Abstract
Hypertrophic cardiomyopathy (HCM) is caused by sarcomere genes and is the most common monogenic cardiovascular disorder. Left ventricular hypertrophy (LVH) is the classic hallmark sign for diagnosis; however, LVH may not manifest until adolescence or later in individuals who are mutation carriers. Genetic testing can identify individuals who carry pathogenic sarcomere mutations and have a clinical diagnosis of HCM—that is, mutation carriers who have not yet manifested LVH but are very likely to develop the disease. Evaluating this new patient subset with state-of-the-art echocardiography has shown evidence of early signs of left ventricular impairment. Tissue Doppler imaging is able to detect mutation carriers who may have altered diastolic function, by identifying individuals with reduced tissue Doppler velocities. This article reviews the findings of using tissue Doppler imaging in HCM and other forms of cardiomyopathy. Understanding this source of HCM presents an opportunity to predict and better manage disease.
Keywords
Understanding the genetic basis of cardiovascular malformations has aided in the development of genetic screening in a number of syndromes. This determination of a genetic source that contributes to cardiac abnormalities has provided an opening for predicting and counteracting inherited cardiac disease.1,2 Hypertrophic cardiomyopathy (HCM) is one example of this process. HCM is an autosomal dominant disorder; it is the most common inherited cardiovascular disorder and is caused by mutations in genes encoding sarcomere proteins.2,3 Echocardiographic features of HCM include left ventricular hypertrophy (LVH) and diastolic dysfunction, and it presents a risk factor for arrhythmias, sudden death, and heart failure. 2 There are other inherited cardiovascular disorders that contribute to LVH and/or reduced diastolic compliance of the left ventricle in the absence of increased loading conditions, such as Becker muscular dystrophy (BMD), Duchenne muscle dystrophy, Fabry cardiomyopathy, and Friedreich ataxia (FRDA). Incorporating genetic testing into the clinical assessment allows early detection of the disease process for those at risk for developing HCM, and while LVH is a hallmark sign of HCM, the development of LVH is often manifested in the third decade of life. 2
This type of genetic screening has allowed a new and important subset of patients considered to have preclinical HCM. The utilization of echocardiography, particularly modalities such as tissue Doppler imaging (TDI), can detect subclinical cardiac manifestations of this genetic disorder and identify individuals who will potentially develop clinical signs of HCM. The recognition of individuals at risk prior to the onset of clinical diagnosis could help document treatment responses in future clinical trials designed to weaken and eventually prevent the development of HCM.2,4 This article describes the current role of TDI in identifying preclinical parameters of cardiac diastolic dysfunction in the absence or presence of LVH in congenital hypertrophic myopathies.
Preclinical Assessment of HCM
Currently, TDI is being used as a measure to identify those individuals with positive markers for a number of inherited cardiomyopathies by documenting structural abnormalities in the myocardium. 4 TDI is an echocardiographic technique that assesses myocardial velocities. By utilizing the apical 4-chamber view, the TDI waveform is obtained, which displays direction and velocity of the annulus as it moves throughout the cardiac cycle. Most important, TDI is used to evaluate global and regional systolic left ventricular (LV) function and abnormal LV relaxation in a variety of conditions.1,4
Genotype refers to information stored in the genetic code of a person, whereas the phenotype represents the recognizable traits, physical and biochemical, associated with the genotype. 5 More than 140 disease-causing mutations have been identified in genes encoding sarcomere proteins, most commonly β myosin heavy chain (~35%), cardiac myosin binding protein C (30%), cardiac troponin T (~15%), and cardiac troponin I. 3 HCM is an autosomal dominant disorder categorized by unexplained LVH, abnormalities of diastolic function, and increased risk for sudden death. It is hypothesized that development of LVH in cases of HCM is a late expression of the disease and that sarcomere mutations cause abnormalities of myocardial biochemistry and function well before obvious cardiac hypertrophy progresses. 6 The echocardiographic findings of LVH, not accounted for by other systemic or cardiac disease, generally serve as the basis for the diagnosis of HCM. The expression of the HCM phenotype typically does not occur until adolescence or thereafter, making the clinical diagnosis of HCM early in life particularly challenging. 3
The histopathologic features of HCM are myocyte hypertrophy, disarray, varying types of fibrosis, and small vessel disease. Myocyte disarray is described as areas of structural disorder of hypertrophied myocytes and nuclear changes. Patients with fibrosis commonly exhibit both macroscopic and microscopic scars due to small vessel disease. The septum appears gray or displays areas of white patchy fibrosis to large white transmural scars. 7 Figure 1 shows an example of severe concentric hypertrophy in the gross pathologic specimen from a patient with HCM as compared with a normal heart. Patients with HCM (Figure 1a) present with markedly increased wall thickness, prominent in the interventricular septum but also present in the free lateral wall. In contrast, normal myocardium (Figure 1b) demonstrates a very orderly arrangement of myocytes.
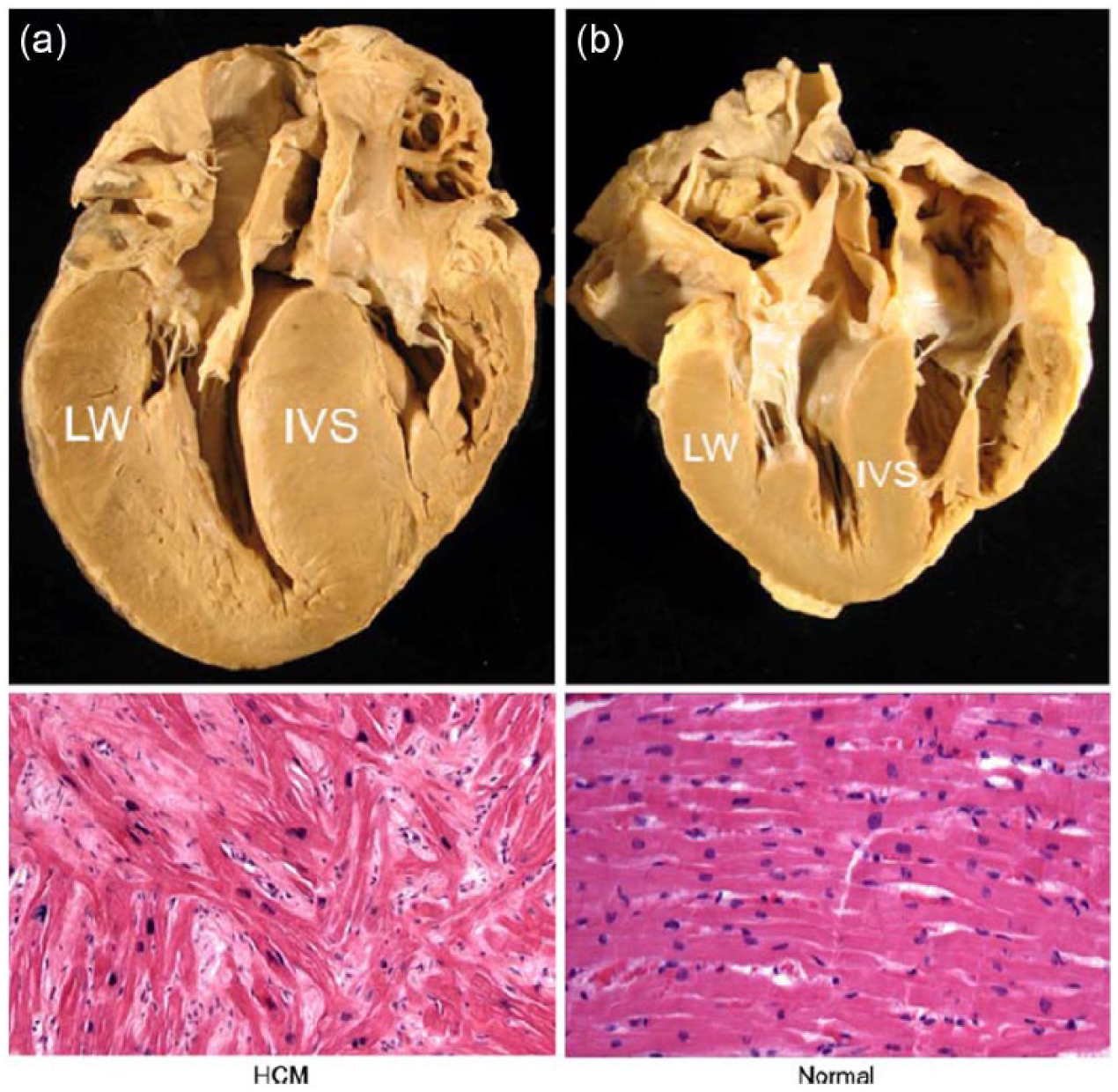
The gross and corresponding microscopic findings of hypertrophic cardiomyopathy (HCM, a) compared to a normal heart (b). With HCM (a) the wall thickness is significantly increased, particularly in the interventricular septum (IVS) but also present in the free lateral wall (LW). Histologic sections stained with hematoxylin and eosin demonstrate myocyte disarray and fibrosis, the pathognomonic features of HCM. In contrast, normal myocardium (b) does not show the marked wall thickening and has a very orderly arrangement of myocytes. (Images are at 100× magnification. Reprinted with permission from: Ho C: Hypertrophic cardiomyopathy: preclinical and early phenotype. J Cardiovasc Transl Res 2013;6(4):662–670.)
Normal Cardiac Morphology Assessment of Preclinical HCM by Noninvasive Imaging
The America Society of Echocardiography and the European Association of Echocardiography have proposed the following cutoff points for a diagnosis of LVH: LV septal wall thickness >0.9 cm for women and >1.0 cm for men. Table 1 lists the America Society of Echocardiography / European Association of Echocardiography guidelines for grading the severity of LVH based on LV septal thickness measurements when values exceed the reference limit. 8 HCM presents very typical echocardiographic features if LVH exists. However, without overt LVH, no 2D or conventional Doppler echocardiographic parameters have been able to differentiate preclinical individuals from their healthy genotype-negative relatives. 2
Classification of the Severity of LVH Based on LV Septal Thickness Measurements. a

Abbreviations: LV, left ventricular; LVH, left ventricular hypertrophy.
Modified from Barbieri et al. 8
Figure 2 shows the inability of standard echocardiographic imaging to identify preclinical sarcomere mutation carriers. The parasternal long axis and apical four-chamber images from a 24-year-old woman with overt HCM in the top panels show marked asymmetric septal hypertrophy and severe systolic anterior motion of the mitral valve, causing LV outflow tract obstruction. Similar images in the bottom panels from her 23-year-old cousin show a normal standard echocardiographic examination without LV hypertrophy or other diagnostic features of HCM. Genetic testing identified a myosin heavy chain (MYH7) gene mutation in both patients, which was the etiology of HCM in the 24-year-old and so demonstrates the limitations of standard 2D echocardiography in identifying preclinical gene mutation carriers. 2
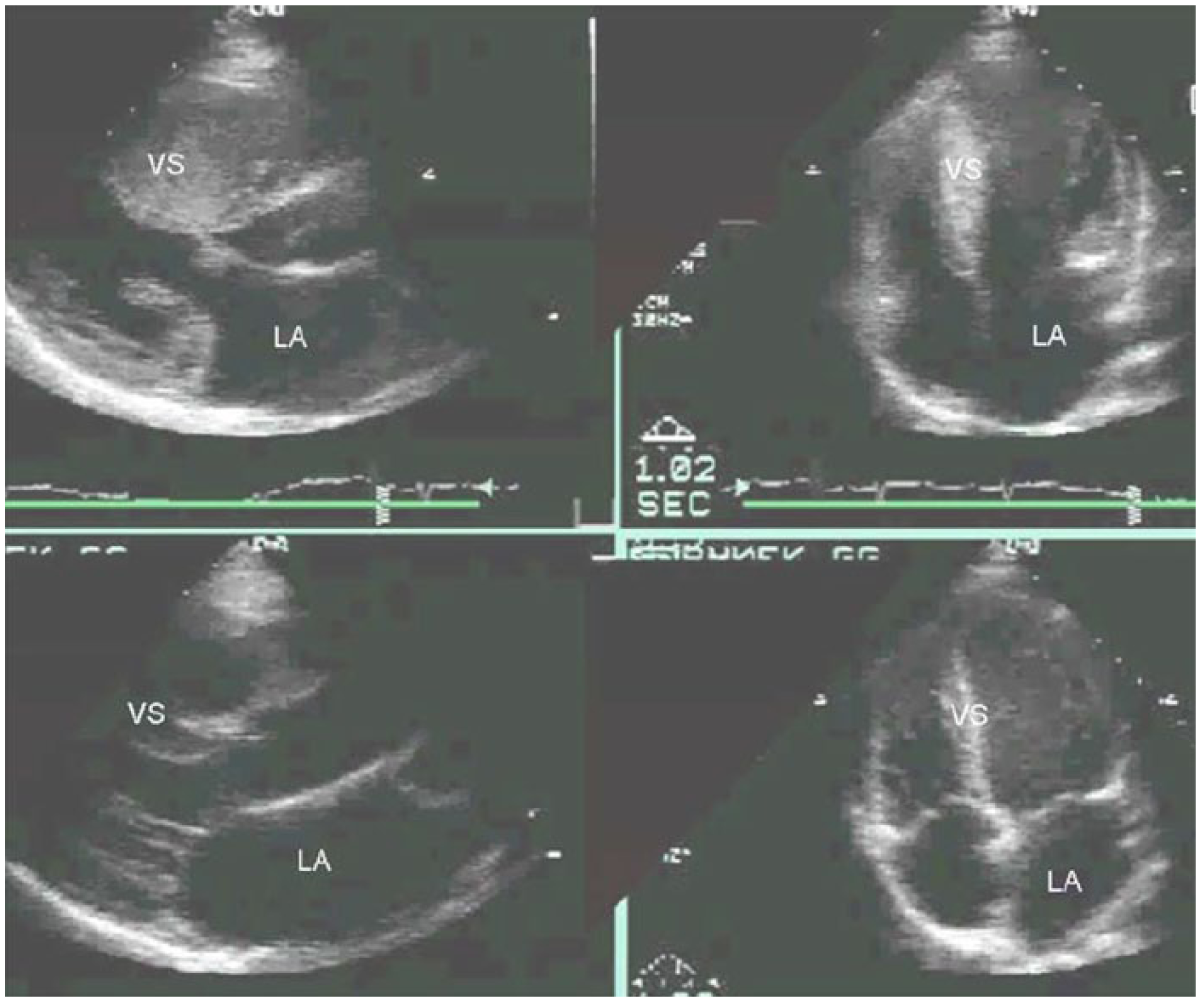
The top panels show parasternal long axis and apical four-chamber images from a 24-year-old woman with overt HCM. There is marked asymmetric ventricular septal (VS) hypertrophy and severe systolic anterior motion of the mitral valve, causing left ventricular (LV) outflow tract obstruction. Genetic testing identified a myosin heavy chain (MYH7) gene mutation as the etiology of HCM in her family. The bottom panels show parasternal long axis and apical four-chamber images from her 23-year-old cousin who had a normal standard echocardiographic examination without LV hypertrophy or other diagnostic features of HCM. Genetic testing showed that she also carries the MYH7 mutation, demonstrating the limitations of 2-dimensional echocardiography in identifying preclinical gene mutation carriers. (VS: ventricular septum; LA: left atrium) (Reprinted with permission from: Ho C: Hypertrophic cardiomyopathy: preclinical and early phenotype. J Cardiovasc Transl Res 2013;6(4):662–670.)
Assessment of Diastolic Function
Early changes in the LV diastolic function can be detected with the use of more sensitive imaging techniques, and studies show that diastolic abnormalities precede the onset of LVH. 2 Conventional echocardiographic assessment of the LV diastolic function depends on the mitral valve Doppler patterns. Left atrial pressure is reflected by the gradient between the left atrium and the LV and is independently and inversely related to ventricular relaxation. 9 However, because mitral inflow patterns are extremely sensitive to preload as diastolic dysfunction worsens, the use of mitral valve inflow patterns to assess diastolic function remains limited.
TDI evaluation of diastolic function has been shown to be less load dependent than standard Doppler techniques, and TDI of the mitral annular has been reported to be effective in the assessment of LV diastolic filling pressures. 7 Unlike conventional transmitral methods, TDI techniques have the potential for load-independent assessments of diastolic function. When the annular TDI waveform is obtained from the apical 4-chamber view, the direction and velocity of the annulus are shown throughout the cardiac cycle.1,3 The components of a TDI waveform are shown in Figure 3. During systole, the lateral mitral annulus moves toward the apex, resulting in a positive waveform on the spectral Doppler display (S′). During diastole, the annulus is first displaced toward the base by the effects of ventricular relaxation (E′). A second negative annular velocity occurs later in the cardiac cycle secondary to atrial systole (A′). The E/E′ ratio, which is calculated by dividing the E-wave mitral filling velocity by the annular tissue E′ velocity, is a measurement that has been shown to be reliable for the estimation of pulmonary artery wedge pressure, an indirect estimate of left atrial pressure.1,3 These measurements can be operator dependent, and for proper diagnostic assessment, obtaining accurate and reproducible TDI measurements is critical. Sonographers must be mindful of the scanning errors that may affect these Doppler signals. Of significance is the proper placement of the sample volume to allow accurate TDI tracings, as inaccurate placement of the sample volume can alter the TDI tracings and measured velocities 1 (Figure 4).
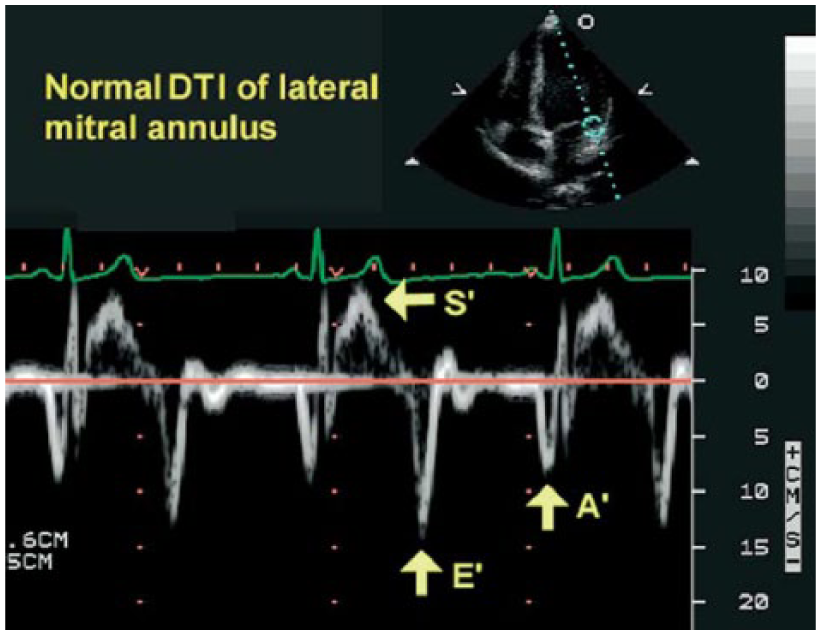
Normal Tissue Doppler Imaging waveform obtained using an apical 4-chamber view from the lateral mitral annulus in a healthy patient during end-expiratory apnea. The spectral Doppler profile is characterized by a single waveform above the baseline (S′) and negative waveforms in early (E′) and late (A′) diastole. (DTI: Doppler Tissue Imaging) (Reprinted with permission from: Hill J, Palma R: Doppler tissue imaging for the assessment of left ventricular diastolic function: a systemic approach for the sonographer. J Amer Soc Echocardiography 2005;18(1):80–89.)
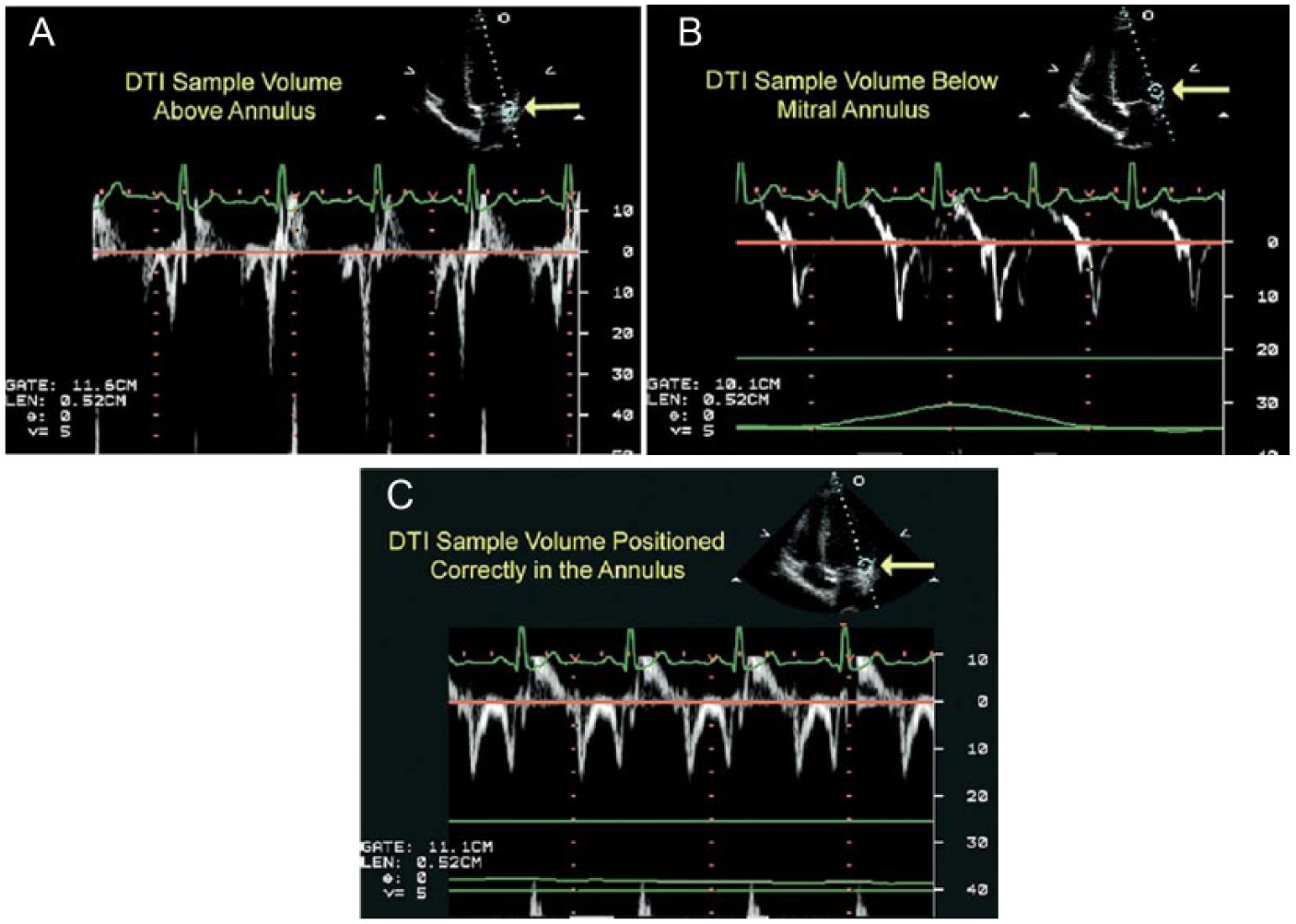
Effects of sample volume position on Tissue Doppler Imaging (labeled DTI in the images) velocities and waveforms. All the Doppler waveforms shown were obtained from same patient. (A) Doppler waveforms obtained with the sample volume located slightly above (on the apical side) the lateral mitral annulus during normal respiration. The resulting waveforms are non-uniform with significant differences in peak E′ and A′ velocities from beat-to-beat. (B) Doppler waveforms obtained with the sample volume placed slightly below (on the ventricular side) the lateral annulus during normal respiration. These Doppler waveforms are also non-uniform, faint, and difficult to measure. (C) Doppler waveforms recorded with proper sample volume placement at lateral mitral annulus during end-expiratory apnea. The waveforms are uniform and consistent, with no beat-to-beat variation of peak diastolic velocities, allowing consistent measurement. (Reprinted with permission from: Hill J, Palma R: Doppler tissue imaging for the assessment of left ventricular diastolic function: a systemic approach for the sonographer. J Amer Soc Echocardiography 2005;18(1):80–89.)
HCM Evaluated With TDI
Abnormal contraction and relaxation of the left ventricle were first shown with pulsed TDI in a transgenic rabbit model of HCM. 10 These results have been confirmed in several human studies, comparing controls with patients with an identified mutation, with or without LVH.3,4,11,12 Ho et al 3 found reduced TDI velocities to be consistently present for a variety of mutations in ß myosin heavy chain, cardiac troponin T, and myosin binding protein C (Table 2). Subjects with the β myosin heavy chain mutation had evidence of abnormal diastolic function by TDI; their early diastolic TDI velocities at each corner of the mitral annulus were lower than those in the control group regardless of the presence or absence of LVH. Of note, the early diastolic velocities at the mitral annulus were significantly reduced by 13% to 19% in the subjects without LVH as compared with the control subjects. Myocardial systolic function did not differ much between the subjects without LVH and the control subjects, but systolic velocities were significantly reduced by 15% to 21% in the subjects with LVH as compared with control subjects.
Myocardial TDI Velocities at the Mitral Annulus. a
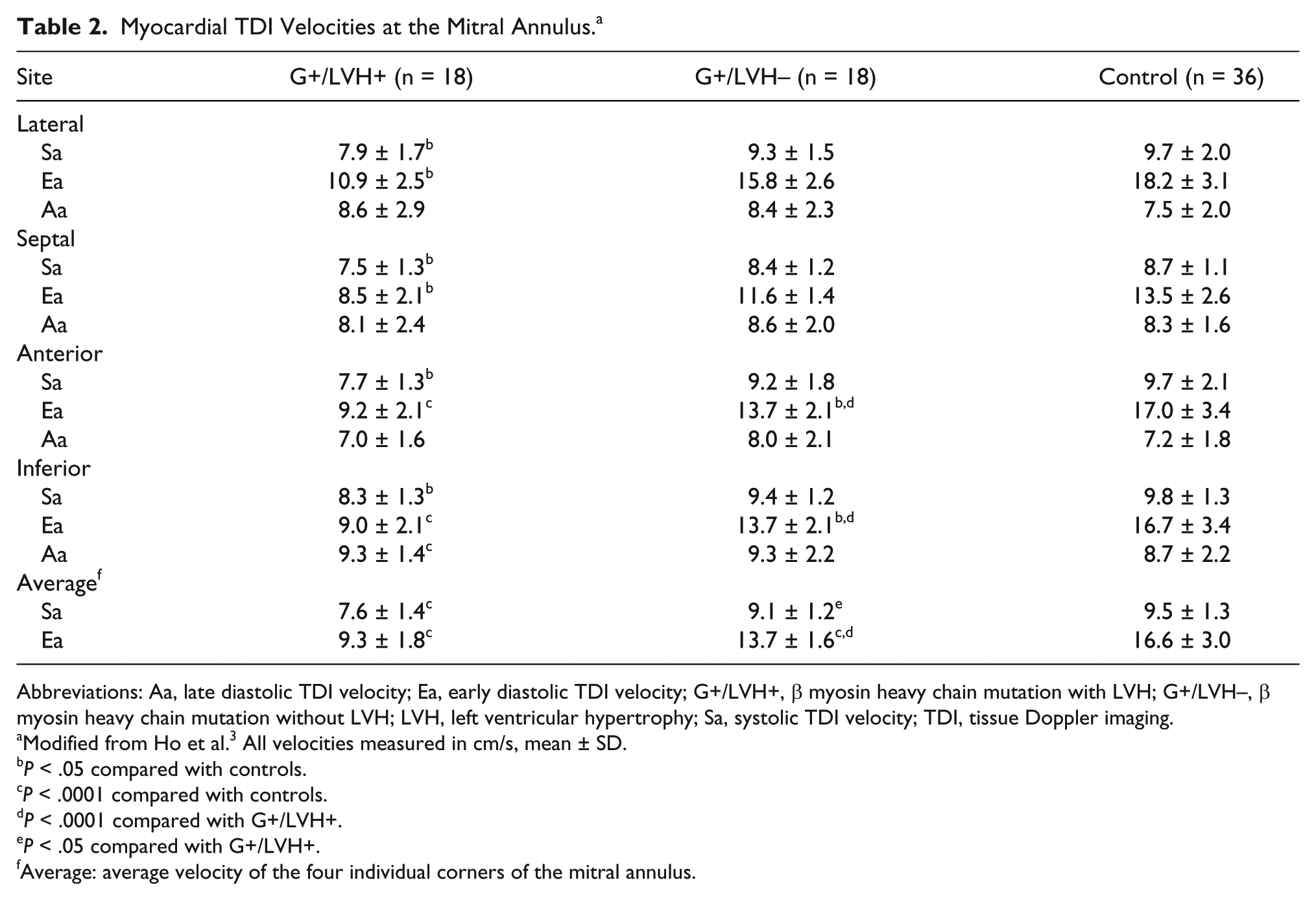
Abbreviations: Aa, late diastolic TDI velocity; Ea, early diastolic TDI velocity; G+/LVH+, β myosin heavy chain mutation with LVH; G+/LVH–, β myosin heavy chain mutation without LVH; LVH, left ventricular hypertrophy; Sa, systolic TDI velocity; TDI, tissue Doppler imaging.
Modified from Ho et al. 3 All velocities measured in cm/s, mean ± SD.
P < .05 compared with controls.
P < .0001 compared with controls.
P < .0001 compared with G+/LVH+.
P < .05 compared with G+/LVH+.
Average: average velocity of the four individual corners of the mitral annulus.
Nagueh et al reported the results of 2D imaging, Doppler echocardiography, and TDI imaging on 30 subjects with familial HCM (FHCM), 13 subjects who were positive for a number of mutations but did not have LVH, and 30 age- and sex-matched controls (all adults; 77% women). 11 Data from the 2D and Doppler echocardiographic studies showed that FHCM patients had an increased septal and posterior wall thickness, LV mass, and left atrial volume; FHCM patients also had a considerably lower E/A ratio, longer isovolumic relaxation time, longer deceleration time, and a larger atrial filling fraction than the other two groups (Table 3). Similarly, systolic filling fraction was increased and atrial reversal–A duration was prolonged in FHCM patients. There were no significant differences in 2D and Doppler indices between the mutation-positive subjects without LVH and controls.
2D and Doppler Echocardiographic Measurements.
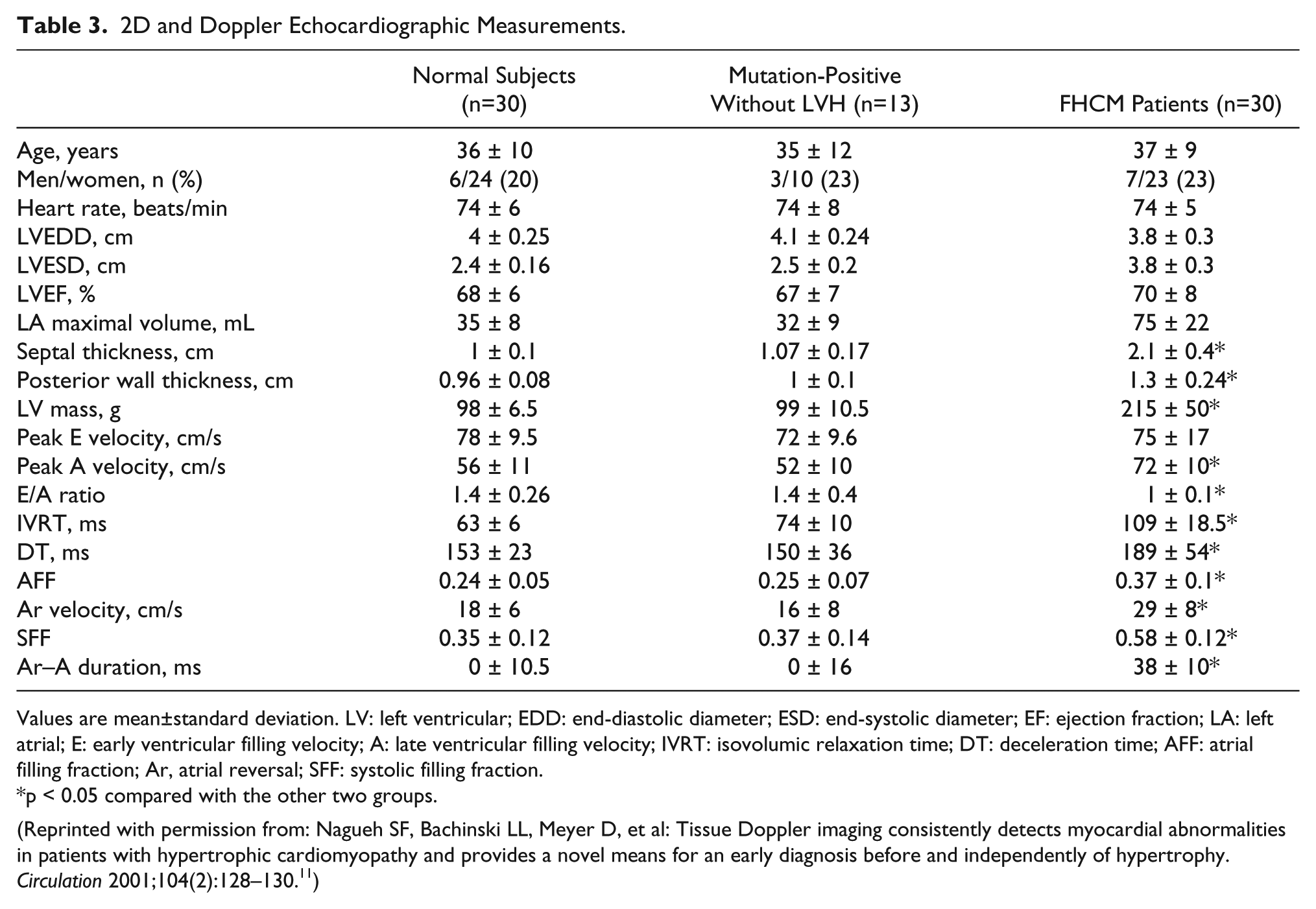
Values are mean±standard deviation. LV: left ventricular; EDD: end-diastolic diameter; ESD: end-systolic diameter; EF: ejection fraction; LA: left atrial; E: early ventricular filling velocity; A: late ventricular filling velocity; IVRT: isovolumic relaxation time; DT: deceleration time; AFF: atrial filling fraction; Ar, atrial reversal; SFF: systolic filling fraction.
p < 0.05 compared with the other two groups.
(Reprinted with permission from: Nagueh SF, Bachinski LL, Meyer D, et al: Tissue Doppler imaging consistently detects myocardial abnormalities in patients with hypertrophic cardiomyopathy and provides a novel means for an early diagnosis before and independently of hypertrophy. Circulation 2001;104(2):128–130. 11 )
With TDI, subjects with FHCM and those who were mutation positive without LVH had reduced systolic and early diastolic velocities at both corners of the mitral annulus as compared with the control group, with TDI velocities diminished the most in the FHCM patients (Table 4). A lateral annular systolic velocity <13 cm/s was shown to have a sensitivity of 100% and a specificity of 93% for differentiating the mutation positives without LVH from the controls. Similarly, a lateral annular early diastolic velocity <14 cm/s had 100% sensitivity and 90% specificity. A septal annular systolic velocity <12 cm/s and early diastolic velocity <13 cm/s both had 100% sensitivity and 90% specificity. Myocardial contraction and relaxation velocities by TDI were uniformly lower in the test subjects, regardless of LVH status, compared with the age- and sex-matched controls. These findings suggested that myocardial dysfunction is an early phenotype that occurs independently of LVH and can be consistently detected by TDI. Figure 5 shows in the upper panel the pulsed TDI recording from a patient with HCM compared with an age-matched control in the lower panel. 4
Pulsed Tissue Doppler Imaging in Hypertrophic cardiomyopathy.
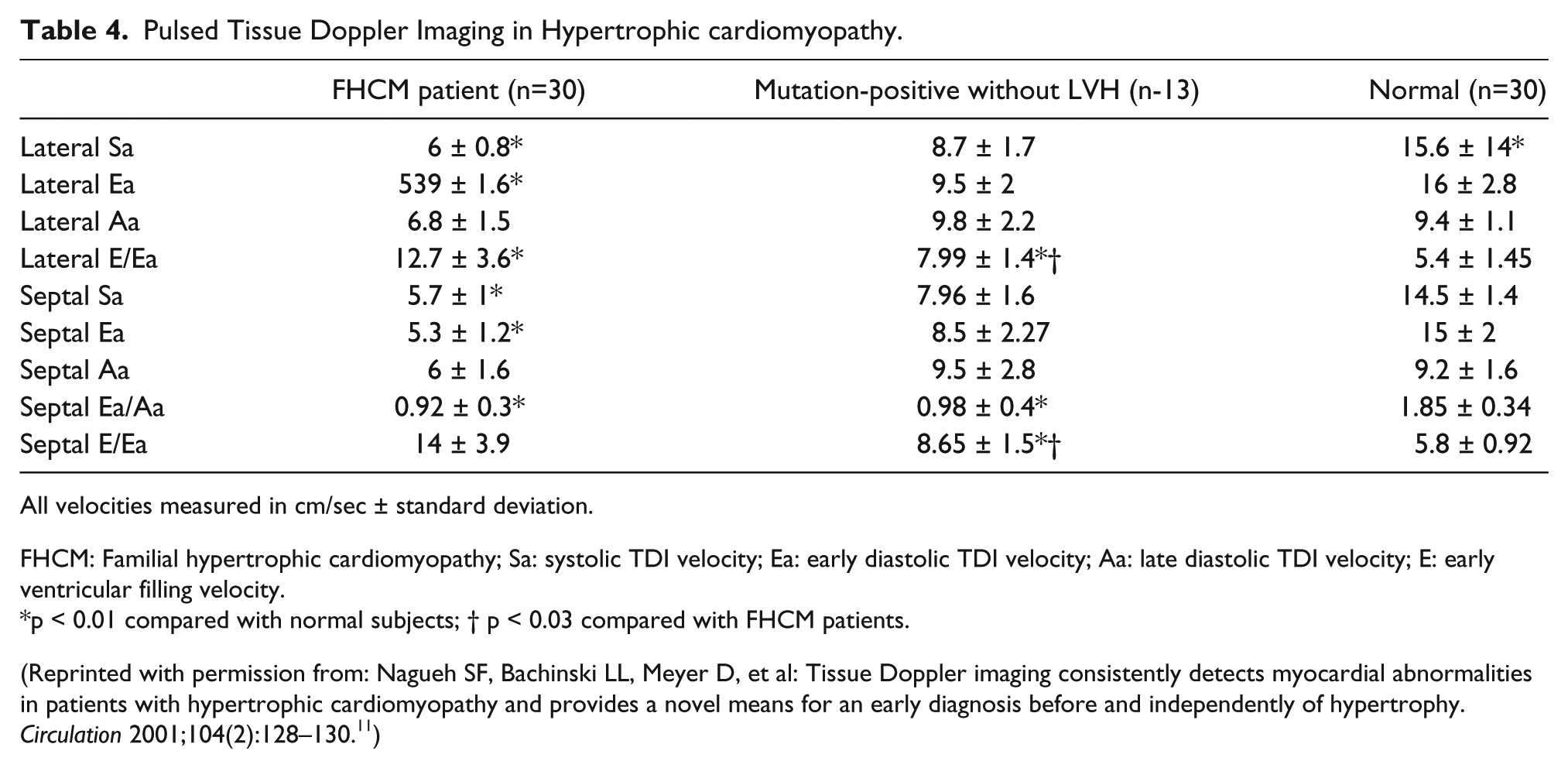
All velocities measured in cm/sec ± standard deviation.
FHCM: Familial hypertrophic cardiomyopathy; Sa: systolic TDI velocity; Ea: early diastolic TDI velocity; Aa: late diastolic TDI velocity; E: early ventricular filling velocity.
p < 0.01 compared with normal subjects; † p < 0.03 compared with FHCM patients.
(Reprinted with permission from: Nagueh SF, Bachinski LL, Meyer D, et al: Tissue Doppler imaging consistently detects myocardial abnormalities in patients with hypertrophic cardiomyopathy and provides a novel means for an early diagnosis before and independently of hypertrophy. Circulation 2001;104(2):128–130. 11 )
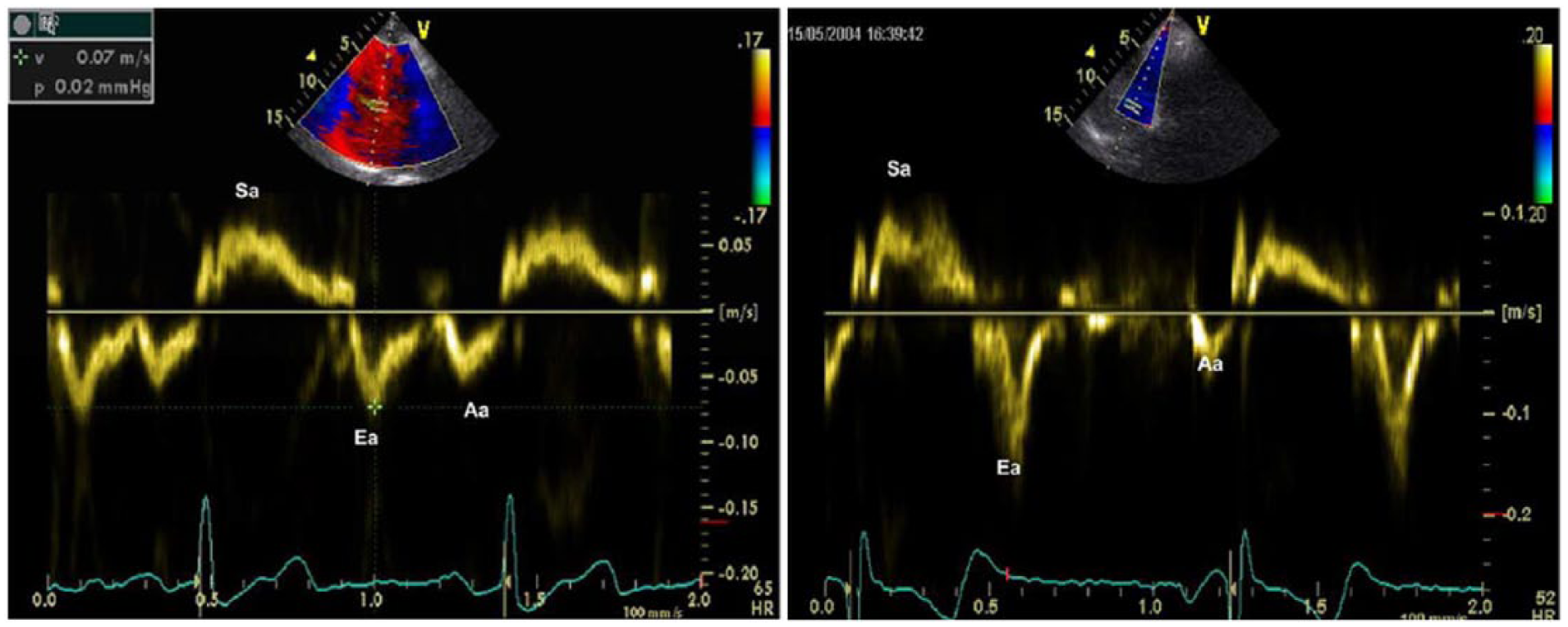
Example of pulsed Tissue Doppler Imaging in a patient with hypertrophic cardiomyopathy (upper panel) and an age matched normal control subject (lower panel). Note the differences in the velocity scale between the patient and the control.
Other Inheritable Disorders Examined With TDI
Fabry Disease
Fabry disease is an X-linked disorder caused by deficiency of lysosomal enzyme α galactosidase A, responsible for the hydrolysis of terminal α galactosyl residues from glycolipids and glycoproteins. The disorder is caused by mutations in the α galactosidase A gene located in the X chromosomal region Xq22. 13 Cardiac involvement is very common and is the most important cause of death in affected patients. 4 Cardiac features include severe LVH that mimics an obstructive or nonobstructive HCM. Preclinical findings of the disease are essential with respect to treatment with an enzyme replacement therapy. When this can be administered in an early stage, early complications can be avoided, such as electrical instability and thromboembolic events. 4
Reports have shown that TDI demonstrates reduced myocardial contraction and relaxation velocities in patients who are positive mutation carriers for HCM, even before the manifestation of LVH.4,13 A recent study evaluated whether patients with cardiac Fabry disease have reduced contraction and relaxation velocities. The study compared 20 patients (10 mutation and LVH positive, 10 mutation positive but LVH negative) with 10 healthy control subjects and showed that all mutation-positive patients had significantly reduced peak systolic, peak early diastolic, and peak late diastolic TDI velocities at both corners of the mitral annulus when compared with control subjects (Table 5). These results suggested that TDI can provide a preclinical diagnosis of Fabry cardiomyopathy, allowing early intervention and management.
Pulsed Tissue Doppler Imaging in Fabry Disease. a
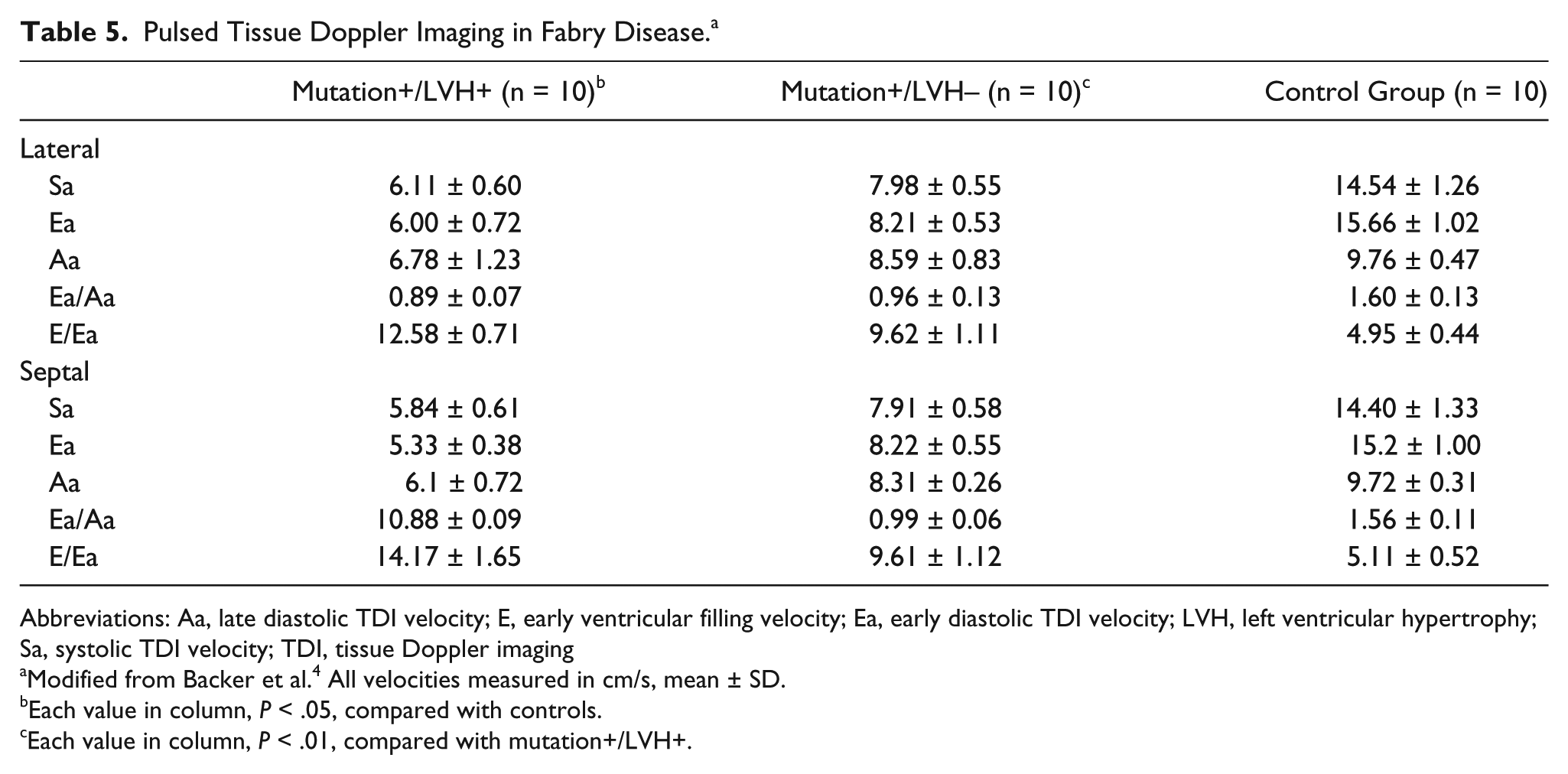
Abbreviations: Aa, late diastolic TDI velocity; E, early ventricular filling velocity; Ea, early diastolic TDI velocity; LVH, left ventricular hypertrophy; Sa, systolic TDI velocity; TDI, tissue Doppler imaging
Modified from Backer et al. 4 All velocities measured in cm/s, mean ± SD.
Each value in column, P < .05, compared with controls.
Each value in column, P < .01, compared with mutation+/LVH+.
Friedreich Ataxia
FRDA is an inherited autosomal neurodegenerative disorder associated with cardiomyopathy and impaired glucose tolerance. The genetic basis for FRDA is an acid α glucosidase lysosomal enzyme instability resulting in loss of a mitochondrial protein, frataxin. The most common echocardiographic abnormality is asymmetrical LV hypertrophy and thickening of the papillary muscles. Dutka et al showed with TDI that myocardial velocity gradients in systole and during the rapid ventricular filling phase of early diastole are reduced in patients with FRDA who are without cardiac symptoms 14 (Table 6).
Tissue Doppler Imaging in Friedreich Ataxia. a
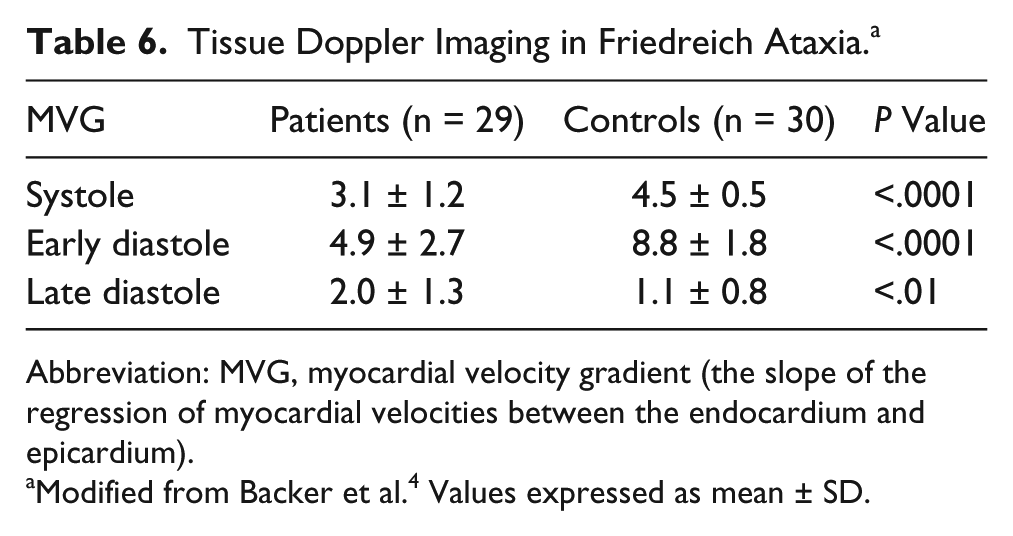
Abbreviation: MVG, myocardial velocity gradient (the slope of the regression of myocardial velocities between the endocardium and epicardium).
Modified from Backer et al. 4 Values expressed as mean ± SD.
Becker Muscular Dystrophy
BMD is an X-linked mutation of the dystrophin gene characterized by skeletal muscle dystrophy and progressive heart failure, which frequently leads to a fatal outcome. 7 Cardiac involvements are not accurately predicted by gene mutation and may occur in patients without muscle weakness; therefore, systematic cardiac examinations are required. TDI during echocardiography allows accurate quantification of regional myocardial function and may be a reliable method for early detection of myocardial dysfunction in BMD. Agretto et al have shown that most of the regional pulsed TDI diastolic indices were decreased in BMD patients and carriers when compared with control subjects4,7 (Table 7). Not only does TDI confirm systolic hemodynamic alterations in BMD patients with reduced LV ejection fraction, but pulsed TDI also detects early segmental systolic abnormalities in BMD patients with normal LV ejection fraction and in female carriers. These results support the theory that TDI may represent an independent marker of early systolic dysfunction versus standard echocardiography.
Pulsed TDI in Becker Muscular Dystrophy. a
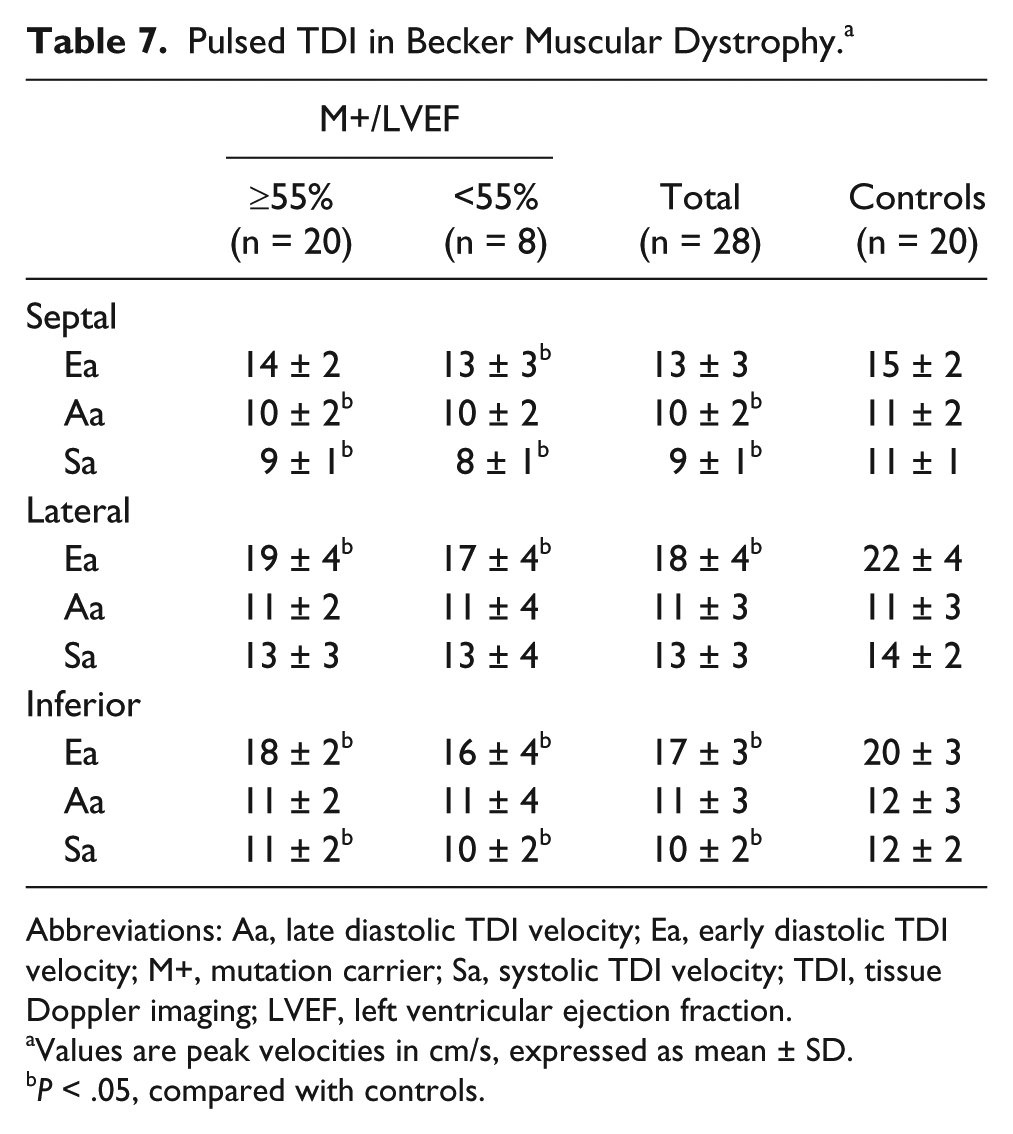
Abbreviations: Aa, late diastolic TDI velocity; Ea, early diastolic TDI velocity; M+, mutation carrier; Sa, systolic TDI velocity; TDI, tissue Doppler imaging; LVEF, left ventricular ejection fraction.
Values are peak velocities in cm/s, expressed as mean ± SD.
P < .05, compared with controls.
Conclusion
Table 8 shows an overview of TDI findings in patients with a variety of genetically linked, inherited cardiomyopathies. It is evident from these studies that TDI can be used for the early detection of functional abnormalities in individuals who have positive genetic testing for cardiomyopathies. The diastolic dysfunction found in preclinical disease appears to be an early consequence of sarcomere mutations, not significant enough to have caused LVH. Tissue Doppler echocardiography may be helpful in increasing our future understanding of the changes in myocardial structure and function in these inherited cardiomyopathies. Early detection of dysfunction in mutation carriers could provide the opportunity for early intervention, including risk stratification and pharmacologic therapies to prevent evolving cardiac phenotypes. 15
Overview of the Different Inherited Cardiomyopathies Studied With TDI.
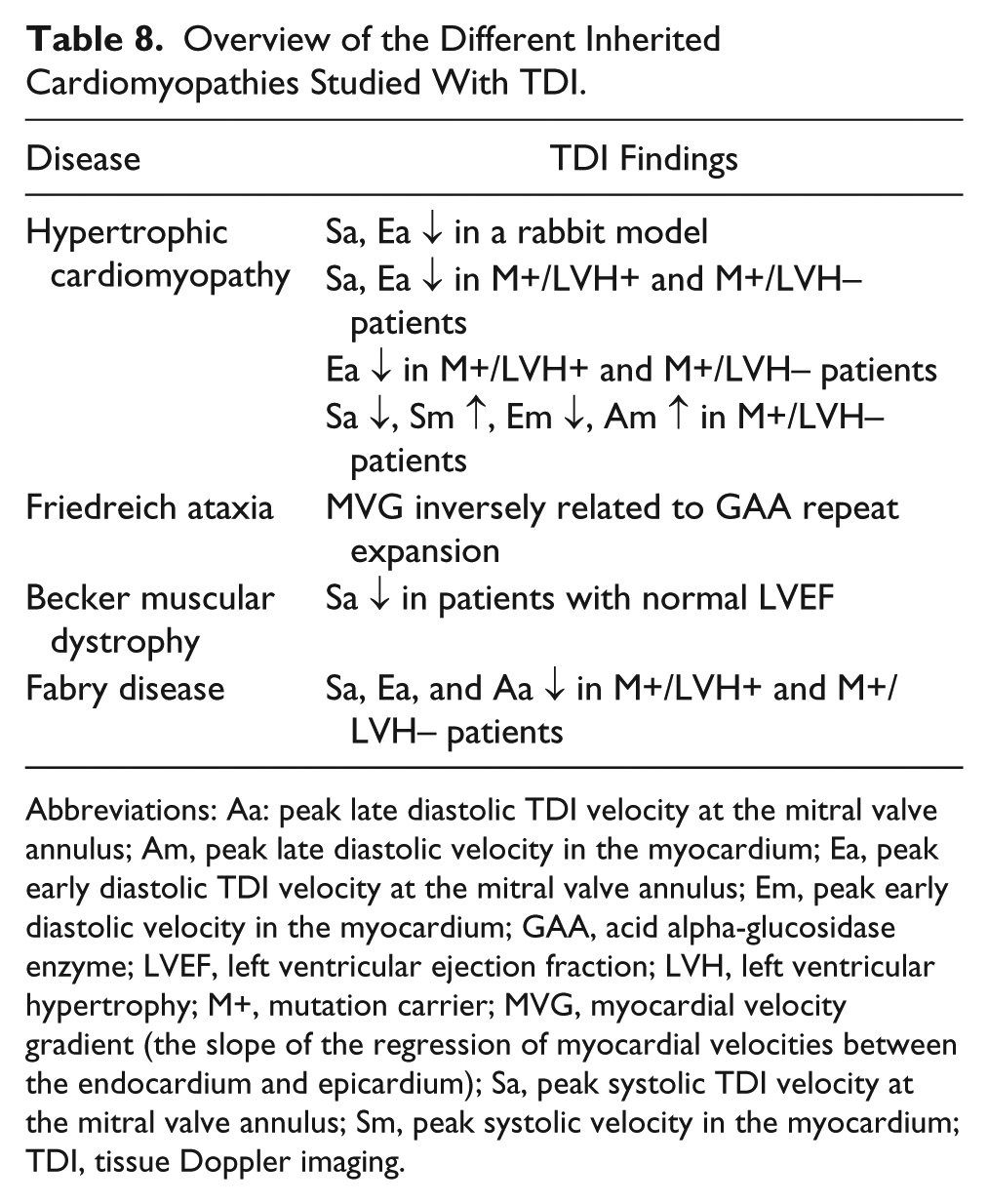
Abbreviations: Aa: peak late diastolic TDI velocity at the mitral valve annulus; Am, peak late diastolic velocity in the myocardium; Ea, peak early diastolic TDI velocity at the mitral valve annulus; Em, peak early diastolic velocity in the myocardium; GAA, acid alpha-glucosidase enzyme; LVEF, left ventricular ejection fraction; LVH, left ventricular hypertrophy; M+, mutation carrier; MVG, myocardial velocity gradient (the slope of the regression of myocardial velocities between the endocardium and epicardium); Sa, peak systolic TDI velocity at the mitral valve annulus; Sm, peak systolic velocity in the myocardium; TDI, tissue Doppler imaging.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
