Abstract
Background
The use of vasopressors to maintain adequate hemodynamics and perfusion remains a fundamental aspect of caring for critically ill patients. 1 The dosing of vasopressors varies widely due to provider preference, heterogeneity of critically ill patients, and gaps in current literature. There are no critical care guideline-based recommendations surrounding optimal vasopressor dosing strategies. Institutions are left to adopt their own approach and may select a weight-based (WB) or non-weight-based (NWB) dosing strategy. 2 Variation between different units or providers in the same hospital may also exist.
Few studies have compared NWB and WB dosing for vasopressors. Studies that have evaluated WB dosing focus largely on using actual (ABW) or total body weight (TBW) as the dosing weight, rather than ideal body weight (IBW). Ideal body weight, which defines a specific weight of a patient in relation to their height, may offer several advantages when used to dose medications. Given the combination of a vasopressor’s hydrophilic properties, small volume of distribution, and rapid clearance, there is a strong scientific rationale that WB dosing with IBW may be superior to TBW dosing.3 -5 Evidence has shown that using IBW when dosing other classes of hydrophilic medications in critically ill patients, such as rocuronium and vecuronium leads to optimal conditions for endotracheal intubation without prolonged paralysis. 6 Dosing strategies using IBW may be able to better predict safer, yet still effective dosing parameters while limiting adverse effects that are compounded by excessive dosing.
A recent retrospective cohort study of 189 patients explored the dose requirements of norepinephrine to achieve goal mean arterial pressure (MAP) in cardiopulmonary intensive care unit (ICU) patients with septic shock. 7 Researchers found that patients receiving WB norepinephrine, using TBW, required lower doses to achieve goal MAP (0.07 µg/kg/min, NWB vs. 0.05 µg/kg/min, WB, P ≤ 0.001) when all dosing was converted to WB. WB dosing also led to lower norepinephrine doses at initiation. There were no significant differences in the duration of mechanical ventilation, ICU/hospital length of stay, or in-hospital mortality. WB dosing led to a longer duration of norepinephrine use. The authors concluded that a WB dosing approach led to similar achievement of goal MAP without additional adverse effects.
Another retrospective, multicenter cohort study compared time to achieve goal MAP with WB dosing, using estimated or measured ABW, and NWB dosing of norepinephrine for patients with septic shock and body mass index (BMI) ≥ 30 kg/m2. 8 There was no difference in the two dosing strategies in time to achieve goal MAP (60 min, NWB vs 58 min, WB, P = 0.28). The study did find a difference in the secondary outcome of time to norepinephrine discontinuation. Patients in the WB dosing group experienced longer time to norepinephrine discontinuation (27 hours, NWB vs. 33 hours WB, P = 0.03). Because ABW was used, the researchers recognize there was higher cumulative dose exposure. Authors recommended using either an NWB or WB dosing strategy for dosing norepinephrine, with a preference toward NWB to reduce exposure to norepinephrine.
Much of the literature regarding WB and NWB dosing has been conducted in septic shock patients with extremes of BMI (underweight, overweight, and obese). A propensity-matched analysis of WB dosing of norepinephrine investigated clinical outcomes of underweight (BMI < 18.5 kg/m2) and morbidly obese (BMI ≥ 40 kg/m2) patients. 9 The primary endpoint was in-hospital mortality. Although the cumulative norepinephrine dose was highest in morbidly obese patients, this group had lower in-hospital mortality than underweight patients (41.4% vs 24.7%, P < 0.001). However, mortality at 1 year after hospitalization was higher in the morbidly obese cohort. Higher norepinephrine exposure in the morbidly obese group was associated with acute kidney injury and development of cardiac arrhythmias.
Radosevich and colleagues compared norepinephrine dosing requirements in obese (BMI ≥ 30 kg/m2) and nonobese (BMI < 30 kg/m2) patients with septic shock. 10 Analyzing 100 patients in each study group, the investigators found that obese patients required lower norepinephrine WB, using actual body weight, doses than nonobese patients at 60 minutes following initiation (0.09 µg/kg/min vs 0.13 µg/kg/min, P = 0.006). However, there was no difference found in the equivalent NWB dose (9 µg/min in obese vs 8 µg/min, P = 0.72). The authors argue that because obese patients required lower WB doses than nonobese patients, an NWB dosing strategy can be safely employed for obese patients.
Consistency of dosing weight selection is well-documented as an important parameter if using WB dosing in critically ill patients. 11 Critically ill patients have significant variability in pharmacokinetics and pharmacodynamics, thus ensuring consistent body weight selection may minimize the risk of errors.
Methods
Historically our institution used an NWB dosing strategy in critical care units for vasopressors including norepinephrine, epinephrine, and phenylephrine. Patients undergoing cardiac surgery used a WB vasopressor dosing approach. In late 2022, our institution implemented a WB dosing strategy for vasopressors to standardize vasopressor dosing among hospital units. We hypothesized that a WB dosing strategy would lead to more appropriate starting doses, more accurate titrations, and faster attainment of goal MAP when compared to NWB dosing. Non-weight-based and WB vasopressor starting and default maximum doses, as well as titration doses and frequencies can be seen in Table 1. Maximum doses could be exceeded at the discretion of the provider.
Institution-Specific NWB and WB Recommended Starting and Default Maximum Doses, as Well as Titration Ranges for Epinephrine, Norepinephrine, and Phenylephrine.

Abbreviations: NWB, non-weight-based; WB, weight-based.
This was a single-center, retrospective, quasi-experimental chart review of patients receiving NWB and WB vasopressor dosing strategies. The study was reviewed and determined to be exempt by the University of Nevada Reno Institutional Review Board.
This before and after study compared patients receiving an NWB dosing strategy from July to September of 2022, to patients receiving a WB dosing strategy from December 2022 through February 2023. Patients receiving a WB strategy used IBW for vasopressor dosing. In patients with an actual body weight that was lower than their IBW, actual body weight was used. For patients under five feet, IBW was calculated by subtracting 1 kg (male 50 kg, female 45.5 kg) for each inch below 60 inches. 12
The study was conducted in three ICUs (trauma/surgical ICU, neurological ICU, and cardiac/medical ICU) within our institution. Patients were included if they were aged 18 years and older, admitted to an ICU, and received norepinephrine, epinephrine, or phenylephrine infusions for greater than or equal to 4 hours. Dopamine was not examined because it was already dosed in µg/kg/min throughout the institution. Known pregnancy, patients receiving a vasopressor upon arrival to our institution, patients already at their goal MAP at time of initiation of a vasopressor, cardiac surgery patients, and prisoners were excluded.
The primary outcome measure was the time to achieve goal MAP in minutes. Secondary outcome measures included MAP at 60 minutes following initiation of the vasopressor, total number of vasoactive agents required, use of vasopressin, duration of mechanical ventilation, in-hospital mortality, hospital length of stay, and discharge disposition. Additional secondary outcomes included an analysis of primary and secondary outcomes for patients with BMI < 18.5 kg/m2, >30 kg/m2, and >40 kg/m2, and those with a diagnosis of septic shock.
Data were collected from the EPIC electronic medical record. Data collected included blood pressure, MAP, vasopressor doses, steroid use, length of stay, discharge disposition, fluid bolus amount received, goal MAP target, sex, age, weight, height, BMI, time of vasopressor initiation, septic shock diagnoses, and comorbidities.
Statistical analysis was performed using SAS® version 9.4. Quantitative variables were assessed using the Student’s T test for all normalized data and the Wilcoxon test for non-normal data. Qualitative data were assessed using chi-square and Fisher’s Exact Tests. A P value of <0.05 was considered significant.
Results
A total of 654 adult patients were identified that received vasopressors for greater than or equal to 4 hours (Figure 1). The most common reason for exclusion was that patients were already at their goal MAP prior to initiation of a vasopressor, followed by patients undergoing cardiac surgery. There were 153 patients included in the NWB dosing arm and 183 patients in the WB dosing arm.
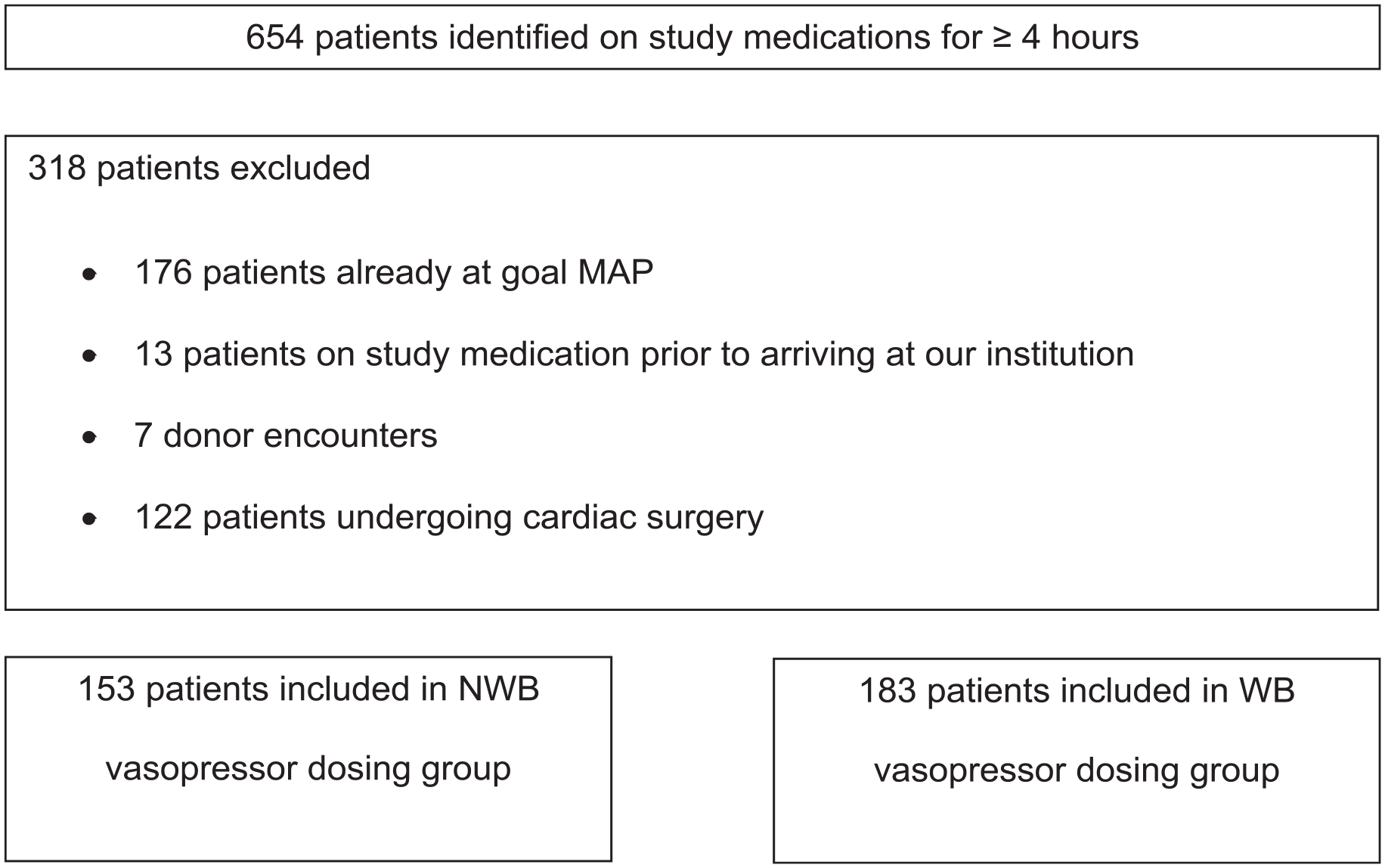
Study population identified with exclusion criteria for both NWB and WB dosing strategies.
Baseline characteristics including age, gender, and comorbidities are shown in Table 2. Most patients evaluated were male and the mean age was in the 60s. There was no significant difference in baseline MAP at the time of initiation of a vasopressor (58 mm Hg, NWB vs 56 mm Hg, WB). Goal MAP did not differ between groups, and both received a similar amount of fluid bolus. The NWB group had more patients with underlying chronic kidney disease. The WB dosing group had a higher percentage of patients who received corticosteroids.
Baseline Characteristics Including MAP at Time of Initiation, Comorbidities, Mechanical Ventilation, Goal MAP, Fluid Boluses, and Receipt of Corticosteroids for the NWB and WB Dosing Groups.
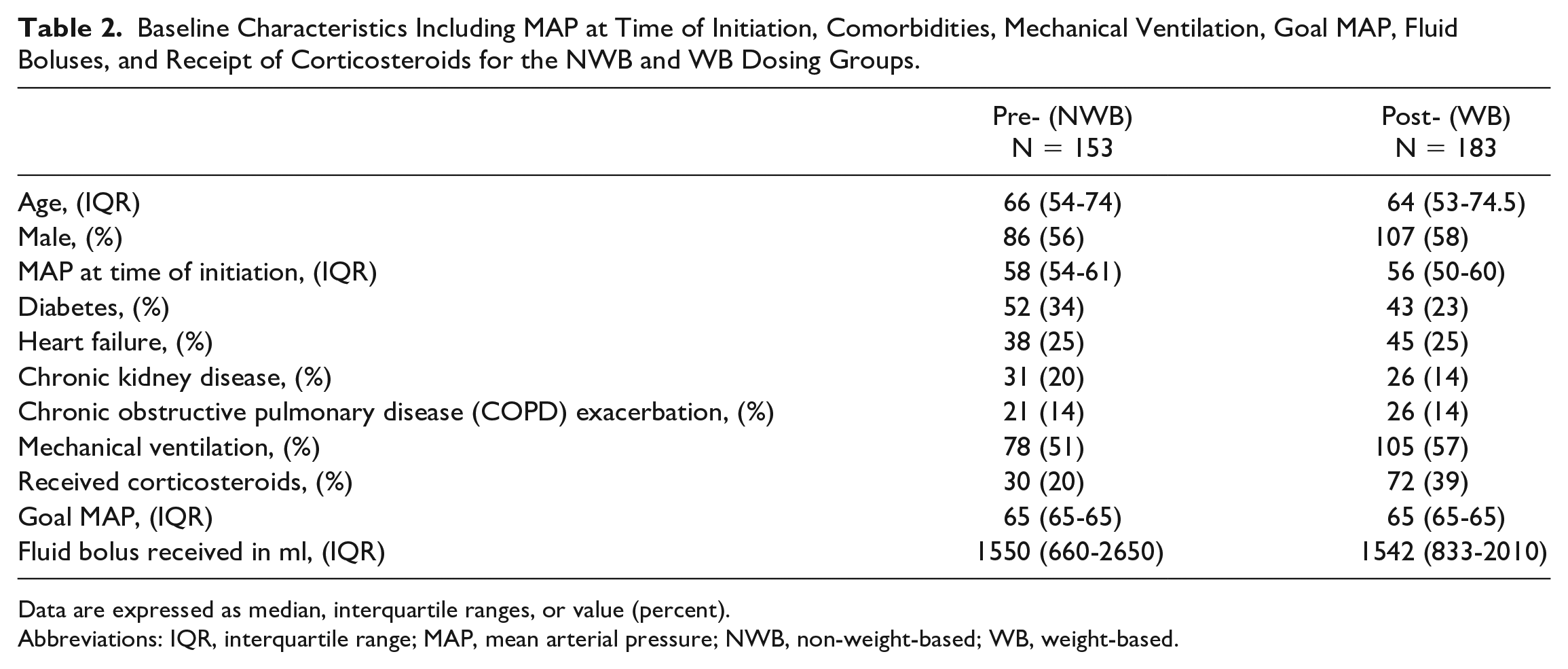
Data are expressed as median, interquartile ranges, or value (percent).
Abbreviations: IQR, interquartile range; MAP, mean arterial pressure; NWB, non-weight-based; WB, weight-based.
As shown in Figure 2, the median time to achieve goal MAP in the NWB dosing group was 24 minutes and 21 minutes in the WB vasopressor dosing group (P = 0.1713).
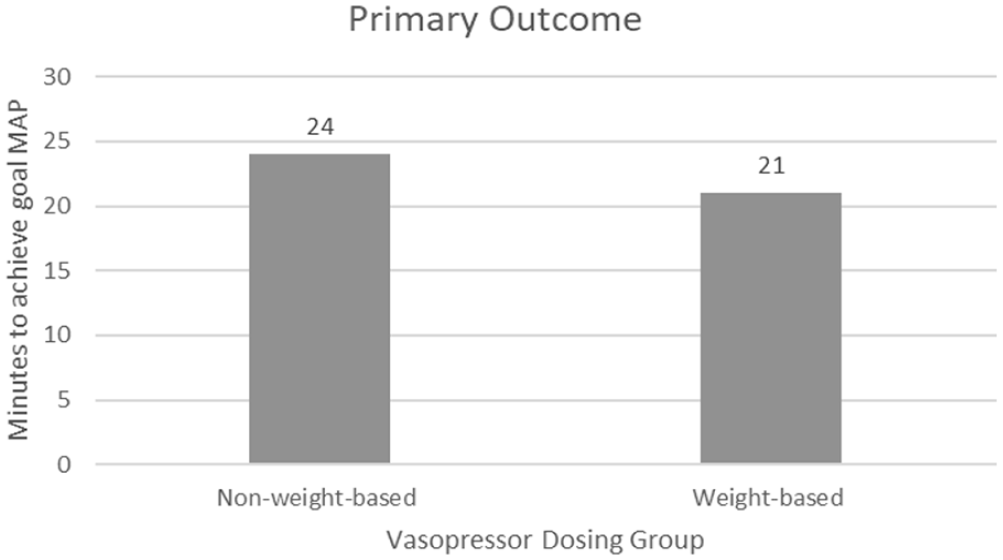
Comparison of time to achieve goal MAP in 153 patients that received NWB vasopressor dosing and 183 patients that received WB dosing. The figure compares the median time to achieve goal MAP in minutes.
Secondary outcomes are shown in Tables 3 and 4. Mean arterial pressure at 60 minutes after the start of vasopressor infusion was similar in both groups. There was no difference in the number of vasoactive agents required including the use of vasopressin. Furthermore, there were no differences in hospital length of stay or in-hospital mortality. Discharge disposition was classified into one of 4 categories: home, hospice, further/long-term care, and expired. There was no difference in discharge disposition between the two groups.
Secondary Outcomes Explored in the Study.
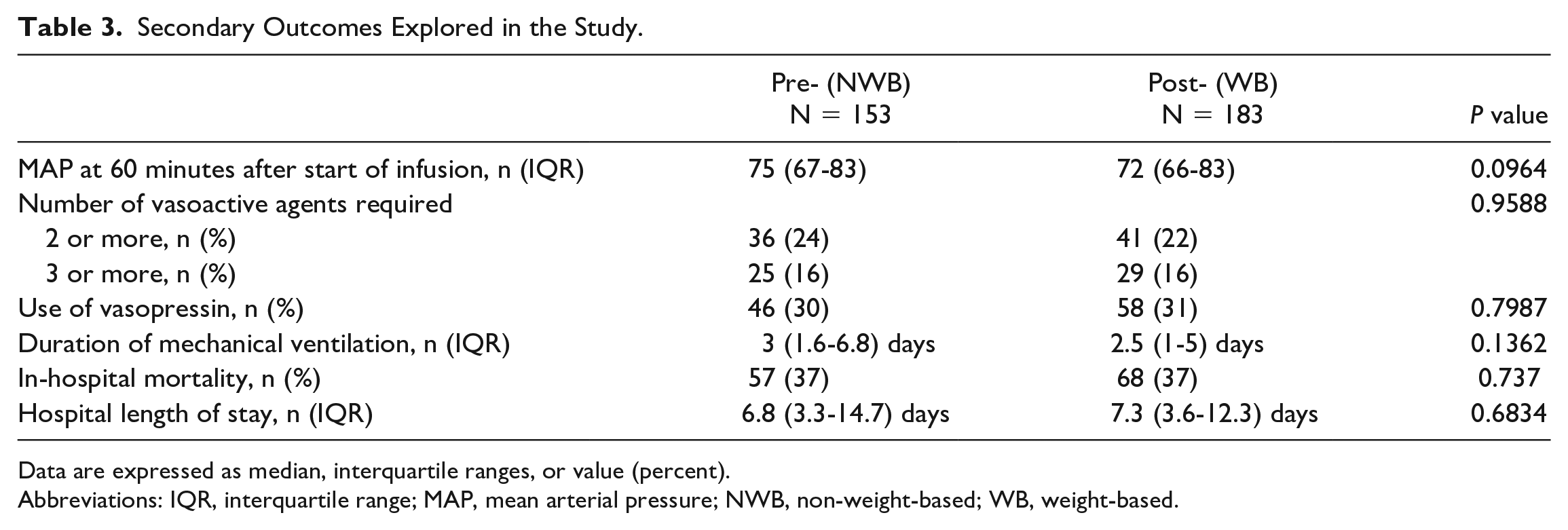
Data are expressed as median, interquartile ranges, or value (percent).
Abbreviations: IQR, interquartile range; MAP, mean arterial pressure; NWB, non-weight-based; WB, weight-based.
Discharge Disposition for 336 Patients Included in the Study.
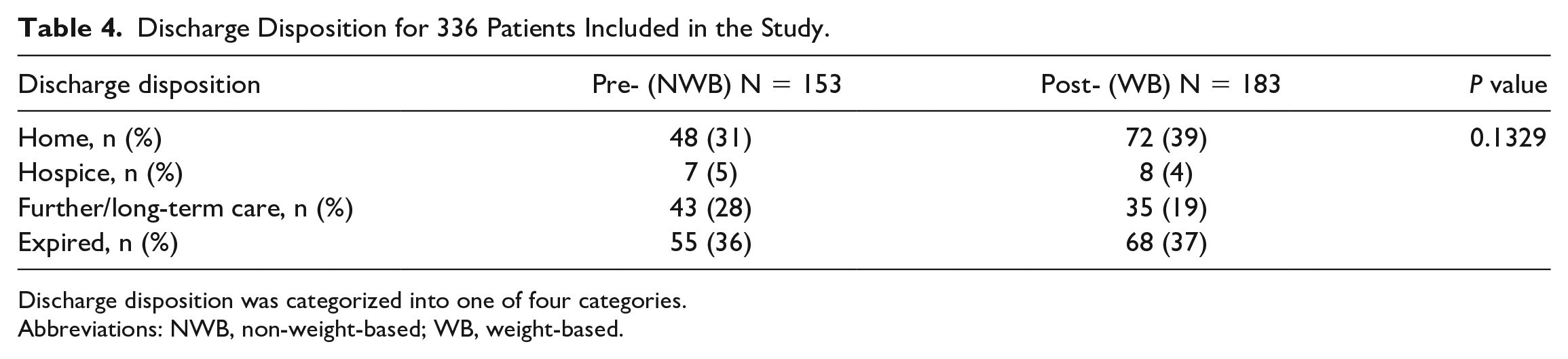
Discharge disposition was categorized into one of four categories.
Abbreviations: NWB, non-weight-based; WB, weight-based.
Subgroup analyses were performed for underweight, overweight, and obese patients (BMI < 18.5 kg/m2, >30 kg/m2, and >40 kg/m2) and those with a septic shock diagnosis. The number of patients included in each subgroup as well as outcomes can be seen in Tables 5 and 6.
Subgroup Analysis of Patients With Extremes of Body Mass Index (BMI < 18.5 kg/m2, >30 kg/m2, and >40 kg/m2).
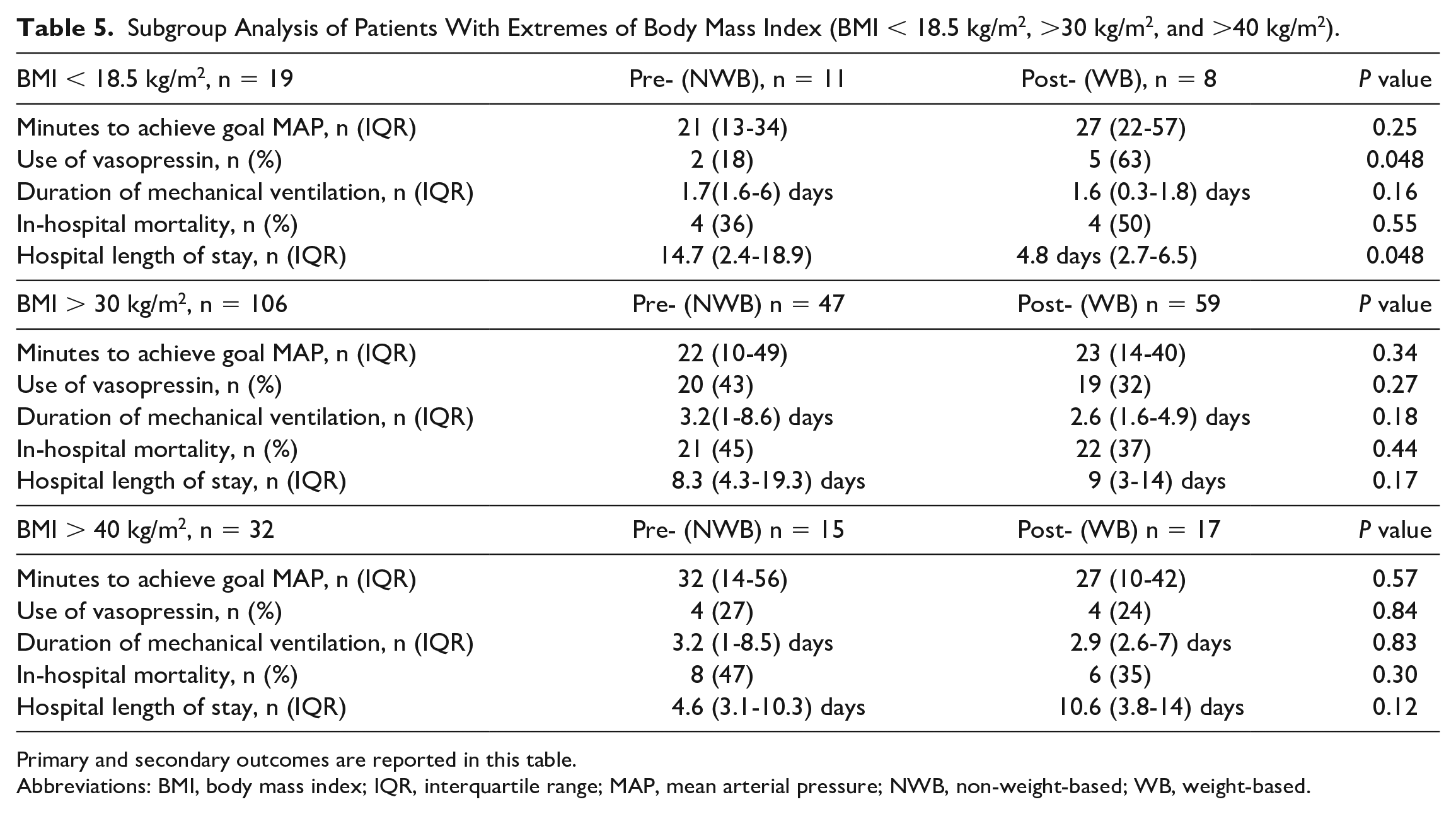
Primary and secondary outcomes are reported in this table.
Abbreviations: BMI, body mass index; IQR, interquartile range; MAP, mean arterial pressure; NWB, non-weight-based; WB, weight-based.
Subgroup Analysis of Patients With Septic Shock.
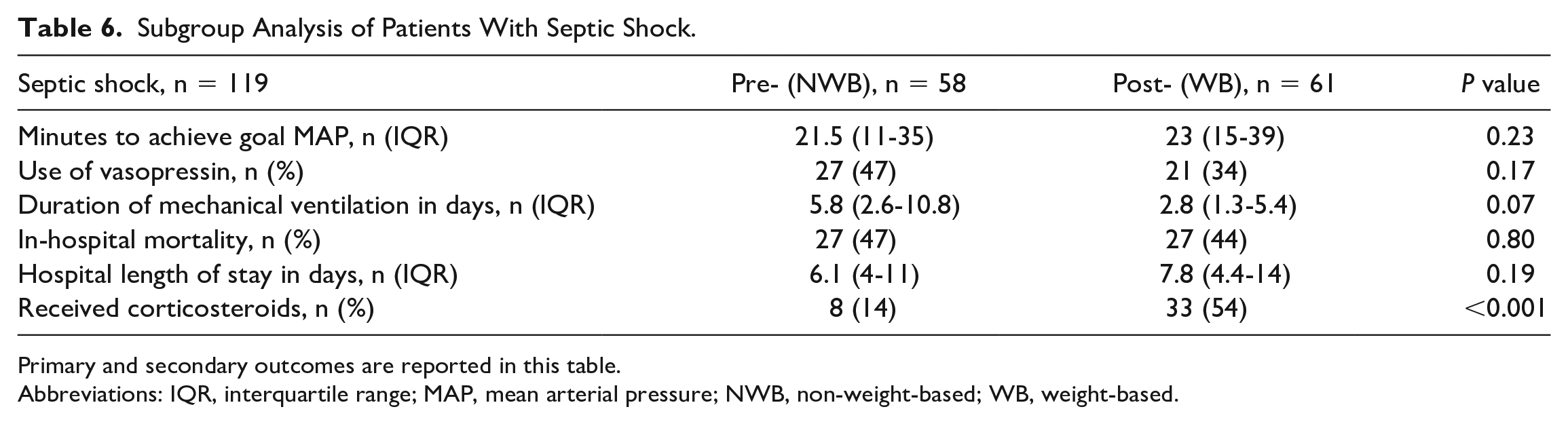
Primary and secondary outcomes are reported in this table.
Abbreviations: IQR, interquartile range; MAP, mean arterial pressure; NWB, non-weight-based; WB, weight-based.
There were no differences in the underweight, overweight, and obese patients for time to achieve goal MAP with either dosing strategy. The underweight subgroup had more use of vasopressin in the WB dosing group. However, this group also had a shorter hospital length of stay than those in the NWB dosing group.
There were 119 patients included in the septic shock subgroup analysis (58 NWB vs 61 WB). About 14% of those in the NWB group received corticosteroids versus 54% in the WB group, P ≤ 0.001. There were no other significant differences between the groups.
Adverse events and safety data were not collected due to restrictions of the electronic medical record.
Discussion
There was no difference in the time to achieve goal MAP when comparing a historical NWB vasopressor dosing approach to a WB dosing method. There were no significant differences in secondary outcomes related to vasopressor dosing strategies.
The subgroup analysis for patients with extremes of BMI showed no difference in the time to achieve goal MAP with either dosing strategy. Patients with a diagnosis of septic shock received corticosteroids more often in the WB vasopressor group. Late adoption of the 2021 Society of Critical Care Medicine’s Surviving Sepsis Guideline which defined the dose recommended to add corticosteroids as norepinephrine or epinephrine ≥0.25 µg/kg/min for at least 4 hours may have influenced this result. 13 While not statistically significant, patients with septic shock used less vasopressin in the WB dosing group, which may be clinically important due to the cost of this agent and warrants further study.
A higher number of patients than expected were excluded due to already being at their goal MAP at the time of vasopressor initiation. A literature review conducted in 2015 concluded that a single fixed value MAP goal is not suitable for most patients. 14 Authors discuss that high MAP goals are associated with adverse effects, and the optimal goal MAP range is likely 65 to 75 mm Hg for most patients. However, the results also promote individualization of MAP goals given comorbidities and clinical course. The addition of a goal MAP range, rather than a target with no upper limit (e.g., titrate to MAP > 65 mm Hg), may be beneficial in avoiding increasing MAP beyond physiologically needed doses and avoidance of premature vasopressor initiation.
Our institution converted to the WB vasopressor approach to align dosing strategies among our hospital units. Standardization of medication concentrations and dosing strategies is endorsed by the American Society of Health-Systems Pharmacists. The Standardize 4 Safety initiative funded by the Food and Drug Administration encourages continuity of medication concentrations across hospital units. Standardization increases patient safety and decreases medication errors. 15
Limitations of the study include retrospective study design and heterogeneous patient population. Bolus-dose vasopressor data were not collected which may have influenced time to hemodynamic stability. A retrospective cohort study examined the time to achieve hemodynamic stability (MAP of 65 mm Hg for at least 6 hours without escalating doses of vasoactive agents) with phenylephrine bolus doses prior to continuous norepinephrine infusion. 16 The analysis found that those who received a phenylephrine push had a higher incidence of hemodynamic stability at 3 hours (28.4% with push vs. 18.8% without push; confidence interval [CI]: 0.9%-18%). In addition, a formal power analysis was not conducted. Interpretation of the subgroup analysis is also limited due to small sample size, so these findings should be considered exploratory in nature. Finally, we recognize a potential confounder in seasonal differences and patient presentation during the timeframes of our study.
Further research should be conducted to assess potential adverse effects of WB vasopressor dosing, as these data were not collected in this study. This study explored the time to achieve the first goal MAP, but future studies may investigate how long patients stayed above their goal MAP. Time to initiation of an additional vasoactive medication may also help to determine if WB vasopressor dosing reduces the total vasoactive medication burden. Other potential confounders should also be researched, such as the use of midodrine to wean vasopressors and inotrope usage.
Conclusion and Relevance
This study adds to the limited evidence that NWB or WB dosing strategies can be used for vasopressor dosing. To our knowledge, this is the first study comparing WB dosing strategies using IBW in comparison to WB. Institutions should adopt a consistent dosing strategy and dosing weight for dosing norepinephrine, epinephrine, and phenylephrine. Consistent weight documentation and selection remain a priority if using WB dosing strategies.
Footnotes
Acknowledgements
The authors have no acknowledgements.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
