Abstract
Budd-Chiari syndrome (BCS) with an intracaval stent is a rare presentation. In most Western patients with BCS, it is predominantly the hepatic veins that are involved, not the inferior vena cava (IVC). In the case presented, the patient had antiphospholipid syndrome with BCS that involved the IVC and required an intracaval stent. Presented with the case study are scanning techniques for BCS and the IVC to help sonographers evaluate the normal and abnormal anatomy and hemodynamics of this system.
Keywords
Introduction
Budd-Chiari syndrome (BCS) is an uncommon disorder. It is estimated to affect about 1 in 100 000 individuals.1 -3 In the Western world, it is more common in females and usually presents in the third or fourth decade of life. 4 The outcome is poor in many cases, and the condition is often misdiagnosed or underdiagnosed. Therefore, a successful diagnostic and therapeutic approach is of vital importance. With routine use of imaging, particularly sonography, BCS has been diagnosed more often and in milder forms.5,6 Sonography is an effective modality for diagnosis and gives accurate anatomic and physiologic information to determine the patency and direction of flow in the hepatic veins, inferior vena cava (IVC), and portal venous system. Duplex ultrasonography of the liver has been shown to have a sensitivity and specificity of approximately 85% to 90% for the diagnosis of BCS.7-9 Sonography is extremely helpful in the documentation of complex intrahepatic venous anastomoses and is an excellent noninvasive test to assess stent patency. This gives the patient the opportunity to begin treatment options earlier, which is key to long-term survival. This syndrome may be managed with anticoagulants, transluminal angioplasty/stenting, portosystemic shunting, and orthotopic liver transplantation. 10
Case Report
A female in her fourth decade presented with a history significant for antiphospholipid syndrome and BCS treated with an intracaval stent. Her laboratory studies showed mildly elevated total bilirubin and aspartate aminotransferase and mildly decreased serum albumin. She was on anticoagulation therapy for life, taking enoxaparin and warfarin daily. She came in for a follow-up examination status post IVC stent placement 13 months prior. Documented information prior to the patient’s IVC stent placement was not available.
The abdominal Doppler sonogram was performed using an Acuson Sequoia 512 (Siemens Ultrasound, New York, New York, USA) with a 4-MHz curvilinear transducer. Gray-scale imaging demonstrated lobulated borders of the liver (Figure 1) with an enlarged caudate lobe (Figure 2) and diminished visualization of the hepatic veins. The patient’s liver showed signs of chronic disease due to inadequate perfusion and drainage. The examination also showed the intracaval stent in the IVC (Figure 3). Color and spectral Doppler analyses demonstrated that the IVC exhibited diminished and turbulent flow, which was predominantly toward the right atrium (Figure 4). The portal vein was noted to have hepatofugal flow (Figure 5). Flow in the right hepatic vein was irregular and bidirectional (Figure 6). The left hepatic vein flow was dampened and showed an artifact secondary to near 90-degree insonation, making it difficult to determine the direction of flow 11 (Figure 7). These findings were felt to be indicative of functional failure of the intracaval stent, preventing the liver from draining properly and causing portal hypertension. At that time, close follow-up without any intervention was recommended.
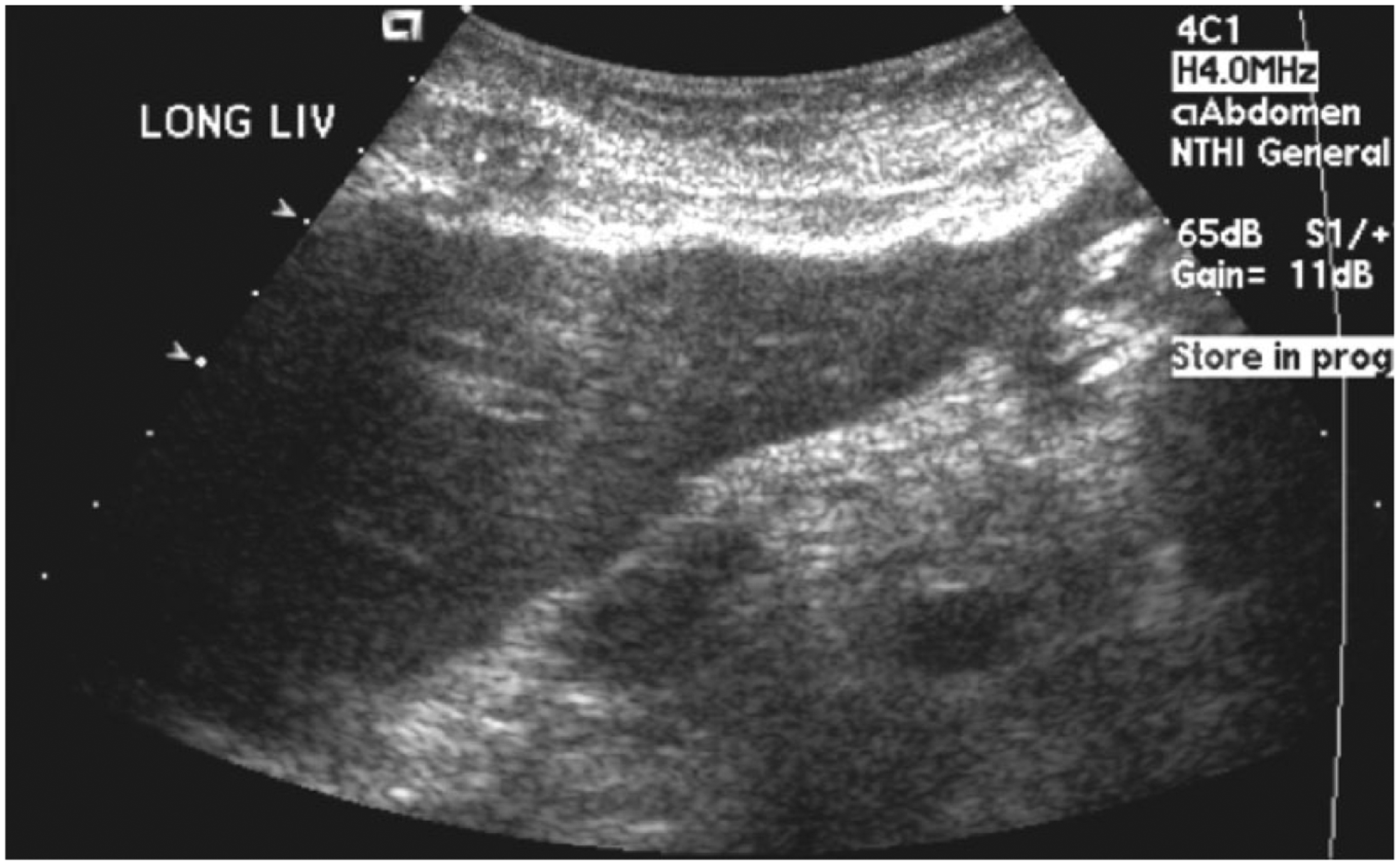
Longitudinal gray-scale image of the liver showing the lobulated borders.
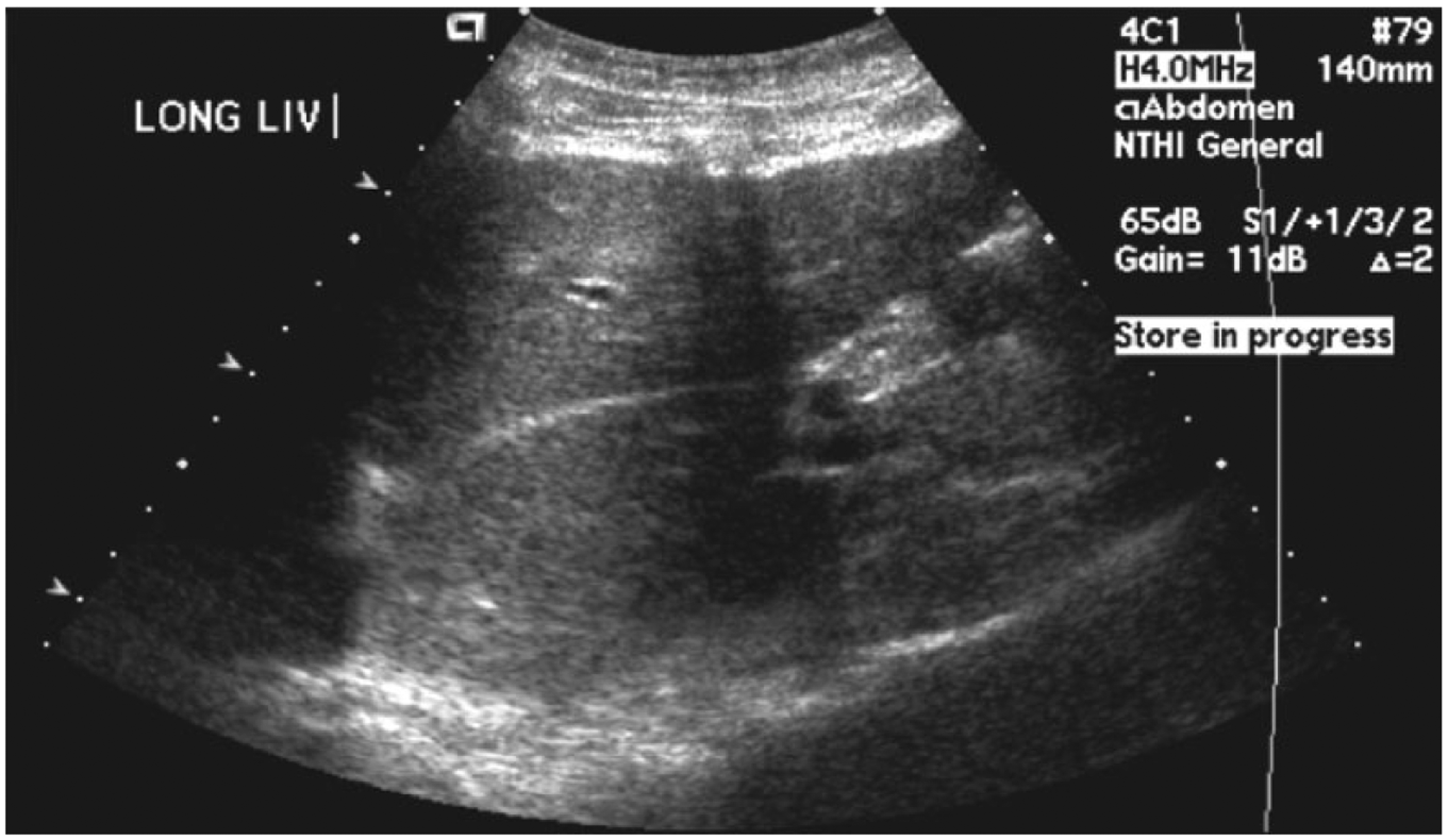
Longitudinal gray-scale image of the liver showing the enlarged caudate lobe.
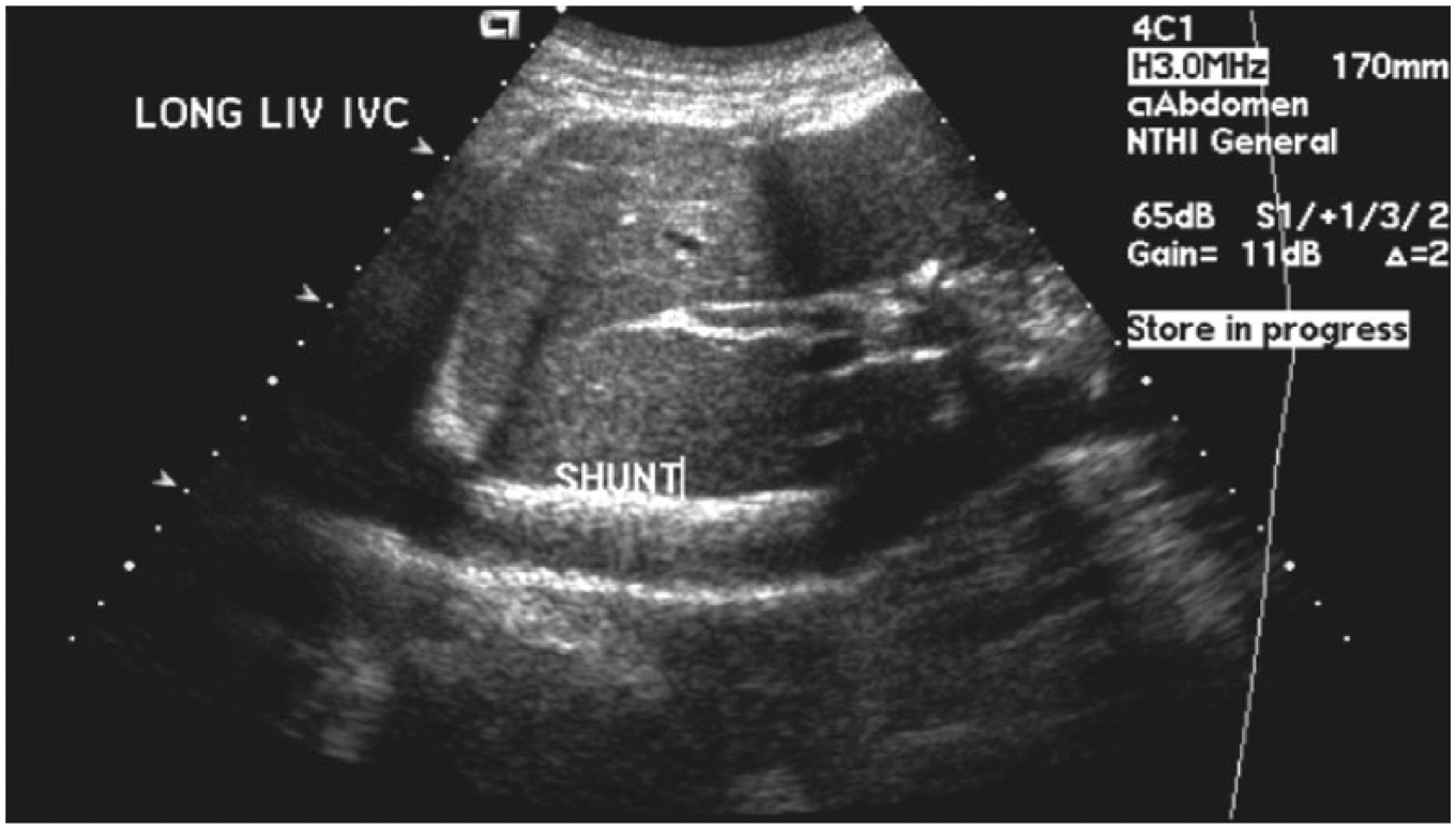
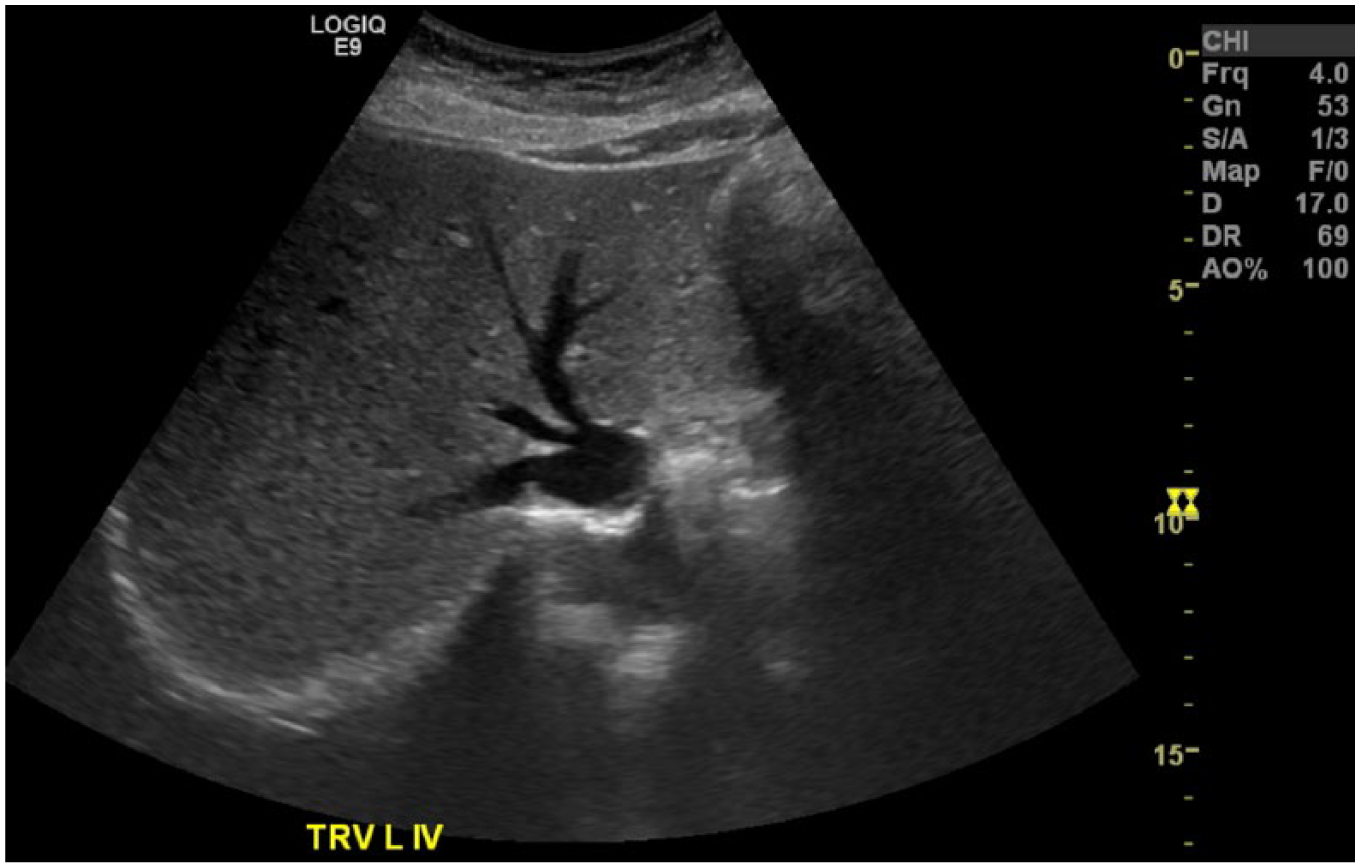
Longitudinal gray-scale image of the liver showing the bright echogenicity of the stent in the inferior vena cava (erroneously labeled
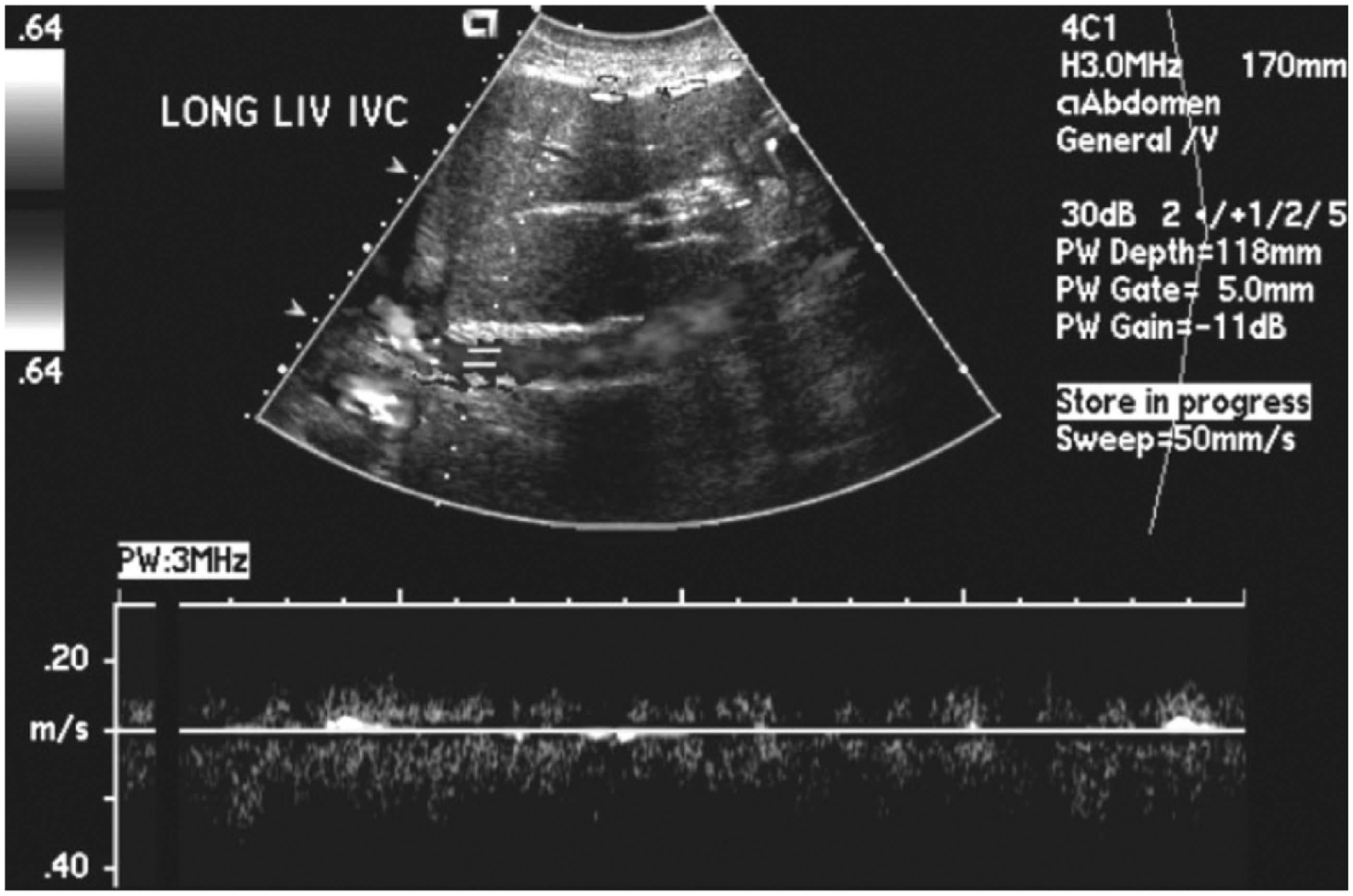
Color Doppler (displayed in gray-scale) and spectral Doppler display of flow in the inferior vena cava showing disturbed and diminished flow predominantly antegrade.
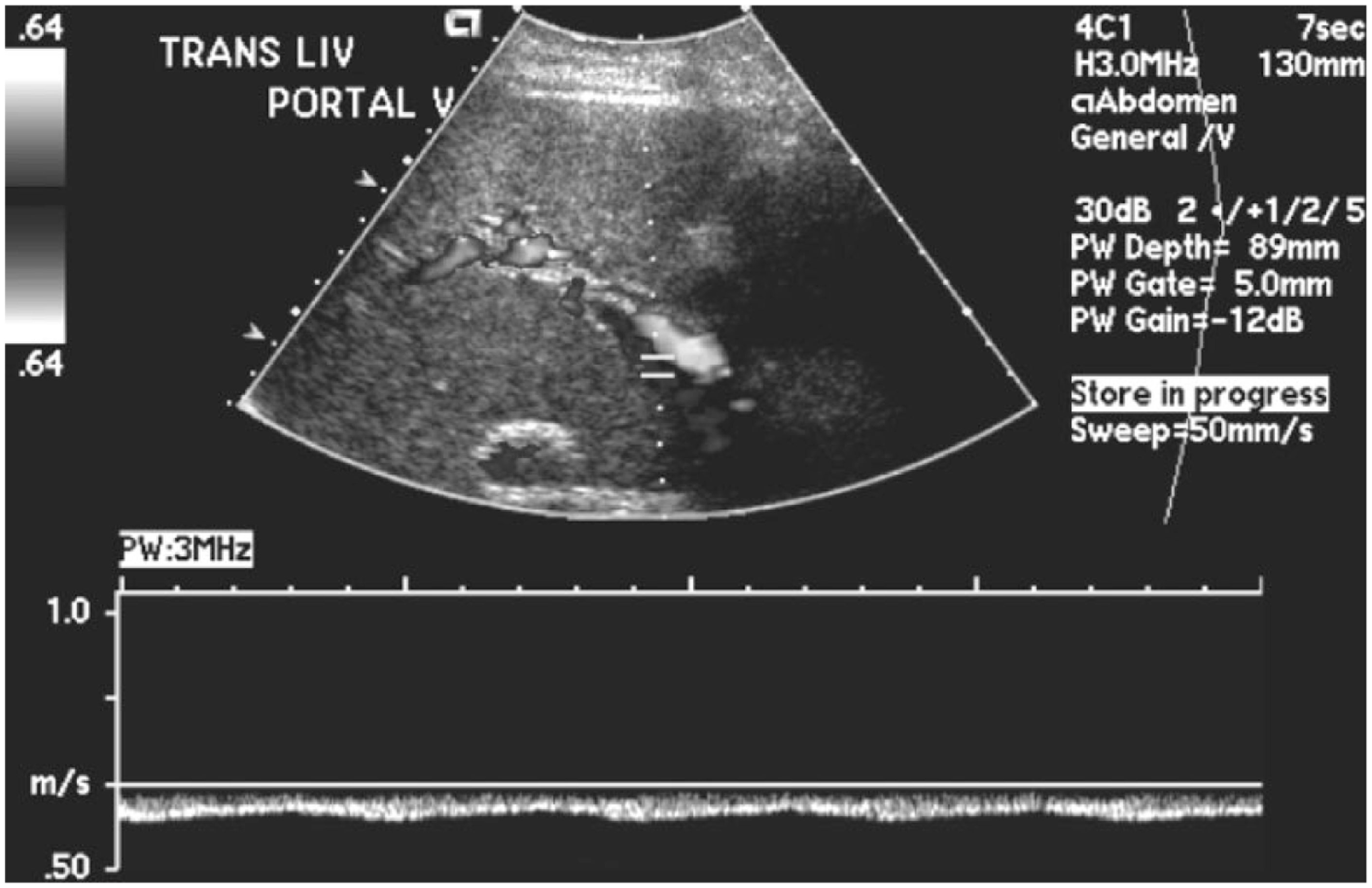
Color Doppler (displayed in gray-scale) and spectral Doppler display of flow in the portal vein showing reversed (hepatofugal) flow.
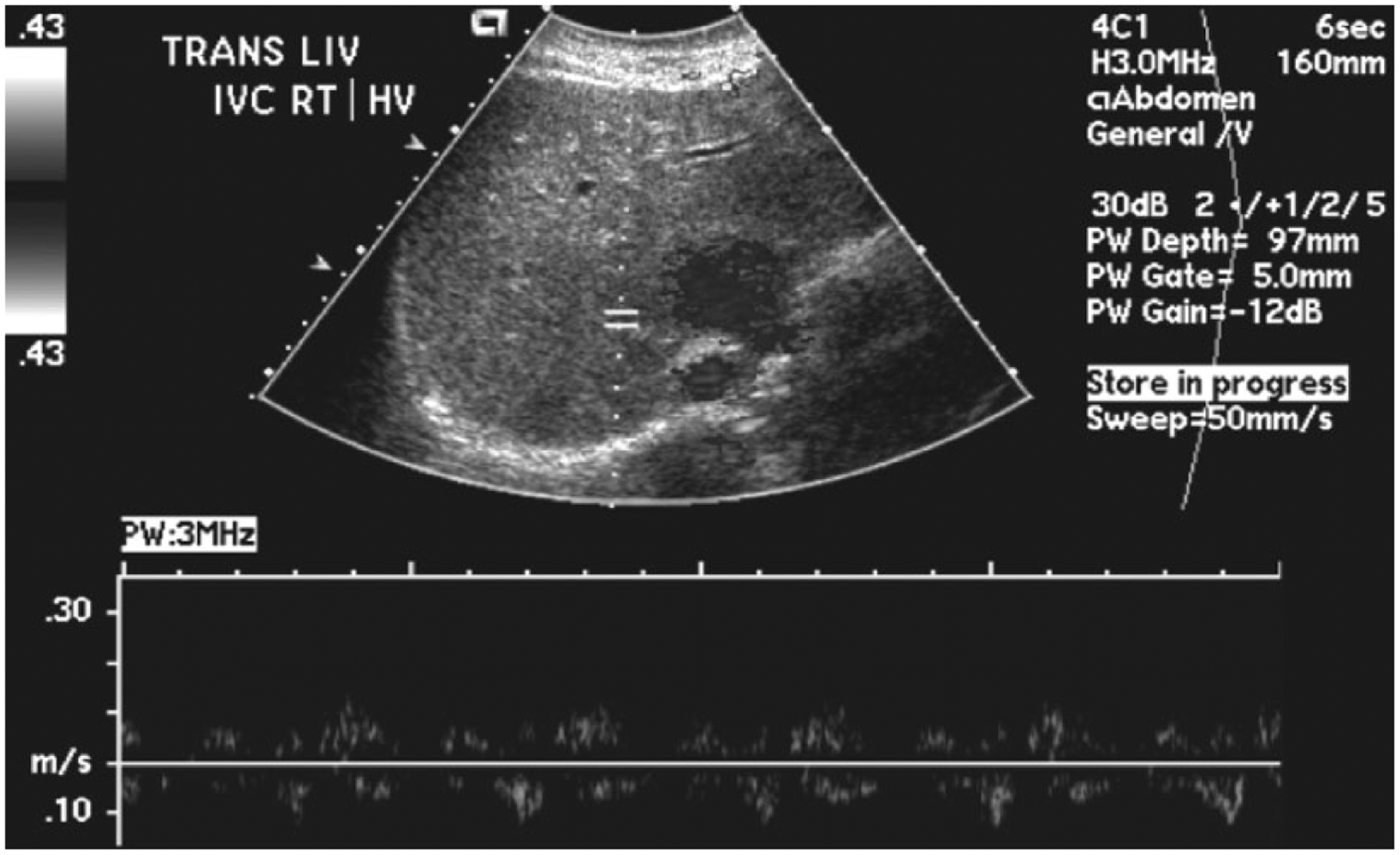
Color Doppler (displayed in gray-scale) and spectral Doppler display of flow in the right hepatic vein showing diminished and irregular flow patterns likely in part secondary to a poor Doppler insonation angle.
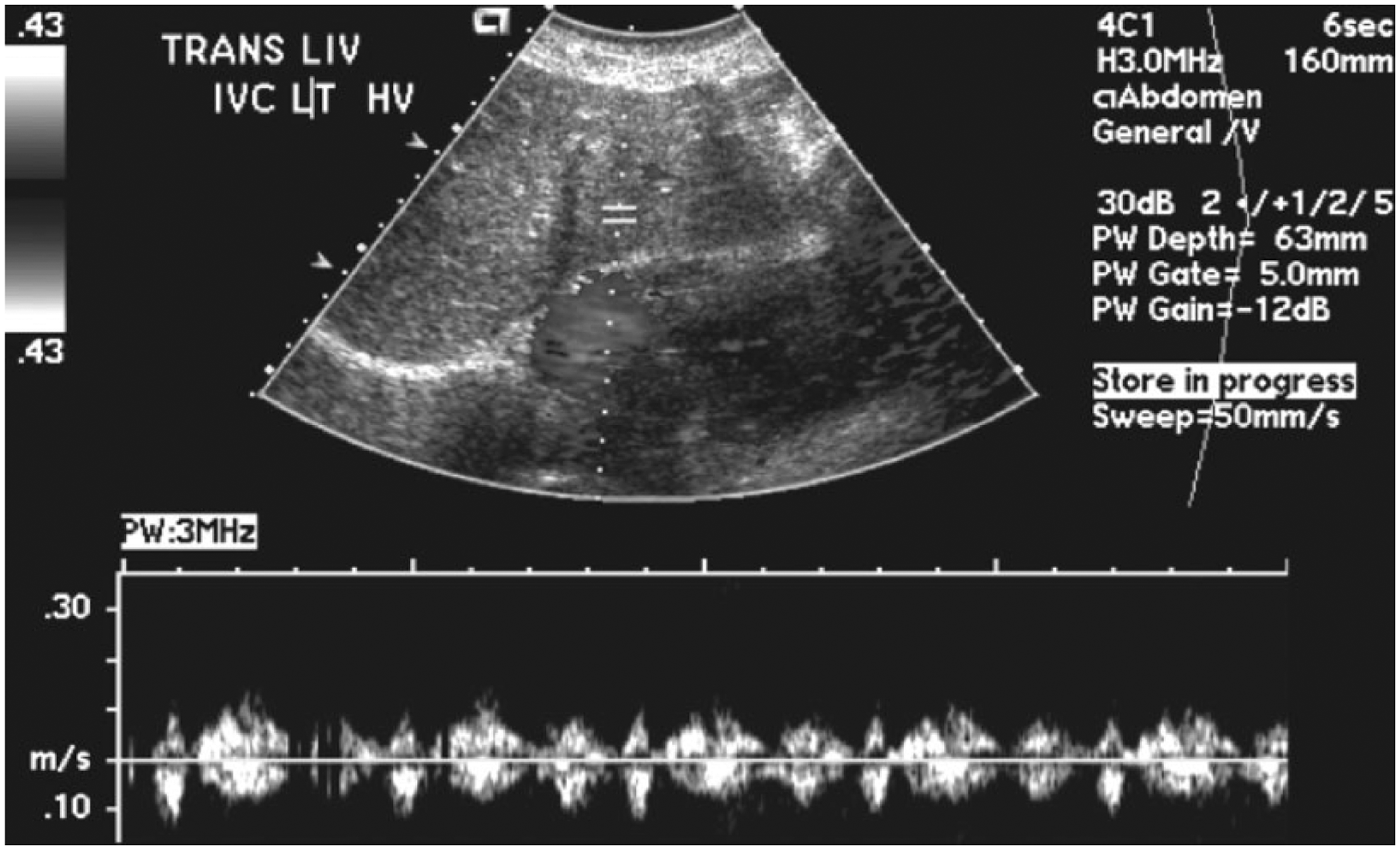
Color Doppler (displayed in gray-scale) and spectral Doppler display of flow in the left hepatic vein showing a dampened waveform with an artifact likely in part secondary to a poor Doppler insonation angle.
A year after this examination (two years after placement of the IVC stent, shown schematically in Figure 8), the patient presented with acute hepatic encephalopathy. This was caused by an increased ammonia level secondary to liver dysfunction. The next step in treatment for this patient would be an evaluation for transjugular intrahepatic portosystemic shunt or liver transplant. However, because of the patient’s comorbidities, it is not certain that she would be eligible for a liver transplant. At this time, no further follow-up is available.
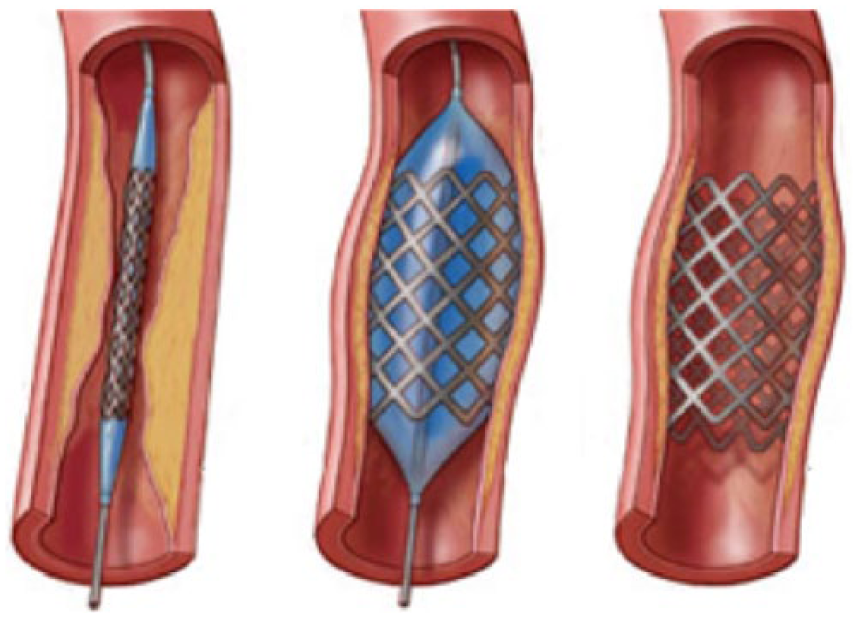
Schematic diagram of the procedure for angioplasty with stenting. (Diagram with permission from Verona Veins at Access Care Physicians. Available from http://www.veronaveins.com/dialysis-access-management.php.)
Discussion
Because BCS remains an uncommon disorder, data regarding its epidemiology are limited. Available literature suggests that it occurs about 1 in 100 000 individuals, is more common in women, and usually presents in the third or fourth decade of life.1-4 Differential diagnoses include portal hypertension, cirrhosis, right-sided heart failure, metastatic liver disease, infectious hepatitis, and many other less common disorders. 6 It is predominantly characterized by hepatic venous flow obstruction. It may be classified as primary or secondary based on its pathophysiology. 12 The primary form is caused by thrombotic obstruction of the hepatic veins, usually as a result of thrombophilia such as antiphospholipid syndrome. The secondary form results in thrombosis of the hepatic veins or IVC caused by extrinsic compression from such sources as pregnancy or tumors or by trauma. It can also be secondary to a congenital anomaly where the IVC is obstructed by webbed or membranous tissue. In approximately 25% to 30% of all cases, the exact cause is never determined. 2 Budd-Chiari syndrome starts with an obstruction of the hepatic outflow; secondary to this, intrahepatic sinusoidal pressure increases, which causes portal hypertension. Reduced hepatic function then leads to necrosis of the hepatocytes, followed by hypoxia and ischemic injury, which ultimately leads to liver failure. 13
Sonographically, the liver is typically large and bulbous in the acute phase of BCS. It appears hypoechoic and the hepatic veins are enlarged in cases of acute thrombosis. In chronic cases, the hepatic veins are usually not visualized and the liver appears hyperechoic and heterogeneous. 14 With BCS, the caudate lobe can become hypertrophied. It has a dual blood supply from the right and left portal veins, and it is the only segment of the liver that has venous drainage directly into the IVC via the caudate vein. 7 Typically in BCS patients, venous drainage of the liver is preserved through the caudate vein. This vein then receives blood not only from the caudate lobe but also from other parts of the liver through collateral vessels, leading to an enlargement of the lobe and the vein. 15 Hypertrophy of the caudate lobe, irregularities of the liver contour, and regenerative nodules are prominent features of chronic BCS. 16 There is often atrophy of the right hepatic lobe as a result of extrinsic compression of the portal blood flow causing it to be either slowed or reversed (hepatofugal). 13 Color Doppler evaluation of the hepatic vascular system may show filling defects that could signify thrombus within the vessels; pulse wave spectral Doppler evaluation may show abnormal blood flow patterns as well.14,17
A review of the current literature showed no published protocol for sonographic follow-up of transluminal balloon angioplasty of the IVC with intracaval stent. Therefore, sonographers should follow standard liver Doppler protocols including gray-scale, color Doppler, and pulse wave spectral Doppler evaluation, taking care to adjust gain and scale settings as needed and including all appropriate views, resistive indices (RIs), and velocity measurements. Table 1 shows a listing of expected normal findings as well as abnormal findings that are likely to be seen secondary to BCS. A typical anterior-posterior measurement is taken of the longest axis of the right lobe of the liver. A normal RI for the hepatic artery should be between 0.55 and 0.70, characteristic of a low resistance runoff. Any measured RI above or below the normal range may represent disease.18,19 If an intracaval stent is in place, the IVC should be thoroughly investigated throughout its length with special attention given to the hepatic vein confluences (Figure 9). The normal IVC measures approximately 2.5 cm and should not exceed 3.7 cm, although its diameter will vary with respiration. The hepatic veins have a normal diameter of 4 to 7 mm and will show predominantly antegrade flow with significant heart rate phasicity, with systolic flow greater than diastolic flow 7 (Figure 10). The IVC and the hepatic veins are strongly affected by both cardiac and respiratory variations. It is important to obtain Doppler signals from these vessels when the patient is asked to stop breathing or to take a small inward breath and stop breathing.5,19 In both the IVC and hepatic veins, an abnormal waveform pattern may result in a flattening of waveform morphology and a loss of phasicity. 20 The caudate vein (when visible) should be evaluated, as a size of 3 mm or greater could be indicative of BCS. 15 The sonographer should also look for intrahepatic collaterals, as these are common with BCS. 16 Reasons for turbulent venous flow include atrial fibrillation, hepatic vein thrombosis, or patient ventilation. Causes of blunted or monophasic venous waveforms include cirrhosis, steatosis, hepatic lymphoma or metastasis, and respiratory expiration or valsalva. Chronic heart failure may contribute to a waveform displaying irregular biphasic flow. 19
Gray-scale transverse image of the liver showing the normal hepatic veins at the confluence with the inferior vena cava. (Image courtesy of Pamela Crawford, RT, RDMS, RVT, El Centro College.)
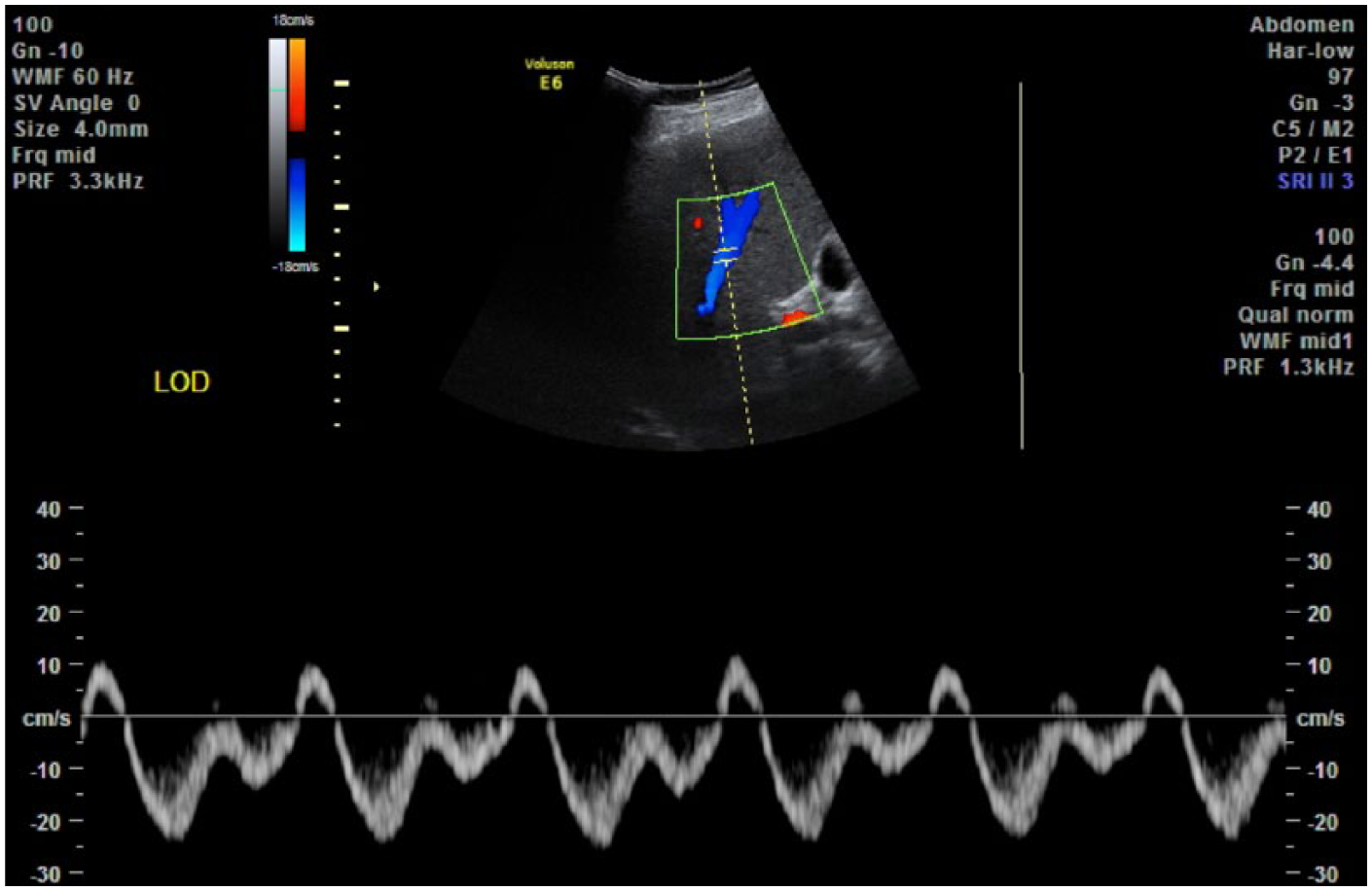
Color Doppler and spectral Doppler display of flow in the normal hepatic vein scanned with the patient in the left oblique decubitus position.
In the examination of this patient, the IVC was insonated with the patient in the supine position. Often, this position can limit the full course of visualization, and the insonation angle in this position may lead to unreliable data. As demonstrated in Figures 11 to 14, some alternate patient positions to use for increased visualization and better Doppler angles of the IVC would be the left posterior oblique, right posterior oblique, and left lateral decubitus positions. For the best results, it is very important to keep the Doppler insonation angle at 60 degrees or less. Doppler measurements are not reliably achieved, and Doppler waveforms are often distorted, at Doppler angles greater than 60 degrees. 21
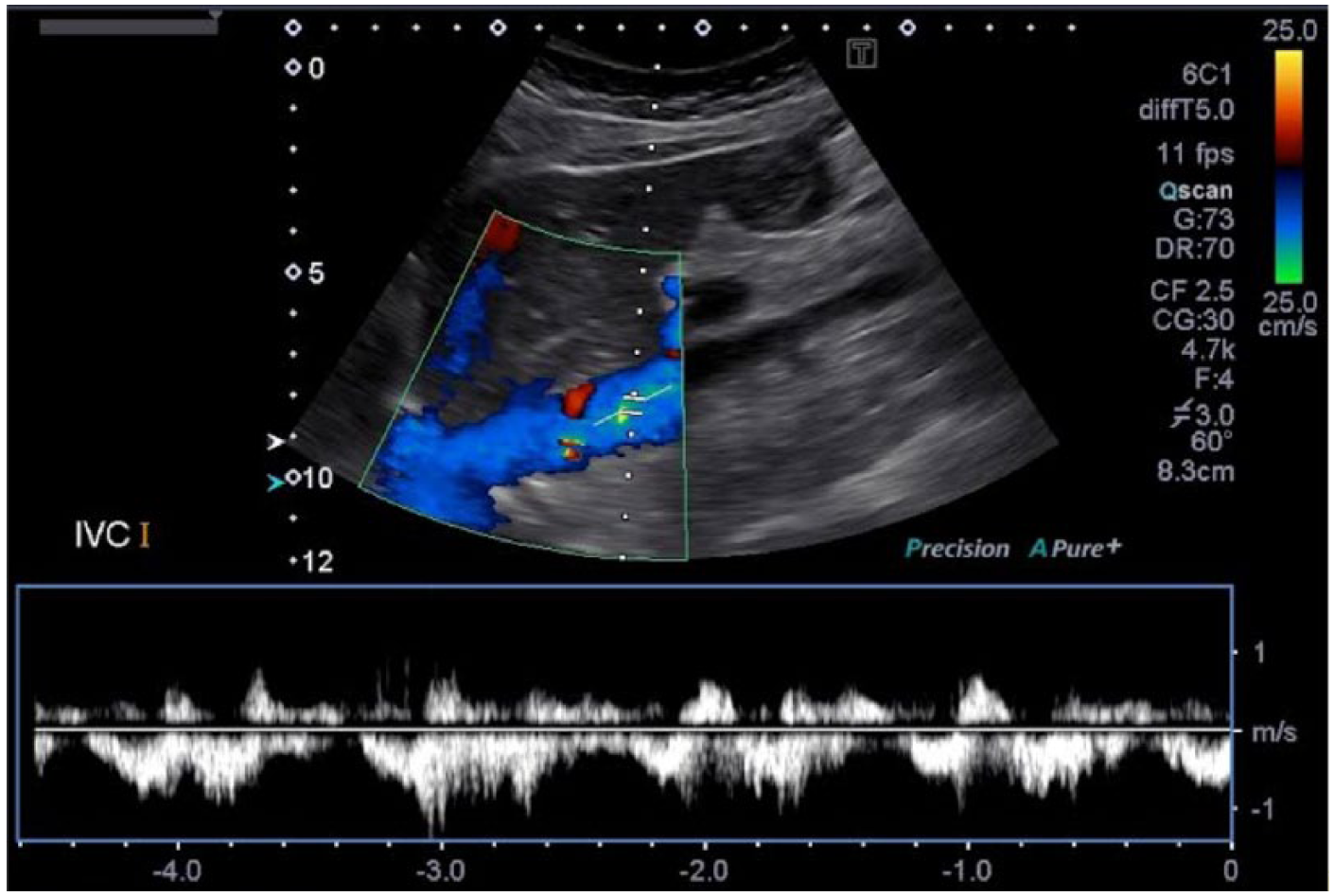
Color Doppler and spectral Doppler display of flow in the normal inferior vena cava scanned with the patient supine.
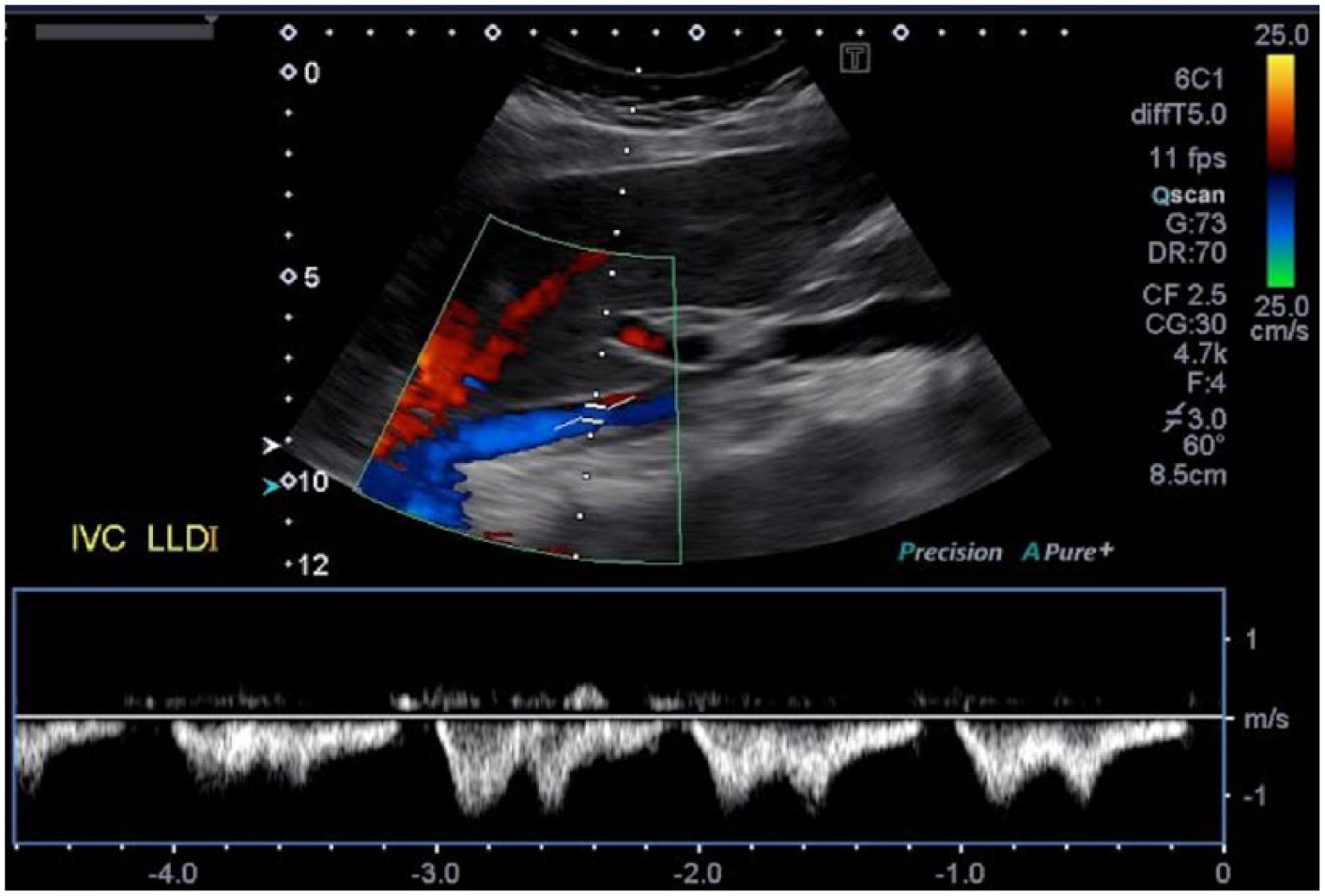
Color Doppler and spectral Doppler display of flow in the normal inferior vena cava scanned with the patient in the left lateral decubitus position.
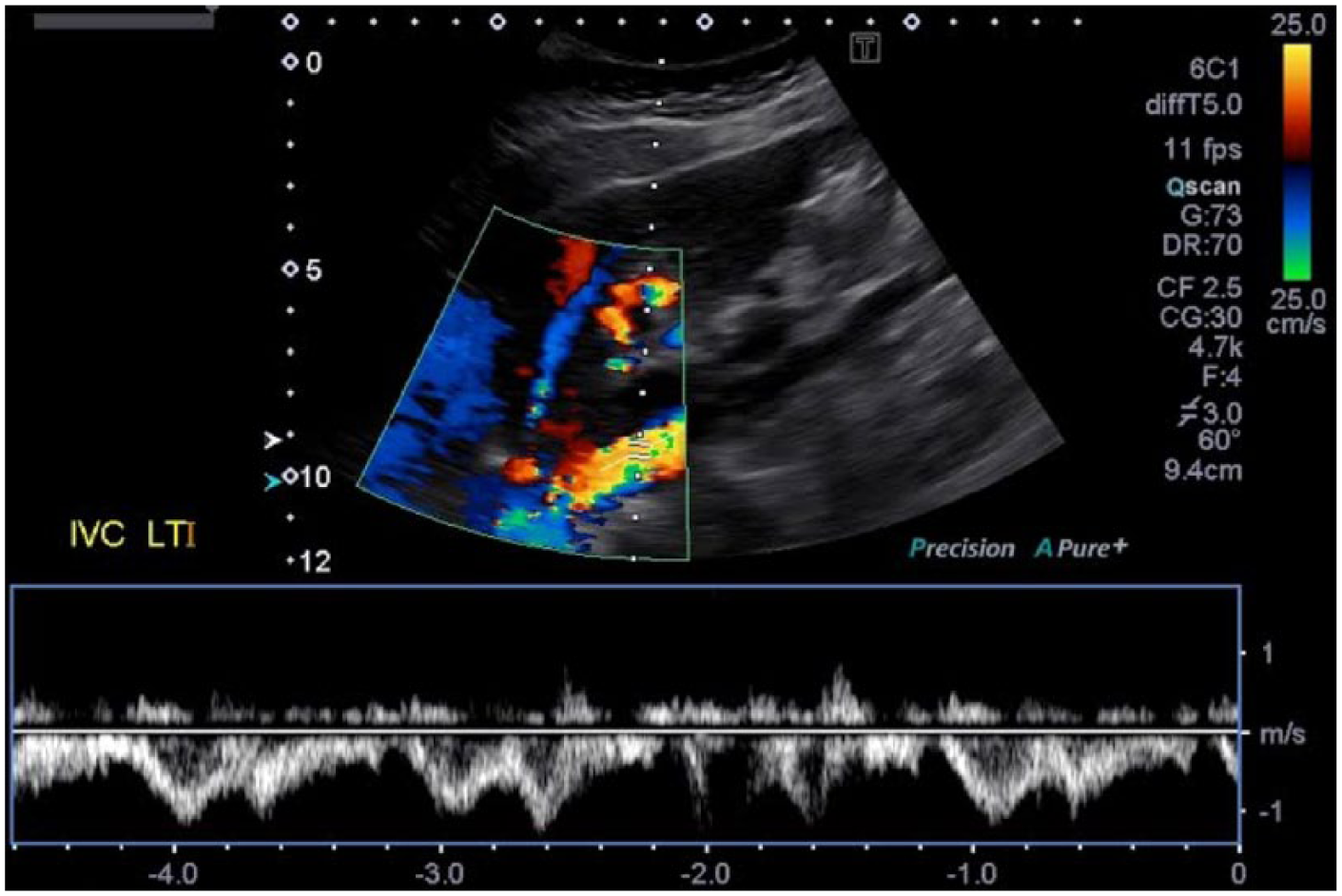
Color Doppler and spectral Doppler display of flow in the normal inferior vena cava scanned with the patient in the left posterior oblique position.
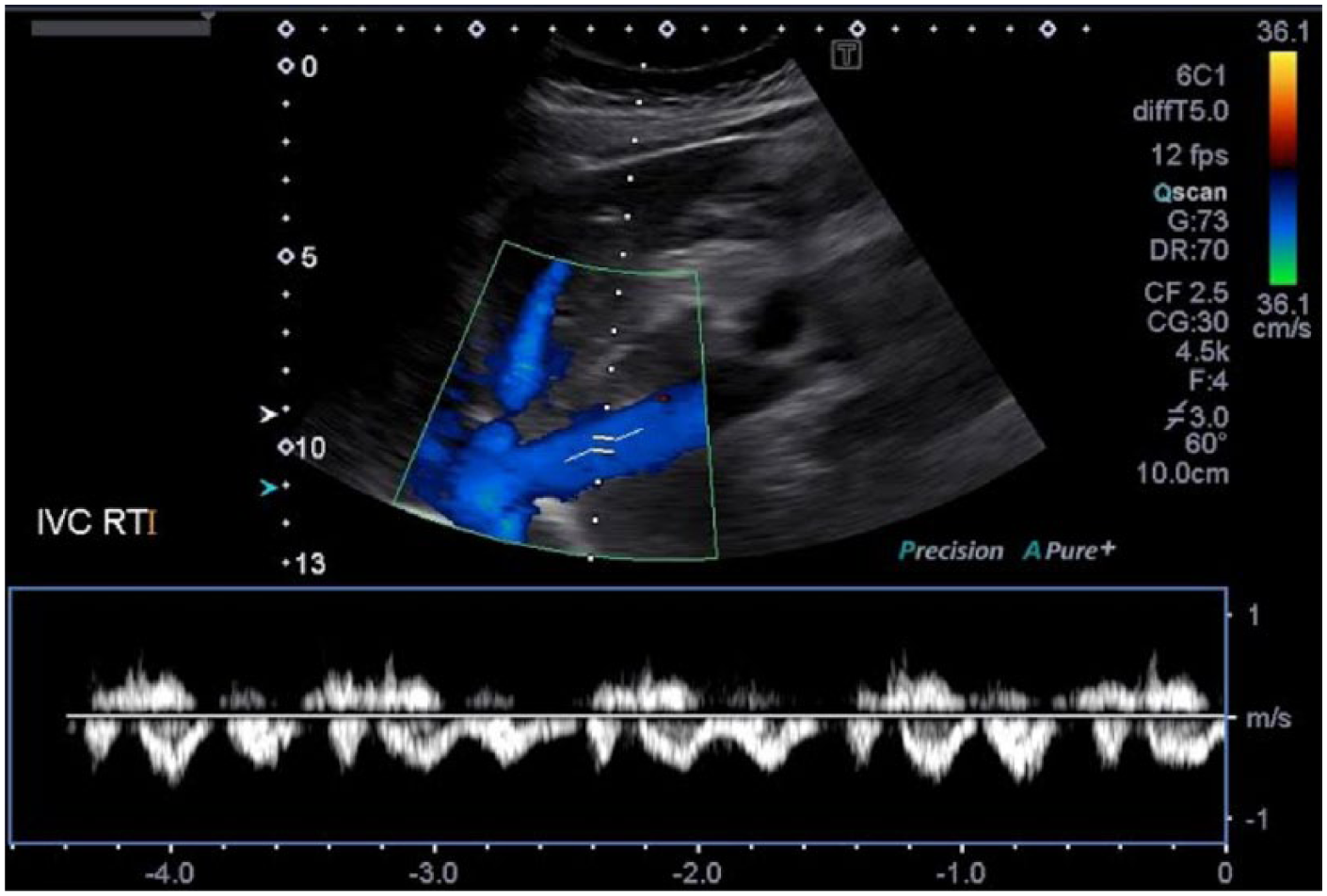
Color Doppler and spectral Doppler display of flow in the normal inferior vena cava scanned with the patient in the right posterior oblique position.
There is a treatment strategy recommended for patients with BCS using a graded approach ranked by order of increasing invasiveness. Anticoagulation therapy is administered first and may be used for short-term and long-term symptomatic benefits. The second form of treatment is angioplasty with or without stenting of short segment venous thrombosis in the hepatic veins or IVC to recanalize the vessel and keep it patent 22 (Figure 8). Portosystemic shunting is considered as the third alternative in patients unresponsive or unsuited to stenting. The procedure involves creating a shunt from the portal vein to the IVC. There are two ways of creating a portosystemic shunt: surgically or percutaneously. In the past, shunts were created surgically during open abdominal procedures using vascular bypass techniques; now, most portosystemic shunts are created percutaneously via a transjugular intrahepatic portosystemic shunt. 23 In this procedure, via percutaneous access through the jugular vein and IVC, a stent is placed communicating directly from the right portal vein to the right hepatic vein. The aim of the shunt is to decompress the congested liver and return portal venous flow to its normal hepatopetal direction. 13 When portal decompression is performed early in the course of BCS, liver damage and the hemodynamic disturbances can be reversed. However, a delay in diagnosis or treatment often results in irreversible liver damage. 24 The fourth and final form of treatment is orthotopic liver transplantation. 25 It is the appropriate treatment in the late stages of BCS, when the liver disease is no longer reversible and progressive hepatic decompensation cannot be stabilized. 24 Prognosis is poor in patients without treatment, with death resulting from progressive liver failure within 3 years from time of diagnosis in virtually all patients. Five-year survival following portosystemic shunting is 78%. 26 The five-year survival rate following liver transplant is approximately 70%.17,27
About a third of patients with BCS have short length stenosis of either the hepatic veins or the IVC. 28 A primary IVC stenosis or occlusion is least common in Western countries, as pure hepatic vein involvement predominates in these locations. Patients with primary IVC involvement are candidates for percutaneous transluminal angioplasty of the IVC, which may be performed via either a jugular or femoral vein approach. Under sonographic or fluoroscopic guidance, a balloon catheter is inserted to dilate the IVC and a stent is then placed into the obstructive portion to support the vessel and keep it patent (Figure 8). Restenosis of the affected segment appears to be more common in patients undergoing primary angioplasty alone rather than in patients undergoing primary stenting. 29 Routine duplex sonographic follow-up should be done to assess for patency and stable placement of stent or for recurrence of BCS symptoms. When anticoagulation therapy (to prevent re-occlusion) and angioplasty (performed early enough) are done together, there appears to be better success in controlling BCS long term. 27
Conclusion
Budd-Chiari syndrome can have multiple causes and is ultimately a lethal condition if untreated; however, there are treatment options available to help extend life as long as possible. As seen in this case, gray-scale, color Doppler, and spectral Doppler sonography are extremely useful in the detection and follow-up of BCS. Sonography can also be used to characterize the location and severity of the condition, which will determine the level of treatment offered for these patients. Proper equipment settings, image optimization, and patient positioning are key to acquiring reliable diagnostic images.
Footnotes
Acknowledgements
I would like to dedicate this article to my family and friends who have supported me during my journey through the Diagnostic Medical Sonography program. I would like to thank Texas Health Presbyterian Hospital of Plano for providing the case images. I extend special thanks to Lynn Schluns, BS, RDMS, RVT, Program Director of Diagnostic Medical Sonography at El Centro College in Dallas, Texas, for her assistance, instruction, and guidance in writing and submitting this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.

