Abstract
Introduction
Gabapentin and pregabalin are commonly prescribed medications that are well tolerated and recommended for the management of neuropathic pain related to diabetes, chemotherapy, postherpetic neuralgia, or spinal cord injury.1 -4 Gabapentinoids are also prescribed for the management of restless leg syndrome, fibromyalgia, seizures, and pruritus.1 -4
Gabapentin and pregabalin dosing can be adjusted based on a patient’s response to therapy. Gabapentin dosing ranges from 100 to 3600 mg daily and pregabalin dosing is 25 to 600 mg daily.1,2 Gabapentin and pregabalin exhibit greater than 90% kidney elimination and adjustments to dose and frequency are recommended for patients with chronic kidney disease (CKD).1,2 For patients with a creatinine clearance (CrCl) below 60 mL/min, a dosage reduction of 50% is recommended for both drugs. Furthermore, with every additional 50% decline in CrCl, the dose should be further reduced by half.1,2,5 Despite these dosing recommendations, gabapentinoids are frequently prescribed at higher than recommended doses and dosing frequencies in patients with decreased CrCl or CKD. 6 Currently, there is limited evidence on the cognitive and physical consequences of higher than recommended chronic gabapentinoid dosing.6 -9
Drowsiness and dizziness are commonly reported adverse effects (AEs) associated with gabapentinoids in addition to hypotension, tremor, confusion, and weakness.1,2,4,10 Gabapentinoids exert the pharmacologic effect on the alpha-2-delta-1 subunit of voltage-gated calcium channels which modulate the presynaptic release of excitatory neurotransmitters. Gabapentinoids are also structurally related to GABA, and although they do not extensively bind at the GABA receptors, they may exhibit GABA-like activity at higher doses. As a result, there is an increase in central nervous system (CNS) depression and a heightened risk of falls.1,2 This is a concern in patients with decreased kidney function taking higher gabapentinoid doses as drug accumulation can increase AEs. 11 Ishida and colleagues found that patients receiving hemodialysis and taking a gabapentinoid had an increased risk of a fall. 6 Another risk with gabapentinoid use is respiratory depression from CNS depression which is multiplied with the use of concomitant medications such as opioids, anxiolytics, and antidepressants.12 -14
A greater frequency of AEs has been reported in patients receiving higher doses of gabapentinoids, but limited evidence can be found for gabapentinoids’ influence on the frequency of AEs in patients with CKD.4,7,10,15,16 The purpose of this study was to determine if patients with decreased CrCl experience differing frequencies of AEs related to their home dose of gabapentin or pregabalin at hospital admission.
Methods
This retrospective cohort study was conducted at a single-center 246-bed hospital in the midwestern region of the United States between January 2018 and June 2019. This study was approved by the center’s Institutional Review Board. Patients eligible for inclusion were adults (≥18 years of age) with gabapentin or pregabalin prescription documentation on the admission medication reconciliation documents. All included patients had at least one serum creatinine (SCr) measured during their hospital stay. Patients were excluded if they were pregnant, immunosuppressed (defined as a diagnosis of active leukemia, myelodysplastic syndrome, HIV, or other active cancer, or receiving chemotherapy), or admitted for opioid or alcohol dependence, and withdrawal. Patients with missing information required to calculate CrCl were also excluded.
The primary outcome was to evaluate the appropriateness of outpatient gabapentinoid prescription according to the patient’s CrCl at hospital admission. Secondary outcomes included individual incidences of AEs related to gabapentinoid use (hypotension, tremor, confusion, drowsiness, weakness, and fall) and the influence of concomitant opioid and psychiatric medications on the frequency of AEs.1,2,15 Estimated CrCl was calculated using the patient’s age, SCr on admission, ideal body weight (IBW), and the Cockcroft-Gault calculation. 17 Adjusted body weight (AdjBW) was used in place of IBW when actual body weight exceeded 120% of IBW. Patients were placed in 1 of 2 groups based on their estimated CrCl of ≥60 mL/min or <60 mL/min. Subgroup analysis also included a comparison among patients with CrCl of 15 to 29 mL/min and patients with a CrCl <15 mL/min. The determination of appropriateness was based on the calculated CrCl and dosing guidance provided by the manufacturers of gabapentin and pregabalin.1,2 The incidence of AEs related to gabapentinoid use was collected for each patient and recorded as present or absent as described in inpatient admission and progress notes. Concomitant opioid or psychiatric medications on admission were also recorded. The frequency of AEs is reported as a percentage of patients that presented with an AE for a CrCl ≥60 mL/min, CrCl <60 mL/min, and concomitant opioid or psychiatric medications. Post-hoc analyses assessed the primary outcome and incidence of AEs related to gabapentinoid use for all groups except for patients with a hospital admission diagnosis of acute kidney injury (AKI).
Data collected from each patient’s electronic medical record (EMR) included demographic data (age, sex, ethnicity, height, and weight), hospitalization (reason for hospital admission, discharge dates, and admitting medical team), and past medical history (diabetes, hypertension, coronary artery disease, peripheral vascular disease, CKD, amputation, and peripheral neuropathy). Adverse effects were identified through descriptions provided in the hospital admission EMR except for hypotension. Hypotension was determined by the admitting diagnosis and recorded blood pressure of less than 90/60 mm Hg. 18 Influence of concomitant antihypertensives was not included in the assessment of hypotension as an AE of gabapentinoids. Serum laboratory chemistries including a complete metabolic panel and blood count were recorded daily when available. Each patient’s vital measurements were recorded as available for up to 10 days following the admission date, with the selection of the lowest daily blood pressure and heart rate for inclusion. Admission, hospitalization, or discharge medications collected included gabapentin, pregabalin, and concomitant central nervous depressants, such as opioids, benzodiazepines, and antidepressants (Supplemental Appendix 1).
Nominal data were analyzed using either Fisher’s exact or chi-square test and continuous data were analyzed using the Mann-Whitney U-test. All tests of significance were 2-tailed and P < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS version 26 (Armonk, NY) and Microsoft Excel 16. Results are reported as number (%), average (±SD), and median (interquartile range [IQR]).
Results
Medication orders for gabapentin or pregabalin identified 585 hospitalized patients for initial screening. The primary reason for exclusion was an admission diagnosis of alcohol or opioid dependence (Figure 1). A total of 286 patients met the inclusion criteria and were grouped according to their CrCl. Baseline characteristics (Table 1) found significant differences between groups for age, height, and weight (P < 0.001, P = 0.042, and P < 0.001, respectively). Patients with a CrCl <60 mL/min were older (median age of 70 years), and a greater proportion of patients were female (55.5%). Patients with a CrCl <60 mL/min had a significantly higher past medical history of hypertension, diabetes, coronary artery disease, or peripheral vascular disease (P = 0.012, P = 0.018, P = 0.020, and P = 0.002, respectively). The proportion of patients prescribed concomitant opioid medications did not differ significantly between groups (P = 0.422), though a significantly lower proportion of patients with CrCl <60 mL/min were prescribed concomitant psychiatric medications (P = 0.008).
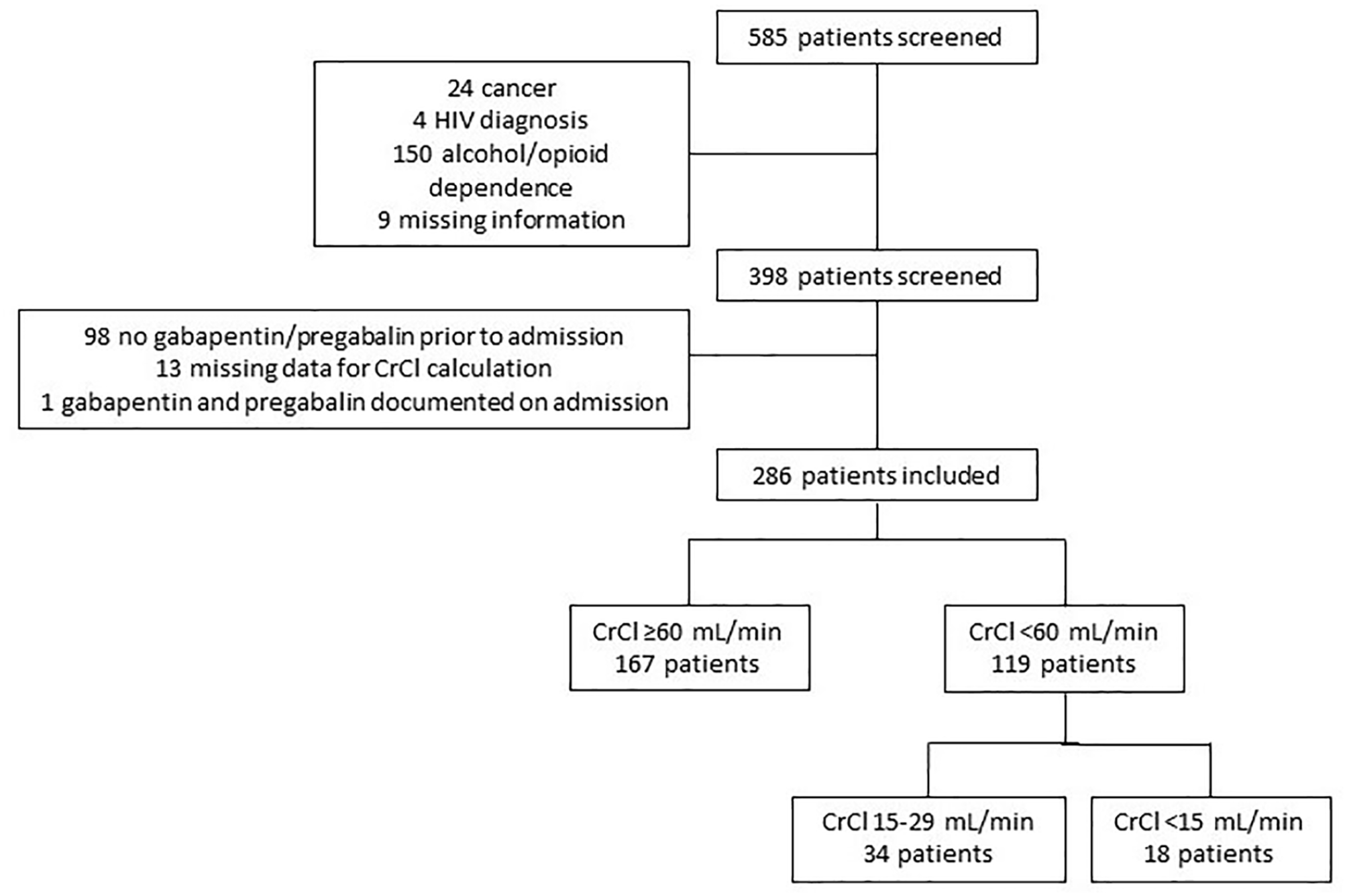
Patient screening and inclusion.
Baseline Characteristics.
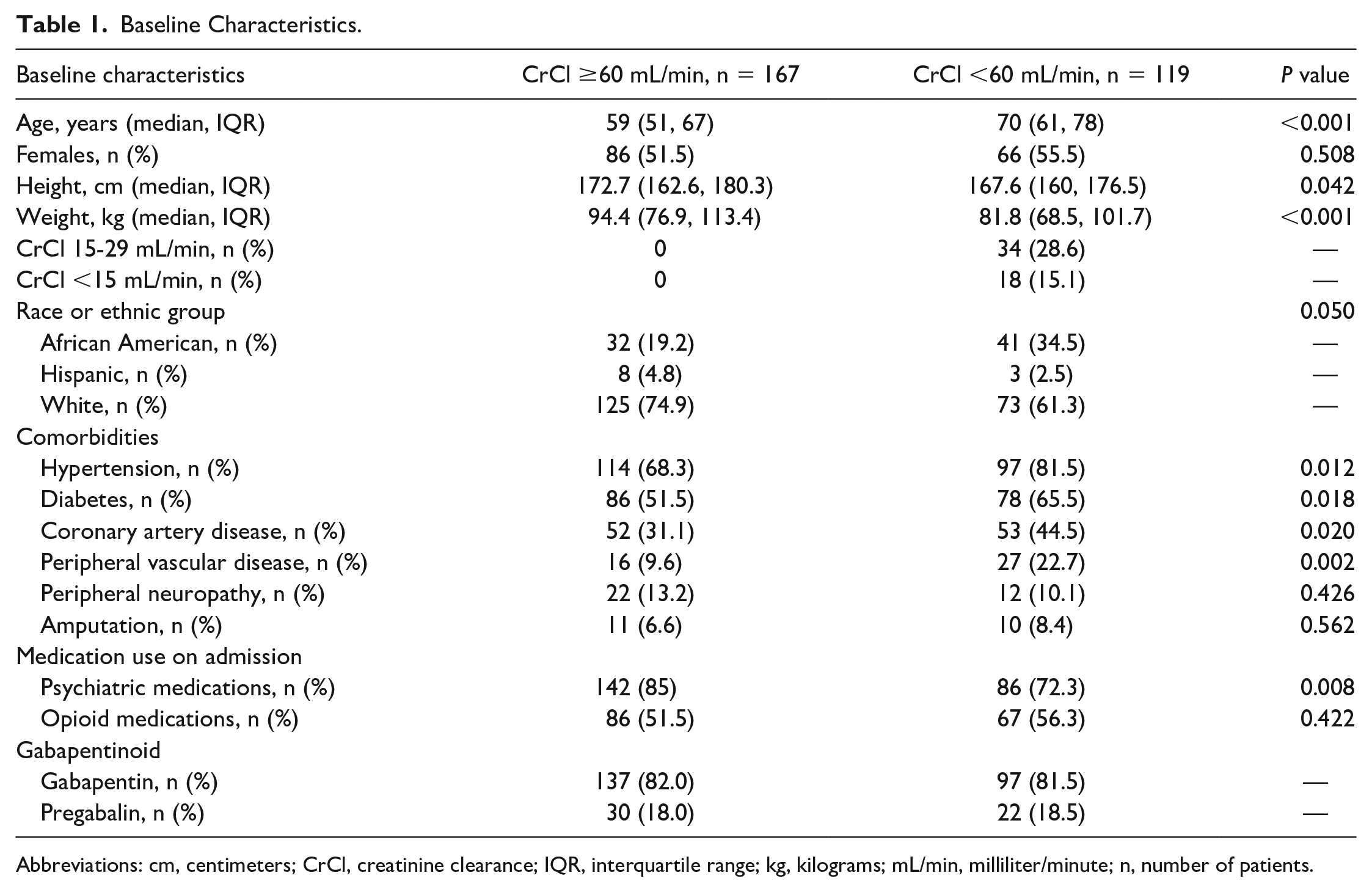
Abbreviations: cm, centimeters; CrCl, creatinine clearance; IQR, interquartile range; kg, kilograms; mL/min, milliliter/minute; n, number of patients.
Gabapentin (n = 234) was prescribed more frequently than pregabalin (n = 52) overall and the average total daily doses (TDDs) for gabapentin and pregabalin were greater for patients with a CrCl ≥60 mL/min (Table 2). An analysis of patients with a CrCl <30 mL/min found nonsignificant differences between average and median TDD of gabapentin and pregabalin between groups with a CrCl 15 to 29 mL/min and a CrCl <15 mL/min (P = 0.436 and P = 0.429, respectively).
Dosing of Gabapentin and Pregabalin According to Creatinine Clearance as Compared to Manufacturer Recommendations.
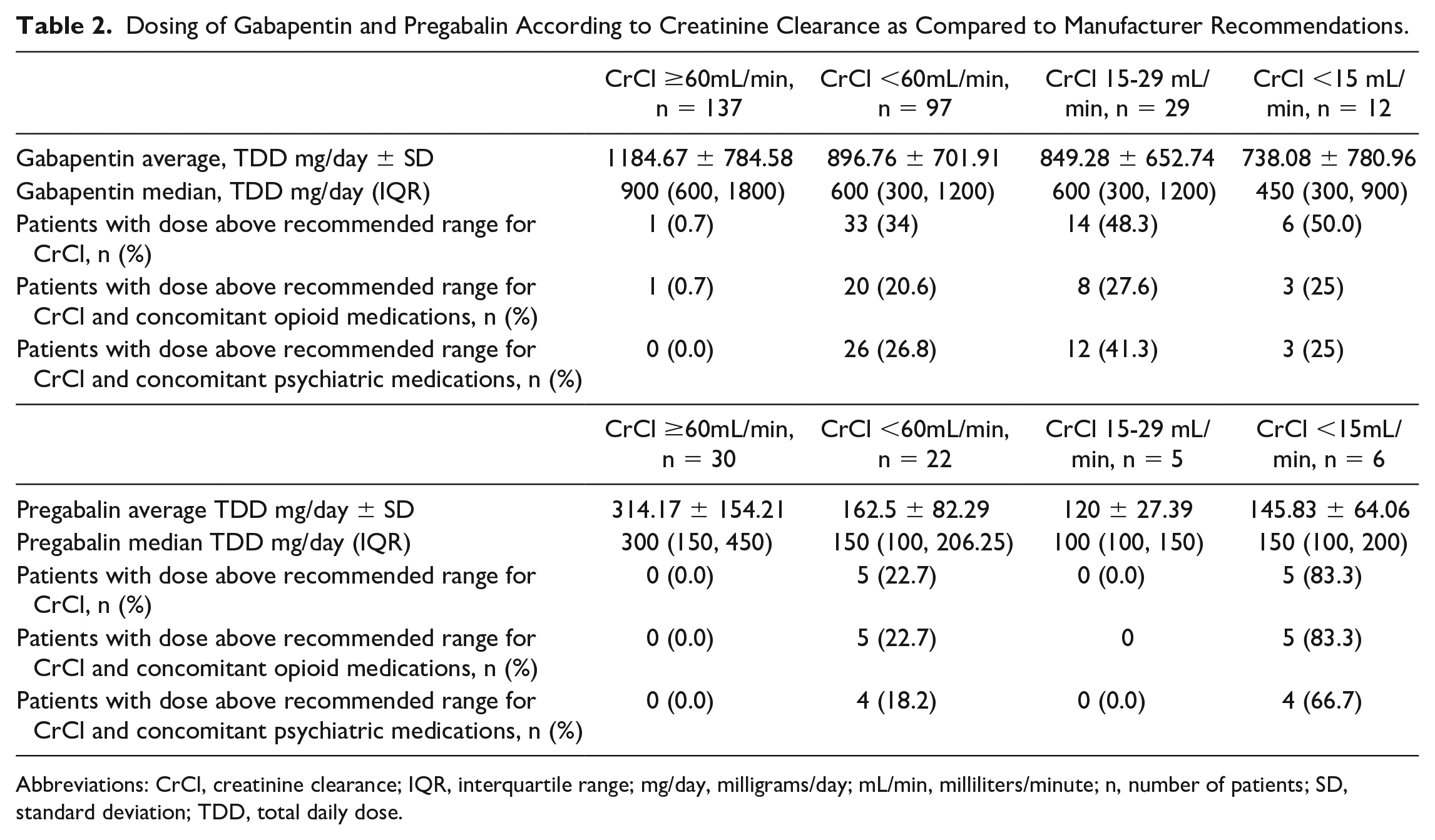
Abbreviations: CrCl, creatinine clearance; IQR, interquartile range; mg/day, milligrams/day; mL/min, milliliters/minute; n, number of patients; SD, standard deviation; TDD, total daily dose.
Patients with a CrCl <60 mL/min were prescribed doses higher than the manufacturer’s recommendations for gabapentin (34%) and pregabalin (22.7%). Nearly half of the patients receiving gabapentin in the 15 to 29 mL/min group and <15 mL/min group were receiving inappropriately high doses while 80% of patients receiving pregabalin in the <15 mL/min group were prescribed inappropriately high doses (Table 2). Inappropriate gabapentin dosing was identified in patients receiving concomitant opioid medications (n = 21, 61.8%) and those receiving concomitant psychiatric medications (n = 26, 76.5%). Pregabalin patients with inappropriate doses, (n = 5, 100%) were receiving concomitant opioids, and (n = 4, 80%) were receiving concomitant psychiatric medications (Table 2).
A smaller proportion of patients with a CrCl ≥60 mL/min presented with an AE in comparison to patients with a CrCl <60 mL/min. Patients with CrCl <60 mL/min presented with a significantly higher number of falls (P = 0.029) while the remaining AEs were not significantly different between groups (Figure 2). Concomitant opioid and psychiatric medications contributed to a higher prevalence of AEs in both groups of patients with CrCl ≥60 mL/min and CrCl <60 mL/min (Figure 2). Notable increases were observed for hypotension, confusion, and falls for patients with concomitant opioid medications and CrCl <60 mL/min. Patients with concomitant psychiatric medications and a CrCl <60 mL/min had a greater frequency of falls compared to those with a CrCl ≥60 mL/min with an increase in confusion and weakness.
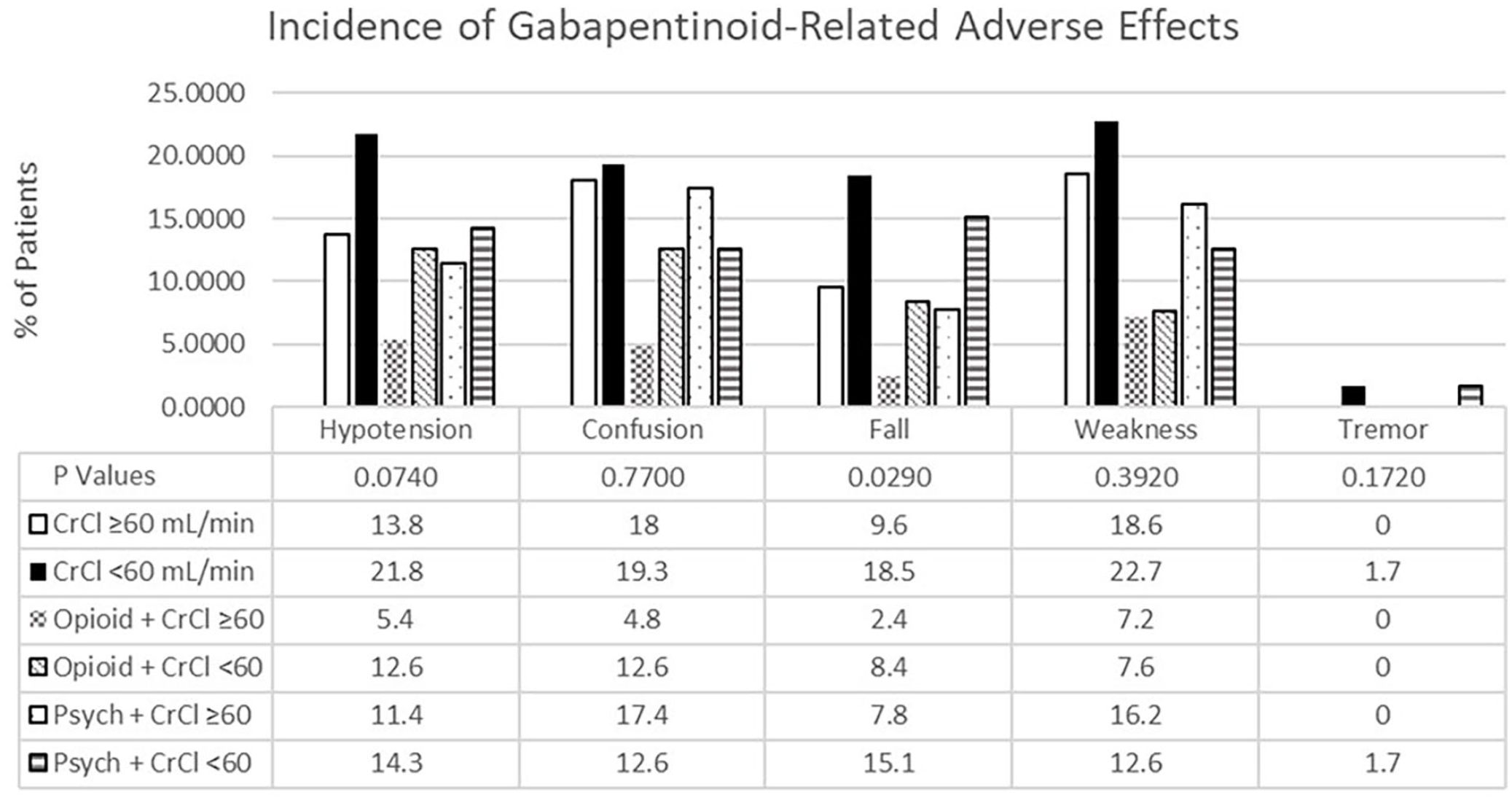
Incidence of gabapentinoid-related adverse effects compared between estimated kidney function. A description of the number of adverse effects reported for groups CrCl ≥60 and <60 mL/min displayed by the percentage of patients in each group. A significant difference between groups was identified for the incidence of a fall (P = 0.029) while all other adverse effects were not statistically significant.
Post-hoc subgroup analysis excluding patients with AKI revealed similar outcomes for baseline characteristics, TDD, and incidence of AEs. In addition, the significance of incidences of hypotension and falls between groups was identified (Supplemental Appendix 2).
Discussion
This study identified patients prescribed higher-than-recommended doses of gabapentin or pregabalin and the frequency of gabapentinoid-related AEs based on kidney function. Prior to hospital admission 22.7% to 34% of patients with CrCl <60 mL/min were prescribed higher-than-recommended doses of gabapentinoids. For patients receiving concomitant opioid and/or psychiatric medications, 18.2% to 26.8% were prescribed higher than recommended gabapentinoid doses. Patients with a CrCl <60 mL/min who were prescribed a gabapentinoid had a significantly higher rate of reported falls and a greater prevalence of gabapentinoid-related AEs at hospital admission. Concomitant opioid and psychiatric medications appear to contribute to the overall prevalence of AEs, particularly for hypotension, confusion, and falls.
These results are alarming and consistent with literature describing the risks of inappropriate gabapentinoid dosing in patients with reduced kidney function. Zand and colleagues retrospectively observed patients (n = 729) with serum gabapentin levels and known medical outcomes. 11 The patients were stratified into 3 groups based on kidney function (eGFR > 90 mL/min [n = 126], eGFR < 90 mL/min [n = 594], and patients receiving dialysis [n = 9]) with the mean daily gabapentin doses of 1922 ± 118, 1830 ± 45.3, and 1254 ± 217.2 mg respectively. This study identified significantly higher serum gabapentin concentrations and an increase in AEs for patients with an eGFR < 90 mL/min or receiving dialysis. Our study used different stratification parameters for kidney function but identified similar results related to gabapentinoid AEs. Ishida and colleagues examined data from the United States Renal Data System for patients receiving hemodialysis and stratified the groups by gabapentinoid dose. This study reported significant increases in altered mental status, fall, and fracture for patients receiving daily doses exceeding gabapentin 300 mg and pregabalin 100 mg. 6 A difference between Ishida and this study is the higher estimated kidney function and most patients were not receiving hemodialysis. However, this study found similar patterns of AEs in patients with a CrCl <60 mL/min as Ishida reported in patients receiving hemodialysis. The AEs chosen for evaluation in this study (hypotension, tremor, confusion, drowsiness, weakness, and fall) were identified through gabapentin and pregabalin package inserts and relevant literature.1,2,15 Thus, this study and current literature demonstrate that unadjusted gabapentinoid dosing based on a patient’s kidney function is likely associated with an increased risk for AEs. 6
The concern of inappropriate dosing is further heightened when considering the concomitant prescription(s) of psychiatric and opioid medications. In an effort to promote safer prescribing practices, the Food and Drug Administration (FDA) in 2019 released a warning about the dangers of CNS depression when using gabapentinoids alone or with opioids and other medications that cause CNS depression. 19 The FDA warning statement identified that the risk of CNS depression was higher in patients with a CrCl <60 mL/min when prescribed a concomitant psychiatric or opioid medication. This study demonstrates real-world data associated with this risk of dose-dependent CNS depression effects in patients receiving a gabapentinoid, CrCl <60 mL/min, and concomitant opioid or psychiatric medications, especially with this study’s finding of an increase in the frequency of falls among patients prescribed concomitant psychiatric medications. Thus, caution should be taken by providers and pharmacists when assessing the appropriateness of gabapentinoid dosing in patients with CrCl <60 mL/min and medications that contribute to incidences of AEs.
This study addresses specified outcomes in patients taking gabapentinoids with impaired kidney function compared to patients with normal kidney function. Minimal exclusion criteria allowed for the inclusion of patients with a wide range of CrCl and gabapentinoid dosing. This study had several limitations including there were no preceding studies to use for a power calculation to guide the number of patients needed for the primary outcomes. This is an important consideration as the primary outcomes were not significant but did show a higher prevalence of AEs in the CrCl <60 mL/min group. Also, there were significant differences in several baseline characteristics that could have been confounders for the frequency of AEs. Initially, patients admitted with AKI were not considered, but a post-hoc analysis found no meaningful changes in the primary results. This study did not examine the potential influence of concomitant antihypertensive medications on the incidence of hypotension. Finally, this was a single-center retrospective study, limiting generalizability outside of this patient population and prescribing practices. Therefore, this study was subject to channeling bias and reliance on accurate documentation of gabapentinoid prescription and AEs in the EMR.
Conclusion
This study identified a high incidence of patients with CKD who were prescribed inappropriately high doses of gabapentinoids. Although this study did not identify a direct relationship between gabapentinoid dosing in patients with CKD to the number of reported gabapentinoid-related AEs at hospital admission an increase in falls was identified. Larger, multicenter studies are needed to characterize gabapentinoid prescribing practices in patients with CKD to better characterize the potential relationship and impact on patient safety.
Supplemental Material
sj-doc-2-pmt-10.1177_87551225231217906 – Supplemental material for Evaluation of Gabapentin and Pregabalin Use in Hospitalized Patients With Decreased Kidney Function
Supplemental material, sj-doc-2-pmt-10.1177_87551225231217906 for Evaluation of Gabapentin and Pregabalin Use in Hospitalized Patients With Decreased Kidney Function by Gwendolyn M. Knowles, Grace E. LaFleur and Mariann D. Churchwell in Journal of Pharmacy Technology
Supplemental Material
sj-docx-1-pmt-10.1177_87551225231217906 – Supplemental material for Evaluation of Gabapentin and Pregabalin Use in Hospitalized Patients With Decreased Kidney Function
Supplemental material, sj-docx-1-pmt-10.1177_87551225231217906 for Evaluation of Gabapentin and Pregabalin Use in Hospitalized Patients With Decreased Kidney Function by Gwendolyn M. Knowles, Grace E. LaFleur and Mariann D. Churchwell in Journal of Pharmacy Technology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
