular dysfunction and etiology is imperative for the subsequent effective treatment of the cardiomyopathy process. One common cause of DCM is an infection from a viral insult that results in inflammation and consequent damage to the myocardium. Viral myocarditis may present as an acute or chronic illness. The clinical presentation of myocarditis demonstrates nonspecific symptomology that is variable between individuals. Accurate diagnosis of viral myocarditis relies upon various diagnostic testing methods to ascertain the origin of the infectious agent and resultant alterations in cardiovascular function. One such diagnostic testing method used in the detection of the presence and severity of DCM associated with viral myocarditis is echocardiography. The following case presentation demonstrates a dilated cardiomyopathy due to an acute myocarditis from Coxsackie B enterovirus evaluated with echocardiography.
Myocarditis is an infectious process that initiates inflammatory changes in the myocardium. This can result in an alteration to both the structure and function of the heart in the absence of ischemia. 1 The spectrum of causative agents of myocarditis is broad and varies from viral, bacterial, and fungal infections to autoimmune diseases and, in rare cases, hypersensitivity reactions to pharmacological agents. Due to the wide range of etiologies, the global prevalence and incidence of myocarditis has been difficult to accurately document, 2 but the most common etiology of myocarditis in developed countries remains myocarditis that is viral in origin. 3 Common viral agents involved in the myocarditis process include the enteroviruses, adenoviruses, and parvoviruses. The following clinical case presentation provides a case report of a patient with viral myocarditis from Coxsackie B (a common enterovirus), which presented as acute congestive cardiac failure (CCF) secondary to dilated cardiomyopathy (DCM).
Case Description
A 61-year-old woman presented to the emergency department of an acute care hospital under direction from a
Echocardiography Findings
The echocardiogram showed severe dilation of the left ventricular (LV) cavity (by American Society of Echocardiography chamber quantification criteria 4 ) with severe reduction in global systolic function: Simpson’s biplane LV volume of 137 mL (corrected for body surface area equal to 87 mL/m2), and the 2D Simpson’s biplane LV ejection fraction (LVEF) was calculated at 19%. Figure 1 and Figure 2 demonstrate Simpson’s four-chamber diastolic and systolic volumes, with Figures 3 and 4 demonstrating Simpson’s two-chamber diastolic and systolic volumes, as well as the biplane volumes and ejection fraction (EF) percentage. The presence of intracavity thrombus was excluded.
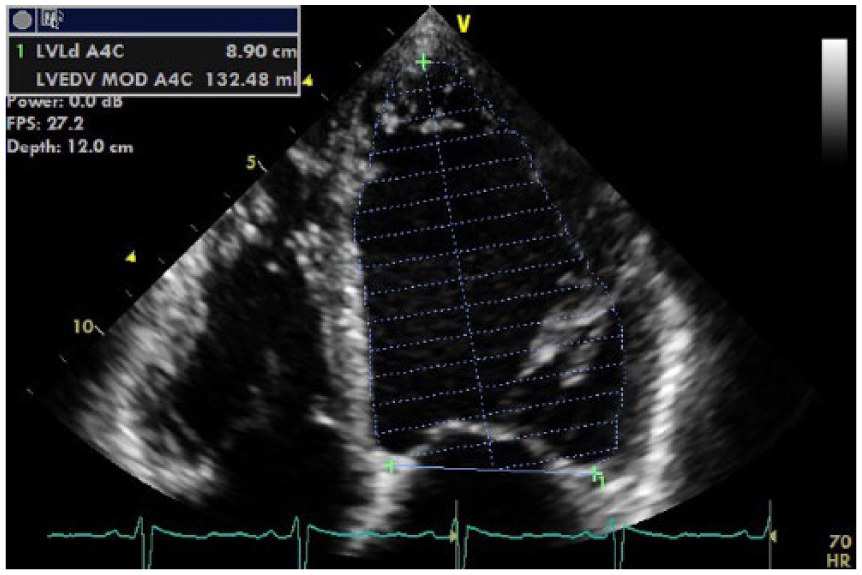
Apical four-chamber view of Simpson’s 2D left ventricular end diastolic (LVED) volume. The value of 132 mL for the LVED volume demonstrates severe dilation (normal, 94 ± 23 mL) (according to Lang et al., 4 measurements exceeding ±1.96 standard deviations or the 95% confidence interval should be classified as abnormal).
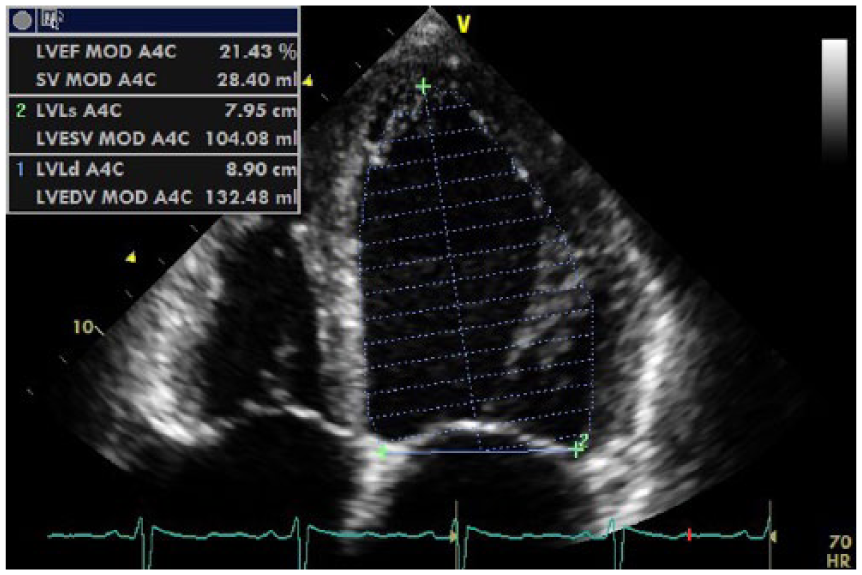
Apical four-chamber view of Simpson’s 2D left ventricular end systolic (LVES) volume. The value of 104 mL for the LVES volume is significantly abnormal (normal, 36 ± 12 mL). 4 This figure also demonstrates a Simpson’s 2D single-plane ejection fraction (EF) for the four-chamber view of only 21% (normal, 62% ± 8%). 4 The value of the four-chamber EF percentage demonstrates a profound decrease in systolic function.
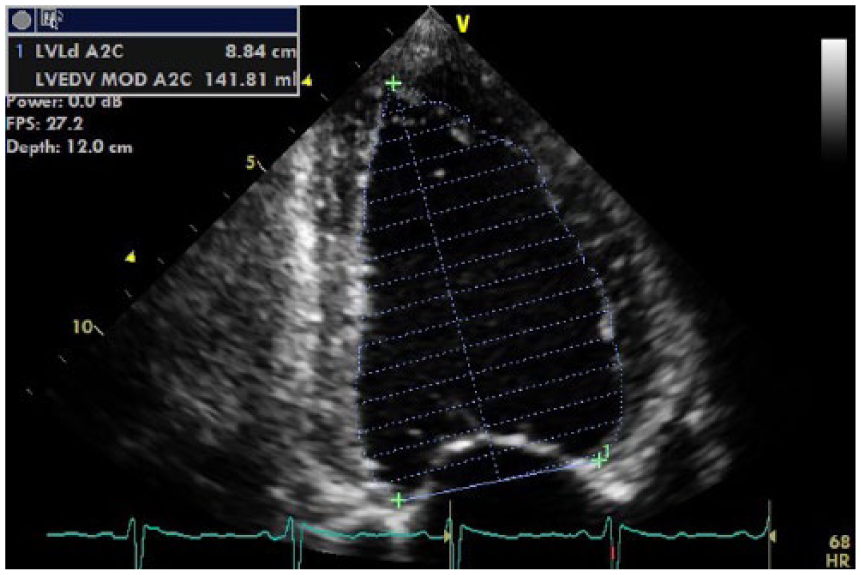
Apical two-chamber view of Simpson’s 2D left ventricular end diastolic (LVED) volume. The value of 141 mL for the LVED volume demonstrates severe dilation (normal, 87 ± 23 mL). 4
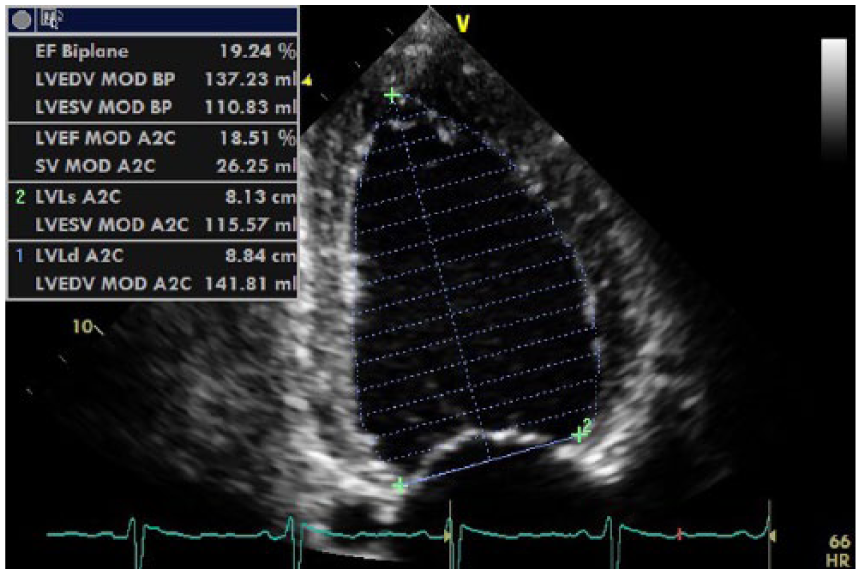
Apical two-chamber view of Simpson’s 2D left ventricular end systolic (LVES) volume. The value of 115 mL for the LVES volume is significantly abnormal (normal, 32 ± 11 mL). 4 This figure demonstrates a Simpson’s 2D single-plane ejection fraction (EF) for the two-chamber view of only 18.5% (normal, 64% ± 6%). 4 This figure also quotes Simpson’s 2D biplane left ventricular end diastolic (LVED) volume and biplane EF percentage. The biplane measurements demonstrate a significant degree of dilation of the left ventricle (biplane LVED volume 137 mL; normal, 76 ± 15 mL), 4 with a significant degree of systolic dysfunction (biplane EF percentage 19%; normal, 64% ± 6%). 4
LV diastolic function was severely impaired and consistent with restrictive physiology, which was evidenced by a mitral inflow deceleration time (MDT) of 155 ms with a mitral E/A ratio of 2.48 (Figure 5) and a global e/e′ ratio of 21.7. Figures 6 and 7 show tissue Doppler imaging (TDI) indicating an increase in the left ventricular end diastolic pressure (LVEDP), and a left atrial (LA) biplane volume/body surface area (BSA) of 56 mL/m2, indicating a reduction in LV compliance. The finding of a restrictive filling pattern is usually associated with heart failure and a poor prognostic outcome. 5
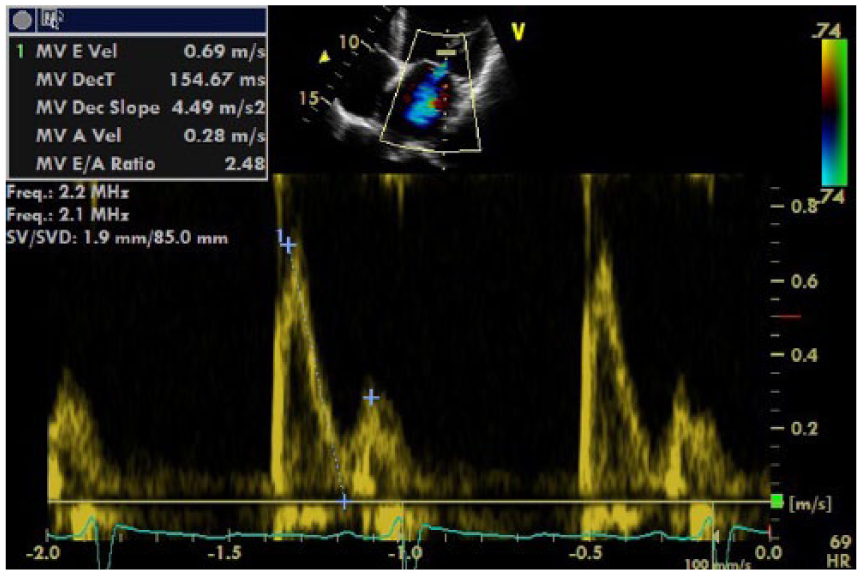
Mitral valve inflow spectral Doppler shows a pattern of restrictive diastolic dysfunction of the left ventricle. The E/A ratio was 2.48, which greatly exceeds the normal reference range for the sex and age of this patient (normal E/A ratio, 0.96 ± 0.32), 5 as well as a shortened mitral deceleration time (MDT) of 154.67 ms (normal, MDT 244 ± 79 ms). 5
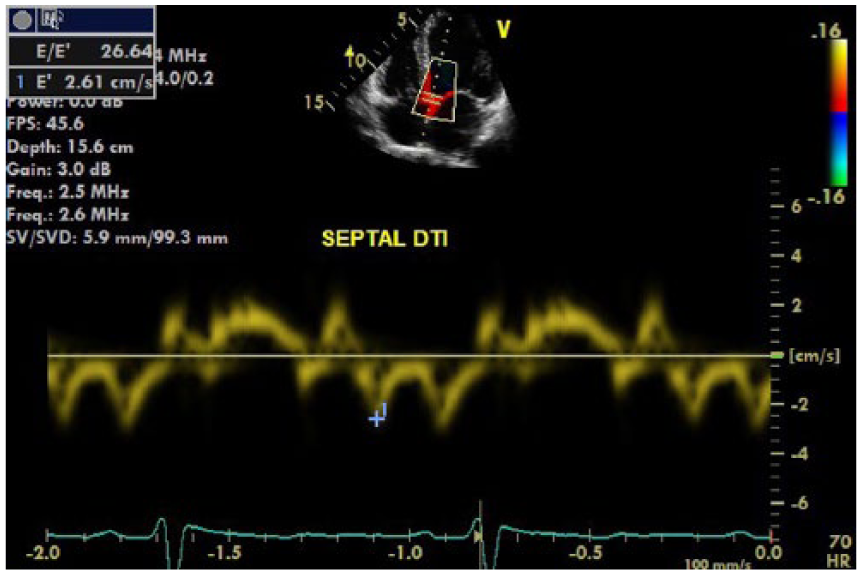
Left ventricular septal tissue Doppler imaging (TDI). The e′ velocity of 2.6 cm/s is significantly reduced (e′ velocity <5 is consistent with severe diastolic dysfunction 5 ) with a significantly increased E/e′ ratio of 26.6 (abnormal ≥15), 5 implying restrictive diastology with elevated left ventricular end diastolic pressure.
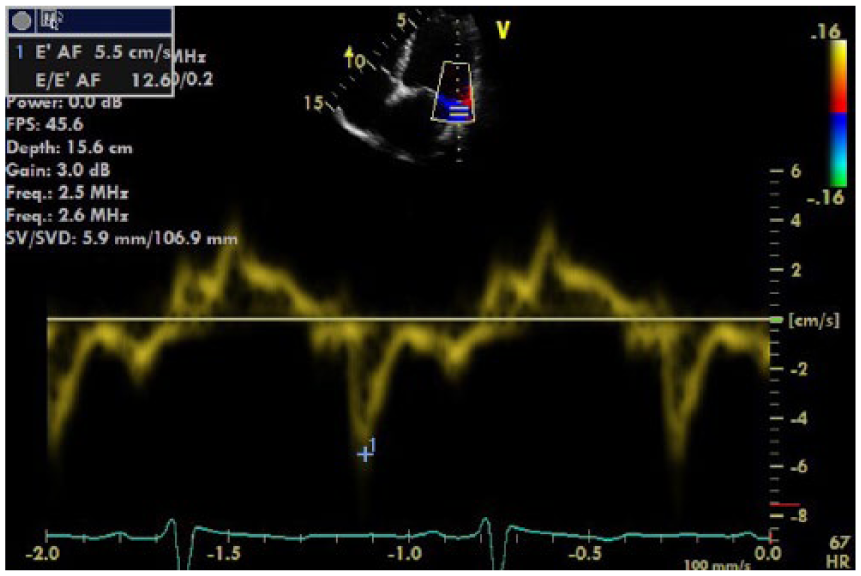
Left ventricular lateral tissue Doppler imaging demonstrating reduction in relaxation and increase in left ventricular end diastolic pressure.
The right ventricular (RV) systolic function was also reduced with an RV systolic annular velocity (S′) of 7.5 cm/s (Figure 8). There was coexistent moderate mitral regurgitation consistent with mitral annular dilation due to the altered left ventricular geometry. The RV systolic pressure was within normal limits (34 mm Hg). However, the accuracy of this measurement was limited in the setting of the impaired RV systolic function. A mean pulmonary artery pressure was unable to be estimated to accurately provide an approximation of the pulmonary conditions. In summary, the echocardiographic findings were consistent with acute biventricular failure initiated by dilated cardiomyopathy associated with acute viral myocarditis.
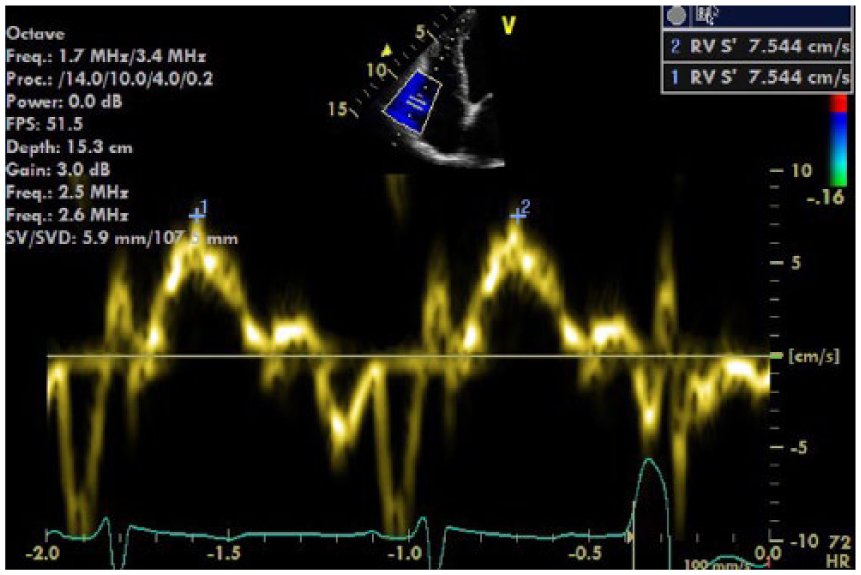
Right ventricular (RV) S′ of 7.5 cm/s demonstrates a reduction in RV systolic function (RV S′ velocity <9.5 cm/s indicates RV systolic dysfunction). 4
Treatment and Outcomes
The patient in this case report was treated with a traditional approach, commencing on a low-dose diuretic (Lasix 20 mg/d) and an angiotensin-converting enzyme (ACE) inhibitor (Coversyl 2.5 mg/d), to which the patient responded well, demonstrating a reduction in shortness of breath. A low-dose beta-blocker (bisoprolol 1.25 mg/d) was initially administered and with a gradual increase over a period of three months.
At the three-month mark, the patient had a follow-up echocardiogram that documented an improvement in LV systolic function from the original EF of 19% to 37% (Figures 9–12), indicating an improvement from severe to moderate LV systolic dysfunction. An improvement in LV diastolic function was also noted, with the patient now demonstrating a pattern of abnormal relaxation (Figure 13). The LVEDP remained relatively unchanged, with only a slight improvement in the septal e′ velocity of 3.5 cm/s with a calculated e/e′ ratio of 20 (improved from 26.6) (Figure 14).
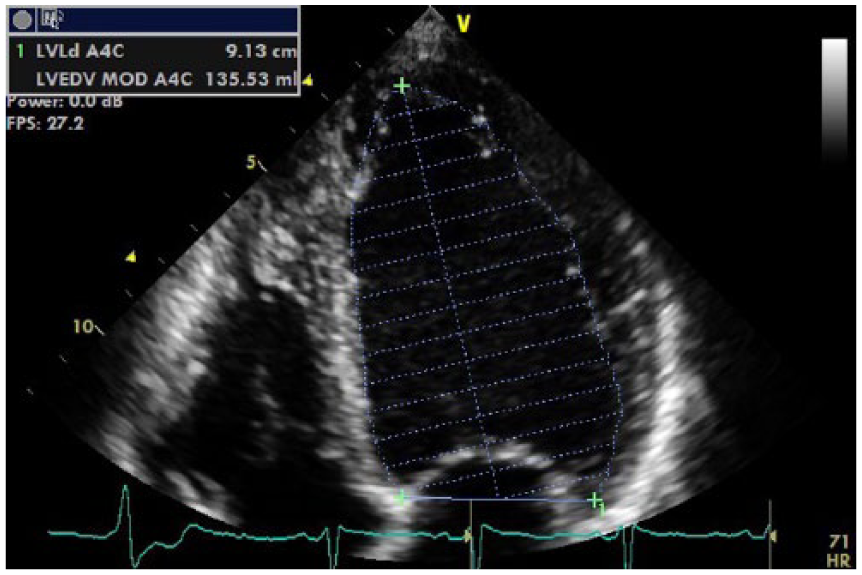
Apical four-chamber view of Simpson’s 2D diastolic volume posttreatment.
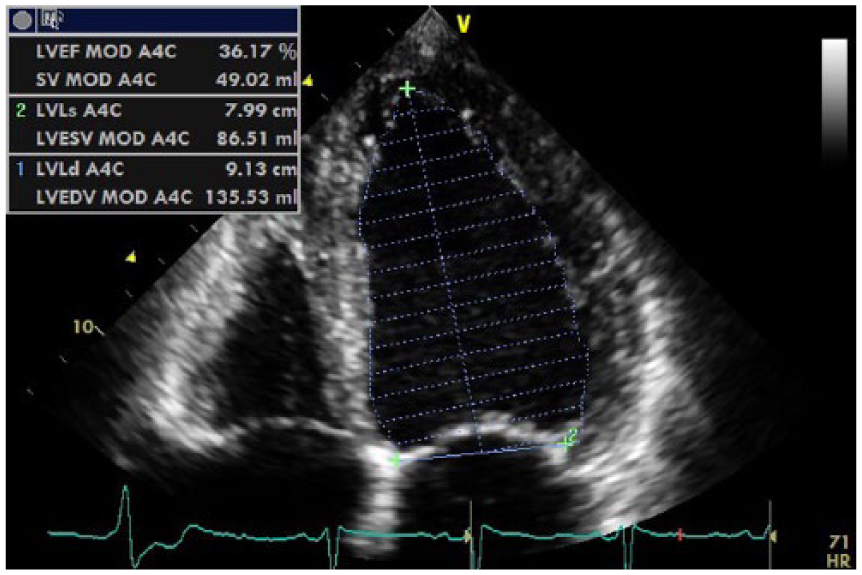
Apical four-chamber view of Simpson’s 2D systolic volume posttreatment.
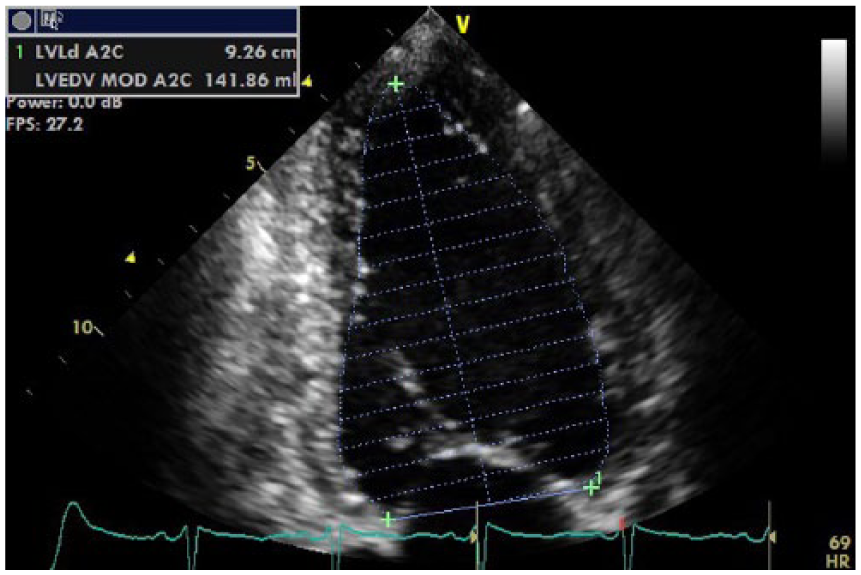
Apical four-chamber view of Simpson’s 2D biplane diastolic volume posttreatment.
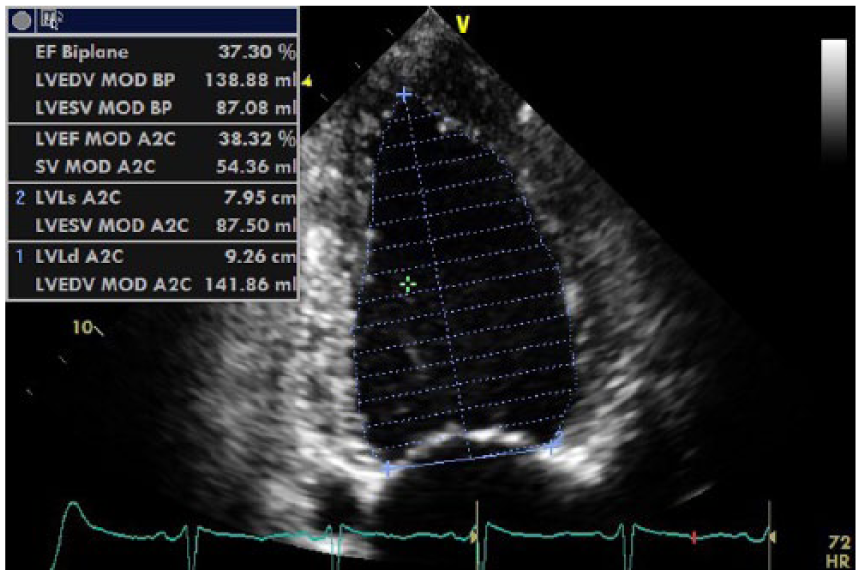
Apical two-chamber view of Simpson’s 2D systolic volume posttreatment This figure quotes Simpson’s 2D biplane left ventricular end diastolic pressure and biplane ejection fraction (EF) posttreatment. The biplane measurements demonstrate improvement in the systolic function of the left ventricle, with the biplane EF percentage increasing from 19% (initial presentation) to 37% (posttreatment).
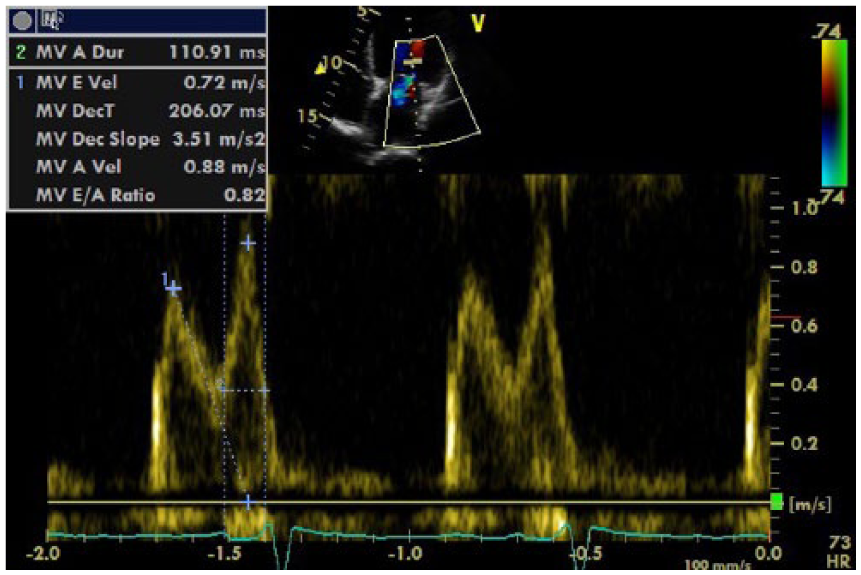
Mitral valve inflow spectral Doppler shows a pattern of abnormal relaxation posttreatment with the E/A ratio of 0.82 and the mitral inflow deceleration time (MDT) 206 ms (mild left ventricular diastolic dysfunction is demonstrated by an MDT >200 ms and an E/A ratio of <0.8). 5 This demonstrates an improvement in the diastolic function of the left ventricle as the pattern of severe diastolic dysfunction (restrictive diastology) has improved to a pattern of mild diastolic dysfunction (abnormal relaxation).
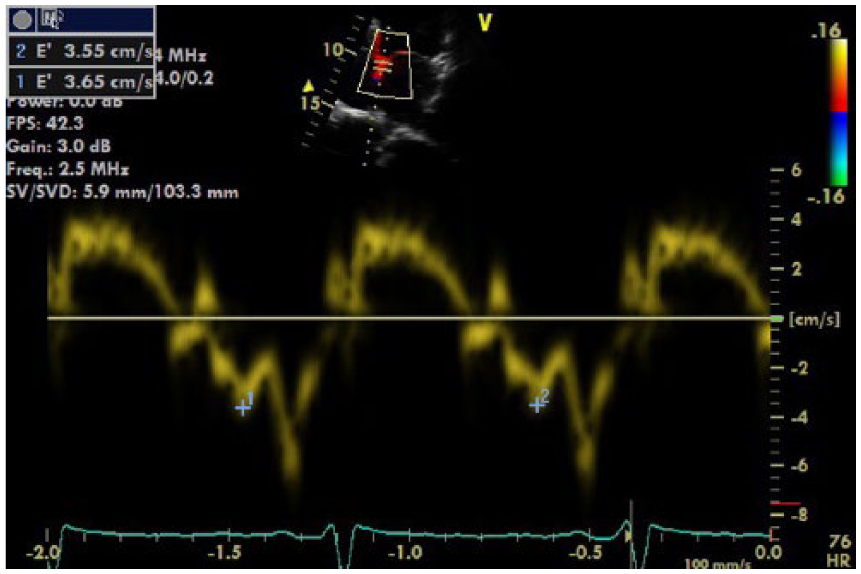
Left ventricular (LV) septal tissue Doppler imaging demonstrating a slight improvement in e′ velocity posttreatment with minimal change noted in the left ventricular end diastolic pressure (LVEDP). This finding is suggestive of an improvement in the degree of LV diastolic dysfunction with only a minor reduction in the LVEDP.
The RV systolic function also demonstrated an improvement with an RV S′ = 9.8 cm/s (Figure 15). The value of the RVSP had not changed (33 mm Hg), but considering that the same value for the RVSP was measured in the setting of the RV systolic dysfunction, it was reported that pulmonary pressures had normalized in the setting of improved RV systolic function.
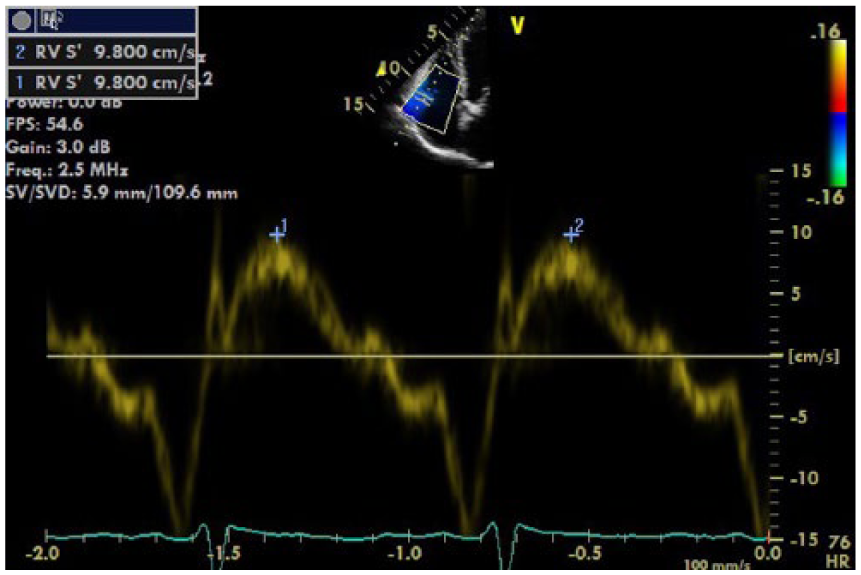
Right ventricular S′ of 9.8 cm/s (normal, >9.5 cm/s) 4 demonstrates an improvement in the systolic function of the right ventricle in response to treatment.
Discussion
DCM can develop during an acute episode of Coxsackie B virus. 6 Common nonspecific presenting symptoms with acute viral myocarditis include dyspnea, fatigue, and upper respiratory tract symptoms, 3 all of which this patient demonstrated. The pathophysiology of viral myocarditis from Coxsackie B has been previously postulated as an immune-mediated destruction of the myocardium via an inflammatory process followed by infiltration of the myocardium with fibrotic constituents. 7
Myocarditis is caused from an external source, such as Coxsackie B enterovirus, that elicits varying immune responses. The current consensus regarding the pathogenesis of viral myocarditis describes a three-phase pathophysiological process. The first phase is an initial acute viral infection that may involve direct cytotoxic destruction of the myocardium. 8 The initial infection results in a secondary activation of the host immune system (a subacute autoimmune response in which inflammatory processes ensue), which is followed by a third and chronic myocardial injury phase.2,3,6,8 Not all patients exhibit all three phases, but when progression to the third phase occurs, the result is the development of DCM due to the inflammatory response that causes fibrotic changes within the myocardium and eventual myocyte necrosis. 2
The approach to treatment for acute heart failure is dependent upon the degree of elevation in LV filling pressure (which is reflected as a volume overload of the LV) and the peripheral consequences of the reduction in cardiac output. Nohria et al. 9 proposed that acute heart failure could thus be classified hemodynamically as wet versus dry (based on whether the patient is volume overloaded or not) and cold versus warm (based on the degree of peripheral tissue perfusion, which is a reflection of the degree of reduction of cardiac output). Treatment of acute heart failure (HF) can be best tailored to the individual patient based on this classification scheme.
The goal of treatment of a patient with acute HF is to normalize the LV filling pressures, which decreases the LV volume overload. This results in reducing the consequences of the congestive component, thereby improving peripheral tissue perfusion by increasing cardiac output. 7
The role of pharmacological treatments in the acute HF patient focuses on the use of loop diuretics to decrease fluid retention (improve volume overload), the use of ACE inhibitors or angiotensin II receptor blockers (ARBs) to improve cardiac output and increase peripheral vasodilation, and the use of beta blockers (due to their cardioprotective effects from an excess of circulating catecholamines). The treatment of patients with acute heart failure due to viral myocarditis with antiviral and/or anti-inflammatory drugs has not been found to improve either short- or long-term outcomes significantly, as many patients with acute HF from myocarditis often improve without immunosuppression therapy. 10
Current pharmacological therapies for the treatment of acute HF are effective in the short term at improving both hemodynamic stability and patient symptomology but are not effective at improving long-term morbidity or mortality. 11 The adverse effects of the current treatment regimen for acute HF relate directly to inappropriate dosage and range from the initiation of cardiac conduction system abnormalities to arterial vasospasm to further deterioration from negative inotropic effects (related to beta blockers) to renal insufficiency, cough, and hypotension (ACE inhibitors). Due to the lack of positive long-term outcomes in acute HF patients, novel approaches to treatment that reduce neurohormonal imbalance (such as V2 arginine antagonists and adjunctive therapy with digoxin) are being considered as a future approach to treatment in these patients. 12
Role of Echocardiography in DCM
There are no specific echocardiographic diagnostic criteria associated with viral myocarditis, but echocardiographic evidence of DCM is well documented, with the echocardiographic features being relatively uniform for a DCM regardless of the etiology of the DCM. 5 Echocardiographic criteria of the presence of DCM include the following:
An LV end diastolic diameter of greater than 112% of the predicated value (corrected for age and BSA) 13 combined with an EF of less than 45%
An LV end diastolic volume corrected by BSA of greater than 75 mL/m2
An increase in LV mass and evidence of LV diastolic dysfunction with associated elevation of left atrial pressure 13
LV wall thickness in DCM may be normal or increased. When LV wall thickness is increased, these cases are more likely associated with severe cases of myocarditis (fulminant myocarditis), in which the increase in wall thickness is related to the active inflammatory process. 3
A useful criterion in the echocardiographic investigation of DCM is the assessment of the pattern of LV systolic function. A pattern of global hypokinesis with a spherical alteration in ventricular geometry is more likely to be associated with myocardial injury from a myopathy process rather than regional abnormalities, which are more likely to be associated with myocardial injury from ischemia. 13 Remodeling of the LV from elliptical to spherical in shape is attributed to a greater increase in the short axis length in comparison to the increase in the long axis length of the LV. 13 Some authors have proposed that the degree of spherical remodeling can be described by the sphericity index (long-axis dimension/short-axis dimension), which in patients with advanced DCM approaches a value of 1. 13 RV systolic dysfunction is also more likely to be associated with a cardiomyopathy process than an ischemic process when there is coexistent LV systolic dysfunction. The presence of biventricular systolic dysfunction in DCM is commonly associated with a poorer prognostic outcome. 14
The use of TDI is an essential parameter in the assessment of diastolic function of the left ventricle. The velocities obtained using TDI reflect the degree of shortening of myocardial fibers in the longitudinal plane of the mitral annulus. The benefit of this measurement is that it can provide an index of LV relaxation in early diastole that is relatively load independent. 5 Patients with diastolic dysfunction will demonstrate a decrease in the e′ velocity due to a decrease in myocardial fiber shortening with a corresponding increase in the E/e′ ratio as the LVEDP increases. 5
The essential cardiac features that should be assessed with echocardiography in a patient with DCM attributed to myocarditis include the assessment of the following5,13,15:
Size of the ventricles
Geometry of the ventricles (spherical vs. elliptical; sphericity index)
Pattern of contractility (regional or global)
Cardiac output via EF estimation
LV wall thickness and LV mass
Loading conditions (i.e., the pulmonary and LV filling pressures)
Associated valvular regurgitation due to chamber dilation
Presence of intracavity thrombus
By assessing the size and function of the ventricles, the severity of the DCM and its complications can be classified and thus the treatment can be tailored for the patient.
Conclusion
As can be seen by the echocardiographic data acquired in this case, the patient presented with a severe DCM as evidenced by severe dilation of the LV, severe impairment of LV systolic function, significant impairment in LV diastolic function, an associated reduction in RV systolic function, and the presence of moderate mitral regurgitation due to LV annular dilation. The role of echocardiography in this patient proved to be of particular use in the investigation of the cause of the presenting symptoms as the patient was subsequently diagnosed with congestive cardiac failure secondary to DCM from acute viral myocarditis from Coxsackie B enterovirus. Due to a definitive diagnosis, appropriate medical treatment was commenced, which improved cardiac function and alleviated patient symptomology.
Footnotes
Acknowledgements
The author thanks Brad Musumeci, co-supervising sonographer, Logan Heart Services, for provision of the echocardiographic images; Dr. Gillian Whalley (Academic Lead and Health Researcher, Unitec Institute of Technology) and Dr. Rebecca Perry (postdoctoral Fellow, The Australian Heart Foundation) for their professional support and editing expertise; and Dr. Naveen Dwivedi, MBBS, FRACP, FCSANZ, cardiologist, Logan Heart Services, for provision of patient clinical information.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
