Abstract
In most cases of common carotid artery (CCA) occlusion, the internal carotid artery (ICA) is also occluded. This case presents a patient with a patent ICA distal to a thrombotic CCA occlusion, likely secondary to cardiac embolization related to chronic atrial fibrillation, with retrograde filling of the extracranial ICA via intracranial collateral flows.
The carotid arteries, as all other arteries in the body, are susceptible to atherosclerotic plaque development. Not uncommonly, there may be atheromatous plaque at the origin of the internal carotid artery (ICA) causing a total occlusion. Rarely, the common carotid artery (CCA) may become occluded, often associated with ipsilateral ICA occlusion 1 ; however, in the case presented below, the ipsilateral ICA and external carotid artery (ECA) remained patent.
The extracranial cerebrovascular duplex and Doppler ultrasound protocol requires imaging the CCA, ICA, ECA, vertebral artery (VA), and subclavian artery (SCA). If significant pathology is demonstrated, the examination may be expanded to include the ophthalmic arteries (OAs). The criteria used to diagnose an occlusion typically include increased echogenicity throughout the course of the vessel, lack of radial pulsation of the vessel, absence of any color flow filling of the vessel, and no detectable spectral Doppler signal throughout the vessel.2–5
Case Report
A woman in her late 80s referred from the Stroke Prevention Clinic presented with transient right arm weakness that had persisted for approximately 6 hours. The patient had a history of atrial fibrillation and was being treated with warfarin. Laboratory analysis demonstrated a subtherapeutic international normalized ratio (INR) of 1.4. Therapeutic levels for this laboratory test that monitors the relative degree of anticoagulation should be between 2.0 and 3.0. 6 At that time, her warfarin dose was readjusted to regulate her INR into the therapeutic range, and she was referred for a carotid duplex sonogram. Her past history included a mild stroke in 2004 with residual left facial asymmetry.
The carotid sonogram was performed by a registered vascular technologist using a Philips IU 22 ultrasound machine (Philips Healthcare, Andover, MA), with the results interpreted by a neurologist from the Neuro Sonography Unit. A linear probe with a 9-MHz center frequency was used. Two-dimensional (B-mode) examination of the left side showed multifocal plaques at the carotid bifurcation and in the proximal ICA. Spectral Doppler analysis demonstrated normal flow dynamics of the left ICA (Figure 1) with normal flow hemodynamics seen in the left CCA and ECA as well. B-mode examination of the right side demonstrated echogenic material within the lumen of the mid and distal CCA with multifocal plaques at the CCA bifurcation and proximal ICA (Figure 2a,b). Spectral Doppler analysis of the proximal right CCA demonstrated a high-pulsatility to-and-fro flow with low velocity, typical of a flow waveform proximal to an occlusion (Figure 3). No flow was detected by color, power, or spectral Doppler in the mid and distal segments of the CCA (Figure 4), even with the spectral Doppler sample volume box size increased. Color Doppler flow assessment of the ICA demonstrated retrograde blood flow. Spectral Doppler analysis confirmed retrograde ICA flow with a peak systolic velocity of only 14 cm/s. Color flow assessment of the ECA showed antegrade flow, which was confirmed by spectral Doppler; the identification of the ECA was confirmed by the presence of an arterial branch and visualization of a temporal tap response (Figure 6). Antegrade flow signals were noted in the vertebral arteries bilaterally. Throughout the spectral Doppler evaluation, note was made of an irregular heart rhythm (Figures 3 and 5).
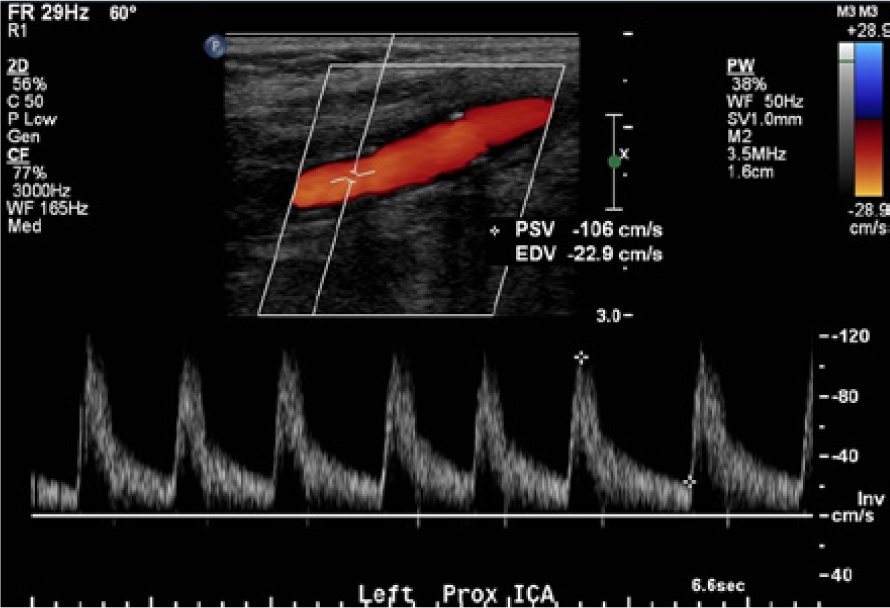
Color and spectral Doppler evaluation of the left internal carotid artery showing normal velocities and a normal low-resistance antegrade flow pattern.
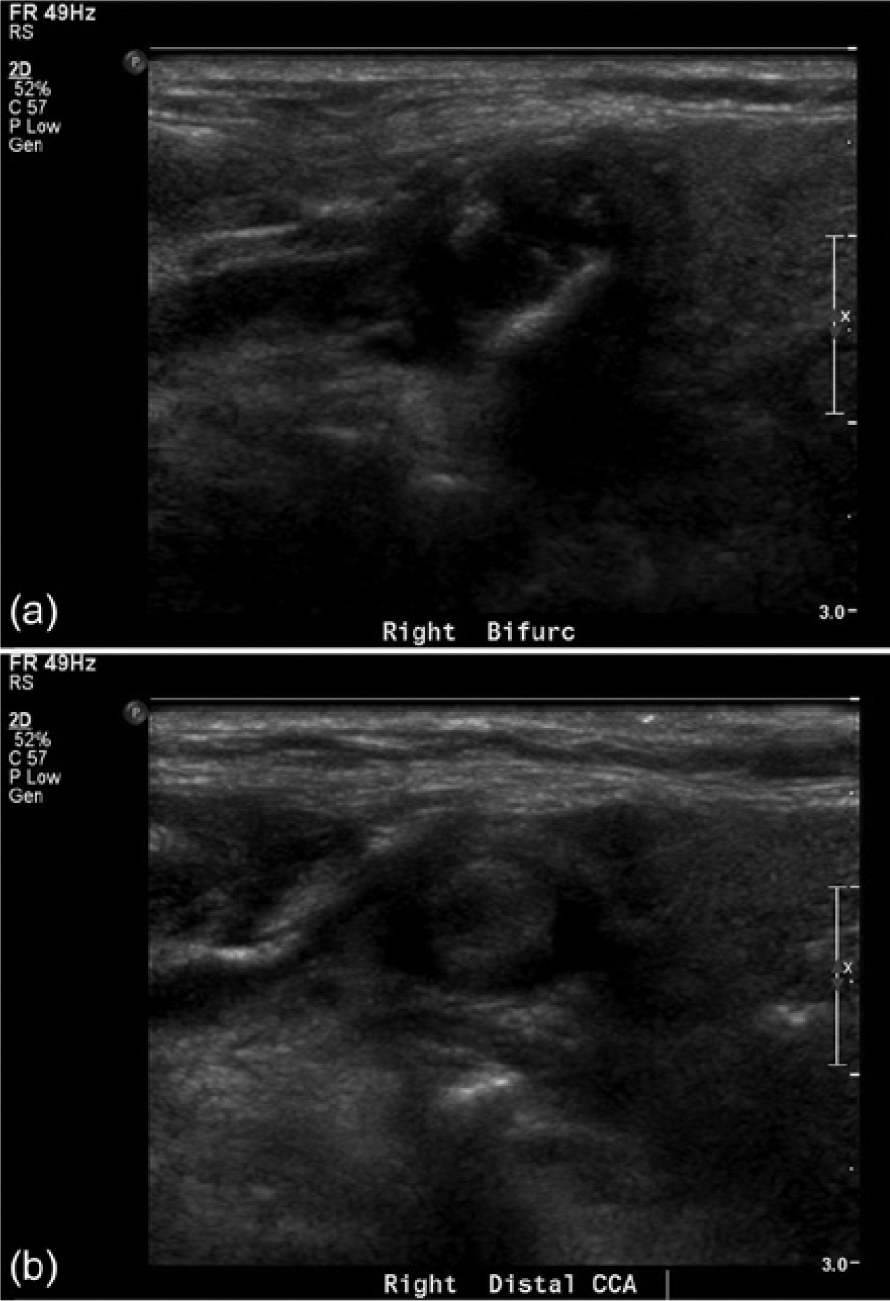
(a) B-mode image of the right carotid artery bifurcation region demonstrating evidence of a diffuse atherosclerotic plaque with heterogeneous echogenicity. (b) Transverse B-mode image of the distal right common carotid artery showing a more homogeneous, somewhat hypoechoic material characteristic of thrombus within the lumen of the vessel.
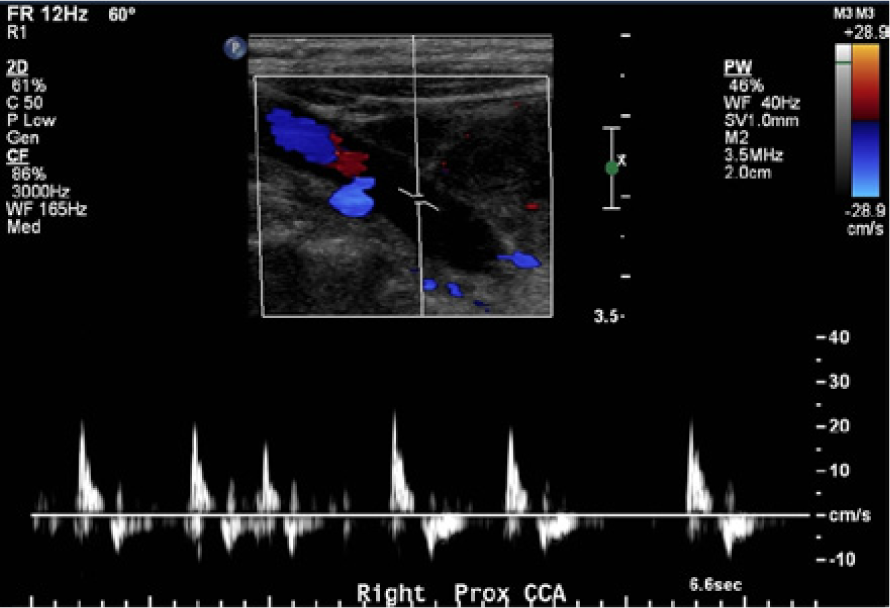
Color and spectral Doppler evaluation of the proximal right common carotid artery showing low velocities with a pulsatile, to-and-fro flow waveform characteristic of a more distal occlusion.
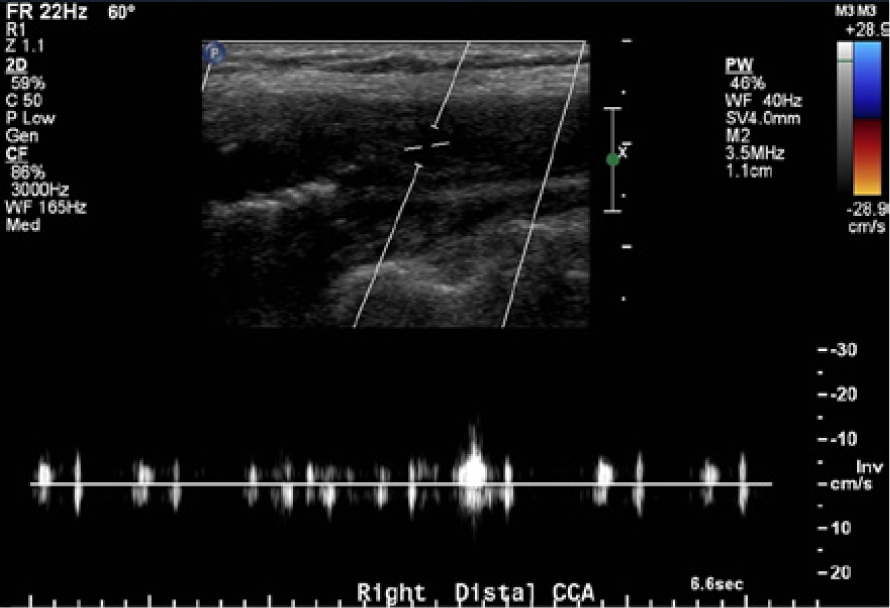
Color and spectral Doppler evaluation of the distal right common carotid artery showing a complete absence of flow. Note the increased sample volume size to maximize sensitivity to any low flow signals.
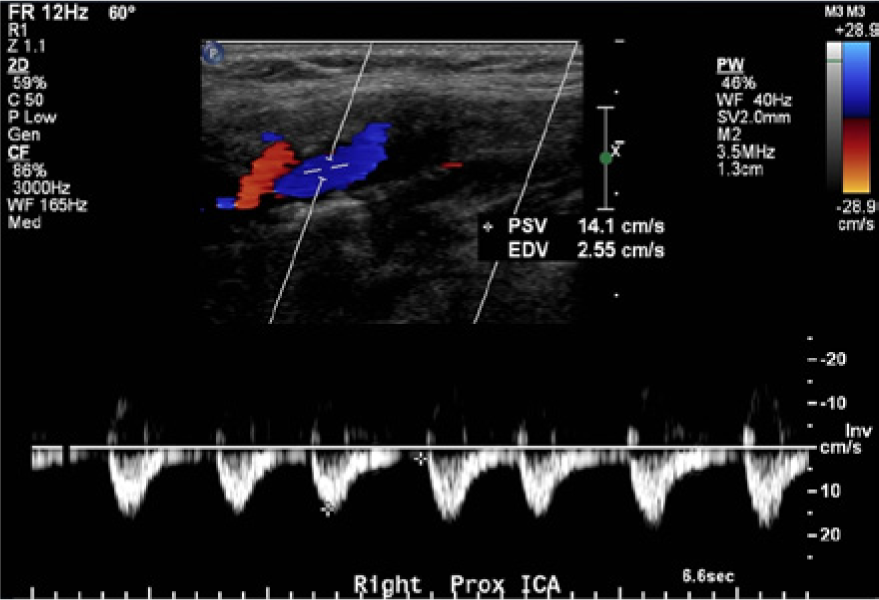
Color and spectral Doppler evaluation of the proximal right internal carotid artery (ICA) showing low retrograde velocities with a blunted, damped flow waveform. Note the antegrade flow direction shown by color Doppler in the external carotid artery just superior to the ICA in the image.
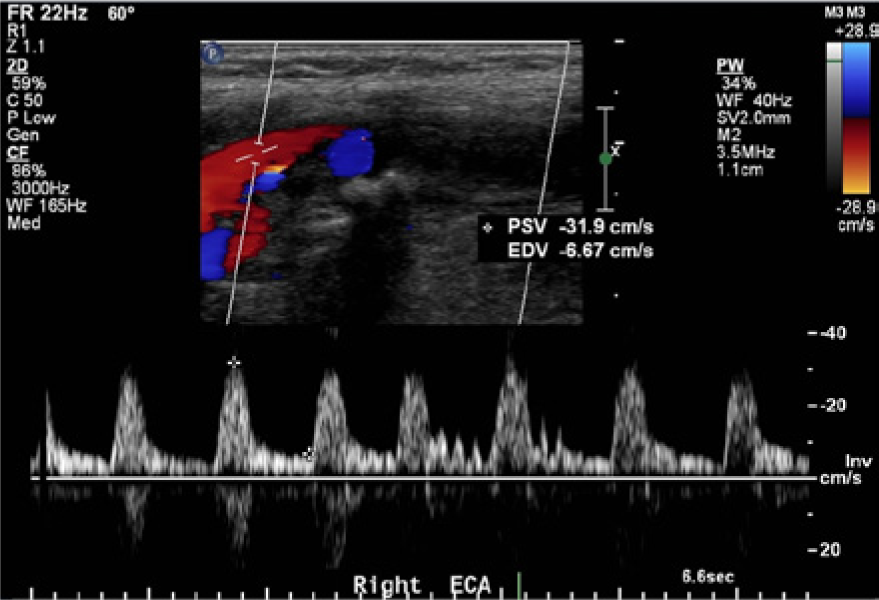
Color and spectral Doppler image of the right external carotid artery (ECA) confirming antegrade flow. Note the branch vessel in the color Doppler image and the temporal tap velocity fluctuations in the spectral display, both characteristic of the ECA.
Based on the results of the carotid sonogram, the patient was sent for a computed tomography (CT) scan of the head without intravenous (IV) contrast and CT angiography (CTA) of the neck and head with IV contrast. The CT and CTA were performed using a General Electric (GE, Piscataway, New Jersey) 64-slice scanner following the departmental stroke protocol. The CT head scan demonstrated no acute abnormality of the parenchyma with evidence of possible chronic ischemic microvascular disease. The CTA of the neck showed left-sided patency with no significant narrowing. Occlusion of the right CCA in the mid and distal segments was demonstrated with the cutoff of IV contrast; more distally, the patency of the ICA and ECA was demonstrated by the presence of IV contrast (Figure 7). The intracranial portion of the CTA showed patency of the anterior communicating artery (AComA); the posterior communicating arteries (PComA) were not seen (Figure 8a,b). While the CTA did not provide direction of flow information, it can be seen that in the right ECA and its branches, the contrast is not as concentrated and is less dense, which indirectly supports the findings of delayed filling via the right ICA. With this alternate collateral pathway for blood flow, the contrast media required additional time to reach the ECA, causing the contrast to be less dense compared with the ICA and contralateral arteries.
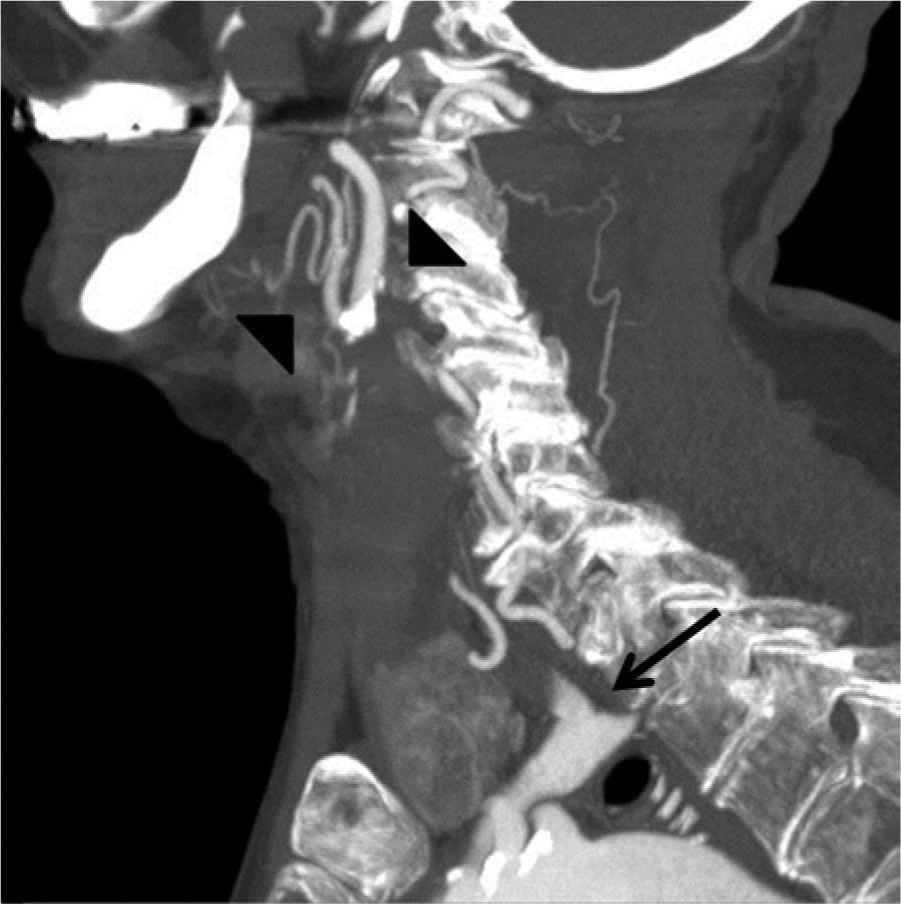
Computed tomography arteriogram of the right common carotid artery and bifurcation in the sagittal plane demonstrating the cutoff at the proximal common carotid artery (black arrow) and the patency of the internal carotid artery and external carotid artery (arrowheads).
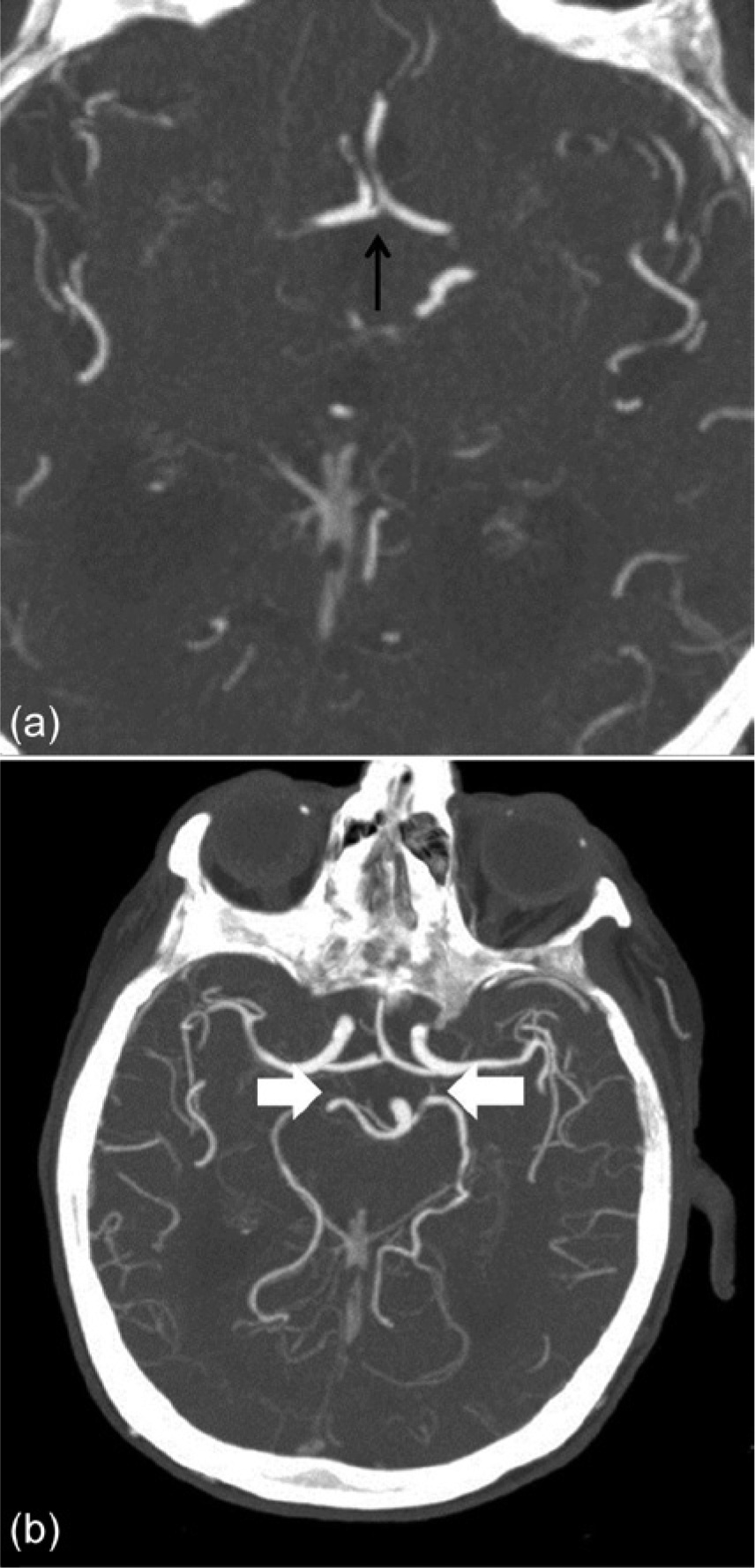
(a) Intracranial computed tomography arteriogram (CTA) showing a patent anterior communicating artery (AComA; black arrow), which is clearly visible and dilated compared with its normal state. (b) Intracranial CTA showing the circle of Willis, again demonstrating a patent, clearly visible AComA, with nonvisualization of either posterior communicating artery (PComA). The white arrows denote the locations where the PComAs should be seen.
Additional diagnostic studies for the patient included an electrocardiogram (ECG), which confirmed the diagnosis of atrial fibrillation, and an echocardiogram. The echocardiogram showed severe left atrial dilation and left ventricular systolic dysfunction; aortic atheromatous disease was also visualized.
Given that the patient’s right-sided symptoms were slowly resolving and believed to be related to a thromboembolic event in the left cerebral hemisphere, with her INR now at a therapeutic level, no significant changes were made in her treatment plan from her previous visit to the Stroke Prevention Clinic. The impression of the Neuro Sonography Unit staff was that she was asymptomatic with regard to the right CCA occlusion, and no interventions were warranted at this time. The patient was scheduled for follow-up with the Stroke Prevention Clinic and advised to follow up with her family physician to monitor her INR and adjust her warfarin dosage appropriately.
The patient returned two months later to the Stroke Prevention Clinic. In that time interval, there had been no recurrence of the transient right arm weakness. Aside from the mild left facial flattening attributed to the prior remote (2004) ischemic event, examination of the cranial nerves revealed no significant injury. The patient remained on warfarin with a therapeutic INR.
In the following months, however, this patient had multiple ischemic events and ultimately presented with delirium and dementia, with a subtherapeutic INR of 1.3. She underwent a CT head scan, which demonstrated an acute infarction of the left middle cerebral and posterior cerebral arterial territories involving the left frontal, parietal, temporal, and occipital lobes. There was no hemorrhagic transformation or midline shift, but the patient did succumb to her ischemic events. All diagnostic studies indicated that the final stroke the patient had occurred in the left cerebral hemisphere secondary to thromboembolic disease, and the right CCA occlusion was not a source of the embolic event(s) that led to the patient’s death.
Discussion
Sonographic examination of the CCA and its major branches, the ICA and ECA, has proven effective for the assessment of stenosis and flow.2,7 For cases of CCA occlusion, color Doppler and spectral Doppler analysis have a reported 97% accuracy, 91% sensitivity, and 99%. 7 Sonography not only demonstrates patency, or lack thereof, in the ICA distal to a CCA occlusion but also allows determination of the direction of flow. 8 The anatomical location of the CCA makes it a relatively easy vessel to access, and if an occlusion is visualized, the Doppler pulse repetition frequency (PRF) can be manipulated to allow for detection of any low-velocity flow distal to the occlusion.2,9 It is not only color and spectral Doppler analysis that contributes to the diagnosis of CCA occlusion. Using B-mode ultrasound imaging, the luminal contents can be imaged, suggesting (but not definitively proving) a complete vessel occlusion. 1 The additional advantages of sonography in examining the carotid arteries are those frequently referred to: it is noninvasive, is relatively easy to use (especially in unstable patients), provides hemodynamic as well as anatomic data, and uses no ionizing radiation or intraluminal contrast.1,10
CCA occlusion is detected in 2% to 5% of the population with cerebrovascular disease.2,3,7,9,10–13 There is little information about the clinical features, neurologic symptoms, etiologies, and pathogenesis of CCA occlusion. 3 Patients who have CCA occlusion can present clinically with a wide range of symptoms, from asymptomatic to severe stroke, and CCA occlusion is not necessarily associated with cerebral infarction or even measurable brain dysfunction.3,10–12 There are many potential causes of CCA occlusion such as atherosclerosis, giant cell arteritis, fibromuscular dysplasia, thrombocytosis, dissection of the CCA or aortic arch, aortic arch aneurysm, iatrogenic occlusion, mediastinal tumors, cardiac embolism, irradiation, trauma, and idiopathic occlusion (of unknown origin).1,2,5,9–11 Of these, atherosclerosis has been shown to be the most common cause.2,3,5,9,11 In the case presented, the presence of atrial fibrillation significantly raises the likelihood of a cardiac embolus, a diagnosis supported by the sonographic evidence of thrombus throughout the vessel with very little or no evidence of any atherosclerotic disease on either side. 3
The triggering or initial site of the origin of the CCA occlusion can vary. The occlusion may begin in the proximal CCA, with antegrade propagation, or the occlusion may begin at the carotid bifurcation with retrograde propagation back to the vessel origin.3,4,7,11 The thrombus typically will extend to the level of the next large vessel reentry point.3,4,7 The prevalence of CCA occlusion tends to be slightly higher on the left side compared with the right, possibly attributable to the differences in arterial length or the direct relationship the left CCA has to the aortic arch.10–12
CCA occlusion is most frequently associated with ipsilateral ICA and/or ECA occlusion. Infrequently, patency is maintained in the distal vessels.1,9 Most often in these cases, ICA perfusion is maintained by flow from collateral circulation through proximal branches filling the ECA.1,8,13 In the least common scenario, rarely seen, retrograde flow can be found in the ICA, being perfused from cerebral circulation, and then into the ECA.1,3,8
Combining the results from the carotid duplex study and the CTA, a blood flow pathway for the case presented can be suggested. Given the complete occlusion of the right CCA, it is likely that the blood flow pathway is antegrade up the left ICA and through the carotid siphon into the anterior cerebral artery, then across the AComA and reversed through the right anterior cerebral artery, ultimately reversing through the right carotid siphon and ICA to supply the right ECA (Figure 9). A potential alternate pathway is retrograde collateral filling of the intracranial ICA via the basilar artery and PComAs7,8; however, in the case presented, the posterior circulation was seen to be noncontributory due to an apparent functionally incomplete circle of Willis, with very small or hypoplastic PComAs, forcing the remaining patent anterior circulation to be the source for collateral flow. This would also explain the severity of the patient’s symptoms and ultimate death secondary to severe stroke. With both anterior hemispheres completely dependent on flow in the left carotid artery system, any disruption of that flow such as would occur with a significant thromboembolic event—and with a right CCA occlusion, any cerebral emboli would go through the left carotid system—would severely impair flow to the entire anterior cerebral circulation.
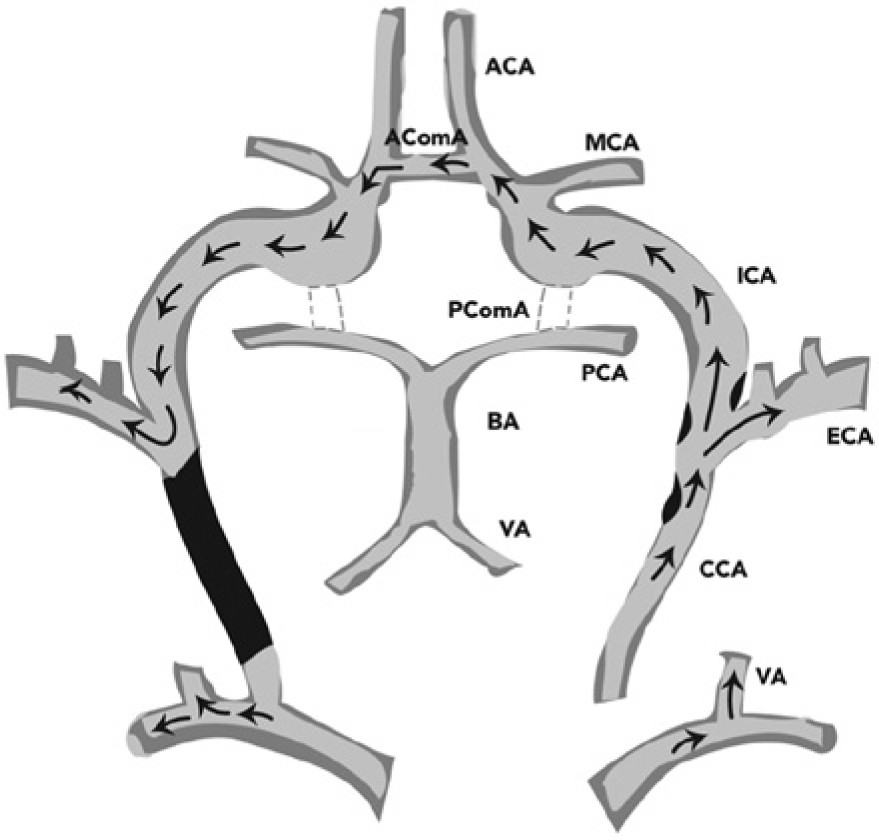
Schematic drawing of the collateral pathway for blood flow in the case presented. Coming off the aortic arch, there is antegrade flow through the left common carotid artery (CCA) and internal carotid artery (ICA) extracranially, which flows through the intracranial ICA into the left anterior cerebral artery (ACA). Blood flow crosses to the right hemisphere via the anterior communicating artery (AComA), with retrograde flows in the right ACA filling the intracranial and extracranial ICA and ultimately leading to diminished but antegrade flow in the right external carotid artery (ECA). The vertebrobasilar system could not provide collateral flows because of the functional absence of both posterior communicating artery (PComAs) (dashed lines). BA, basilar artery; MCA, middle cerebral artery; PCA, posterior cerebral artery; VA, vertebral artery.
When analyzing the vessels distal to a CCA occlusion, it is important to note not only the presence but also the direction and velocity of flow. It will be found that distal to the occlusion, the velocities of the ICA and ECA, if patent, are significantly lower compared with the patent contralateral side. 14 This does not necessarily indicate that the there is insufficient collateral flow but reflects that blood flow velocity is dependent not only on the arterial pressure but also on the degree of peripheral resistance. In the case presented, the velocity in the retrograde ICA was significantly lowered, with a peak systolic velocity of only 14 cm/s and a blunted, damped waveform. As explained by Dermitzakis et al, 7 who observed flow reversal in the contralateral ICA after endarterectomy, the blood flow to the brain from the contralateral side is unobstructed and creates a pressure difference via the collateral pathway of the circle of Willis that would be sufficient to overcome the resistance of the ipsilateral side and produce retrograde flow in the ICA, similar to a subclavian steal syndrome.11,13 Both circumstances might be considered a steal phenomenon where blood is “stolen” from the intracranial circulation to perfuse an extracranial territory. 13
With CCA occlusion being a relative rarity, treatment typically is planned on an individual case basis. 13 Most of the signs and symptoms are related to those of transient ischemic attacks (TIAs) and stroke. Depending on the source and the degree of the occlusion, appropriate management can be provided and treatment plan developed. A treatment plan may be appropriate because many of these patients are at risk for further TIAs or stroke because of associated hemodynamic disturbances, cerebral or ocular hypoperfusion, or emboli from the stump of the occlusion.7,13 It is imperative to note the patency and flow direction of the distal vessels for treatment planning, as patients who present with isolated CCA occlusion typically have a better outcome than those with an associated ICA occlusion.2,3 Depending on the circumstances, the treatment plan may vary from a surgical approach, revascularization from a bypass graft or thrombectomy, or medical management with anticoagulation.3,9
Conclusion
Sonography is a very valuable tool to assess the carotid arteries, especially due to its ability to provide functional, hemodynamic information as well as anatomic data. 13 In the event of a CCA occlusion, it is particularly important to note the patency of the more distal vessels, with attention to the analysis of any distal flow patterns.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
