Abstract
Background:
Inpatient use of intravenous iron has been increasing. Ferric gluconate is traditionally given once daily. Twice-daily dosing provides faster iron repletion, but there are limited data to support the safety of twice-daily dosing.
Objective:
The aim of this study was to investigate the safety of twice-daily dosing for ferric gluconate compared with daily dosing.
Methods:
This was an institutional review board-approved retrospective observational study of hospitalized adult patients who received intravenous ferric gluconate 250 mg daily or twice daily between January 1 and April 3, 2022. The primary composite safety outcome included hypotension, infusion reaction, rapid response alert, or escalation in level of care. Secondary outcomes included total amount of iron received, hospital length of stay, and changes in laboratory values.
Results:
A total of 126 patients were included in this study, with 63 patients in each group. The primary outcome occurred in 29 patients (46%) in the twice-daily group compared with 36 patients (57.1%) in the daily group (relative risk = 0.81; 95% CI, 0.57-1.13;
Conclusions:
In this retrospective single-center study of hospitalized adult patients, those who received intravenous ferric gluconate twice daily did not experience an increased rate of a composite safety outcome of hypotension, infusion reactions, or escalation in level of care compared with those with daily dosing.
Introduction
With increased drug costs and the desire to curb drug expenditures in inpatient pharmacy, trends in medication use are constantly being evaluated. The inpatient use of intravenous iron products has increased in recent years for several reasons, including updated recommendations for use in heart failure and continued need in patients with chronic kidney disease, malabsorption conditions, and profound iron-deficiency anemia.1 -3 Regardless of the indication for intravenous iron, there are several formulation options. Selection of an intravenous iron formulation can be based on adverse effects, dosing regimen, availability, and cost. Iron dextran and ferric carboxymaltose often can be given as a one-time infusion, whereas iron sucrose, ferumoxytol, and ferric gluconate require multiple smaller doses for full repletion.
Another consideration in the selection of intravenous iron formulation is the adverse-effect profile of each formulation. Administration of various forms of intravenous iron can lead to hypotension, infusion-related reactions, kidney injury, and (rarely) anaphylaxis. 4 Iron dextran in particular has been associated with a risk of anaphylactic reaction in its older, larger-molecular-weight formulation. 4 From 1992 to 1996, ferric gluconate had a reported rate of 3.3 allergy episodes per million doses per year, whereas iron dextran had a rate of 8.7 allergy events per million doses per year in 1995. 5 Newer formulations of iron dextran, as well as other formulations of intravenous iron, are often better tolerated. A meta-analysis of 97 trials reviewed intravenous iron for all indications and found no increase in serious adverse effects associated with intravenous iron and a decreased incidence of gastrointestinal adverse effects compared with control formulations (eg, oral iron, no iron, placebo, or intramuscular iron). There was, however, a higher rate of infusion reactions in the intravenous iron group. 6 A study comparing ferric gluconate with placebo in hemodialysis patients found the rates of total adverse events and serious adverse events to be similar between the 2 groups. The adverse effects of ferric gluconate also were much less severe in nature than those of iron dextran in this study. 7 Nevertheless, safety is always a consideration when administering intravenous iron regardless of the formulation.
At this large academic medical center, the 2 formulations of intravenous iron on the adult formulary are iron dextran and ferric gluconate. Iron dextran is recommended if rapid repletion is needed, whereas ferric gluconate is the preferred agent for most patients due to its lower cost. Although the Food and Drug Administration-approved dose of ferric gluconate is 125 mg, the maximum dose used in practice at this institution is 250 mg with a frequency of daily or twice daily. A study of 123 adult patients with heart failure found that ferric gluconate dosed at 250 mg daily was associated with a low frequency of adverse reactions, thus supporting the 250-mg dose. 8
A prospective single-arm open-label study of 13 class III or class IV heart failure patients receiving intravenous ferric gluconate twice daily showed that this dosing regimen allowed for an increase in hemoglobin without significant hemodynamic changes. The most common adverse effects reported in this study were gastrointestinal in nature (ie, nausea, constipation, and abdominal discomfort occurred in 3 patients each), with reports of injection-site reactions in 5 patients. 9 Although there are limited data to support the 250-mg twice-daily regimen, this dosing strategy may be preferred to help decrease hospital length of stay, particularly for patients whose primary barrier to discharge is iron repletion. The aim of this study was to further investigate the safety of twice-daily dosing for ferric gluconate compared with daily dosing.
Methods
This institutional review board-approved retrospective study included adult patients admitted to a large academic medical center who received intravenous ferric gluconate between January 1 and April 3, 2022. All patients who were 18 years of age or older, admitted to the hospital, and receiving ferric gluconate during this time frame were included regardless of their indication for intravenous iron supplementation. Patients were excluded if their ferric gluconate frequency differed from once or twice daily, if they received only 1 dose, or if their individual doses were <250 mg.
The primary composite safety outcome included hypotension, the use of medications to treat anaphylaxis, infusion reactions as noted in the patient’s chart, or transfer to an intensive care unit. Hypotension was defined as a decrease in mean arterial pressure of 10 mm Hg or more from pre-infusion to post-infusion blood pressure. The medications used to treat anaphylaxis or infusion reactions at this institution include epinephrine, diphenhydramine, albuterol, hydrocortisone, and methylprednisolone. Secondary outcomes included changes in ferritin, hemoglobin, transferrin saturation, and transferrin concentrations from baseline to after administration of all ferric gluconate doses; total amount of ferric gluconate received; and hospital length of stay.
Patients were identified via a search using Tableau data visualization software. Primary and secondary outcomes were extracted from the electronic medical records. Infusion reactions were identified via manual chart review of the notes in the electronic medical records and included a review of the allergies documented in the patient’s chart. Blood pressures within 3 hours prior to each dose of ferric gluconate were compared with blood pressures within 3 hours after the same dose. Laboratory values prior to the administration of ferric gluconate were considered as baseline values. The post-iron laboratory values were from the first available draw after the final dose of ferric gluconate. Microsoft Excel and SPSS version 29 were used for data collection and statistical analysis. The primary composite outcome, hypotension, and use of the anaphylaxis kit were compared using
Results
A total of 212 patients were identified for inclusion consideration. Seventy-six patients were excluded due to a frequency other than daily, twice daily, every 12 hours, or every 24 hours. Ten patients were excluded because their doses were <250 mg. After exclusion, each group consisted of 63 patients (Figure 1). At baseline, the 2 groups were similar in weight and race (Table 1). The twice-daily group was older than the daily group (median age 66 vs 53 years) and had a lower percentage of males (40% vs 61.9%). Fewer patients in the twice-daily group had ICD-10 codes for heart failure (44.4% vs 63.5%) and chronic kidney disease (12.7% vs 39.7%) compared with the daily group.

Patient selection. Patients were identified via Tableau data visualization software and excluded based on frequency other than daily or twice daily or a dose <250 mg.
Baseline characteristics.

IQR, interquartile range
The primary composite safety outcome occurred less frequently in the twice-daily group compared with the daily group (29 [46.0%] vs 36 [57.1%]); however, this was not a statistically significant difference (relative risk [RR] = 0.81; 95% CI, 0.57-1.13;
Primary outcomes.
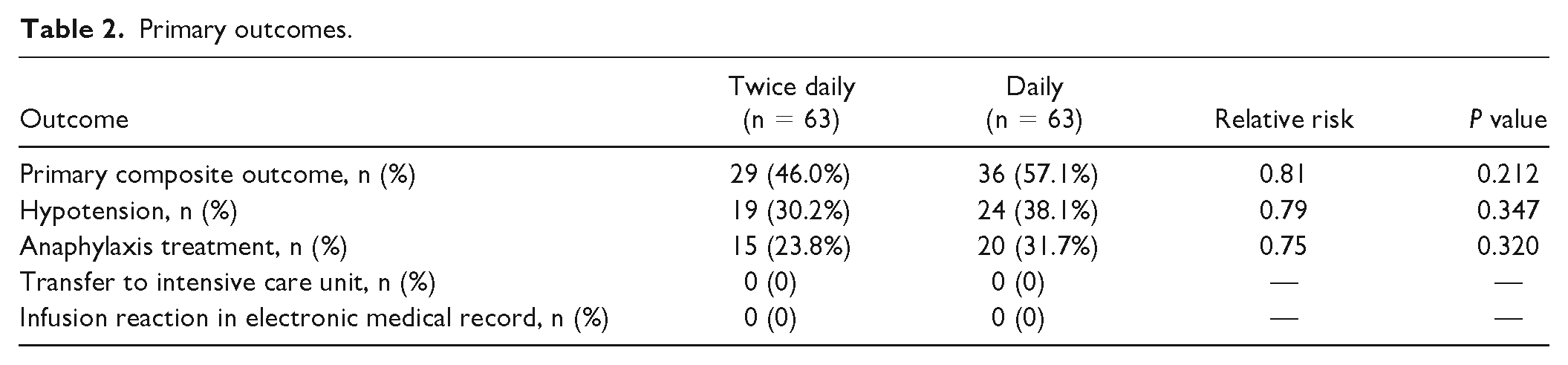
Table 3 summarizes the secondary outcomes of this study. Of the patients who had both pre- and post-ferric gluconate laboratory values available, iron concentrations increased by 16 to 24 μg/dL in both groups, and transferrin saturation increased by about 6% to 7% in both groups. Hemoglobin increased by a median of 0.3 g/dL (IQR, 0.375-1.075 g/dL) in the daily group and 0.0 g/dL (IQR, –0.6-0.8 g/dL) in the twice-daily group, but this difference was not statistically significant (
Secondary outcomes.

Discussion
In this retrospective single-center study, a difference in a composite safety outcome for patients receiving ferric gluconate twice daily compared with the standard daily dosing regimen was not seen. There also were no differences between the 2 regimens in a variety of efficacy outcomes, although these results are limited. These results reflect a lack of safety concerns for the continuation of our current practice of twice-daily dosing of ferric gluconate in adult patients to accelerate intravenous iron repletion. A larger study, perhaps a randomized, controlled trial, would be beneficial in providing further information on this subject.
It is important to note that the administration of a medication from the anaphylaxis kit does not necessarily mean that a patient has experienced anaphylaxis or an infusion reaction. On further investigation of the patients who received administrations of medications associated with the anaphylaxis kit (15 patients in the twice-daily group and 20 patients in the daily group), it was noted that only 1 patient received medications that were deemed to be possibly associated with the administration of ferric gluconate. For this patient in the twice-daily group, epinephrine and diphenhydramine were administered, but there was no documentation of an infusion reaction in the patient’s chart, and the patient went on to receive 2 additional doses of ferric gluconate.
This study, due in part to its retrospective nature, had several limitations. First, because patients were not randomized to the 2 groups, there was no way of ensuring that the 2 groups would be similar at baseline. Age, sex, and proportion of patients who had heart failure or chronic kidney disease at baseline differed between the 2 groups, which may have affected the outcomes. In addition, the investigators were unable to ensure that all the intended laboratory tests were obtained for each patient, which limited the number of patients in whom the secondary outcomes could be studied. Although a difference in length of hospital stay was noted, this may be due to selection bias on the part of provider. Providers may have chosen the daily-administration option for patients who were more likely to have a prolonged length of stay, and providers may have favored the twice-daily dosing in patients expected to be discharged sooner. Lastly, this study had a relatively small sample size. However, this study had a larger number of subjects compared with the limited available literature on twice-daily dosing of ferric gluconate and thus adds to the body of evidence supporting this dosing regimen. 9
In conclusion, this retrospective single-center study in hospitalized adult patients demonstrates that there were no significant safety differences between twice-daily and daily administration of ferric gluconate. These results do not raise any concerns regarding the continued use of twice-daily dosing of ferric gluconate.
Footnotes
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
