Abstract
Highlights
Utilization of dalbavancin was associated with an observed increase in treatment completion and was not found to result in a statistically significant increase in cost of care at our organization.
Background
Infections associated with injection use are an important consequence of the opioid epidemic and hospitalizations related to this have increased, with some hospitals experiencing a 15-fold increase in the past decade.1,2 In particular, persons who inject drugs (PWID) have a substantially increased risk for bloodstream and deep-seeded infections.3 -5 In addition, persons who use drugs (PWUD) may be more likely to delay pursuit of medical attention leading to further progressed, more severe infections.3 -6 For these reasons, this patient population often receives extended courses of antibiotics, and first-line treatment options are limited to a few intravenous (IV) options. 7 Extended durations of IV antimicrobials often involve discharging patients with longstanding IV access for antibiotic administration, with the access to be maintained by the patient or visiting nurse. There have long been concerns with discharging PWID or patients with any variety of substance use disorders with IV access related to potential misuse or inappropriate care of the access.7,8 Consequently, PWUD may have to be maintained in the hospital or postacute care facilities to ensure adherence.
There is a paucity of effective strategies that abate challenges specific to PWID therapy completion, with only utilization of oral antibiotics with high bioavailability and attempts at daily infusions prevailing as potential avenues for treatment completion. However, these strategies give rise to their own set of challenges that may negate their utility. Among prescribers, there is a documented concern that utilization of oral agents may lead to a decline in adherence, which has been substantiated by clinical data.9,10 While daily infusions remove the necessity of indwelling catheters, they may create challenges if a patient has issues with transportation or lack of safe and stable housing and this method is associated with high rates of treatment failure. 7 An option that could allow for infrequent administration is needed to obviate these concerns and facilitate compliance in PWUD. One such option is dalbavancin (DAL), a long-acting single infusion lipoglycopeptide antibiotic with a half-life of 346 hours. 11
While DAL is approved only for bacterial skin and skin structure infections, data supporting the potential use of DAL for more challenging to treat infections, such as Staphylococcus aureus blood stream infections (SABSI), osteomyelitis, and endocarditis have been published.12 -17 In addition, DAL use has been associated with benefits particularly in PWID, including decreased hospital stay and reduction in patients leaving against medical advice (AMA).17 -20 While the utility of DAL has been demonstrated, hesitancy to adopt its use in health care systems might exist due to the cost-prohibitive nature of DAL and perceived issues with insurance reimbursement. However, several studies have suggested significant cost savings based on projected cost avoidances.17 -23
In 2019, a retrospective review was conducted to assess whether there may be a role for the use of DAL at Baystate Medical Center (BMC) with the aim being reduction of inpatient length of stay (LOS). Seventy-six of these patients were identified as having IV drug use in their past medical history (PMH). Of the 76 patients, 16 patients had antibiotic courses that could have been completed within 14 days of discharge. For the purposes of this evaluation, we assumed each patient would have received DAL 14 days from planned end of treatment date and it was calculated that 115 inpatient hospital days would have been saved. While the potential reduction in LOS was promising, the most impactful finding of this research was the discovery that only 29 of the 76 patients (38%) completed their full course of IV therapy. As a result of these findings in 2020, a new DAL institutional pathway was introduced at BMC, with the goal of improving completion of IV therapy in PWUD and reducing hospital LOS.
The objective of our current study is to determine the cost effectiveness of this DAL institutional care pathway in a population of PWUD in actual, measured health care utilization data as well as the effect on treatment completion.
Methods
DAL Institutional Care Pathway Methods
Dalbavancin eligibility criteria were developed with collaboration between the antimicrobial stewardship pharmacists (ASPs) and infectious disease (ID) Physicians at BMC to select for patients who could not comply with first-line treatment options due to inability to care for longstanding IV catheters or to be discharged to a postacute care facility (Table 1).
Eligibility Criteria of the Dalbavancin Institutional Care Pathways.

Abbreviation: ID, infectious disease.
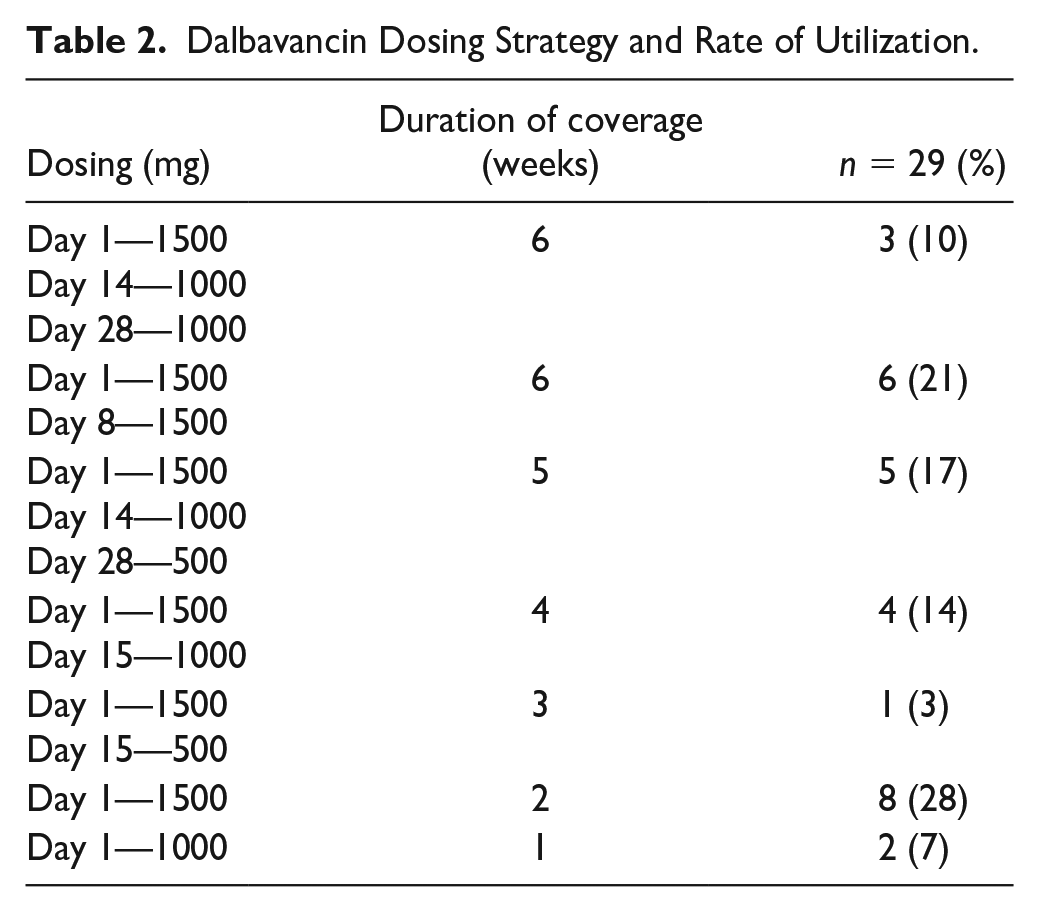
Patients with bacteremia and other serious gram-positive infections being followed by ID Consult Services were assessed for DAL candidacy by ASP. If patients were found to meet eligibility criteria, ASP would establish a dosing schedule (Table 2). Antimicrobial stewardship pharmacist also collaborated with Case Management and Regional Medical Authorization Coordinators for Infusion Services for an insurance benefits investigation, to schedule infusion appointments, and to establish effective posthospital discharge communication. Finally, education about DAL and necessary steps to complete treatment were provided to the patient by ASP, ID physicians, and case management.
Dalbavancin Dosing Strategy and Rate of Utilization.
Cohort Study Methods
Study location, design, and eligibility
This is a retrospective cohort study of PWUD receiving DAL at BMC, a 700-bed academic medical center, with comparison of microcost data between the 2 cohorts. All patients receiving an inpatient dose of DAL were reviewed, and only patients with a history of substance abuse were included. Data were collected on demographics, characteristics of infection and care, and rate of treatment failure with DAL. Cost and health care utilization information were also assessed for PWUD treated with DAL and a random sample of PWUD with SABSI treated prior to the institutional availability of DAL. Cost data for both groups were provided by the BMC finance department based on actual itemized hospital bills including the entire admission cost and revenue data from the described patients.
Data extraction
Data were collected via chart review though BMC’s Clinical Information System (CIS) to determine patient demographics, treatment plan, and progression of care. Clinical outcomes included 30-day and 90-day mortality, 30-day and 90-day readmission, 30-day and 90-day readmission due to infection, recurrence of infection, loss to follow-up, and treatment failure.
Definitions
Deep-seated infections
Osteomyelitis or septic arthritis, prosthetic joint infection, infective endocarditis, or other endovascular infection.
Treatment failure
Infection-related readmission, lack of clinical response necessitating change to an alternate agent with gram-positive spectrum of activity, or if antibiotics were used beyond the previously established end date of therapy recommended by ID at ID follow-up appointment or during readmission for that infection.
Compliant
Received all intended doses of DAL or were up-to-date with DAL doses when failure occurred.
Non-compliant
Did not receive all intended doses of DAL according to the established treatment schedule established by ASP. This includes patients who were lost to follow-up.
Data analysis
Cost information of the DAL population was compared with 20 randomly selected PWID with SABSI who were managed before the institutional availability of DAL. Both cohorts’ cost information was obtained via review of financial records for each admission. Direct cost represented the costs to the department and was variable depending on volume. Net revenue denotes reimbursement received from each case. Subtracting the direct cost from the net revenue yielded the contribution margin (what the case contributed to the bottom line). Microsoft Excel was used to perform descriptive statistics on the DAL population, Student’s t-test was utilized to compare cost data between the DAL and non-DAL patients. This study was approved by the BMC Institutional Review Board.
Results
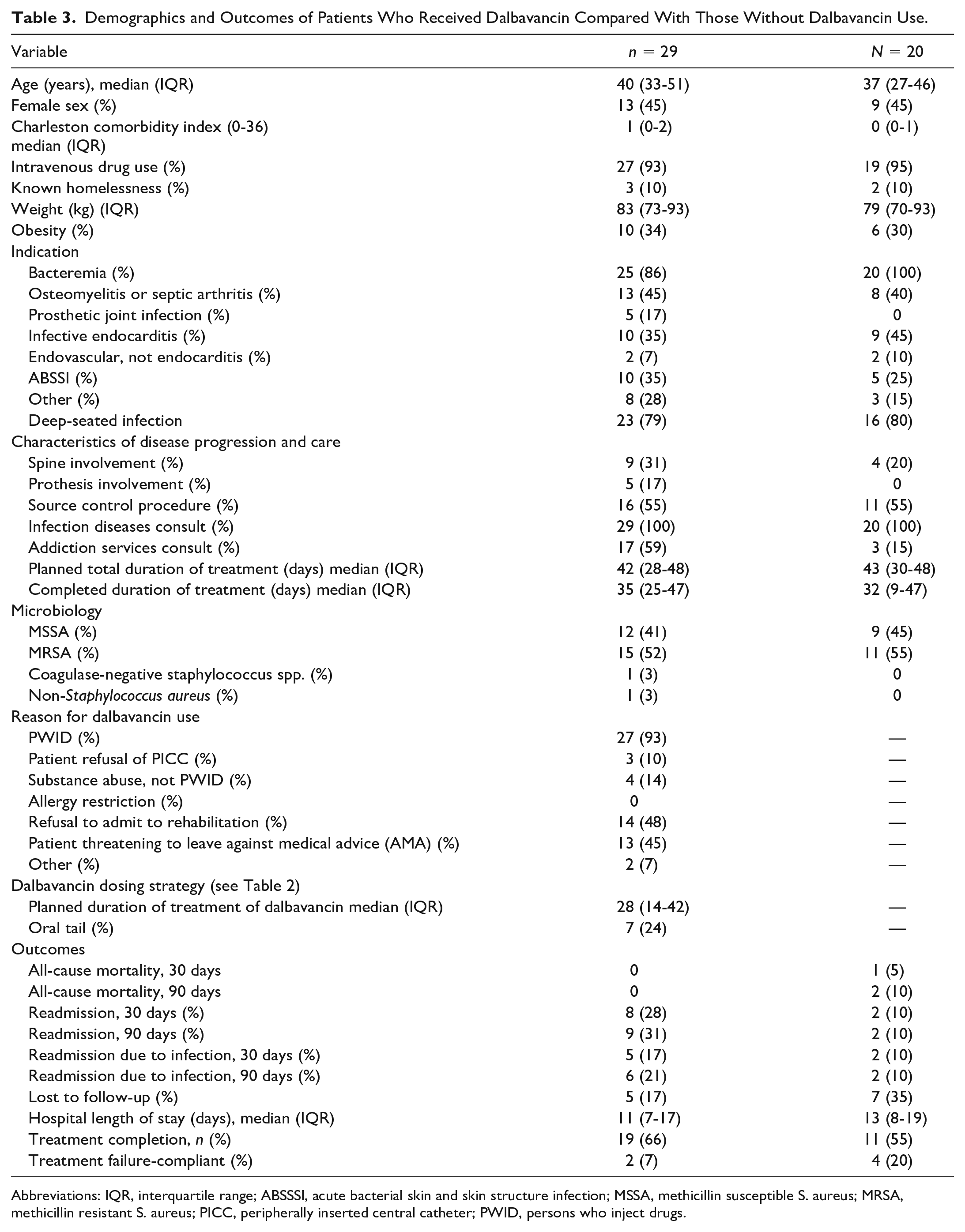
From June 2020 to January 2022, 38 patients received a dose of DAL. Nine patients who did not have substance use noted per chart review were excluded from the cohort study. Demographics are described in Table 3. Most patients were male (55%) in both groups with a median age of 40 in the DAL group compared with 37 in the non-DAL group. The majority of patients in both groups were intravenous drug user (IVDU) (93% in the DAL group and 95% in the non-DAL group) and this was also the primary reason for selection of DAL (93%). In the DAL cohort, S. aureus accounted for 27 (93%) of isolated pathogens, with 12 (41%) being resistant to methicillin. In the control group, S. aureus accounted for 100% of pathogens, as required by the selection criteria, with 11 (55%) patients having methicillin resistant S. aureus.
Demographics and Outcomes of Patients Who Received Dalbavancin Compared With Those Without Dalbavancin Use.
Abbreviations: IQR, interquartile range; ABSSSI, acute bacterial skin and skin structure infection; MSSA, methicillin susceptible S. aureus; MRSA, methicillin resistant S. aureus; PICC, peripherally inserted central catheter; PWID, persons who inject drugs.
Twenty-five (86%) DAL patients were bacteremic during their admission compared with 20 (100%) in the non-DAL group. Twenty-three patients (79%) and 16 (80%) in the DAL and non-DAL groups, respectively were considered to have deep-seated infections. The most common diagnoses included osteomyelitis or septic arthritis, which was present in 13 patients (45%) in the DAL group and 8 (40%) in the non-DAL and infective endocarditis in 10 patients (35%) managed with DAL and 8 (45) without. The listed indications are not mutually exclusive.
Nine (31%) DAL patients had spine involvement compared with 4 (20%) non-DAL patients and 5 (17%) DAL patients had prosthetic involvement; none of the non-DAL patients had prosthetic involvement. Every patient on and off DAL had an ID consult whereas 17 (59%) DAL and 2 (15%) non-DAL patients received consultation by addiction services. For the DAL group, the median overall planned treatment duration was 42 days while the median DAL treatment duration was 28 days. The median completed duration of treatment was 35 with 19 (66%) patients having completed the intended duration of treatment. In the non-DAL group, the median planned treatment duration was 43 days and the median completed days was 32. Dosing strategies utilized for the administration of DAL are represented in Table 2.
Patient outcomes are depicted in Table 3. The mortality rate at 30 and 90 days was 0 in the DAL cohort compared with one patient expiring (5%) within 30 days in the non-DAL group. In the DAL group, 5 patients (17%) were readmitted due to infection within 30 days and 6 patients (21%) were readmitted due to infection within 90 days; for the non-DAL group, 2 (10%) of patients were readmitted and both were due to infection. For the DAL and non-DAL groups, respectively, 5 (17%) and 7 (35%) patients were lost to follow-up. For DAL, treatment failure occurred in 2 (7%) patients in the compliant group and 8 (28%) in the non-compliant group. In the non-DAL group, treatment failure was observed in 4 (20%) of compliant patients compared with 7 (35%) who were non-compliant. The median LOS (Table 3) was 11 days in the DAL group compared with 13 days in in the previous study’s non-DAL group (P = 0.12); the current study’s non-DAL group also had a median of 13 days.
Patients who received DAL yielded median direct cost of $13 518 (Table 4) with a reimbursement median of $21 758 resulting in a contribution margin of $7180 (IQR = $2753-$11 754). Comparatively, patients without DAL use had a median direct cost of $11 567 with a median reimbursement of $18 222 producing a median contribution margin of $6655 (IQR = $1833-$17 668). There was no statistically significant difference between direct cost (P = 0.47), reimbursement amount (P = 0.71), and overall contribution margin (P = 0.47).
Comparative Cost Data Between Patients Who Received Dalbavancin and Patients With SABSI Who Received Standard Care.

Abbreviation: IQR = interquartile range; SABSI = Staphylococcus aureus blood stream infections.
Discussion
Providing high-quality care to PWID with serious infections poses a challenge due to the preclusion of standard of care antibiotics due to concerns with adherence and upkeep of long-term IV access. While guidelines and historical treatment data typically guide our care plans for deep-seated infections, they are often difficult to implement safely and complete successfully in PWUD. Dalbavancin, while not first line for any of the infections commonly seen in this patient population, offers an alternative that may increase the chance of patients receiving full treatment. The efficacy of DAL has been established with several studies looking at many types of infections, including deep-seated infections.12 -17 While the purpose of our study was not to assess efficacy, the observed low rate of treatment failure is promising—as our population consisted almost entirely of complicated, difficult to treat infections.
In addition to being an effective option, other benefits to DAL use have been observed. Dalbavancin has been shown to have the potential to decrease LOS, rates of patients leaving AMA, and progression of infection.17 -23 In an era of hospital-bed shortages related to the COVID-19 pandemic, the benefit of shorter LOS cannot be understated. In a study conducted by Morrisette and colleagues, they saw a reduction in LOS of 9 days with DAL or oritavancin use, and this difference was more profound in PWUD with a difference of 20 days. 17
While DAL is a promising option for this patient population, it is the cost that often leads to restricted use and underutilization of this option. However, several studies suggest a potential for cost savings with this agent. Streifel and colleagues, based on their observed reductions in LOS, surmised a theoretical mean cost-saving per patient of $40 414. 21 However, only less than half of the patients were PWUD. Vazquez Deida et al 22 selected specifically for vulnerable patients (with most having substance use disorders) and were able to see high rates of clinical success (81%) with a cost avoidance of $9600 per patient. However, this was selective for patients who only required 1 dose of DAL that was given inpatient—ensuring perfect compliance, and potentially limiting the opportunity of DAL to complete larger portions of therapy. In the previously mentioned retrospective cohort by Morriessete et al, 17 a cost minimization analysis was performed to predict a health care savings of $19 555 in non-PWUD and $40 455 in PWUD. Alexander et al 24 studied a population of 66 patients, most of whom had deep-seated infections, and projected a cost savings of $1.1 million in their population when DAL was used. Similarly, Leuking et al 25 estimated a cost savings of $1.2 million dollars, based on a projected avoidance of 566 inpatient days across 40 patients. Their population included a majority of complicated infections and 67.5% of their patients had substance use disorders. Importantly, when selecting for PWUD with a higher likelihood of leaving AMA—it can be difficult to rely on cost data that assume hospitalization for the entire treatment duration. In our study, we measured actual LOS and were only able to show a modest decrease in hospital LOS from a median of 13 to 11 days. Our study adds to the previously reported data by comparing cost of care in actual, measured health care dollars in a complicated patient population that consists mostly of PWID, many of whom required multiple DAL doses.
Our patient population of DAL recipients consisted of the complicated infections that are often seen in PWUD with 86% having had bacteremia during their stay and 79% having a deep-seated infection with 90% caused by S. aureus. The median duration of planned therapy being a 6-week course. Because of the complexity of these infections and the required treatment duration for most, it is unsurprising that over half of these patients required more than 1 dose of DAL. In addition, the necessity of multiple doses likely expounds the relatively high rate of lost to follow-up compared with other studies. While the lack of mortality seen our patient population is encouraging, this was an anticipated finding as all of these patients were deemed clinically stable enough for discharge—as required in the DAL Pathway eligibility criteria (Table 1). What is most encouraging is that 66% of patients completed therapy, which was much improved from the 38% completion rate previously observed in a similar patient population of PWID at BMC.
In terms of cost data, we were unable to corroborate the theoretical substantial cost savings seen in other literature. However, we were able to show no statistically significant difference in direct cost, reimbursement rates, or contribution margins with real, measured cost data. Numerically, it can be seen that the contribution margins were similar, with DAL use resulting in higher amount of $7180 compared with the $6655 seen without its use.
When considering adaptation of a similar program at other institutions, several potential barriers to implementation and limitations of this study should be acknowledged. Our results are largely affected by selection bias, as ID trained pharmacists and physicians selected patients who would benefit most from DAL. Provider discretion was used, and alternative therapy was pursued in patients who did not express a commitment to return for follow-up dosing. This is also a potential down-side to the program as it can be time intensive to screen these patients. At BMC, only a select few pharmacists were trained to do so, therefore, the labor was concentrated on a few individuals. In addition, this was a retrospective, single center study at an institution that is part of a large health care system, which limits generalizability to smaller health care systems. It is also possible that some patients were readmitted to other hospitals in the area, and we are unable to account for this, however, our health care system does represent the largest care provider in the area. Finally, this was not a large pharmacoeconomic study with considerations of more of the complex variables related to cost. For example, we did not consider additional revenue gained from higher-bed turnover by tracking revenue from subsequent utilization of resources based on reduced LOS. However, this suggests that our estimate is conservative and additional cost savings related to DAL use may have occurred that were not accounted for. Therefore, utilization of a similar clinical pathway is likely to not be significantly more expensive than other options currently in use, such as oral antibiotics on discharge, with a potential to increase rates of completion of intended duration of treatments.
Future research should include a wider range of economic variables and should be done in different institutions to enhance the generalizability of these findings.
In conclusion, this was a retrospective cohort study that characterized patients enrolled in our DAL institutional pathway and determined the difference in treatment completion and cost between patients with and without DAL use. Dalbavancin patients had a 0% rate of mortality, 31% rate of 90-day readmission, and 7% rate of treatment failure in compliant patients. When DAL was used, there was no statistically significant difference in the contribution margin that averaged at $7180 and $6655 for DAL and normal care, respectively (P = 0.47). Dalbavancin use in select patients with infections requiring long-term use of antibiotics may be a viable treatment option, with no statistically significant difference in contribution margins.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
