Abstract
Background
Safe and effective administration of intravenous (IV) antibiotics in a resource-optimized fashion is essential to meet the needs of today’s cost-constrained healthcare systems. Moreover, in a pandemic setting, when healthcare resources are under increased and continual strain, finding ways to relieve even a small portion of that burden becomes doubly important. 1
The MINI-BAG Plus Container System (Baxter HealthCare Co., Deerfield, IL) and VIAL-MATE Adaptor (Baxter HealthCare Co.) are widely used point-of-care systems for the administration of intravenously delivered drugs and drug products. The MINI-BAG Plus Container System contains a sterile, nonpyrogenic solution (ie, 0.9% sodium chloride injection or 5% dextrose injection) with an integral drug vial adaptor and is used for IV administration after admixture of the diluent with a single-dose powdered drug vial. 2 The VIAL-MATE Adaptor is a device that connects a standard 20-mm single-dose drug vial to an IV solution container allowing pharmacists or healthcare practitioners to mix the contents immediately before administration of the reconstituted drug to the patient. 3
The MINI-BAG Plus Container System and VIAL-MATE Adaptor aim to expedite and improve the ease of administration of IV drugs. Studies have demonstrated that, compared with conventional methods, the MINI-BAG Plus Container System can reduce the time to preparation of IV drugs,4 -6 the costs associated with drug preparation,4,6,7 and the risk of microbiological contamination. 5 Similarly, the VIAL-MATE Adaptor has been shown to be associated with drug-preparation time savings, compared with standard reconstitution methods, and to lead to fewer problems with drug reconstitution. 8 There is a need for cost-effective and convenient alternative modalities like these to facilitate the administration of antibiotics not just within hospitals but also in the outpatient setting, for patients no longer requiring hospitalization. 9 However, it is imperative to ascertain that drug vials are physically compatible with point-of-care systems and that there are no physicochemical changes in the properties of medications that could potentially reduce their potency during treatment. 10
Cefiderocol is the first siderophore cephalosporin antibacterial agent approved in the United States for the treatment of hospital-acquired or ventilator-associated bacterial pneumonia and complicated urinary tract infections caused by susceptible Gram-negative microorganisms. 11 In Europe, cefiderocol is approved for the treatment of patients with infections caused by susceptible Gram-negative bacteria with limited treatment options. 12 Cefiderocol has demonstrated potent in vitro activity against a broad range of Gram-negative bacteria, but not against Gram-positive bacteria or anaerobes. 13 The recommended dosing regimen of cefiderocol is 2 g every 8 hours over a 3-hour IV infusion, with dose adjustments as required depending on renal function; 11 the minimum administration volume for a 2 g dose is 100 mL. The product is supplied as 1 g of white to off-white sterile lyophilized powder for reconstitution in single-dose, clear-glass vials sealed with a rubber stopper and an aluminum seal with flip-off cap. 11 Reconstituted and diluted cefiderocol is stable for up to 6 hours at room temperature, including the infusion time, or for up to 24 hours at 2°C to 8°C while being protected from light. 11 Because cefiderocol is stable at room temperature or 4°C, it is an ideal candidate for use with point-of-care administration systems.
Objectives
This study was conducted to investigate the physical compatibility of the commercially available MINI-BAG Plus Container System and VIAL-MATE Adaptor with the drug product vials used for cefiderocol.
Methods
The physical compatibility of cefiderocol 1 g vials with both the MINI-BAG Plus Container System and the VIAL-MATE Adaptor was assessed at room temperature using predefined criteria, selected based on the operating instructions of the devices and elements judged by the investigators to indicate successful functioning of the assembled system (Table 1).
Predefined Physical Compatibility Criteria for Cefiderocol Vials With the MINI-BAG Plus Container System and the VIAL-MATE Adaptor.
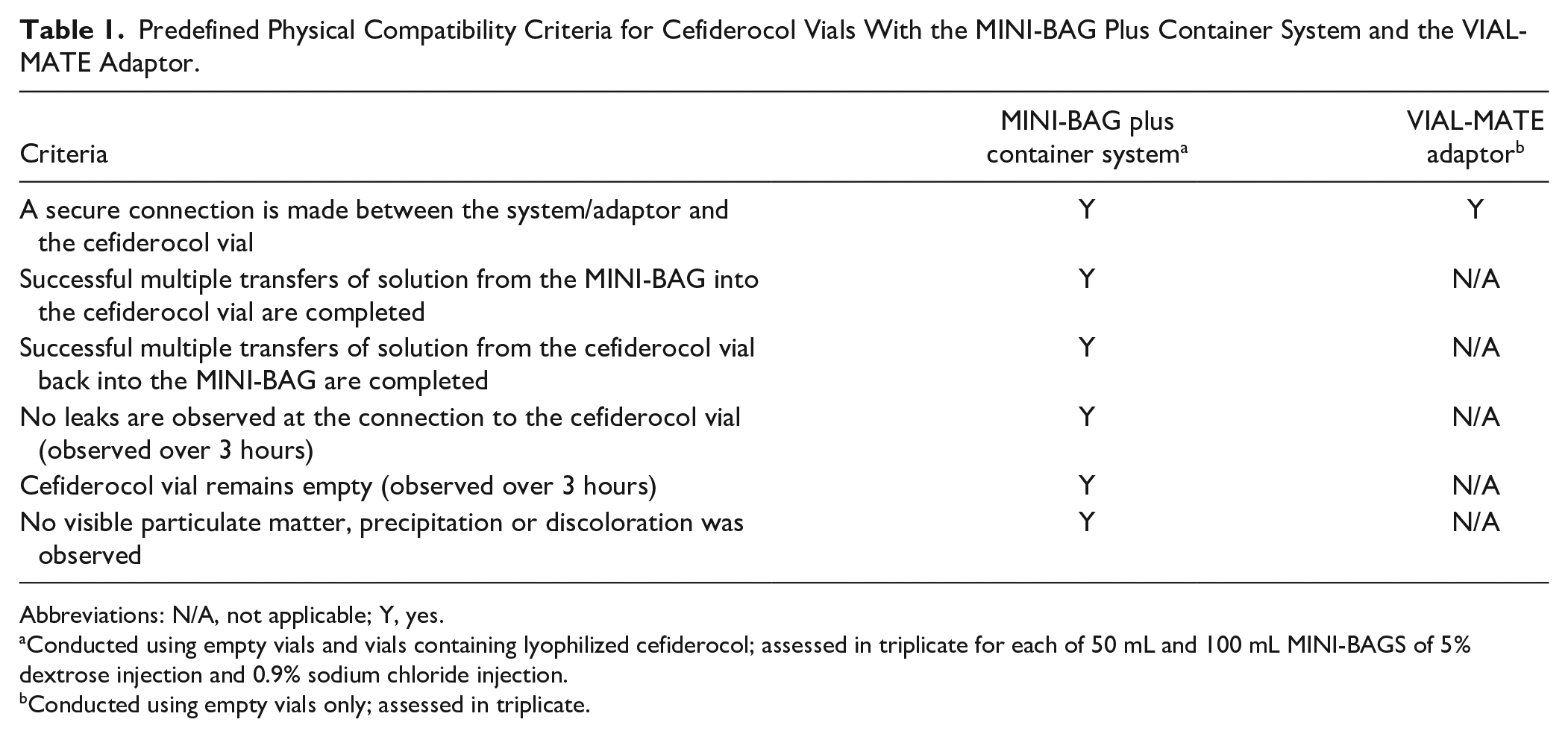
Abbreviations: N/A, not applicable; Y, yes.
Conducted using empty vials and vials containing lyophilized cefiderocol; assessed in triplicate for each of 50 mL and 100 mL MINI-BAGS of 5% dextrose injection and 0.9% sodium chloride injection.
Conducted using empty vials only; assessed in triplicate.
MINI-BAG Plus Container System Physical Compatibility
Testing was performed at 2 centers, 1 investigating empty vials (Shionogi Inc.), the other investigating vials containing lyophilized cefiderocol powder (Shionogi & Co., Ltd.). At each center, 2 individuals took part in the study: 1 individual was responsible for performing all connections and the other was responsible for investigating the integrity of all the connections. The cefiderocol vials were stored at 2°C to 8°C (36°F to 46°F) and protected from light in their carton until time of use. Empty vials were stored at room temperature.
Qualitative physical compatibility was assessed in triplicate with 50 and 100 mL MINI-BAGS of 5% dextrose injection or 0.9% sodium chloride injection. The MINI-BAGS and the empty vials or the vials containing lyophilized cefiderocol were connected according to the manufacturer’s instructions. All vials were attached to the MINI-BAG Plus Container Systems individually. A breakaway seal in the tube between the vial adaptor and the diluent bag was broken using both hands. In step 1 of the mixing process, the vial was held upright and the MINI-BAG, held directly above it, was squeezed until the vial was approximately half-full of the diluent (Figure 1a). The vial was swirled gently to mix the drug with diluent. In step 2, the system was inverted, so that the vial was held upside down in one hand and the MINI-BAG supported beneath it with the other hand. The bag was squeezed rhythmically to force air into the vial and, between each squeeze, to allow the contents of the vial to drain into the bag. These 2 steps were repeated until mixing and transfer was completed, ensuring that no liquid was left in the vial. The vial was not removed from the bag. At this stage, the mixture was ready for administration to the patient. The MINI-BAGs with connected cefiderocol vials were hung for 3 hours (Figure 1b); the solution was inspected visually for particulate matter, any precipitation, and discoloration, as well as for leaks from the vial connections and/or leaks back into the vial.
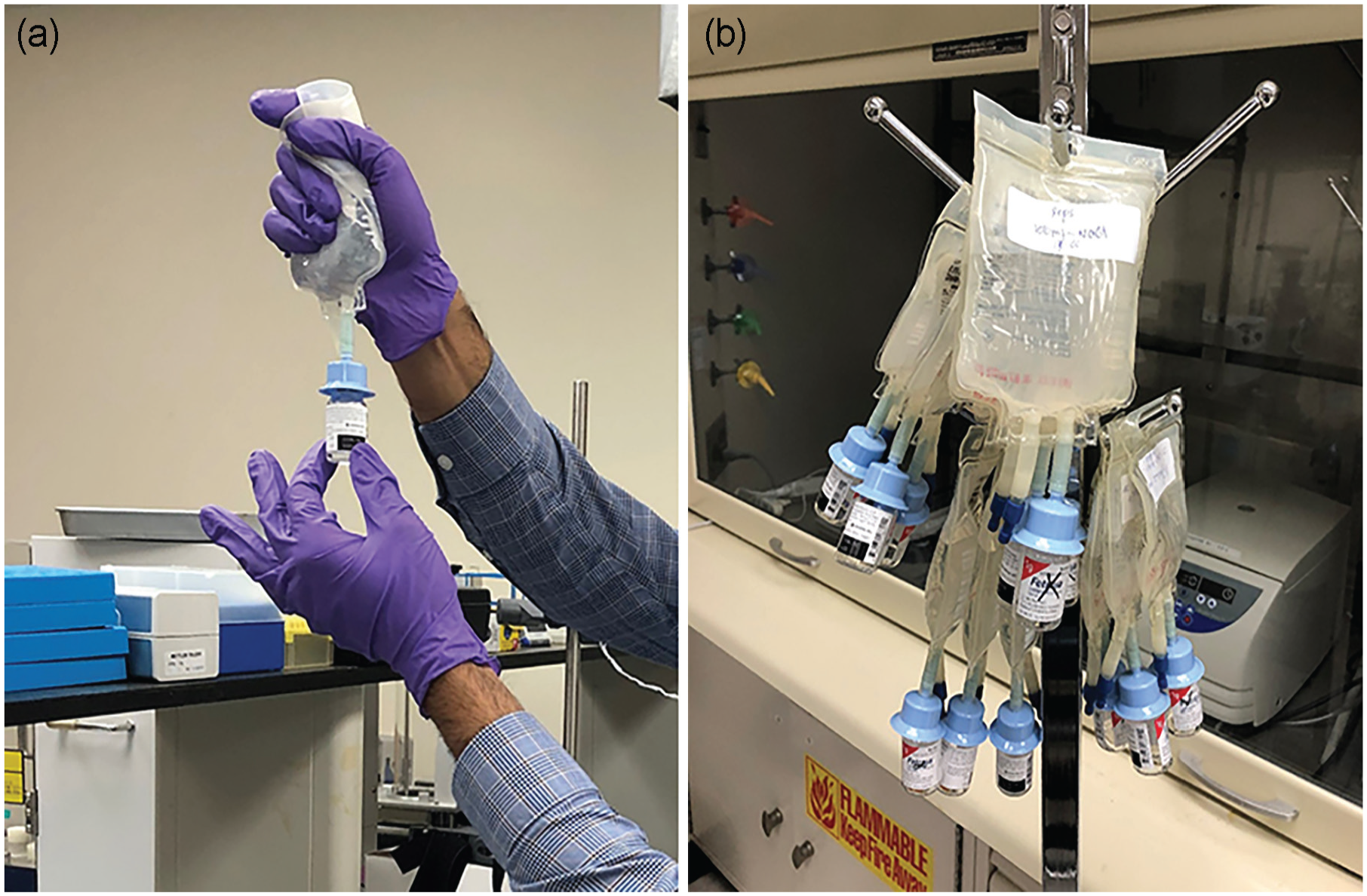
Physical compatibility between the MINI-BAG plus container system and cefiderocol vials. (a) Transfer of 5% dextrose injection or 0.9% sodium chloride injection into vials of cefiderocol. (b) Vials of cefiderocol connected to the MINI-BAG, observed over a period of 3 hours.
VIAL-MATE Adaptor Physical Compatibility
Testing was performed by 2 individuals at 1 center on empty cefiderocol vials only (Shionogi Inc.): 1 individual was responsible for performing all connections and the other was responsible for investigating the integrity of all the connections. Empty vials were stored at room temperature. The physical compatibility of the vials with the VIAL-MATE Adaptor was assessed in triplicate. With the upright vial placed on a hard surface, the VIAL-MATE Adaptor was firmly pushed straight down onto it, without twisting, until the device audibly clicked into place, at which point the connection was deemed to be secure (Figure 2).
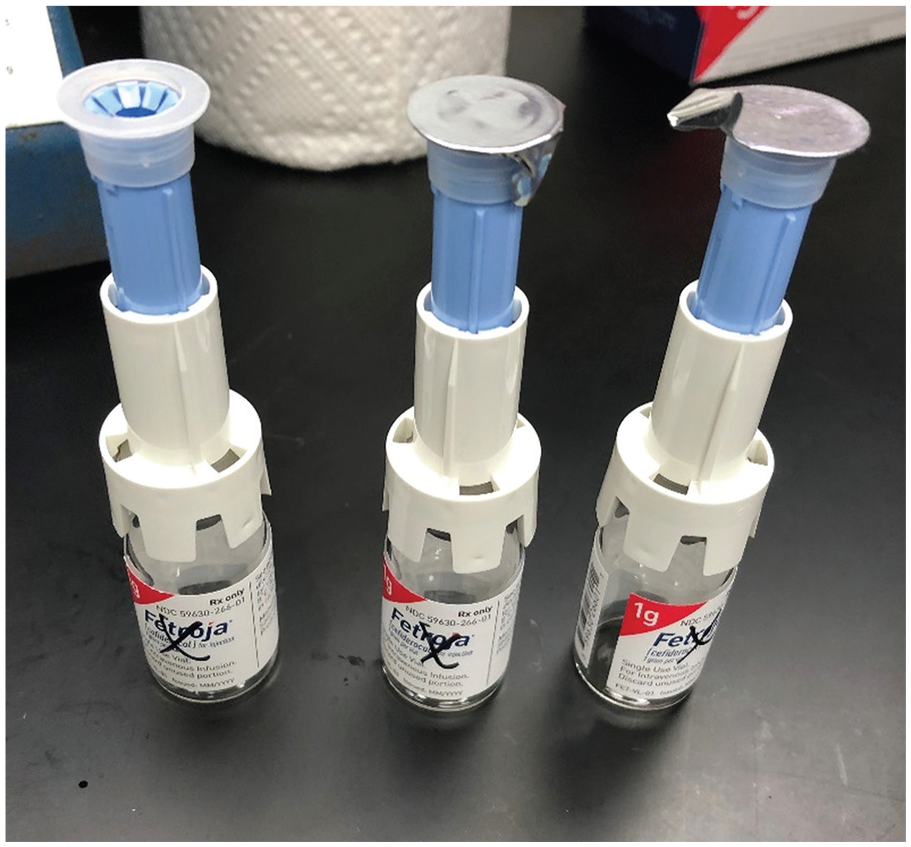
Physical compatibility between the VIAL-MATE adaptor and cefiderocol vials.
Results
In the first part of the investigations, all predefined criteria to establish physical compatibility between the cefiderocol vials and the MINI-BAG Plus Container System were met (Table 1). Thus, a secure connection between the MINI-BAG and the cefiderocol vial was achieved, and the diluent could be transferred and drained from the MINI-BAG into the cefiderocol vial several times. Cefiderocol was successfully reconstituted in either 5% dextrose or 0.9% sodium chloride for both the 50 mL and 100 mL MINI-BAGs. All reconstituted cefiderocol was transferred successfully into the MINI-BAG, and no backward leaking into the vial or from the connections was detected visually over a 3-hour period. In addition to successful fluid transfer between the vial and MINI-BAG Plus Container System and the lack of leaking during and after vial connection, there was also no particulate matter in, or precipitation or discoloration of, the prepared solution over 3 hours.
In the second part of the investigations, a secure connection between the VIAL-MATE Adaptor and the cefiderocol vials was demonstrated. While some force was required to connect the adaptor to the vial, the amount needed was not excessive and should be well within the capability of most operators.
Discussion
A secure connection between the MINI-BAG and the cefiderocol vial was achieved, without leaking or the presence of particulate matter or precipitation in the prepared solution over 3 hours. In addition, a secure connection between the VIAL-MATE Adaptor and the cefiderocol vials was demonstrated.
Cefiderocol, with a recommended dosing regimen of 2 g every 8 hours over a 3-hour IV infusion, 11 has documented compatibility with Y-site administration. 14 The fact that cefiderocol is stable for up to 6 hours at room temperature, including the infusion time, following reconstitution 11 underlines its suitability for use in both hospital and outpatient parenteral antibiotic treatment settings 15 with the MINI-BAG Plus Container System and VIAL-MATE Adaptor systems.
The use of MINI-BAG Plus Container System and VIAL-MATE Adaptor systems in the reconstitution and delivery of un-reconstituted cefiderocol immediately prior to administration, may increase the efficiency of cefiderocol administration in hospitals, outpatient settings or long-term healthcare facilities. In addition, because these systems minimize environmental and personal exposure, they reduce the risk of contamination, a feature that has been noted with conventional preparation methods. 5
Limitations of the study include its small size, although there are no reasons to suspect that the results would be different with a greater number of tests or investigators. Other potential challenges of the study, including a reliance on subjective assessment of drug compatibility and operator bias in systems’ handling, are also inherent in the use of many drug delivery systems.
Conclusion and Relevance
In this study, cefiderocol 1 g vials were shown to be compatible with the MINI-BAG Plus Container System and the VIAL-MATE Adaptor. The findings provide confidence that cefiderocol can be used easily and effectively with these systems across the range of treatment settings, including where resources are limited.
The MINI-BAG Plus Container System and VIAL-MATE Adaptor have widespread utility across a range of different drugs and therapeutic areas. They are used for the administration not only of drugs that require reconstitution prior to use,16,17 like cefiderocol and other antibiotics, but also ready-to-use products, 18 which can offer favorable safety, reduced waste and, in some cases, economic advantages, and may have particular applications for antimicrobial use in the outpatient setting. The results from this small study support the use of these established systems but should not be extrapolated beyond cefiderocol; the compatibility of other products should be confirmed on an individual basis.
Footnotes
Acknowledgements
Medical writing and editorial support were provided by Adrienn Kis and Joanne Shrewsbury-Gee, Highfield, Oxford, United Kingdom, sponsored by Shionogi & Co., Ltd., Osaka, Japan.
Authors’ Note
The data were presented at American Society of Health-System Pharmacists Mid-year Congress, December 5-9, 2021; virtual meeting: Lynch L, Rathod D, Manrodt R, Chen Z, Fam D, Slover C, Nguyen S, Kung F, and Ohno K. Cefiderocol for injection Compatibility testing using the MINI-BAG Plus Container System and the VIAL-MATE Adaptor. Poster 1077024.
Author Contributions
DR, JM, and ZC contributed to conceptualization and data collection. All authors contributed equally to the interpretation of the data, drafting, reviewing, and revision of the manuscript drafts; and approved the final draft for submission.
Declarations of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of Shionogi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The design and conduct of the study, data collection, and interpretation were funded by Shionogi Inc., Florham Park, NJ, USA, and Shionogi & Co., Ltd., Osaka, Japan.
