Abstract
Keywords
Background
Antimicrobial resistance (AMR) has become one of the most alarming public health issues in the 21st century. 1 It complicates the management of various infectious diseases regarding prevention or treatment as microorganisms are no longer susceptible to antimicrobials. 1 Antimicrobial resistance is a natural phenomenon in which microorganisms acquire different mechanisms to overcome being inhibited or killed by antimicrobials. 2 Several factors contribute to the emergence of AMR, such as overuse of antimicrobials, poor adherence, inappropriate dosing, frequency, or duration. 2 Worldwide organizations, such as the World Health Organization (WHO) and the Centers for Disease Control and prevention (CDC) have been exerting a lot of efforts and taking initiatives to improve the use of antimicrobials in the community and hospital settings. 1 The CDC estimated that approximately 2 million people are affected by antibiotic-resistant bacteria, with 2,300 people dying as a result of the infection. 3 In a systematic review in 2019, an estimated 4.9 million deaths were associated with bacterial AMR in 204 countries. 4 The WHO defines rational drug use (RDU) as follows: “Patients receive medications appropriate to their clinical needs, in doses that meet their own individual requirements, for an adequate period of time, and at the lowest cost to them and their community.” 5 Implementing RDU is crucial to limit the emergence of AMR; thus, several policies are issued to guide and promote RDU in different health settings. 5 In attempts to ensure implementation of RDU, drug utilization evaluation (DUE) has been encouraged. 6 Several guidelines have been published to be used as a guide for DUE programs in hospitals. 6 All these interventions have been carried out as an attempt to overcome AMR, improve drug prescribing and utilization, and thus improving patients’ outcomes.
Ciprofloxacin is one of the fluorinated quinolones related to nalidixic acid introduced in the 1980s. 7 It is a widely used fluoroquinolone antibiotic in various conditions, such as urinary tract infections (UTIs), sexually transmitted diseases, skin and bone infections, gastrointestinal infections, and lower respiratory tract infections. 7 It is a broad spectrum antibiotic against gram negative bacteria, such as Escherichia coli, Salmonella species (spp.), Shigella spp., Neisseria spp., Moraxella catarrhalis, Haemophilus spp., and Pseudomonas aeruginosa.7,8 Moreover, it has antimicrobial activity against gram positive bacteria, such as Staphylococcus spp., Streptococcus spp., Mycobacterium tuberculosis, and Listeria monocytogenes.7,8 It produces its antimicrobial effects through the inhibition of deoxyribonucleic acid (DNA) gyrase, preventing DNA replication or transcription, thus inhibiting growth. 9 Concerning safety profile of ciprofloxacin, it has been demonstrated that it has a favorable tolerability.7,8 Most of the adverse effects are mild, necessitating withdrawal only in 1.5% of the patients.7,8 These include gastrointestinal symptoms, mainly nausea, vomiting, abdominal pain, and diarrhea; central nervous system events, such as anxiety, nervousness, tremors, and insomnia; or dermatological side effects, such as pruritus or rash.7,8 Rarely, more serious side effects, such as heart rate–corrected QT interval (QTc) prolongation, tendinopathy, and dysglycemic events have been reported in very few patients.10 -12 Dosing of ciprofloxacin is usually twice daily in doses ranging from 250 to 750 mg per dose.7,8 Duration of treatment usually ranges from 7 to 14 days. Shorter durations as 3- or 5-day regimens are indicated in UTIs.7,8 Bone and joint infections require longer duration of therapy (4-6 weeks) depending on the severity of the infection.7,8
Fluoroquinolones—including ciprofloxacin—are one of the most widely used antimicrobials possessing many advantages as they are nontoxic, well-tolerated, effective, and have a broad-spectrum activity.13 -15 Moreover, they have excellent oral bioavailability, permitting their use in the treatment of various infections.13 -15 Unfortunately, overuse/misuse of fluoroquinolones have led to the emergence of bacterial resistance. Studies showed that resistance against ciprofloxacin have been emerging, especially in Escherichia coli (E. Coli) UTIs. 16 Resistance rates were higher in hospital versus community setting, and varied by region and country being higher in developing countries. 16 This is attributed to the empirical use of ciprofloxacin in UTI. 16
The aim of this study is to evaluate the use of ciprofloxacin in the community setting in terms of appropriate prescribing, dosing, frequency, and duration of use, and to detect serious adverse drug reactions that might occur as a result of inappropriate use of ciprofloxacin.
Methods
Study Design
A cross-sectional, retrospective study to evaluate ciprofloxacin use was conducted over the period from September 2021 to February 2022. The study was conducted by community pharmacists in 5 community pharmacies in Egypt. This study is registered in clinical trials.gov (NCT05916105) and conducted in accordance with the declaration of Helsinki.
Study Population
Patients prescribed oral ciprofloxacin during the period from September 2021 to February 2022 were taken up for study. The sample population was determined depending on drug use evaluation guidelines.
Eligibility Criteria
Patients prescribed oral ciprofloxacin during the study period were included in the study. Those prescribed IV (intravenous) ciprofloxacin or another fluoroquinolone as an alternative or a combination to ciprofloxacin were excluded.
Sample Size
The size of the sample population was determined according to the Joint Commission on the Accreditation of Health care Organization (JCAHO) criteria that mandate a sample size of 5%, if the average number of cases per quarter is greater than 600, and at least 30, if quarter cases are less than 600.
Data Collection
Data were collected by community pharmacists from the pharmacies patients’ records in a data collection sheet including the set of variables identified. Adverse events were identified using the GSQ65 (General Symptoms Questionnaire), physical examination, and lab tests. All precautionary actions were applied to preserve data con-fidentiality.
Study Variables
Independent variables:
• Patients’ demographics: age, gender, and pregnancy/lactation
Dependent variables:
• Department/indication
• Dose
• Frequency
• Duration
• Adverse events
• Drug interactions.
Statistical Analysis
Descriptive statistics were performed for the collected data. The responses were displayed as frequency (counts and/or percentage). Data were categorized and analyzed using software statistical computer package SPSS version 25.0 (SPSS Inc, Chicago, Illinois) and Excel 2013.
Results
A total of 1476 patients’ records were identified through the pharmacies database and screened for eligibility, out of which 978 records did not meet criteria. Then, 498 records were assessed for data collection, where 235 records were discarded due to missing data (diagnosis, frequency, and duration of use). Hence, 263 records were included in the study initially, out of which 112 records were discarded due to failure to document adverse events as a result of loss of follow-up. Finally, a total of 115 patients’ records were included and analyzed (Figure 1).
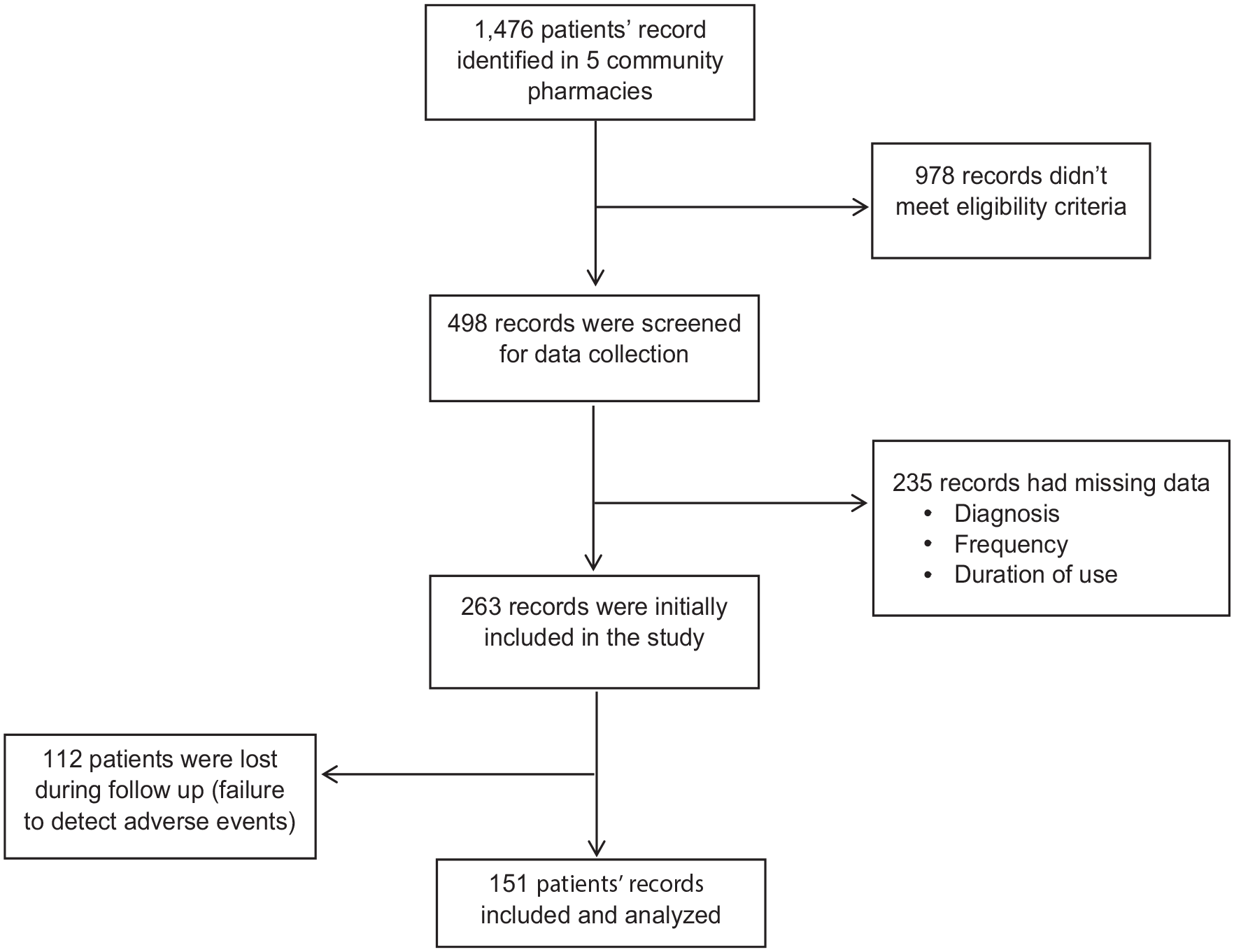
STROBE flow diagram for cross-sectional study.
Age and Sex Distribution of Patients With Ciprofloxacin Indication
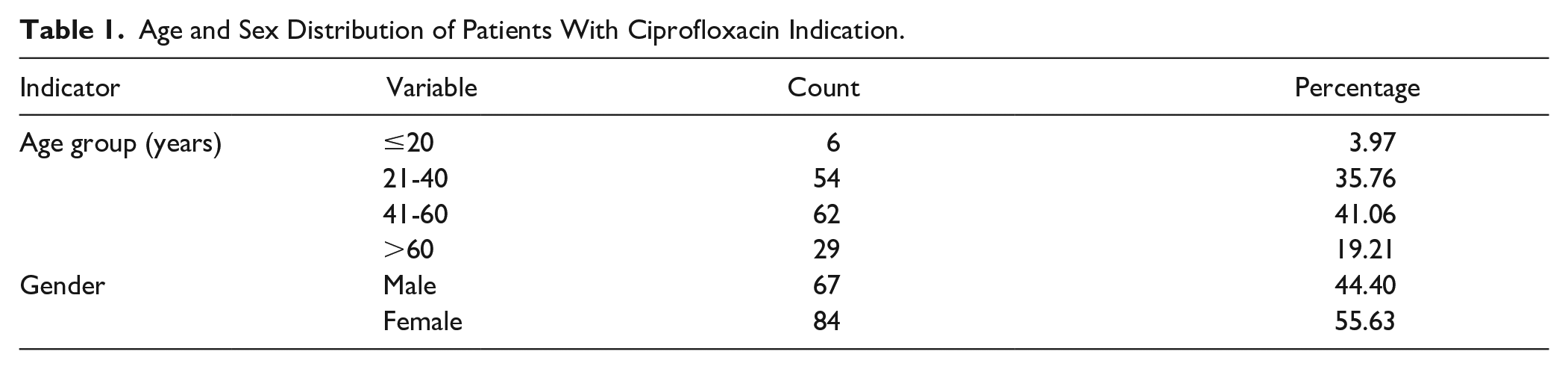
A total of 151 patients’ records indicated for ciprofloxacin were included in the study. Out of 151 patients’ records collected, 67 (44.4%) were those of men and 84 (55.6%) were those of women who were neither pregnant nor lactating. Age ranges were identified as ≤20, 21-40, 41-60, and >60 years. Six patients were ≤20 years of age, 54 patients were between 21 and 40 years of age, 62 patients were between 41 and 60 years of age, and 29 patients were >60 years of age (Table 1).
Age and Sex Distribution of Patients With Ciprofloxacin Indication.
Appropriateness of Ciprofloxacin Prescription
Out of 151 patients’ records, 146 (96.69%) ciprofloxacin prescriptions were appropriate, based on international guidelines and only 5 (3.31%) prescriptions were not in line with the guidelines.
Indications of Ciprofloxacin
Ciprofloxacin was indicated in various disease conditions. Figure 2 summarizes different indications in which ciprofloxacin was prescribed. The most common indications of ciprofloxacin were 18.50% for pulmonary infections, 16.34% for infectious diarrhea (dysentery), and 13.73% in various conditions of internal medicine department. Other indications included surgical site infections (9.65%), orthological (bone) infections (9.15%), cardiac condition (7.84%), urological diseases (7.84%), dermatological (6.84%) and gynecological infections (6.84%), and prostatitis (3.27%).
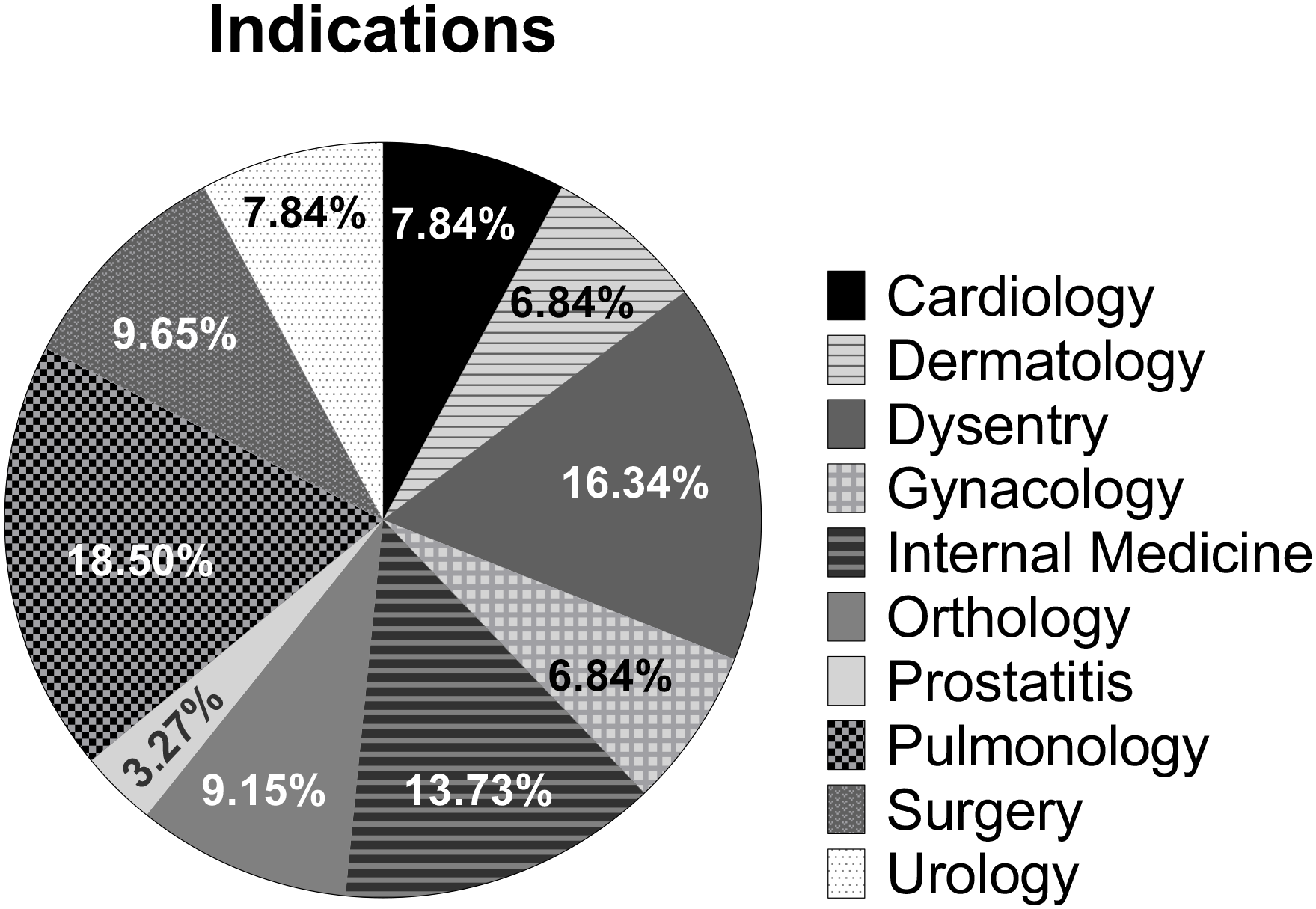
Percentage of ciprofloxacin use based on indications.
Dosage Regimen (Dose, Frequency, and Duration) of Ciprofloxacin
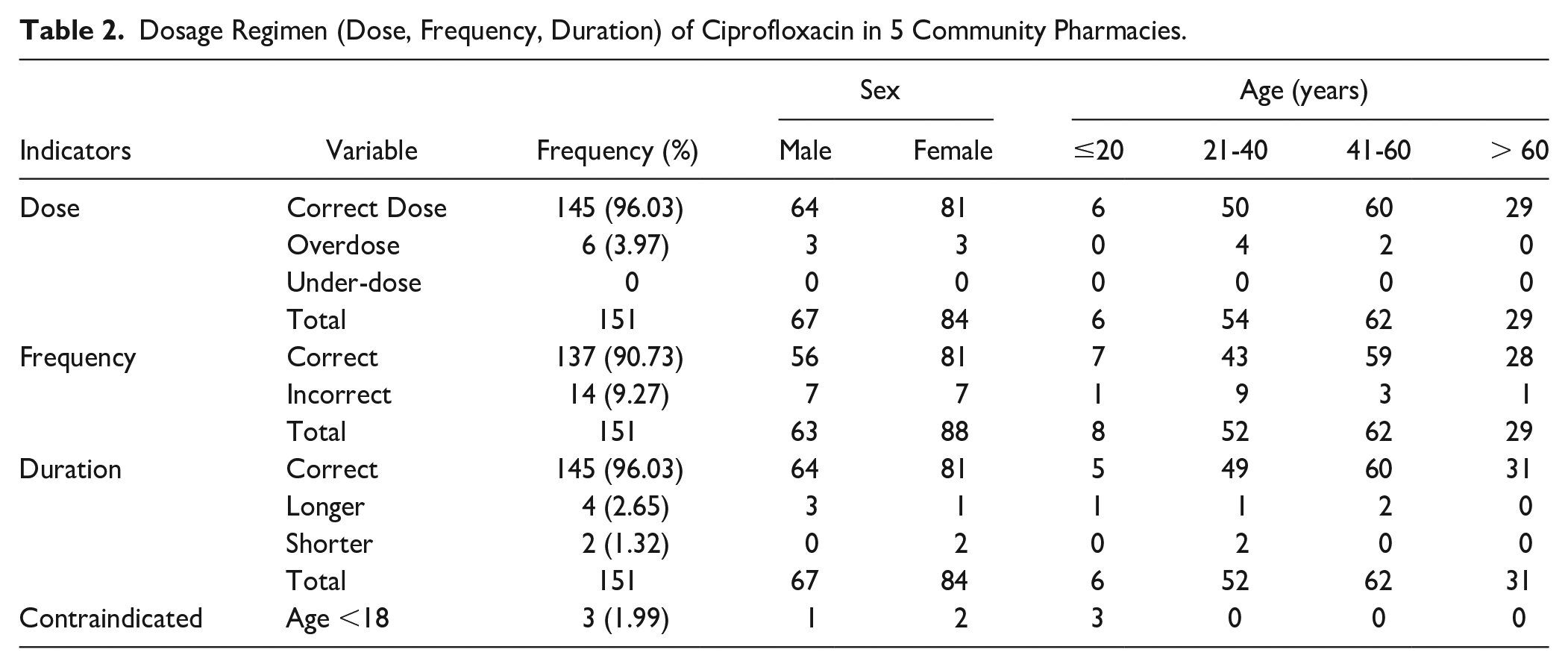
It was found that out of 151 patients’ records collected, 145 (96.03%) contained correct ciprofloxacin dosing, whereas 6 (3.97%) contained overdosing. There were no underdosing regimens. Furthermore, 137 (90.73%) indications were with correct frequency of administration, whereas 14 (9.27%) were with incorrect frequency. Concerning duration of use, 145 (96.03%) records were correct duration, 4 (2.65) were longer duration, and 2 (1.32) were shorter duration (Table 2).
Dosage Regimen (Dose, Frequency, Duration) of Ciprofloxacin in 5 Community Pharmacies.
Ciprofloxacin Use Against Contraindications
There were no pregnant and lactating patients for whom ciprofloxacin was indicated in the study, but it was indicated for 3 (1.99%) children whose ages were ≤18 years, which is against contraindications.
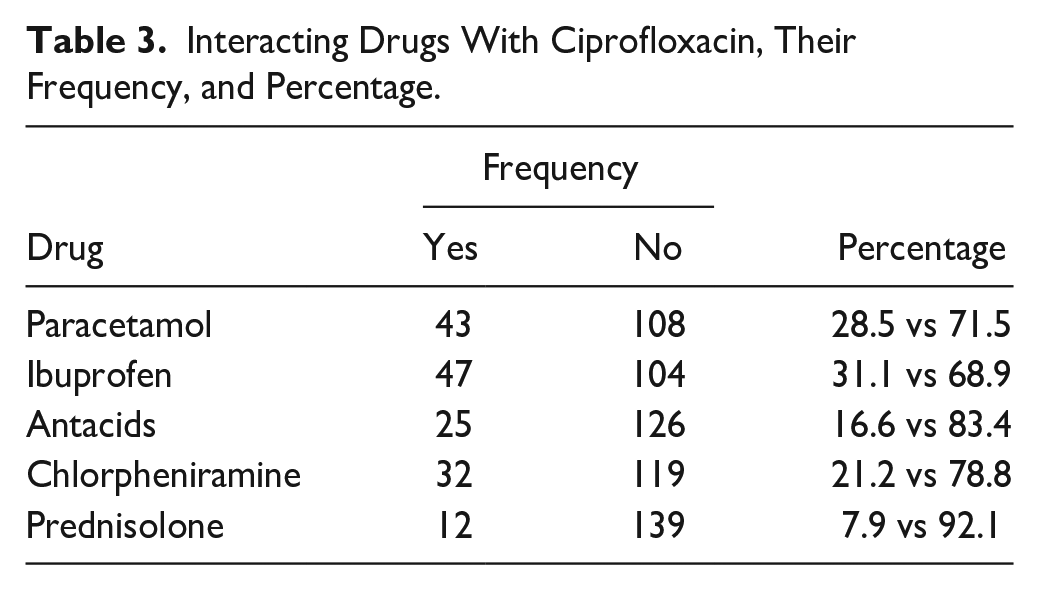
Drug Interactions With Ciprofloxacin
Twenty-six percent of patients’ records contained one or more potentially interacting drugs with ciprofloxacin. These include 43 records with acetaminophen (28.5%), 47 records with ibuprofen (31.1%), 25 records with antacids (16.6%), 32 records with Chlorpheniramine (21.2%), and 12 records with prednisolone (7.9%; Table 3).
Interacting Drugs With Ciprofloxacin, Their Frequency, and Percentage.
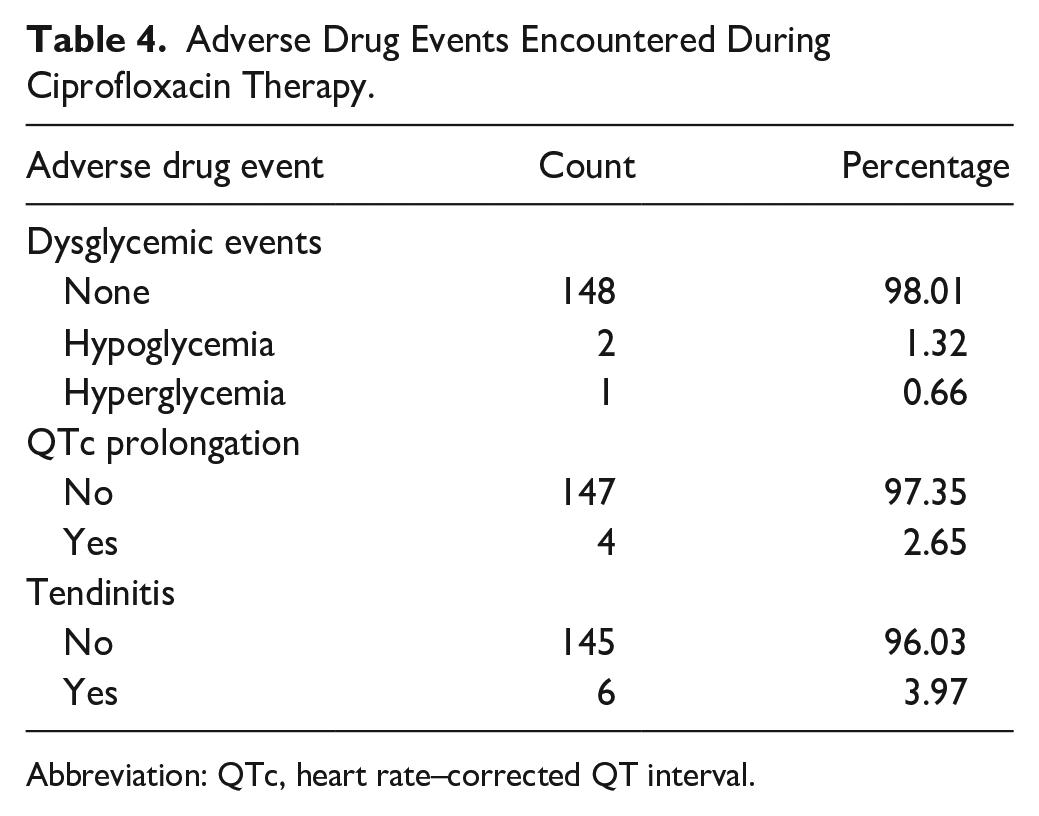
Serious Adverse Events Encountered During Ciprofloxacin Therapy
Serious adverse events have been identified as dysglycemic events, tendinitis/tendinopathy, and QTc interval prolongation. Dysglycemia has occurred in a total of 3 patients; 2 hypoglycemia (1.32%) and 1 hyperglycemia (0.66%). Tendinitis occurred in 6 patients (3.97%) whereas QTc prolongation occurred in 4 patients (2.65%; Table 4).
Adverse Drug Events Encountered During Ciprofloxacin Therapy.
Abbreviation: QTc, heart rate–corrected QT interval.
Discussion
Antimicrobial resistance has become an alarming issue threatening public health and complicating therapeutic management.1 -3 Many initiatives have been undertaken to offer solutions for minimizing AMR, one of which is the Global Antimicrobial Resistance and Use Surveillance System (GLASS) implemented by the WHO since October 22, 2015. 5 Rational drug use, which is the appropriate prescribing of medications in the adequate dose for an adequate duration to the right patient, is an important measure to limit AMR. 5
To ensure RDU is implemented, DUE should be undertaken in all health care settings (eg, hospitals, community pharmacies, and nursing homes). Drug utilization evaluation ensures the safe and effective use of medications to improve patients’ health. 6 Continual improvement in the appropriate use of drugs has the potential to lower the overall cost of care. Several guidelines have been established to ensure the implementation of DUE through implementing drug utilization review (DUR).6,17
Ciprofloxacin is a widely used fluoroquinolone antibiotic for the treatment of various disease conditions.7,8 Dosing regimens (dose, frequency, and duration) differ according to the indication it is prescribed for.7,8 Resistance to ciprofloxacin have been well established long ago. 9 Yet recent studies report increasing resistance to certain pathogens, such as Pseudomonas aeruginosa and E. Coli.16 -18 The spread of ciprofloxacin resistance imposes serious health issues due to its widespread use. Thus, DUE of ciprofloxacin is needed to ensure its appropriate use, according to international guidelines.
Our study showed that 96.69% of ciprofloxacin prescriptions were for the correct indications according to international treatment guidelines. This finding is similar to the results of Goldman 19 and Biru et al 20 that stated that 95% of patients had appropriate justification for use according to criteria. In contrast, Feroche and Alemu 21 showed that 86.5% of the ciprofloxacin prescribing was not in accordance with the WHO criterion.
Different doses of ciprofloxacin vary according to age groups and different conditions. Subclinical doses lead to ineffective control of infections, whereas overdose leads to toxicity. Thus, caution should be taken when prescribing ciprofloxacin to ensure appropriate management of patients. In our study, 96.03% of prescriptions had correct doses, whereas 3.97% were overdose. There was no underdosing in any of the prescriptions. This is similar to the findings of Biru et al, 20 which stated that 92.5% of records were correct dose and 7.5% were overdose. Yet, in contrast, Goldman’s 19 study demonstrated that from 95 regimens only 81% ciprofloxacin dosing were correct, 5.7 % had suboptimal dosing, whereas 13.3% were over dose. Feroche and Alemu 21 showed that 87% of the practices had the correct dose, which is lower than the percentage in our study.
Prolonged duration of ciprofloxacin therapy might be a factor for resistance, thus, appropriate duration of use according to the indication must be used. Our study revealed that 96.03% of the records had correct duration of use, 2.65% had longer duration, and 1.32% had shorter duration. These findings are similar to that of Feroche and Alemu, 21 which had demonstrated that 92.5% of the prescriptions had correct duration, 3% had longer duration, and 2.25% had shorter duration. On the contrary, the findings of Biru et al 20 demonstrated that only 52.5% of the records had correct duration, whereas 45% had longer duration and 2.5% had shorter duration.
There are a number of contraindications against the use of ciprofloxacin, such as pregnancy/lactation, children aged ≤18 years, and hypersensitivity. Our study demonstrated that 1.99% of records were for children ≤18 years, which is an absolute contraindication and contrary to the study of Biru et al, 20 which showed that in 10% of the cases the age group was <18 years. There were no pregnant/lactating women or documented hypersensitivity in our study or in the study of Biru et al. 20
Drug interactions might affect the efficacy and safety of therapy. This might be due to reducing the levels of the drug or decreasing its elimination from the body. There are various mechanisms by which interactions can take place, thus caution should be taken when combining two or more drugs together. In our study, most frequently prescribed drugs with ciprofloxacin are analgesics (acetaminophen, ibuprofen), antacids, chlorpheniramine, and prednisolone. According to patients’ records, 28.5% contained acetaminophen, 31.1% contained ibuprofen, 21.2% contained chlorpheniramine, 16.6% contained antacids, and 7.9% contained prednisolone. Biru et al 20 showed that 43.5% of records contained paracetamol and diclofenac each, while 1.25% contained antacids. Interaction with prednisolone might increase risk of tendinopathy/tendinitis; thus, caution should be taken when combining both drugs. 22 Combination with ibuprofen might potentiate the risk of central nervous system stimulation, such as tremors, involuntary muscle movements, or seizures. 23 Administration of ciprofloxacin with antacids containing Al-Mg (aluminum and magnesium) might lead to decrease in ciprofloxacin absorption, thus dose spacing is recommended. 24
We evaluated some of the serious adverse events that the patients encountered during their therapy with ciprofloxacin. These included QTc prolongation, tendinopathy/tendinitis, and dysglycemia. Dysglycemia occurred at 1.99%; 1.32% of patients had hypoglycemia and 0.66% had hyperglycemic events. Previous studies demonstrated the occurrence of dysglycemia among ciprofloxacin.25 -27 El Ghandour and Azar 25 demonstrated that ciprofloxacin has the ability to cause hypoglycemia. Mohr et al 26 showed that both hypoglycemia and hyperglycemia take place with ciprofloxacin therapy, with higher rates of hypoglycemia and more frequently in diabetic patients. The mechanism by which ciprofloxacin causes hypoglycemia can be explained through a stimulation in insulin release and inhibition of pancreatic h-cell adenosine triphosphate (ATP)-sensitive K+ channel.28,29 However, the mechanism by which ciprofloxacin causes hyperglycemia is not fully clear. Furthermore, QTc prolongation is a serious adverse event that might precipitate torsade de pointes. Studies show that it is unlikely for ciprofloxacin to cause QTc prolongation unless the patient has risk factors that might precipitate torsade de pointes.10,30,31 In our study, 2.65% of patients had prolonged QTc interval but no torsade de pointes or other types of arrhythmias have developed. Tendinopathy/tendinitis is a serious yet rare side effect of ciprofloxacin therapy. We evaluated the incidence of tendinopathy in our study and we found out that 3.97% of patients had tendinopathy/tendinitis. Several case reports demonstrated the incidence of tendinopathy/tendinitis.12,32 Risk of tendinopathy/tendinitis increase when ciprofloxacin is used in combination with oral corticosteroids as in patients with asthma or chronic obstructive pulmonary disease (COPD). The mechanism by which ciprofloxacin causes such an adverse event can be due to inhibiting tendon cell proliferation and causing cell cycle arrest at the G2/M phase, stimulating matrix-degrading protease activity from fibroblasts and exerting an inhibitory effect on fibroblast metabolism, and inhibiting tenocytes outgrowth from the explants, migration of tenocytes, through the suppression of FAK (focal adhesion kinase) phosphorylation.33 -35 Although these adverse events are rare, yet their adverse outcomes on patients necessitates the awareness of clinicians.
Study Limitations
Unavailability of adequate literature on the subject matter of the study and poor patient medical history taking practice are a limitation to the study. Moreover, relatively small sample size and short duration of the study period might affect the generalization of the results.
Conclusion
Drug utilization evaluation of ciprofloxacin is necessary to ensure safe and effective prescribing; hence, optimal patient outcomes. Our study proved that prescribing of ciprofloxacin was according to international treatment guidelines. Dosing regimen was appropriate according to age group and indications. Duration of use was appropriate for the disease condition, with few prescriptions containing longer duration that might lead to AMR. Ciprofloxacin use in combination with possible interacting drugs is an important issue that should be addressed. Serious adverse events due to ciprofloxacin therapy is a critical issue that clinicians should be aware of. More studies with a larger sample size and longer duration should be encouraged to better evaluate the results.
Footnotes
Acknowledgements
The authors are very grateful to all personnel working in community pharmacies for helping in collecting data, for their assistance, and useful contributions.
Author Contributions
GD and RW reviewed the literature and constructed the study design. Collection of clinical data was performed by GD. All authors wrote, revised, and approved the final manuscript.
Data Availability
All relevant data are available in this article. The data presented in this study are available upon request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved and conducted in accordance with the declaration of Helsinki. This study is registered in clinical trials.gov, with code no. NCT05916105.
Consent to Participate
Consent was obtained from all participants in this study.
Consent to Publish
Not applicable.
