Abstract
The objective was to determine the image quality of 3D diagnostic medical sonography (DMS) in children with hydronephrosis. 3D DMS was assessed based on 24 pediatric patients. Image quality was evaluated by two radiologists and a sonographer in terms of rib shadowing, cut-off parenchymal edges, motion artifact, and overall quality. The interreader reliability and relation between image quality and other variables were calculated. The results were based on images of 32 hydronephrotic kidneys. The average quality scoring of the images was quite high. Rib shadowing, cut-off edges, and motion artifact were present in the majority of the cases. The interreader reliability for overall quality, rib shadowing, cut-off, and motion was quite high. There was a correlation between the Society for Fetal and Neonatal Urology’s hydronephrosis grade and higher cut-off edges. Larger kidneys were more likely to show cut-off, motion, and lower quality scores. In this cohort of infants and toddlers with hydronephrotic kidneys, 3D DMS demonstrated good image quality; however, artifacts were attributed to kidney size and severity of hydronephrosis.
Introduction
Hydronephrosis is the dilatation of the renal pelvis and calices and has been reported to be identified in 1.4% to 4.5% of fetuses.1,2 It remains present in 50% of children diagnosed prenatally. 2 Hydronephrosis suggests obstruction, often of the ureteropelvic junction, but does not confirm it; therefore, further imaging is required to establish anatomy and function. 3 Renal sonography, typically two-dimensional, is the accepted first imaging method to confirm hydronephrosis. 4 Sonography helps determine renal size, echotexture, degree of caliectasis, parenchymal thickness, presence of a duplicated system, and ureteral dilatation in children. In addition to sonography, patients with concerning imaging findings, decreased renal function, or suspected anomalous organ development undergo renal function testing (i.e., nuclear medicine diuretic renograms). 5 However, there is no consensus on the extent and mode of postnatal imaging after a prenatal diagnosis of hydronephrosis. 6 Newer applications of two-dimensional diagnostic medical sonography (2D DMS) include semiautomatic quantification of hydronephrosis severity. 7 This is postprocessing imaging software that uses the raw data from imaging acquisition to calculate multiple parameters such as size of the collecting system, depth of calices, thickness of parenchyma, geometric shape, circularity ratio, and curve descriptors. These measurements are used to identify thresholds of clinically significant abnormal washout times at nuclear medicine diuretic renograms, with initial results reporting up to 100% sensitivity. 7 Postprocessing sonographic imaging data could potentially avoid up to 62% of all nuclear medicine studies currently performed in children with hydronephrosis. 7 However, because standard 2D imaging can result in incomplete visualization of the collecting system due to geometric limitations, 3D DMS could provide volumetric evaluation and more complete visualization of the collecting system.8,9 3D DMS also has the potential to decrease imaging time by requiring a single place sweep with reconstruction in multiple planes at a later time. Hence, there is renewed interest in 3D DMS to support automated segmentation of the kidneys and collecting systems, which promises to be a more appropriate technology for this purpose than traditional 2D DMS.
In the hydronephrotic kidney, 3D DMS has been used to calculate individual and split renal parenchymal volume.10–13 However, in initial reports from Europe, scanning was performed by a pediatric radiologist with more than 20 years of experience and findings were not validated by additional users. 8 Different from those initial reports, it is common in the United States for studies to be performed by sonographers. Moreover, the quality of 3D DMS in hydronephrotic kidneys in children has not been reported. The objective is to determine the image quality of 3D renal sonography in children with hydronephrosis using current technology in a standard clinical setting.
Materials and Methods
All patients’ parents provided consent for this Health Insurance Portability and Accounting Act–compliant prospective study, which was approved by the hospital’s institutional review board. A convenience sample of children with ages from 2 weeks to 3 years with hydronephrosis diagnosed in utero was identified, and 3D DMS volume data sets were obtained during their scheduled visit for renal sonography. The studies were performed by one of three senior sonographers using a sonography system with an X 6-1 matrix probe (iU22; Philips Medical Systems, Bothell, WA, USA). Each sonographer received additional training for the specific use of the system and 3D application. 3D data were acquired with the transducer positioned over the midline of the kidneys in the longitudinal axis and using a wide angle sweep (90 degrees). The focal zone depth was adjusted over the renal hilum using a fixed 5-cm focal zone width. The system allowed for the use of harmonic imaging, which was routinely but not universally used. Sonographers followed the hospital’s standard renal and bladder protocol during this study and obtained 1 to 3 volume data sets in the longitudinal view of each kidney in a maximum of three attempts.
The quality of the images was then evaluated in terms of (a) shadowing from adjacent ribs, (b) cut-off parenchymal edges, and (c) motion artifact, using a 4-point Likert-type scale for each category (e.g., 1 = excellent image quality; 2 = good image quality with minimal rib shadow/cut-off/motion; 3 = fair image quality with moderate rib shadow/cut-off/motion; 4 = poor quality/not adequate for diagnosis). Overall quality scores of 1 (excellent; Figure 1), 2 (good image quality), 3 (fair image quality; Figure 2), and 4 (not adequate for diagnosis) were also assigned. All of the scores were assigned by two fellowship trained pediatric radiologists and a senior sonographer during a single session.
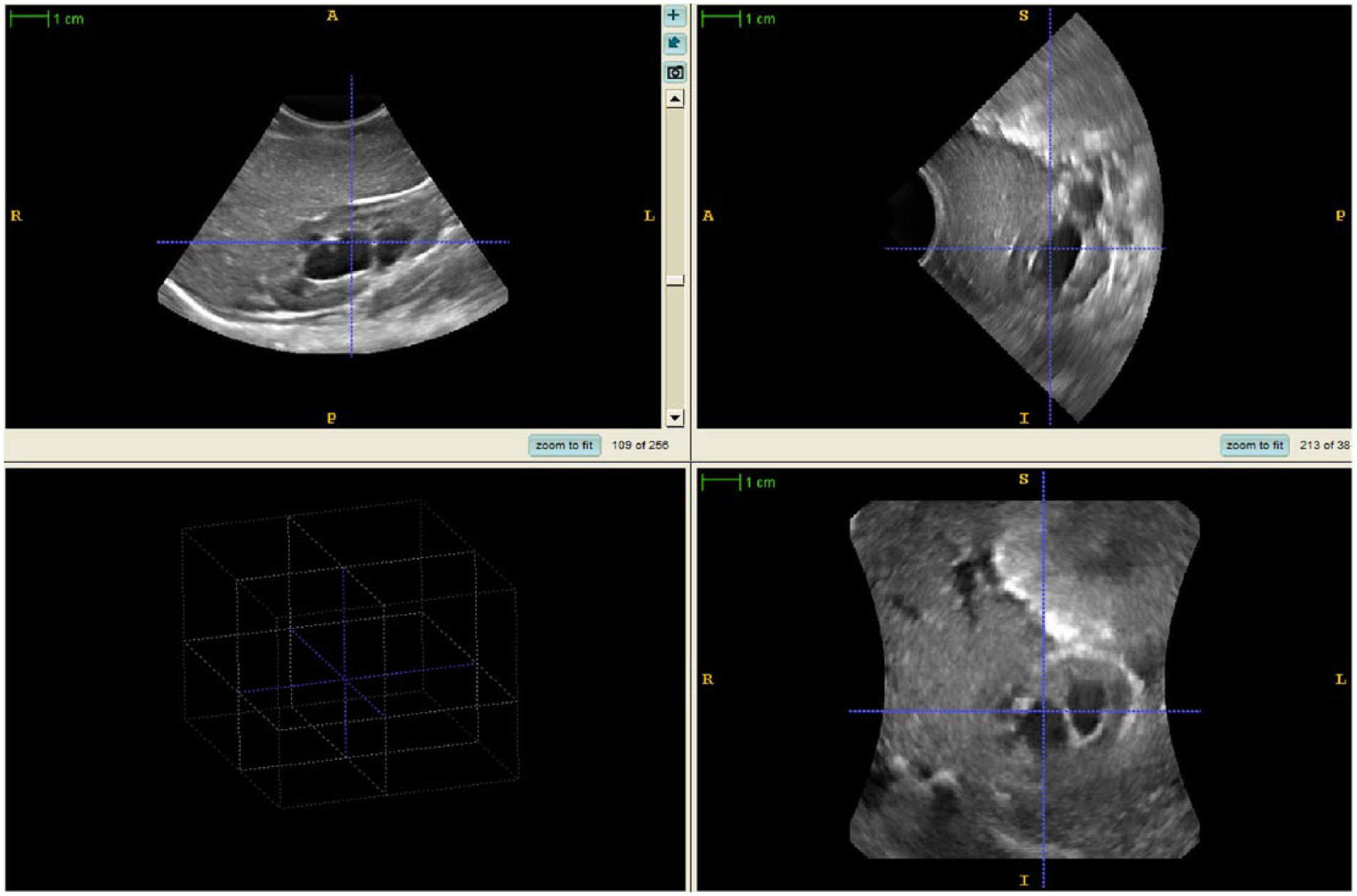
Three-dimensional sonographic image demonstrating excellent image quality of a moderately (grade 3) hydronephrotic right kidney in an 8-month-old male patient.
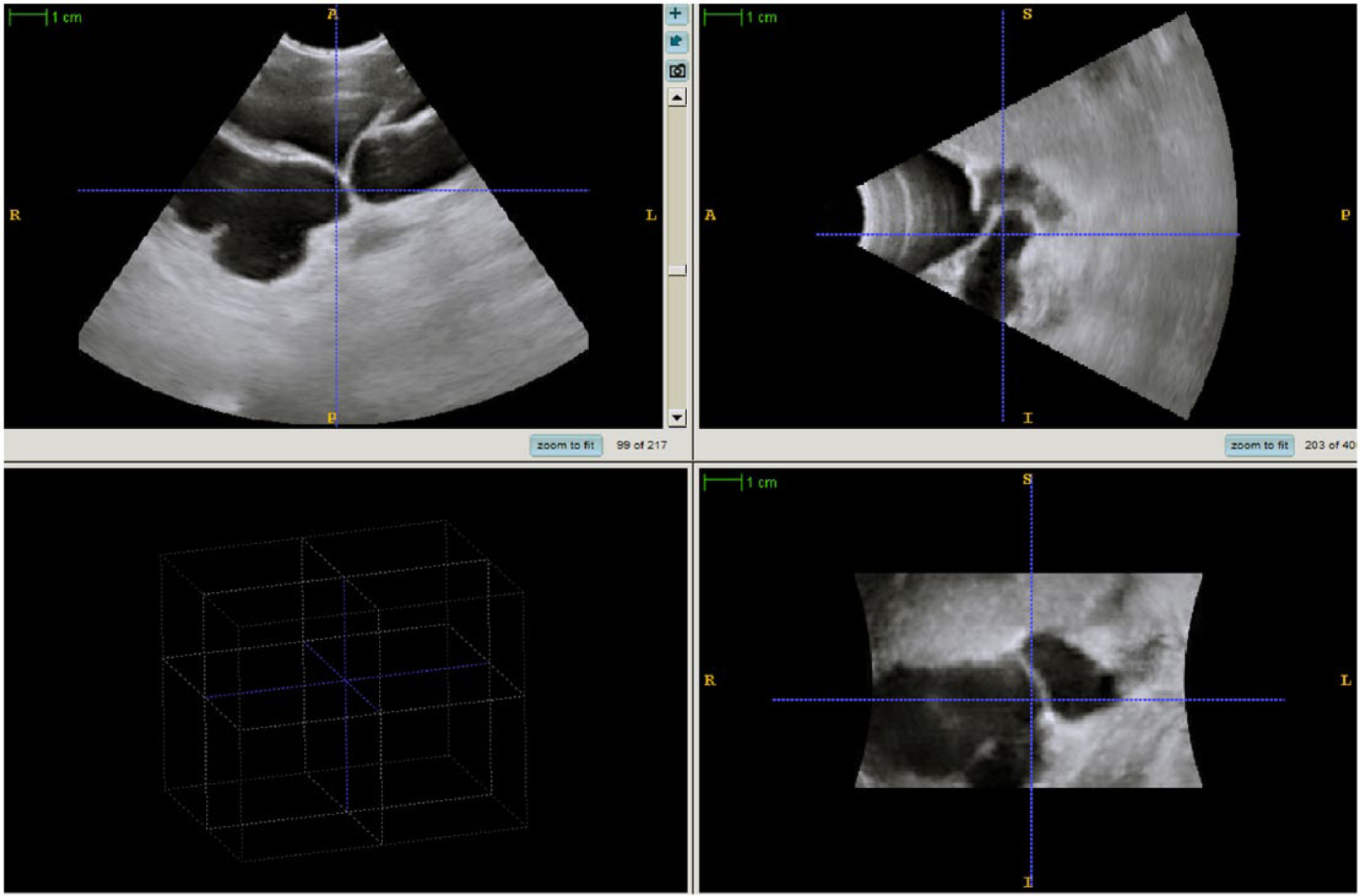
Three-dimensional sonographic image demonstrating significant cut-off of a severely (grade 4) hydronephrotic left kidney in a 6-month-old male patient.
Descriptive analysis of patients’ demographics and hydronephrosis grading (using the Society of Fetal Urology [SFU] system 13 ) as well as the average quality scores from the readers was performed. The interreader reliability was determined using Light’s kappa statistics. 14 Agreement was characterized as slight (k = 0-0.20), fair (k = 0.21-0.40), moderate (k = 0.41-0.60), substantial (k = 0.61-0.80), and almost perfect (k = 0.81-1.00). 15
Then, the relation between image quality and SFU hydronephrosis grading, affected side, and renal length was determined using Fisher exact test. The STATA 14.0 software (StataCorp LP, College Station, TX, USA) was used for data analysis. Statistical significance was established at P ≤ .05, a priori.
Results
Twenty-four patients (19 boys and 7 girls; ages 2 weeks to 3 years, mean 12 ± 10 months) with congenital hydronephrosis between May and November 2015 were enrolled and 32 3D DMS volume data sets of the affected kidneys (15 right and 17 left) were acquired. One patient had a solitary kidney; one kidney was shown to be a multi-cystic dysplastic kidney and was therefore excluded. The remaining kidneys were excluded because they did not demonstrate hydronephrosis (n = 14). Kidney lengths ranged from 5.1 to 10.6 cm (mean 7.3 ± 1.31 cm). The hydronephrosis severity distribution using the SFU grading system was as follows: 11 (34.4%) grade 4; 10 (31.3%) grade 3; 3 (9.4%) grade 2; and 8 (25.0%) grade 1. Baseline characteristics are summarized in Table 1.
Baseline Characteristics of the Study Sample.
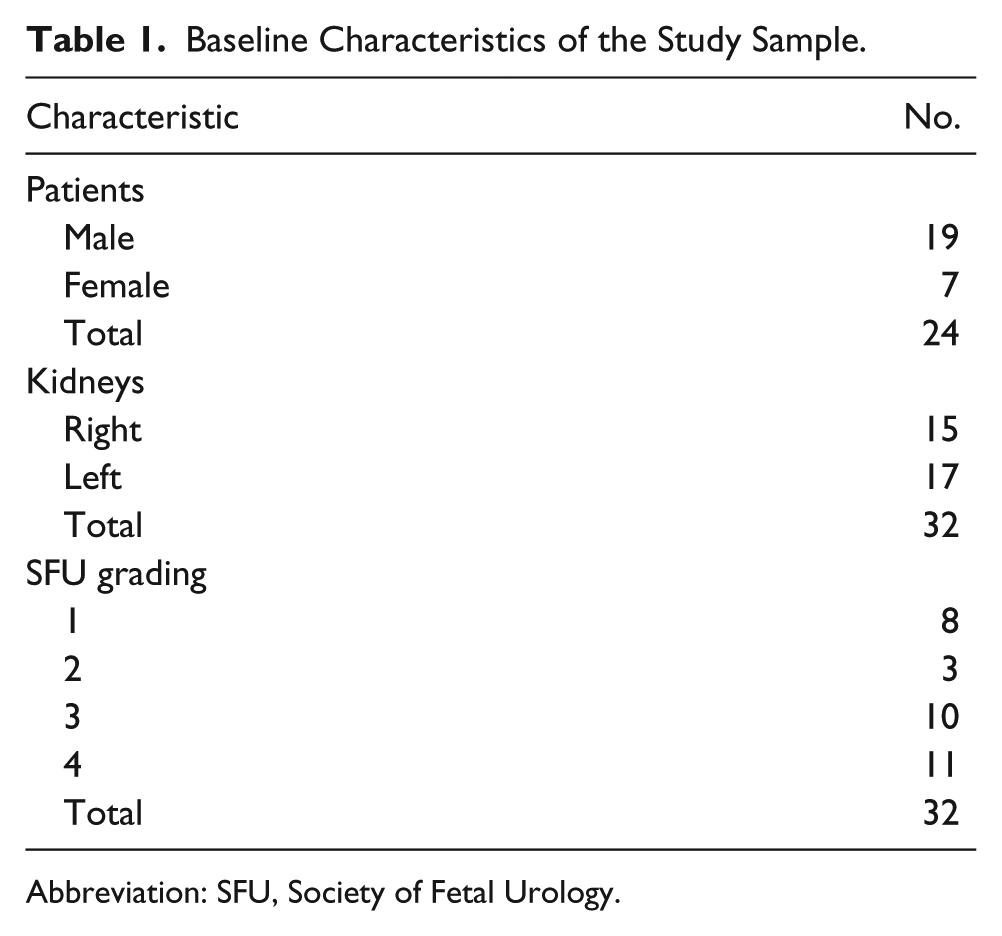
Abbreviation: SFU, Society of Fetal Urology.
The average quality scores of the 3D DMS images were excellent in 4 (12.5%) cases (Figure 1), good in 17 (53.1%) cases, and fair in 11 (34.4%) cases (Figure 2). None of the cases were deemed nondiagnostic. Rib shadowing was present in 24 (75.0%) cases, minimal in 12 (37.5%), moderate in 10 (31.3%), and severe in 2 (6.3%) cases. Cut-off edges were seen in 25 (75.0%) cases, considered minimal in 15 (46.9%), and were moderate in 10 (34.4%) cases. Motion artifact was present in 16 (50%) cases, evaluated to be minimal in 14 (43.8%), and moderate in 2 (6.3%) cases.
The interreader reliability was substantial for rib shadowing, cut-off edges, and motion scores (k = 0.73, k = 0.77, and k = 0.7, respectively). For the overall quality score, the reader reliability was also substantial (k = 0.8). There was a significant correlation between SFU hydronephrosis grade and higher cut-off edges (P = .04). There was no significant relation between the SFU hydronephrosis grade and rib shadowing (P = .35), motion (P = .08), or overall quality score (P = .46). No significant correlation between the kidney side and rib shadowing (P = .29), cut-off edges (P = .36), motion artifact (P = .35), or lower quality scores (P = .24) was found. Kidneys measuring more than 6.5 cm were more likely to show cut off (P = .01), present motion artifact (P = .03), and have lower quality scores (P = .006).
Discussion
In this convenience cohort of infants and toddlers with hydronephrotic kidneys, 3D DMS demonstrated excellent or good quality images obtained in a majority (65.6%) of cases. In our series, none of the cases were of nondiagnostic quality, which is an encouraging finding if these imaging technologies are incorporated into routine clinical care for patients with hydronephrosis. However, artifacts were almost universal, with the majority of cases showing at least one type of artifact. Substantial interreader reliability in all quality scores was also found, which validates our overall scores used for correlations. In the scoring scale adopted for the study, images showing minimal cut-off of the edges of the renal parenchyma could still be deemed of excellent quality because the depiction of the collecting system was excellent. Even though most cases demonstrated only minimal artifact, this could represent a potential challenge for the automation of the renal segmentation and analysis, since most software technologies are sensitive to imaging artifacts.
Artifacts were more common on kidneys measuring more than 6.5 cm. This is intuitive for cutting off the edges of the kidneys; the larger the kidney gets, the harder it is to include its entirety in a single volume. Moreover, it might be harder for the sonographer to anticipate the exact borders of the kidneys during the 3D sweeps in larger kidneys. Changing the settings of the acquisition from a 5-cm focal zone to a larger one might solve this issue. Although this option was not available in the current state, we anticipate that it will become available in the future as the 3D ultrasound hardware and software used continue to improve.
Beyond the artifacts, the study shows that 3D DMS of the hydronephrotic kidney in children with diagnostic-adequate image quality is achievable. This is important because studies have shown that 3D DMS is better suited for semiautomated or automated parenchymal volume calculation and collecting system segmentation and analysis. 16 Parenchymal volume calculations from 3D DMS, which have been reported to have accuracies comparable to those from dimercaptosuccinic acid scintigraphy and magnetic resonance urography, can result in decreased use of computed tomography, magnetic resonance imaging, and nuclear medicine diuretic renograms.12,13,17 Similarly, high-quality 3D DMS depiction and segmentation of the collecting system, which may have the potential to better demonstrate complex anatomy, might result in a decreased need for fluoroscopic evaluation of the collecting system or magnetic resonance urography. 18 In addition, 3D DMS provides a multiaxial demonstration of the entire kidney. 18
Recently, renewed interest has emerged for 3D DMS imaging in infants with hydronephrosis. This may be due in part to providing more accurate and reproducible quantitative imaging analyses that help to identify patients who could safely avoid diuretic mercaptoacetyltriglycine renography and the ionizing radiation dose that is associated with these tests. 9 Images of postprocessing output are presented in Figure 3. Finally, 3D ultrasound may be able to improve the utility of sonography in follow-up by calculating interval renal parenchymal volume growth and changes in split volume for guiding treatment decisions with improved standardization and documentation.8,18
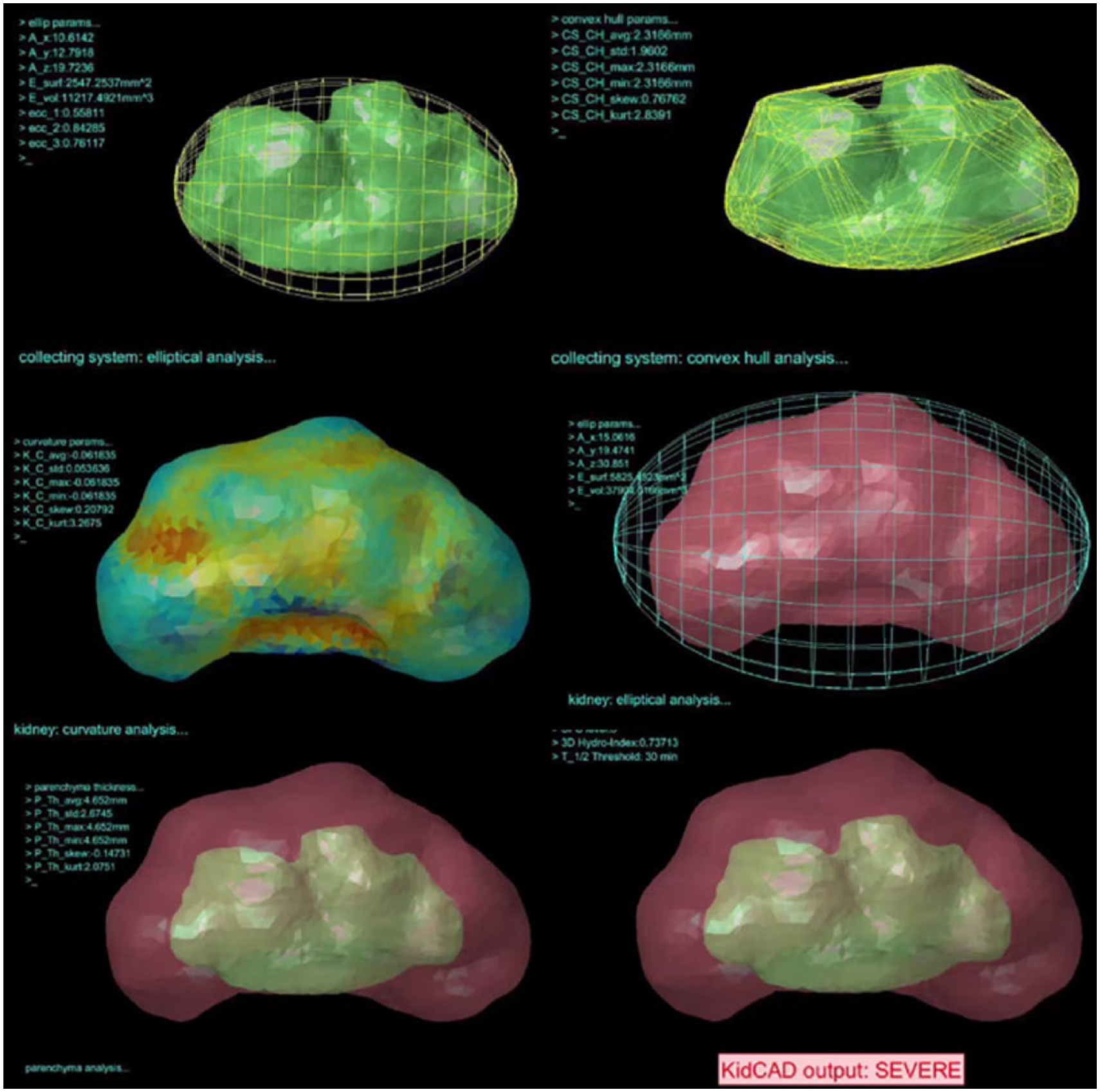
Postprocessing results from three-dimensional sonographic image demonstrating a final determination of severe hydronephrosis requiring additional evaluation with diuretic mercaptoacetyltriglycine renography.
This study has several limitations: (1) This is a convenience sample of a small cohort of clinical patients. Due to the research design, these data cannot be generalized to a larger population. (2) The sample is limited due to the number of kidneys evaluated. However, our study is, to our knowledge, the first to explore the feasibility of 3D DMS in one of the most common findings in pediatric sonography. (3) In addition, time required for obtaining 3D volumes was not quantified. However, because of the highly variable length of sonographic examinations encompassed within the well-documented operator dependency, 19 we do not believe that it would be clinically significant.
Future research from this group will focus on the performance of 3D DMS compared to 2D DMS for the identification of patients who can safely be followed up conservatively and differentiation from those who require functional testing. Nuclear medicine or dynamic magnetic resonance urography would be the functional testing used for comparison in a randomized cohort of patients to avoid selection bias.
In conclusion, 3D DMS of hydronephrotic kidneys in infants and toddlers was feasible in this cohort of patients and demonstrated good image quality in a majority (65.6%) of the cases. Sonographic imaging artifacts are common with this examination and may be related to kidney size.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
