Abstract
Solubility parameters are effective factors for controlling membrane preparation based on hydrophilic/hydrophobic polymers. Hansen solubility parameters (HSPs) of a polysulfone/polyethylene glycol (PSU/PEG) in increasing PEG molecular weights and binary coagulation bath of solvent/non-solvent mixtures are investigated. The results revealed that with increased PEG molecular weight in the casting solution, the relative energy difference (RED) of the solution increases, and subsequently its instability increases too. On the other hand, by increasing the solvent in the coagulation bath, RED decreases. Findings demonstrated that by increasing PEG molecular weight to 15,000g/mol and increasing solvent to 60 wt% in the coagulation bath, an interconnected sponge-like structure with maximum mean pore size (120 nm) was achieved. With increased PEG molecular weight, membrane morphology tended toward to a sponge-like structure due to delayed demixing in the phase inversion stage. Furthermore, pure water permeability and rejection capability of membranes exhibited that with increasing RED of casting solution and reducing RED of coagulation bath, the highest permeability (2199 L/m2h and the lowest rejection capability (33%) of the membrane were attained. This study confirmed that, for the PSU/PEG system, by controlling the solubility parameters, RED value of casting solution and coagulation bath, membrane characteristics can be controlled.
Keywords
Introduction
Several outstanding properties such as blood and biocompatibility, high thermal stability, pH resistance and sterilization capability, make polysulfone (PSU) a favorite polymer for preparing membranes used in biomedical and water purification applications.1–3 Despite multiple polar aromatic rings, PSU is a hydrophobic polymer. Therefore using just PSU in membrane preparation may produce problems such as limited permeability, insufficient fouling resistance, and diminished lifetime. 4 Adding hydrophilic additives such as polyvinylpyrrolidone (PVP) and PEG is a simple and commercial way to improve PSU membrane hydrophilicity.5,6 Moreover, further hydrophilicity and pore former roles, PVP and PEG together with some materials like dextran, bovine serum albumin (BSA), etc. are common solutes in the membrane industry to examine rejection capability and molecular weight cut-off.7–9 The properties and performance of the membranes are highly dependent upon various parameters related to the casting solution and coagulation bath during the phase inversion process.10–12 The thermodynamics of the casting solution is regarded as a determinative factor for the stability of polymeric solutions.13,14 Several parameters such as main polymer, solvent, non-solvent, additives, and ambient conditions, can influence the casting solution characteristics due to the change in thermodynamics of solutions.15–17 Furthermore, thermodynamics and kinetics of phase inversion are well-known phenomena that have impressive effects on the formation of membrane microstructure and other properties including mean pore size and porosity. Both polymeric solution and coagulation bath are effective factors controlling the thermodynamics and kinetics of phase-inversion process. Further to this traditional prospect, other main and basic approach such as solubility parameters are effective factors in predicting final formed membrane characteristics. 18 Hildebrand and Hansen solubility parameters have a strong relationship with the development of stable chemical solutions. 19
Hansen solubility parameters due to the differentiation of solubility parts such as disperse (

The RED number is used as a parameter to evaluate the compatibility of two materials. 20 If RED is lower than 1, it means the solvent could dissolve the polymer, otherwise, it could not dissolve it. When RED declines to 0, it means that the solvent had a great potential for solubility of defined material. 21 Hansen solubility parameters and RED are valuable parameters for evaluating casting solutions and coagulation baths for membrane preparation via the phase inversion method.
Unlike some polymers such as PVP that are compatible with PSU in each composition and molecular weight, PEG in some molecular weights and concentrations can expose instability behavior when blended with PSU. This instability can affect the phase-inversion process and the characteristics and performance of the final formed membrane. Idris et al. 22 studied how PEG molecular weight affects the polyethersulfone membrane. They showed that with increasing PEG molecular weight, the membrane water permeability would increase due to forming larger pores in the membrane structure. Chakrabarty and coworkers 23 showed that with high molecular weight PEG in the PSU membrane, water permeability, pore size, and porosity would increase. Kim and Lee 24 demonstrated that PEG has a pore former role in the PSU membrane. They showed that this behavior correlates with the changes in thermodynamics and kinetics in the phase inversion process. Ma et al. 25 showed that as the PEG molecular weight; porosity, water permeability, and the PSU-based membrane hydrophilicity would increase effectively.
There are limited reports evaluating the instability of PSU/PEG systems at high PEG molecular weights and this study is one of the few scientific works to assess why the PSU/PEG blend is unstable in some molecular weights toward the membrane preparation process. Herein, Hansen solubility parameters and RED of PSU/PEG at different molecular weights of PEG were estimated and their effects on the properties of the final formed membrane were investigated. In the current study, further solubility parameters of PSU/PEG casting solutions; solubility parameters and RED of coagulation bath at different compositions were examined.
Experimental
Materials
Polysulfone (Ultrason S6010) was supplied by the BASF Company (Germany). Polyethylene glycol (PEG) at 3,000, 15,000, and 35,000g/mol, Dimethylacetamide (DMAc), and all other solvents; Tetrahydrofuran, Dimethyl sulfoxide, Dimethyl formamide, Dichloromethane, Acetone, Methyl-2-pyrrolidone, Cyclohexanol, 1-Butanol, Methanol, Butyl acetate, Chloroform, Methyl ethyl ketone, 1,4-Dioxane, Ethylene dichloride, Ethyl acetate, Chlorobenzene, Ethanol, Carbone tetrachloride, Propylene carbonate, Benzene, Hexane, Toluene, Nitroethane, Diethylene glycol, Diacetone alcohol, Trichloroethylene, Acetophenone, Isophorone, and Ethanol amine used to determine polymeric blend solubility were obtained from Merck Co. (Germany). Polyvinylpyrrolidone (PVP) at molecular weights included PVP-K17 (10,000g/mol), PVP-K30 (50,000g/mol), and PVP-K90 (1,200,000g/mol) for the rejection experiment assay were purchased from Rahavard Taamin Co. (Iran), plus PVP with 360,000g/mol supplied from VWR Co. (USA).
Membrane preparation
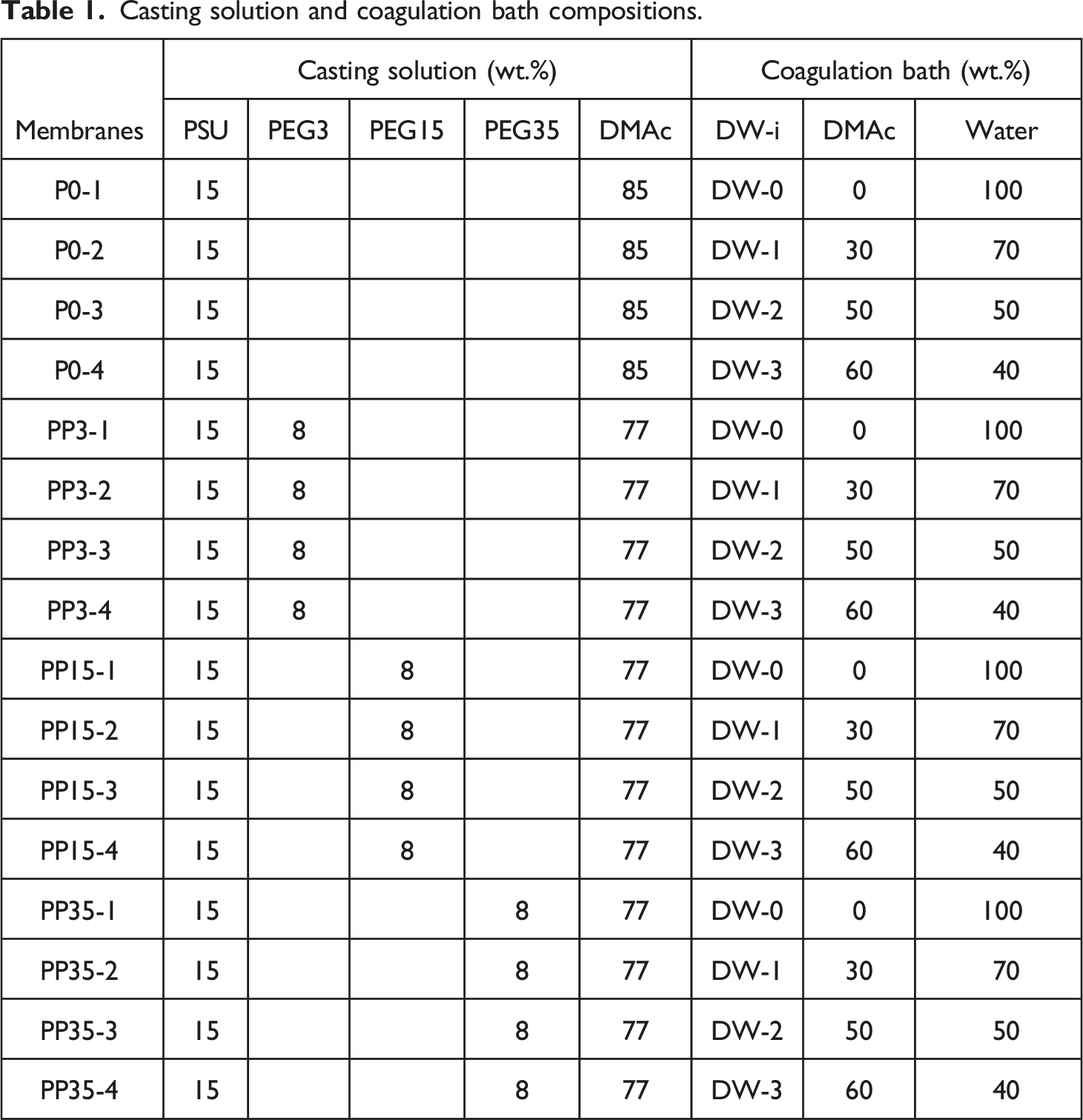
Casting solution and coagulation bath compositions.
Determination of hansen solubility parameters
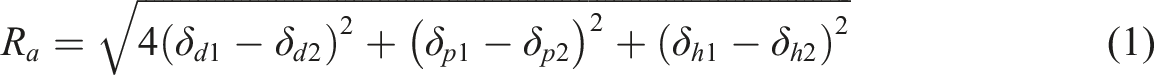




After determining
Membrane water flux


Membrane morphology
Membrane cross-section morphology was observed by a scanning electron microscope (SEM) VEGA TESCAN instrument (Czech Republic) at 1 kx and 5 kx magnifications and 20 kV device voltage. To achieve a defect-less cross-section, membranes were broken in liquid nitrogen and fixed on a holder. The gold sputtering process was employed for the formation of a thin layer of gold on the samples and finally, membrane morphology was observed by the instrument.
Rejection experiment


Membrane mean pore size
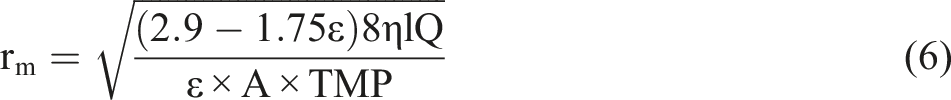

Results and discussion
Evaluation of solubility parameters
Solubility parameters (
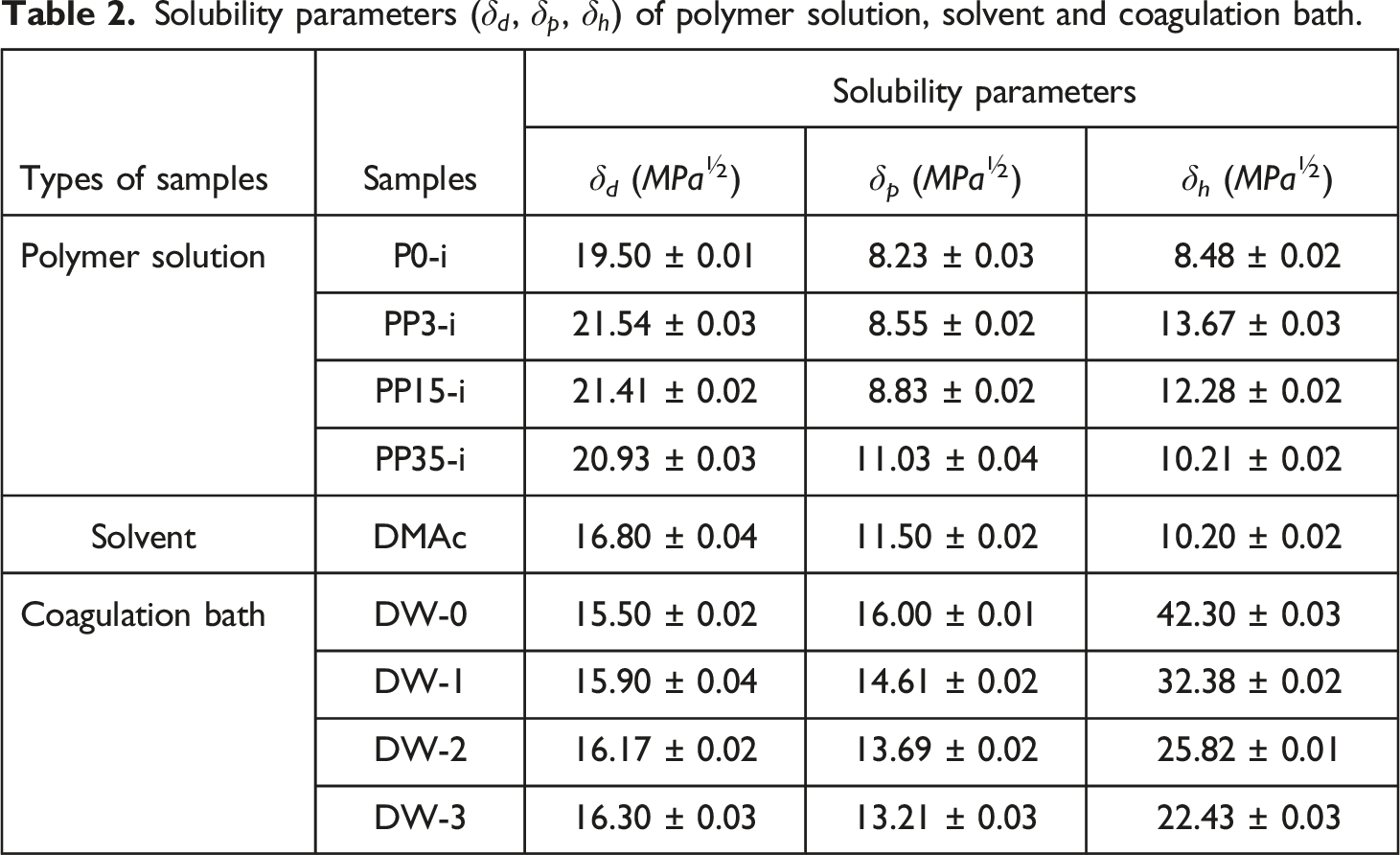
After determining the solubility index, the Hansen sphere of the polymeric blend (PSU/PEG) was drawn, then R0 and solubility parameters for each polymeric system were measured. As represented in Figure 1 and Table 3 by introducing PEG 3000 in the casting solution, both R0 and Ra increased. R0 increased from 8.03 for nascent PSU (P0) to 10.52 for PP3, and Ra increased from 6.54 for P0 to 10.51 for PP3. The results confirmed that R0 decreases as the PEG molecular weight increases. R0 decreased from 10.52 for PP3 to 9.81 for PP15 and to 7.61 for PP35. On the other hand, Ra decreased from 10.51 for PP3 to 9.82 for PP15 and to 8.27 for PP35. At first sight, it seems that as Ra decreases, the PEG and PSU miscibility should be better at a higher molecular PEG weight. But RED is a more accurate parameter for the prediction and evaluation of solution behaviors. Hansen solubility sphere of polymeric blends: (a) PP3, (b) PP15, (c) PP35, and (d) overall diagram of PP3, PP15, and PP35. Solubility parameters, Ra and RED of different coagulation bath. *R0 is the polymer (P0-i, PP3-i, PP15-i, and PP35-i) Hansen sphere.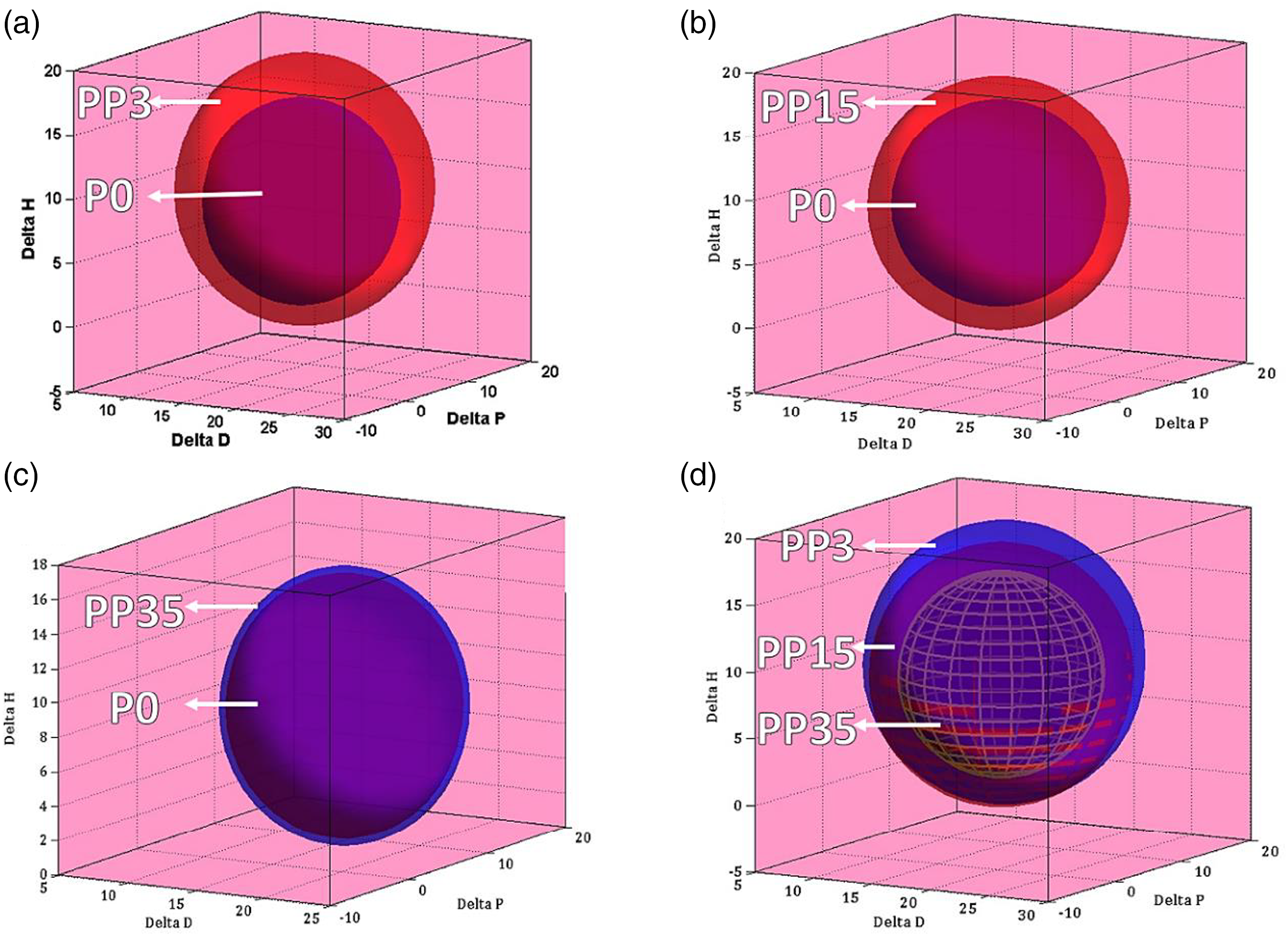
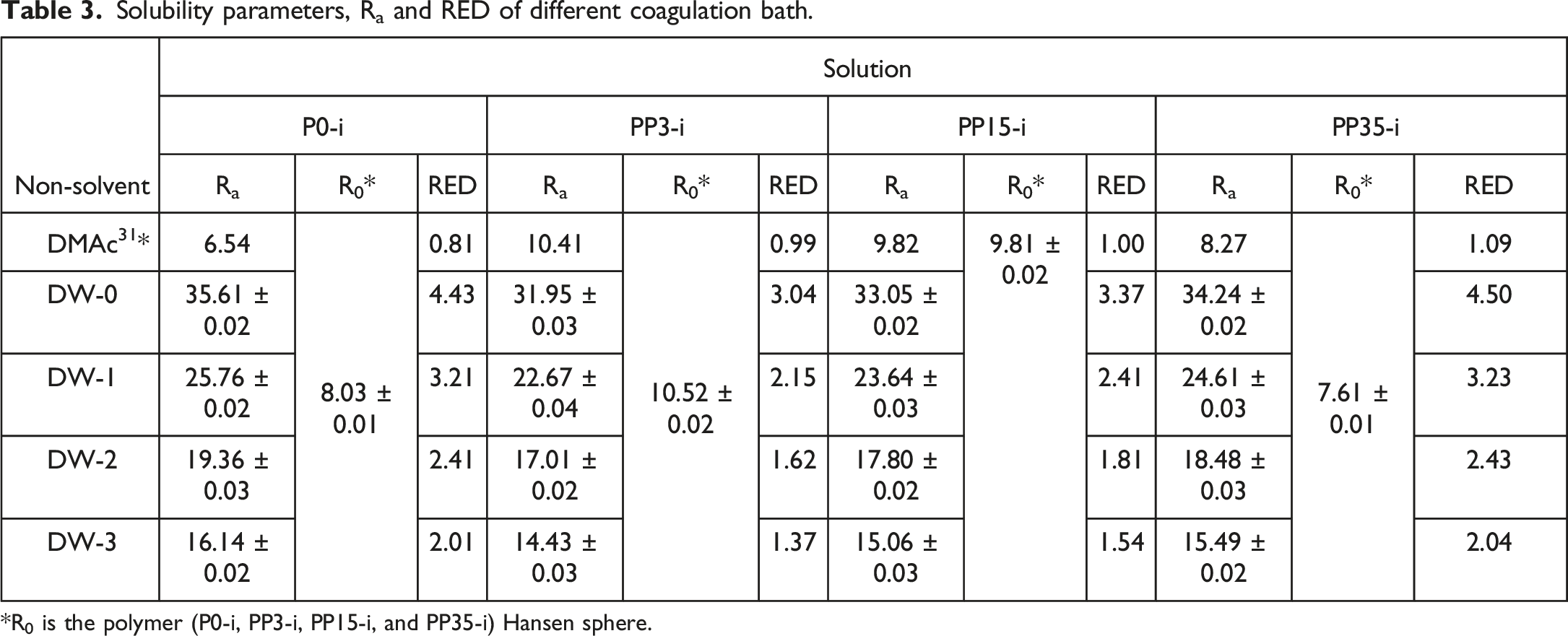
Table 3 presents the R0, Ra and RED. As reported in this table, as the PEG molecular weight increased from PP3 to PP35, the casting solution RED increased from 0.99 for PP3 to 1.00 and 1.09 for PP15 and PP35, demonstrating less casting solution stability at higher molecular weights of PEG. Due to the presence of hydrophilic moieties such as ether and hydroxyl groups, PEG is a hydrophilic polymer.
30
By comparison with P0, both polar interactions including polar-polar interaction, and hydrogen bonding interaction were increased for PP3, PP15, and PP35. While, as the PEG molecular weight increased, the number of –OH end groups would decrease. Therefore, solubility parameters of hydrogen bonding decreased as the PEG molecular weight increased. Indeed, as PEG molecular weight increased, the polymeric solution stability would significantly decrease. Then, in such a condition, an insignificant trigger turns the casting solution into an unstable condition, and consequently, phase separation may take place in the casting solution. As PEG molecular weight increases, the polymer chain ether groups increases, therefore polar part of solubility parameters can be increased. Table 3 presents the calculated Hansen solubility parameters (
The coagulation bath conditions are an important stage in the membrane preparation process.32,33 Water (DW-0) with a 4.43 RED is known as the strongest non-solvent for PSU membrane preparation.
34
According to Table 2, water has the highest value of polar part and hydrogen bonding of HSP. DMAc used as a common solvent for the preparation of PSU membrane, has a RED of 0.81.
35
Therefore, by considering the power of the coagulation bath based on both solvent (DMAc) and non-solvent (water) RED, it is expected that it is possible to adjust the coagulation bath power. Table 3 and Figure 2 represent with the addition of DMAc in the coagulation bath, RED was decreased effectively so that for P0-i this value decreased from 4.43 when only pure water (DW-0) was used as non-solvent, to 3.21 (DW-1), 2.41 (DW-2), and 2.01 (DW-3). Also, by increasing DMAc amounts in the coagulation bath, polar-polar and hydrogen bonding interactions decreased. The effect of coagulation bath composition on the RED value.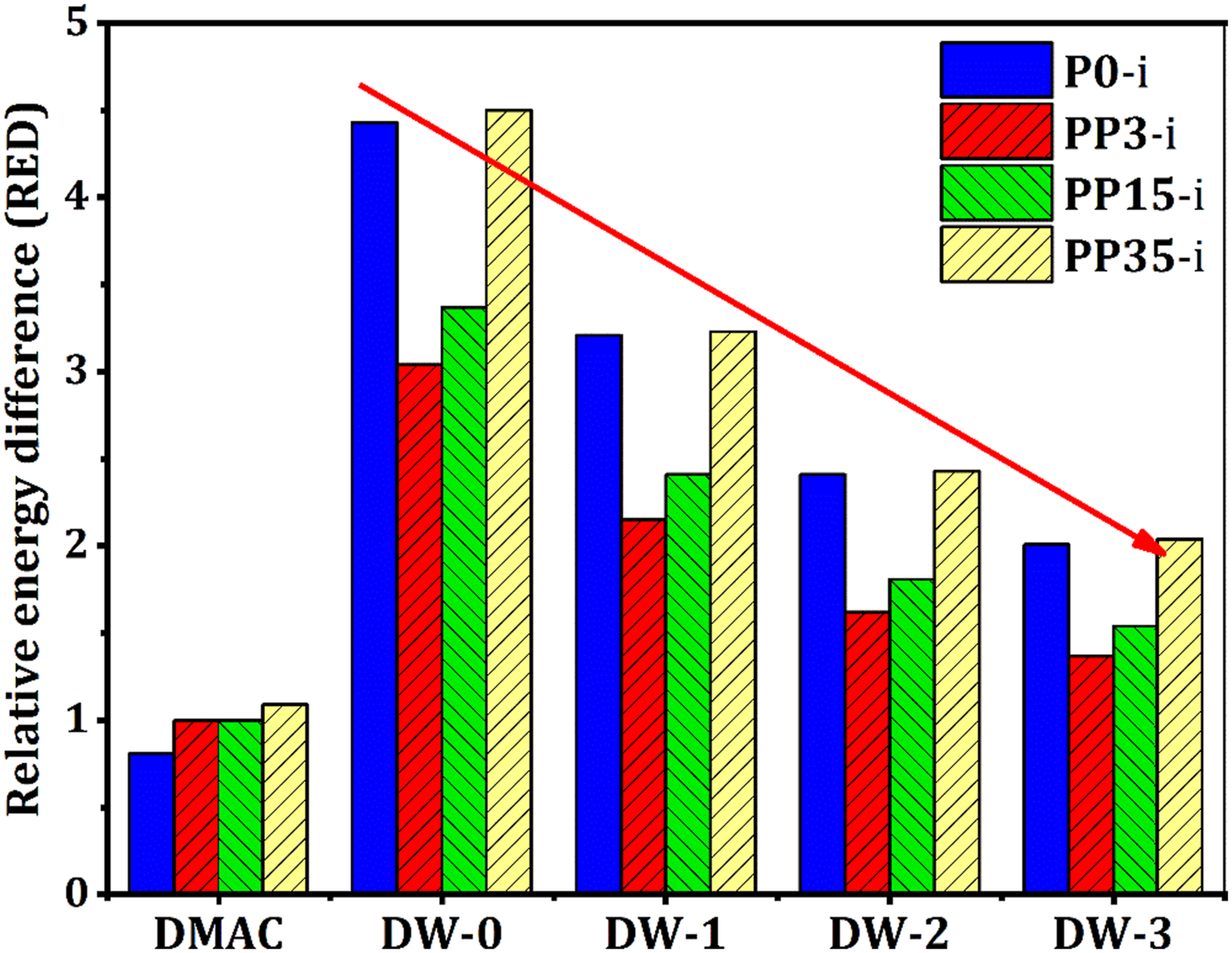
A decrease in RED value of the coagulation bath leads to a reduced coagulation bath power and consequently a slower solvent/non-solvent de-mixing in the phase inversion stage. Thus, it is believed that Hansen solubility parameters and the RED index have a strong effect on how the membrane forms due to the change in kinetic phase inversion. Based on the obtained results, for PP3-i, adding DMAc in the coagulation bath led to decreased RED from 4.43 for DW-0 to 3.21 for DW-1 and 2.01 for DW-3. This trend was revealed for PP15-i and PP35-i (Figure 2 and Table 3). Therefore, by considering the solubility parameters and RED of casting solutions and binary mixture of coagulation baths, controlling and predicting the final formed membrane characteristics is possible.
Membrane morphology
Several parameters can influence the membrane morphology and its performance.36,37 Casting solution composition and coagulation bath are two main causes that have a severe effect on the final formed morphology.38,39 Previous studies reported that adding PEG resulted in forming larger pores in the PSU membrane structure.25,40 These works demonstrated that the membrane morphology is strongly related to the casting solution and coagulation bath solubility parameters. Figure 3 shows that as the PEG molecular weight increases in the casting solution, the membrane morphology changed to a sponge structure. This change in morphology is attributed to the solution viscosity, the solubility parameter differences, and the RED. The membrane morphology greatly depends on the preparation condition.
31
Figure 3 also shows that as the DMAc in the coagulation bath increases, the membrane structure changes to a semi-sponge or full-sponge structure. As mentioned previously, as the DMAc concentration in the coagulation bath increases, its RED decreased from 4.43 to 2.01 for P0-i, 3.04 to 1.37 for PP3-i, 3.37 to 1.54 for PP15-i, and 4.50 to 2.04 for PP35-i. As the RED for the coagulation bath decreased, non-solvent power and subsequently the speed of the phase inversion process decreased. In such a condition, due to the nucleation and growth mechanism, membrane morphology turned to a sponge structure with a decrease in top layer thickness. For P0-i, morphology changed from a channel-like to a finger-like structure in various coagulation baths (Figures 3(a)–(d)). As well as, for the PP3-i (Figures 3(e)–(h)), and PP15-i (Figures 3(i)–(l)) membrane series, the channel-like structure turned to the finger-like structure. One considerable issue is that when DW-2 and DW-3 used as coagulation bath, sponge regions between channels and fingers walls were extended which proved the nucleation and growth mechanism was the factor controlling the membrane morphology. For PP35-i, the membrane structure changed from a semi-sponge structure to a full-sponge structure at the higher DMAc levels in the coagulation bath (Figures 3(m)–(p)). These results determined that solubility parameters and RED of the coagulation bath significantly affect the final membrane structure. Thus, one can conclude that by knowing the solubility parameters and RED of the coagulation bath, the predicting the membrane morphology is possible. On the other hand, increasing PEG molecular weight has the same effect as increasing the DMAc solvent in the coagulation bath. The casting solution viscosity is an important factor in how the membrane morphology is formed. The viscosity for the four casting solutions was: • P0-i, 7,300 mPa.s • PP3-i, 12,700 mPa.s, • PP15-i, 18,600 mPa.s • PP35-i, 30,300 mPa.s. The effect of PEG molecular weight (P0-i (a)-(d), PP3-i (e)-(h), PP15-i (i-l), PP35-i (m-p)), and coagulation bath composition (a, e, i, m (DW-0), b, f, j, n (DW-1), c, g, k, o (DW-2), d, h, l, p (DW-3)) on the formation of membranes morphology.

Indeed, as the PEG molecular weight increases, leads to more polymeric chain entanglement. Therefore, solvent/non-solvent exchange takes longer, thus, membrane morphology would extend with nucleation and growth mechanism, 41 and the sponge structure would develop.
Membrane mean pore size
The membrane mean pore size estimates indicate that solubility parameters and RED have a significant effect on pore size. Higher RED relates to higher casting solution instability, thus, PEG has a non-solvent-like behavior. As casting solution stability decreases, the PSU and PEG chains interaction decreases, and in the phase inversion stage, PEG tends to leave the PSU chain and migrate into the coagulation bath. Therefore, PEG at higher molecular weight due to the higher hydrodynamic radius can form larger pores in the membrane structure. Figure 4 shows that for the non-solvent DW-0, the mean pore size increased from 28 nm for P0-1 to 51 nm for PP3-1, and to 80 nm for PP15-1. While for PP35-1, a lower 70 nm mean pore size was achieved due to a more packed sponge-like structure versus PP15. This mean pore size response was also observed when DW-1, DW-2, and DW-3 were used as non-solvent. The Hansen Solubility Parameter (HSP) shows that water has high hydrogen bonding and polarity. Thus when adding DMAc, the polar part decreased slightly, hydrogen bonding decreased tangibly, and the disperse part increased. Therefore, the non-solvent interaction with the polymeric part would increase, and consequently, a delayed phase inversion process would occur. Figure 4 shows that the P0-i mean pore size increased from 26 nm for the non-solvent DW-0 to 33 nm for DW-1, to 36 nm for DW-2, and to 38 nm for DW-3. With increasing DMAc amounts (DW-0 to DW-3) in the coagulation bath and subsequently decreasing of RED value, mean pore size was increased to 51 nm, 55 nm, 65 nm, and 69 nm for PP3-i series, 80 nm, 82 nm, 98 nm, and 118 nm for PP15-i series, 70 nm, 72 nm, 75 nm, and 77 nm for PP35-i series. This data shows that by reducing coagulation bath RED, the final formed membrane mean pore size would increase. Indeed, as the coagulation bath RED decreases, phase inversion duration takes more time, and then a symmetric membrane with larger pores and a thin skin layer is obtained. However, based on the acquired results, the larger mean pore size can be achieved when a coagulation bath with a low RED value and a casting solution with a high RED value (PEG middle molecular weight) are used simultaneously. Mean pore size of membranes.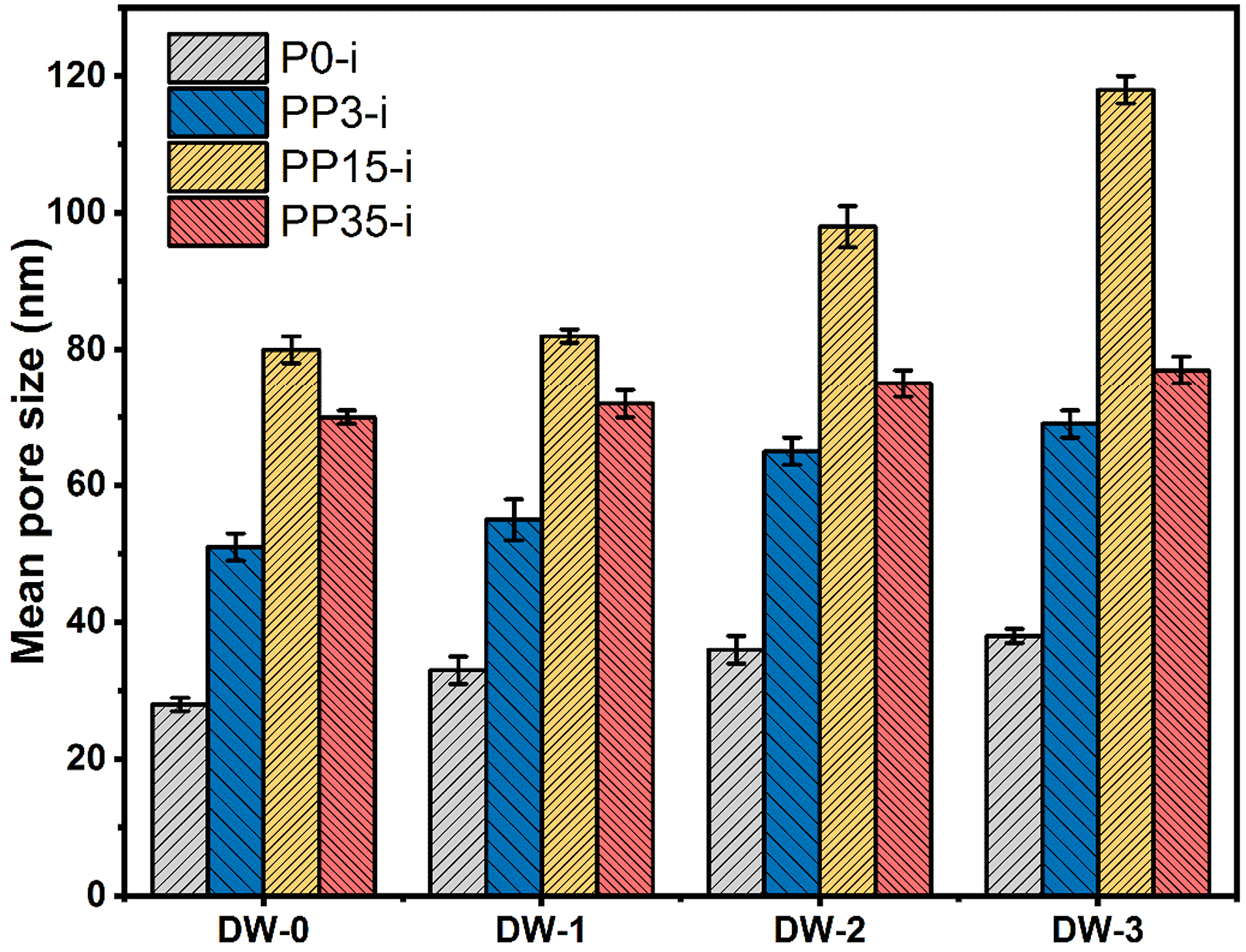
Pure water permeability
Pure water permeability is related to several factors; mean pore size, porosity, thickness, morphology, and hydrophilicity.42,43 Adding PEG greatly effects the membrane flux. Figure 5 shows that adding PEG3,000 increases the water flux from 229 L/m2h for P0-1 to 637 L/m2h for PP3-1. Also, at 15,000g/mol PEG molecular weight the water permeability increased to 1414 L/m2h for PP15-1. For PP35-1, water permeability decreased to 1045 L/m2h. It seems all PEG grades have a role as pore former in the membrane. With increasing PEG molecular weight from 3000 to 15,000, due to the increase in PEG hydrodynamic radius, the PEG removal from the polymeric film in the phase inversion process produces a larger mean pore size. While about PEG35,000, due to more PEG chain entanglement with the PSU matrix, removing PEG from the cast film is slower than for PP15-i, therefore the obtained mean pore size is smaller than for the PP15-i membranes. As Figure 5 shows, the maximum water permeability was achieved for PP15-i. Pure water permeability of membranes.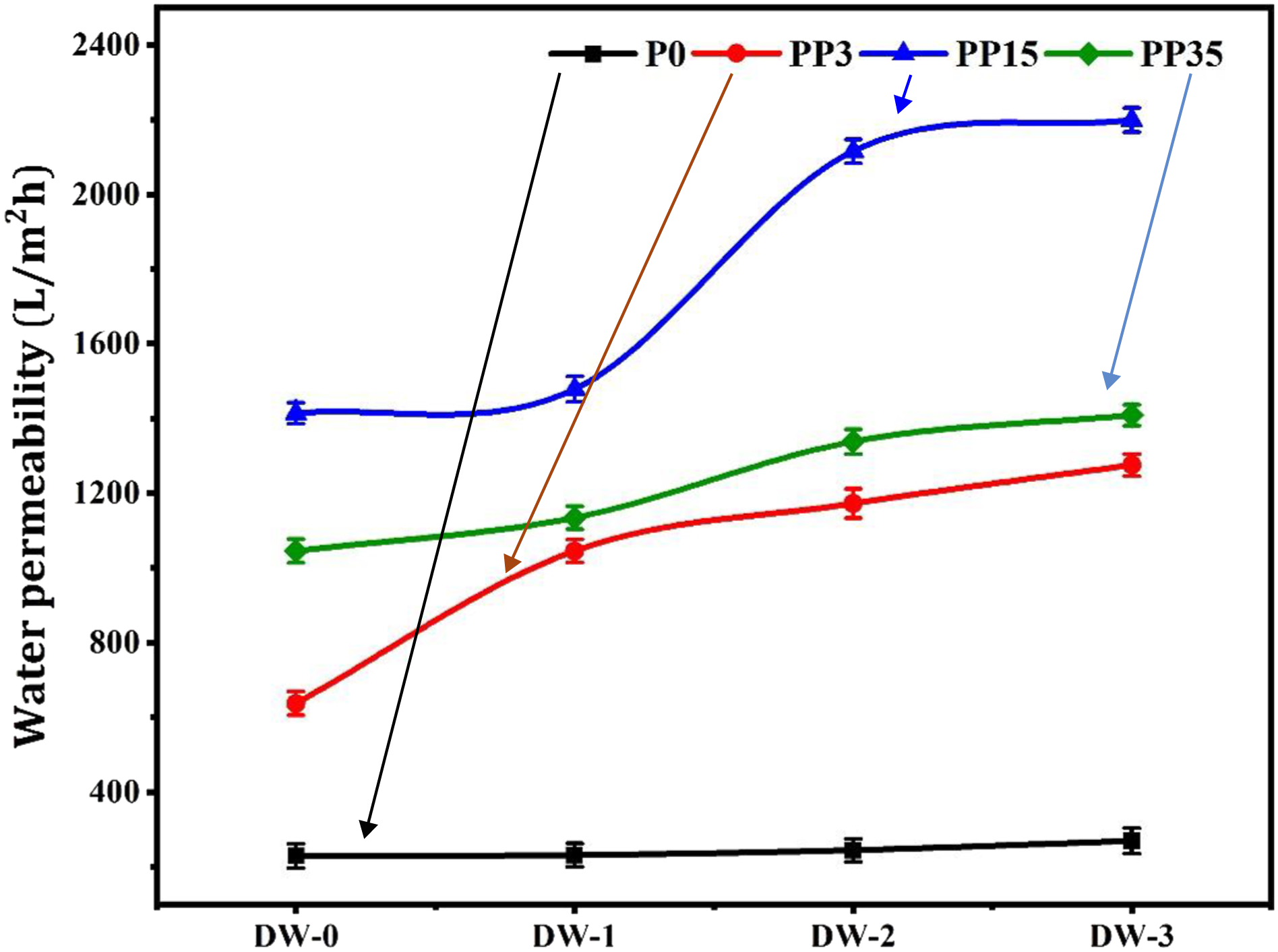
Also how the coagulation bath composition affects the water permeability of membranes was evaluated. The results showed that for reduced RED parameter in the coagulation bath, the water permeability increased. Figure 5 shows the maximum water permeability was reached with 60 wt% DMAc in the coagulation bath (DW-3), wherein its RED parameter was 2.41 when membranes were prepared from P0. Moreover, for other membranes that have PEG in various molecular weights, the maximum water permeability was when DW-3 was used as a coagulation bath. For example, for PP15-4, water permeability was 2199 L/m2h. These results confirmed that with a decreased coagulation bath RED leads to forming a more porous membrane with desired interconnectivity, resulting in a higher water flux.
Rejection experiment
A membrane rejection capability is strongly related to its characteristics including mean pore size and morphology.
44
As revealed in previous sections, as the casting solution RED increases and the coagulation bath RED decreases, a membrane with a larger pore size is obtained. The rejection experiment shows that membrane separation capability is greatly dependent on the membrane morphology which is affected by the casting solution characteristics. This current work demonstrates that the solubility parameters of the polymeric solution and even the coagulation bath significantly affect the final membrane formation. To evaluate the membrane rejection capability, an aqueous PVP solution with molecular weights of 10 kDa, 50 kDa, 360 kDa, and 1200 kDa was used as solute. Figure 6(a) shows that for P0-1 more than 90% solute rejection with 360 kDa and 1200 kDa is obtained. With 50 kDa and 10 kDa the rejection capability is lower than 80% and 70%. With decreasing coagulation bath RED and larger mean pore size, the rejection capability decreased. For other membranes (PP3-i, PP15-i, and PP35-i) similar behavior is observed while the overall rejection capability of PP15-i is lower than PP3-i and PP35-i (Figure 6(b)–(d)). Indeed, for PP15-i due to the larger mean pore size, rejection capability is lower than the others and the lowest rejection was for PP15-4. As mentioned previously, the high casting solution RED and low coagulation bath RED led to forming a larger mean pore size. Rejection results of membranes; (a) P0-i, (b) PP3-i, (c) PP15-i, and (d) PP35-i.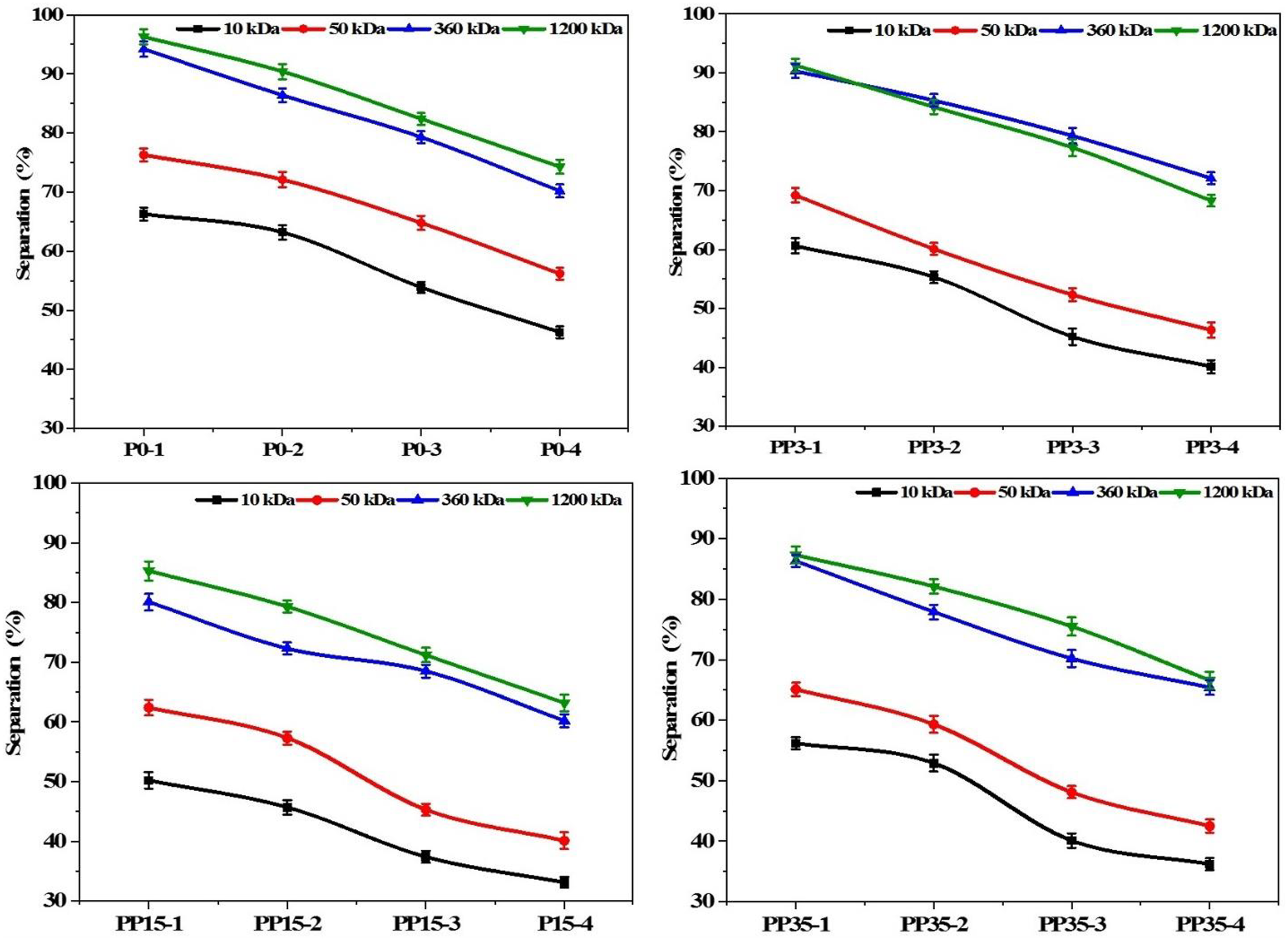
The Einstein-Stocks relation
45
(equation (7)) relates the average molecular weight of each PVP to its particle size. The calculated particle diameters are: • PVP 10 kDa 3.96 nm • PVP 50 kDa 10.28 nm • PVP 360 kDa 33.13 nm • PVP 1200 kDa 67.65 nm.
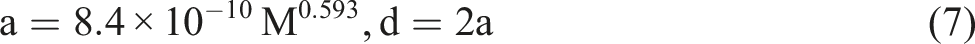

From Figure 6 and equation (7), one can conclude that PP3-1 had over 90% rejection for particle diameters smaller than 67.65 nm. Moreover, this membrane had more than 60% rejection for 10.28 nm diameter particles. As mentioned above, by increasing the coagulation bath RED, a larger mean pore size membrane was obtained for PP3-2 to PP3-4. For PP3-4 more than 74% rejection was obtained for 67.65 nm diameter particles, and 41% for 10.28 nm diameter particles. For PP15-i membranes due to the larger mean pore size, rejection capability was poorer than the PP3-i series. However, for PP15-1, 85% rejection was achieved for 67.65 nm particles and less than 50% rejection for 10.28 nm particles. For PP15-4 due to the larger pores, rejection capability was poorer than others; the maximum 67% separation was achieved for 67.65 nm particles and for less than 33% 10.28 nm particles. But for PP35-i with its smaller mean pore size versus the PP15-i series, rejection capability was more than the PP15-i and less than the PP3-i series. For PP35-1, 86% rejection was achieved for 67.65 nm particles, while for 10.28 nm particles, 55% rejection was achieved. For PP35-4 with a larger mean pore size, 72% rejection was achieved for 67.65 nm particles and 40% rejection for 10.28 nm particles. Therefore, one concludes that the highest water permeability and the lowest rejection capability were obtained for the PP15-i series, and highest the rejection was achieved for PP3-i and then the PP35-i series.
Conclusion
Thermodynamics is an important parameter in the phase inversion process during membrane preparation. How the membrane formation relates to the solubility parameters was discussed in detail. Indeed, solubility parameters are considerable factors that are hidden in the thermodynamic phenomenon which can control the membrane formation process. The casting solution stability impacts the membrane formation with its specific structure. Unlike some hydrophilic additives such as PVP, adding PEG to the PSU casting solution makes it less stable and even unstable. The PEG molecular weight and concentration impact this issue. In this study, Hansen solubility parameters of PSU/PEG blends were evaluated and correlated with increasing PEG molecular weight in casting solution, Ra and R0 of polymeric systems changed from 6.54 to 10.51 for Ra, and 8.03 to 10.52 for R0. It is shown that increasing the PEG molecular weight from 3000 to 35,000g/mol, the casting solution stability decreased impressively. Increasing PEG molecular weight from 3000g/mol to 15,000g/mol, the membrane mean pore size increased from 51 to 80 nm, but by more enhancement in the molecular weight, due to more physical polymeric chain entanglement, a membrane with dense microstructure and therefore smaller mean pore size till 70 nm was obtained. In addition, a DMAc/Water solvent/non-solvent mixture was used as a coagulation bath and its composition was evaluated based on the Hansen solubility parameters and RED index. The results showed that as the RED increased from 0.81 to 1.09 in the casting solution that instability was increased, while for coagulation bath RED was decreased from DW-0 to DW-3 for each polymeric system; 4.43 to 2.01 for P0-i, 3.04 to 1.37 for PP3-i. 3.37 to 1.54 for PP15-i, and 4.50 to 2.04 for PP35-i. The results revealed that with these properties of casting solution and coagulation bath, adjusting the membrane with an approximately symmetric structure with open pores would possible. Therefore, the Hansen solubility parameters, and relative energy difference (RED) of casting solution and coagulation bath correlate with the membrane characteristics such as mean pore size, morphology, and water flux.
Footnotes
Acknowledgments
This work is based upon research funded by Iran National Science Foundation (INSF) under project No. 4001244.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is based upon research funded by Iran National Science Foundation (INSF) under project No. 4001244.
