Abstract
A case study is provided of dasatinib-induced pulmonary arterial hypertension (PAH) in a patient with chronic myeloid leukemia. This condition resolved completely within 2 months of drug discontinuation. Transthoracic echocardiography (TTE) data were obtained throughout the recovery process. After 30 months of dasatinib treatment, a woman in her 30s developed orthopnea and signs of right heart failure (leg edema, hepatomegaly, and weight gain). Transthoracic echocardiography indicated elevated mean pulmonary artery pressure, severely impaired systolic and diastolic right ventricular functions, and dilation of the right ventricle and atrium. Once dasatinib was discontinued, clinical symptoms improved rapidly, and follow-up TTE 2 months later showed normal right heart function. Treatment with an alternative tyrosine kinase inhibitor was initiated and has continued without recurrence of PAH. This case suggests that dasatinib, which inhibits a broad spectrum of tyrosine kinases, could cause reversible PAH; therefore, careful cardiopulmonary evaluation by TTE is necessary.
Pulmonary arterial hypertension (PAH), which is associated with a number of diseases, is characterized by a mean pulmonary artery pressure at rest of ≥25 mmHg. This is caused by a progressive increase in pulmonary vascular resistance and can lead to right ventricular failure. 1 There are many distinct PAH subtypes, including drug- and toxin-induced PAH. 2
Chronic myeloid leukemia (CML) is caused by the genetic abnormality known as the Philadelphia (Ph) chromosome, which includes the fusion gene BCR/ABL. This gene encodes an activated tyrosine kinase (TK) that dysregulates cell proliferation, differentiation, and apoptosis. Historically, CML was treated with chemotherapeutic agents such as hydroxyurea, busulfan, and interferon, but in 2001, TK inhibitors (TKIs) were approved as therapy for CML, dramatically improving survival rates because of their specific inhibitory activity against BCR/ABL. 3 Dasatinib is a TKI with markedly higher affinity than imatinib for BCR/ABL kinase. 4 Although dasatinib is generally well tolerated, it has been associated with pleural effusion and PAH in some patients, 5 possibly because it exhibits a broader spectrum of TK inhibitory activity than other clinical TKIs. It has also been reported that specific PAH treatments are less effective for dasatinib-induced PAH.6,7 Clinical and functional improvements have usually been reported after dasatinib discontinuation, although most patients did not exhibit complete hemodynamic recovery within a brief follow-up period. This report presents a case of dasatinib-induced PAH that resolved completely within a short period after drug discontinuation and describes echocardiographic parameters of right heart function during the course of the disease. Echocardiography was a valuable modality for the detection of PAH and the follow-up evaluation of a patient with hematologic malignancy.
Case Report
An Asian female in her late 30s with no past medical history was diagnosed with chronic myeloid leukemia (CML) by leukocytosis with a periodic health examination. Initial treatment using imatinib (Gleevec) did not result in a major molecular response, 8 so the decision was made to begin treatment using nilotinib (Tasigna) due to the patient’s desire to have children. Unfortunately, severe side effects warranted alteration in treatment strategy, and the patient was placed on dasatinib (Sprycel) at the recommended dose (100 mg/d). After five months of dasatinib treatment, a major molecular response was attained, and the treatment regimen continued until approximately 30 months when the patient complained of dyspnea on effort. A chest radiograph (CXR) revealed bilateral pleural effusion, which is considered to be a known adverse effect of dasatinib. In spite of being placed on diuretic therapy (furosemide [Lasix®] 20 mg/d), the patient’s symptoms became more profound over the course of the next 2 months, developing orthopnea, signs of mild hepatomegaly, lower limb edema, weight gain of 7 kg, and a decrease in right lung sounds.
On admission, the patient’s blood pressure was 102/64 mmHg, sinus rhythm with rate of 80 beats/min, and oxygen saturation was 96% in room air. The CXR revealed prominent bilateral pulmonary arteries, pleural effusion, and cardiomegaly (Figure 1b). The electrocardiogram (ECG) exhibited right-axis deviation, T-wave inversion in the right precordial leads, and deep S waves in the lateral precordial leads, suggesting a right heart overload (Figure 1e). Blood tests revealed elevated brain natriuretic peptide (291.3 pg/ml), glutamic oxaloacetic transaminase (56 IU/l), lactate dehydrogenase (380 IU/l), creatinine (0.95 mg/dl), and white blood cell count (13 480/ml, with a lymphocyte fraction of 67.7%), indicating lymphocytosis. Arterial blood gases were within normal limits. A contrast-enhanced computerized tomography scan of the chest, abdomen, and lower extremities demonstrated moderate bilateral pleural effusion, pericardial effusion, and dilatation of the pulmonary trunk (34 mm), but no signs of pulmonary thromboembolism, parenchymal lung involvement, or deep vein thrombosis were found. Furthermore, no sign of pulmonary thromboembolism was found on lung perfusion scintigraphy. Other specific etiologies of PAH were excluded, including portal hypertension, human immunodeficiency virus infection, congenital heart disease, connective tissue diseases, and left heart diseases.
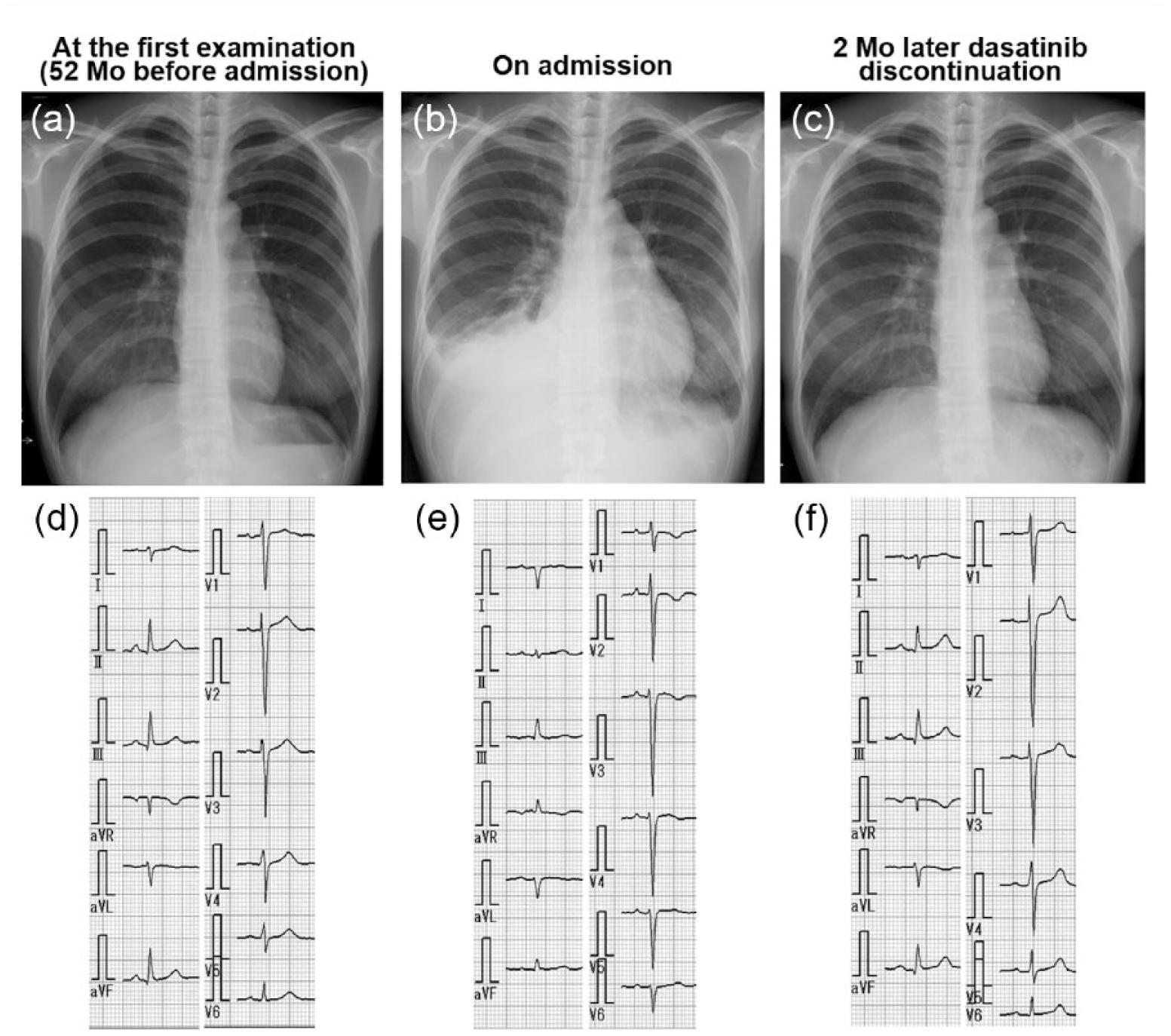
Serial changes in the chest radiograph and electrocardiogram. (a, d) Examination at the first visit 52 months previously. (b, e) On admission. (c, f) Two months after dasatinib discontinuation.
Table 1 shows the results of transthoracic echocardiography (TTE) obtained by a CX50 system with a S5-1 transducer (Philips Medical Systems, Andover, MA). Echocardiography showed a dilated right ventricle and atrium with a significant ventricular septal flattening in both the diastolic and systolic phase, suggesting right heart pressure overload (Figures 2a, 2b, and 2c). Tricuspid annular plane systolic excursion (TAPSE, 15 mm), fractional area change (FAC, 23%), and transtricuspid E/A ratio (0.32) were all below normal parameters, suggesting severely depressed right ventricular systolic and diastolic function. There was mild tricuspid regurgitation (TR) without structural abnormality of the tricuspid valve, and the estimated mean pulmonary artery pressure (PAP) and systolic PAP were 43 mmHg and 72 mmHg, respectively. Systolic PAP was estimated by adding the right atrial pressure (RAP) to the peak TR-pressure gradient. Right atrial pressure was estimated from the diameter of the inferior vena cava and the presence of inspiratory collapse. 9 Mean PAP was estimated using the following equation: mean PAP = 1/3 × systolic PAP + 2/3 × diastolic PAP. 9 Elevation of mean PAP was confirmed with another formula: mean PAP = 4 × (early PR velocity)2 + estimated RAP 9 ; this resulted in mean PAP = 42 mmHg. Left ventricular systolic function was normal, with no regional wall motion abnormality (Table 1). Averaged E/e′ (5.4) and maximal left atrial volume index (24 ml/m2) were normal, whereas septal e′ was only 7.6 cm/s due to a right ventricular contribution to the septal velocity signals. 10
Patient Results Based on Transthoracic Echocardiography, During the Course of the Disease.
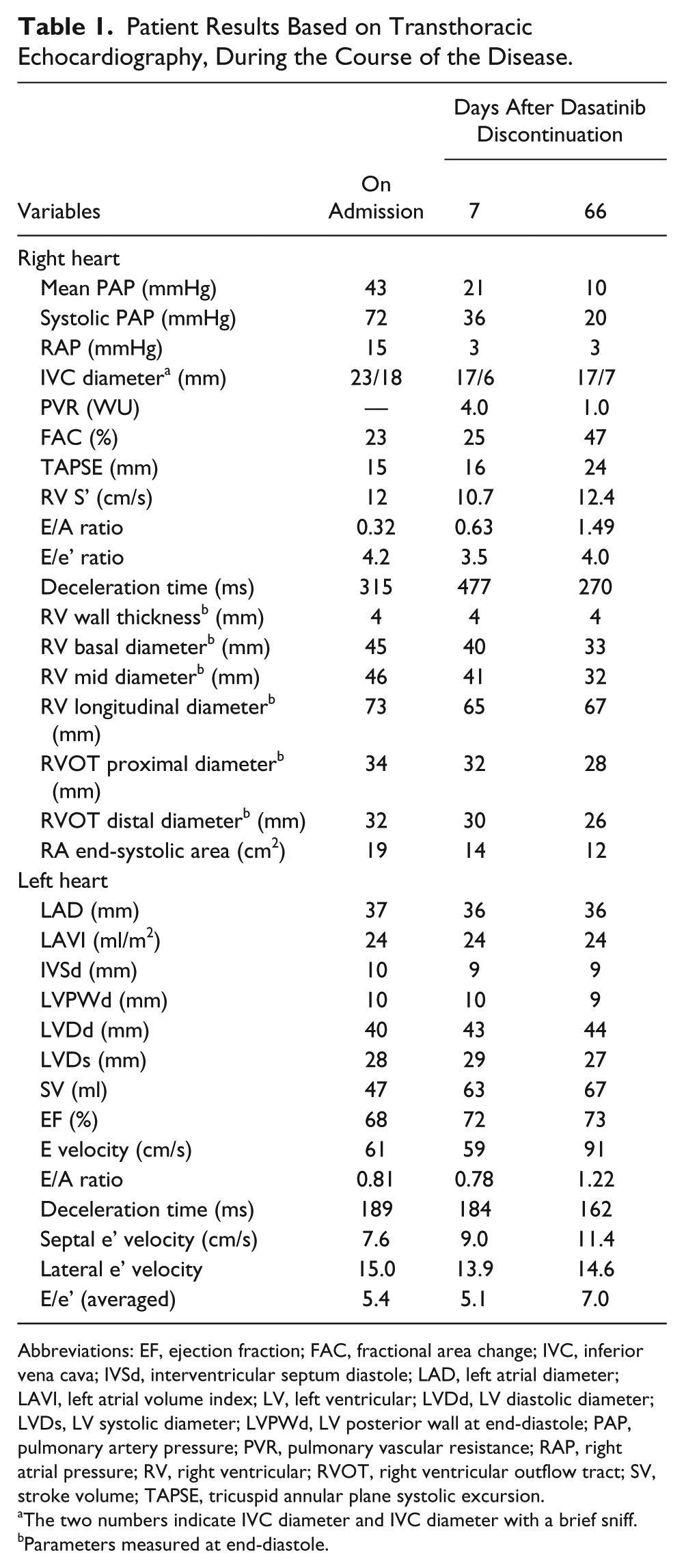
Abbreviations: EF, ejection fraction; FAC, fractional area change; IVC, inferior vena cava; IVSd, interventricular septum diastole; LAD, left atrial diameter; LAVI, left atrial volume index; LV, left ventricular; LVDd, LV diastolic diameter; LVDs, LV systolic diameter; LVPWd, LV posterior wall at end-diastole; PAP, pulmonary artery pressure; PVR, pulmonary vascular resistance; RAP, right atrial pressure; RV, right ventricular; RVOT, right ventricular outflow tract; SV, stroke volume; TAPSE, tricuspid annular plane systolic excursion.
The two numbers indicate IVC diameter and IVC diameter with a brief sniff.
Parameters measured at end-diastole.
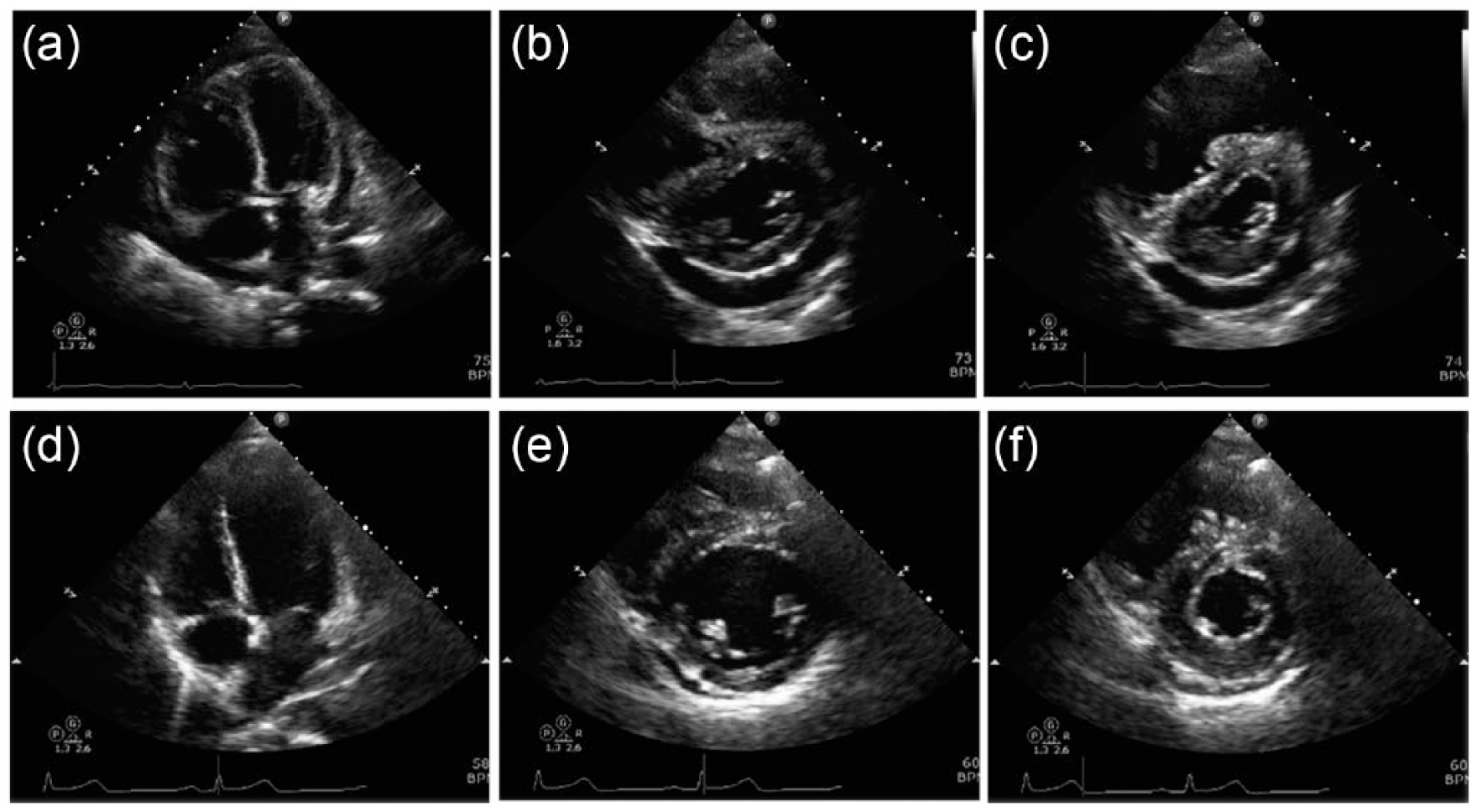
Two-dimensional transthoracic echocardiography images upon admission (upper panels) and two months after dasatinib discontinuation (lower panels). (a, d) Four-chamber views at the end-diastolic phase. (b, e) Short-axis views at the end-diastolic phase. (c, f) Short-axis views at the end-systolic phase.
The mitral E/A ratio (0.81) was also lower than normal values, indicating an impaired relaxation mitral filling pattern because of reduced left ventricular filling rather than diastolic dysfunction. 10 Although there was moderate pericardial effusion, the respiratory variations in transmitral and transtricuspid flow were 13% and 21%, respectively, suggesting no cardiac tamponade. Despite elevated pulmonary artery pressures and presence of pericardial effusion, the patient declined right heart catheterization.
Pulmonary arterial hypertension possibly related to dasatinib therapy was diagnosed, and dasatinib was permanently discontinued; furosemide treatment was increased to 40 mg, and spironolactone treatment (25 mg) was started. Symptoms rapidly improved to World Health Organization functional class I. Follow-up TTE 1 week after dasatinib discontinuation showed decreased mean and systolic PAP (Table 1). Pulmonary vascular resistance (PVR) was 4.0 WU, as estimated by PVR = (mPAPecho − PAWP) / COecho, 11 where mPAPecho was the mean PAP, PAWP the pulmonary artery wedge pressure (estimated as 10 mmHg), 11 and COecho the cardiac output. Two months after discontinuing dasatinib, all echocardiographic parameters were normal (Table 1 and Figure 3), and CXR and ECG data were almost restored to baseline (Figure 1); these results were consistent with our diagnosis of dasatinib-induced PAH. Later, imatinib was started again, and the patient remained stable.
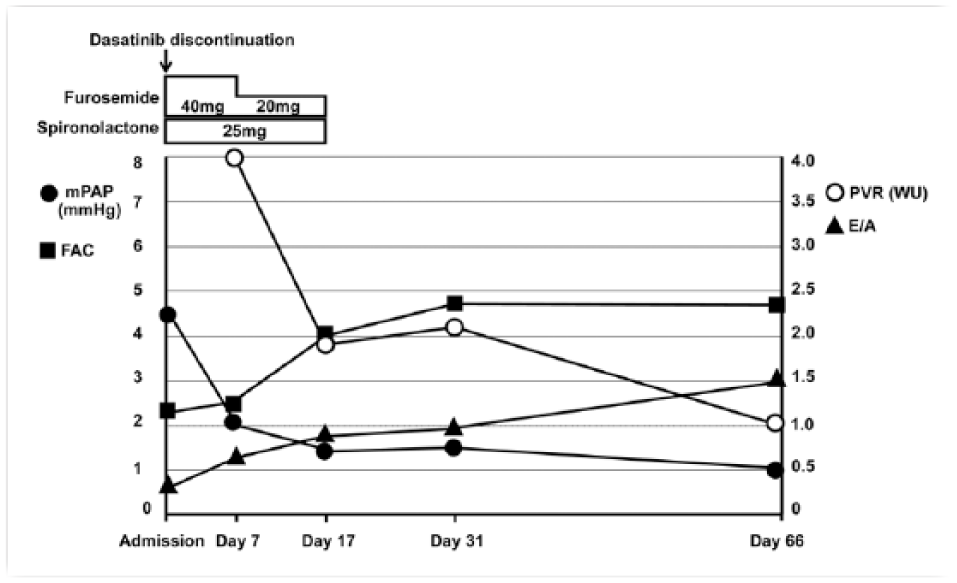
Summary of right heart functional parameters obtained by transthoracic echocardiography and treatment over the two months following the discontinuation of dasatinib.
Discussion
Dasatinib, a therapeutic agent developed for CML and Ph+ acute lymphocytic leukemia (ALL), has recently been reported to induce pulmonary parenchymal changes, pleural effusion, and PAH.5,6,12–15 According to the French pulmonary hypertension registry, the lowest estimated incidence of PAH occurring in patients exposed to dasatinib was 0.45%, whereas no incidental cases of PAH have been reported among patients on imatinib or nilotinib. 6
The mechanism of dasatinib-induced PAH is currently unclear. Although both imatinib and dasatinib inhibit the platelet-derived growth factor (PDGF) receptor pathway, imatinib has been shown to improve PAH, whereas dasatinib can induce PAH.5,6,12–15 Dasatinib exhibits a broader spectrum of TK inhibition than other clinical TKIs, such as imatinib and nilotinib. 16 The main difference in the inhibitory profile between dasatinib and other TKIs is the additional inhibition of Src kinases. 16 These are crucial for smooth muscle cell proliferation and vasoconstriction. Inhibition of Src kinases, which degrade activated PDGF receptors, can increase the signaling of PDGF and other growth factors. 17 Thus, dasatinib-associated PAH may result from Src-kinase inhibition and the resultant overactivation of growth factor signaling pathways in the vasculature.
In a previous case series, the median delay between the initiation of dasatinib and a diagnosis of PAH was 34 months (range, 8–48 months). 6 In line with this, our patient developed PAH 30 months after the initiation of dasatinib. Although the patient presented with significant PAH and moderate pericardial effusion, her vital signs were stable, and there was no indication of cardiac tamponade, suggesting slow progression of PAH. Although it is not certain why PAH arises as a late complication of dasatinib therapy, previous reports have suggested that PAH progression was slower in patients with CML than in those with ALL. One hypothesis is that it may be associated with the combined cytotoxic chemotherapy prior to dasatinib initiation.6,18 Clinical and functional improvements have been observed after discontinuing dasatinib. However, most patients failed to demonstrate complete hemodynamic recovery over subsequent months. Montani et al. 6 reported that the majority of patients did not demonstrate complete clinical and hemodynamic recovery after a median follow-up of 9 months (range, 3–36 months) and that no patients reached normal mean PAP (≤20 mmHg) after dasatinib discontinuation.
Furthermore, a limited response to vasodilator treatment has been reported. 6 Groeneveldt et al. 7 reported that dasatinib-induced PAH was unresponsive to phosphodiesterase type 5 inhibition and was only reversed by discontinuing dasatinib. Unlike these previous cases, this patient exhibited complete resolution of the PAH within 2 months after dasatinib discontinuation, without the use of vasodilators. This rapid resolution is remarkable following long-term treatment with dasatinib (30 months); normalization of pulmonary hemodynamics was not observed after 36 months in a previous study. 6 Although it is unclear why the present case showed such rapid recovery, it is speculated that this patient had no preexisting risk factors for cardiotoxicity from TKIs. Documented cardiovascular adverse events from TKIs include left ventricular dysfunction with resultant heart failure, conduction abnormalities, QT prolongation, coronary artery disease, venous thromboembolism, and hypertension in addition to PAH.19,20 Generally, the incidence and severity of cardiovascular adverse events are related to the underlying cardiovascular risk of the patient. Patients with a history of hypertension, diabetes, prior cardiovascular disease, or prior chest radiation are at higher risk.21–23 Further investigation is needed to reveal the exact mechanisms for dasatinib-associated PAH and the associated risk factors. Indeed, no predictive factors for dasatinib-related toxicity, such as clinical comorbidities and risk alleles, have been identified. 24
In conclusion, dasatinib treatment requires careful attention to cardiopulmonary adverse effects, particularly PAH. Echocardiographic parameters such as right heart dimension, FAC, TAPSE, and transtricuspid E/A ratio, as well as systolic and mean PAP, provide a noninvasive assessment of right heart function in patients with hematologic malignancy. This case study illustrates the importance of routine cardiovascular and pulmonary evaluation by TTE and monitoring of clinical manifestations during dasatinib treatment.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Arinobu Tojo reports research grants from Bristol-Myers Squibb. The other authors declare that there are no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
