Abstract
Acute generalized exanthematous pustulosis (AGEP) is a rare severe cutaneous adverse reaction in neonates and is most commonly drug-induced. Vaccine-associated cases are uncommon in this age group. We report a term male neonate who developed clusters of non-follicular pustules on erythematous plaques approximately 6 hours after receiving a birth dose of hepatitis B vaccine. The infant remained clinically stable, without fever or systemic involvement, and had no exposure to concomitant medications. The rapid onset following a single exposure, characteristic morphology, and prompt resolution were highly suggestive of AGEP. Laboratory investigations and skin biopsy were not performed due to rapid clinical improvement. Infectious etiologies and transient neonatal pustular dermatoses were considered less likely based on the clinical course. Patch testing with the same vaccine lot was positive, supporting a type IV hypersensitivity mechanism. The eruption resolved almost completely within 48 hours with topical hydrocortisone, without complications or recurrence.
Keywords
Introduction
Acute generalized exanthematous pustulosis is a rare severe cutaneous adverse reaction characterized by the sudden onset of numerous sterile, non-follicular pustules on an erythematous base. Although most cases are triggered by medications, AGEP has also been reported following infections and, less commonly, vaccination. 1 Reports of vaccine-associated AGEP in neonates are limited. We describe a neonate who developed a pustular eruption shortly after hepatitis B vaccination, with clinical features strongly suggestive of AGEP.
Case Presentation
A term male neonate with a birth weight of 3.1 kg received a birth dose of hepatitis B vaccine (Herber Biovac HB, 0.5 ml, intramuscular injection in the thigh; lot number 3C0111/0) within the first 24 hours of life. Approximately 6 hours after vaccination, he developed clusters of pinpoints, non-follicular pustules on mildly edematous erythematous plaques localized on the back (Figure 1). He was alert, feeding well, afebrile (36.8°C), with no irritability or systemic symptoms. No concomitant medications had been given and there were no signs of infection; family history was relevant only to maternal food allergy (urticaria to shellfish).

Multiple pinpoint pustules scattered on an erythematous base, approximately 6 hours after vaccination.
Examination revealed multiple superficial pustules, apparently sterile on gross inspection, scattered over sharply demarcated erythematous plaques, without mucosal involvement or lymphadenopathy. Given the rapid clinical improvement and stable condition, laboratory investigations and skin biopsy were not performed. Based on the typical morphology, close temporal relationship to a single exposure, and rapid response to supportive care, AGEP was considered the most likely diagnosis. 1
Topical hydrocortisone 1% twice daily was started immediately after assessment. The lesions improved markedly after 24 hours and were almost completely resolved by 48 hours, leaving only a delicate peripheral collarette of desquamation; systemic corticosteroids or antibiotics were not required, and no complications were observed (Figure 2).
Same site 48 hours after initiation of topical hydrocortisone 1%, showing near-complete resolution of the eruption.
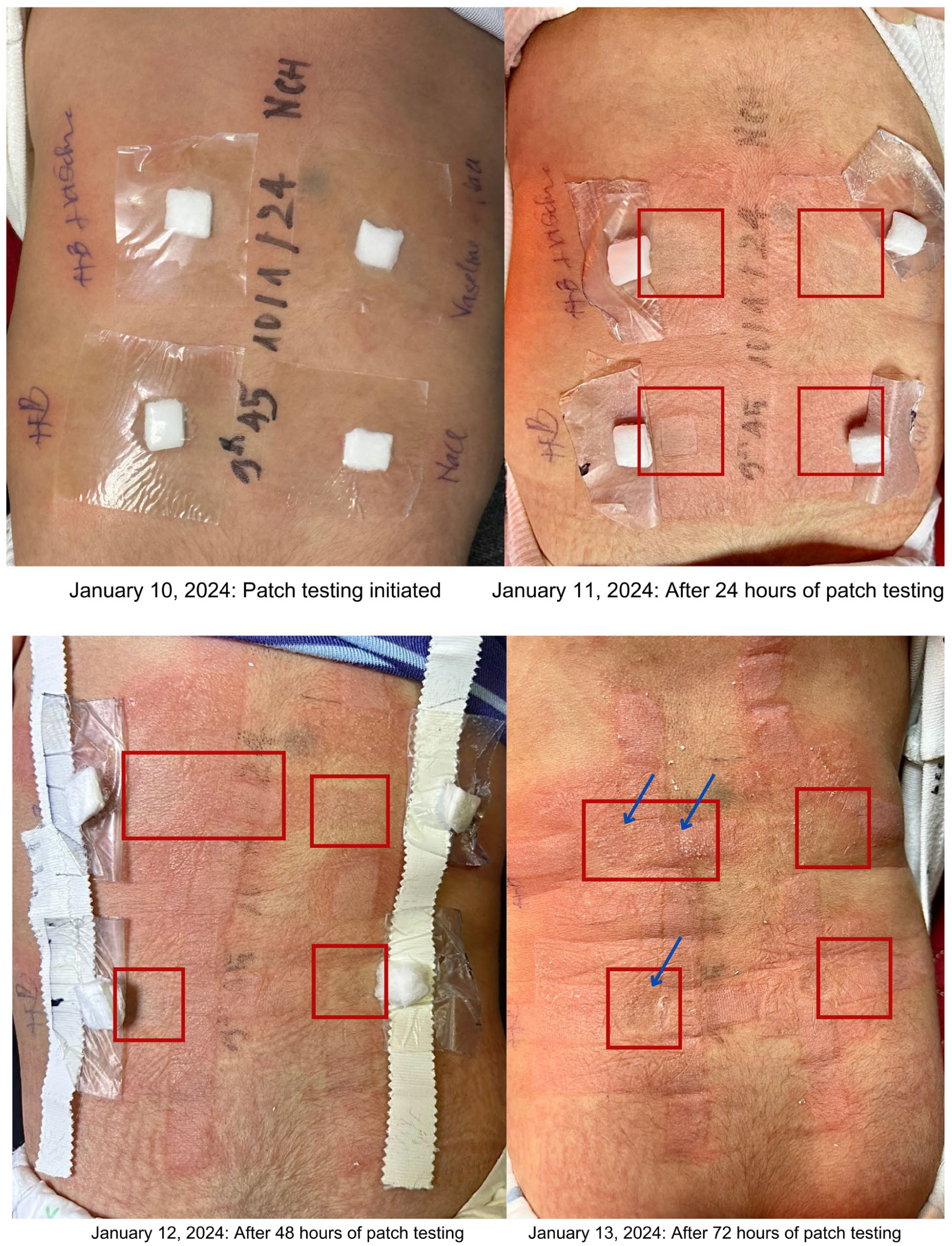
At the 2-month follow-up visit, delayed-type hypersensitivity testing was performed using the same vaccine lot. Patch testing (read at 24, 48, and 72 hours) was positive, supporting a type IV hypersensitivity mechanism and strengthening the causal link with the vaccine. 2 No recurrence or long-term sequelae were observed.
Discussion
AGEP is a severe cutaneous adverse reaction most commonly triggered by drugs and less frequently by infections or vaccines.1,3 Its pathogenesis involves T-cell–mediated responses, neutrophil chemotaxis (interleukin-8), and the interleukin-36 axis. 3 Vaccine-related AGEP has been reported after DTP, MMR, influenza, and COVID-19 vaccines, but, to our knowledge, AGEP in a neonate immediately after the birth dose of hepatitis B vaccine has rarely been documented.3 -5
The differential diagnosis of pustular eruptions in newborns is broad and includes both noninfectious and infectious conditions. 6 Neonatal candidiasis is an important infectious cause to consider, as it may present with widespread pustules and can be associated with mucosal involvement or systemic illness. Other infectious causes include bacterial pustulosis and viral infections such as herpes simplex virus.6,7 In the present case, the absence of fever, systemic features, mucosal involvement, and the rapid spontaneous resolution made infectious etiologies unlikely.
Although histopathologic confirmation was not available, the clinical presentation, short latency after a single exposure, characteristic morphology, rapid resolution, and positive delayed type hypersensitivity testing strongly support AGEP as the most plausible diagnosis. However, the lack of skin biopsy represents a limitation and precludes definitive diagnostic confirmation.
Conclusion
This case illustrates that AGEP can occur in neonates shortly after hepatitis B vaccination. Clinicians should consider AGEP in infants presenting sudden-onset pustular eruptions following immunization, while carefully excluding infectious causes. Documentation of vaccine lot numbers and, when feasible, delayed hypersensitivity testing may assist in guiding future vaccination decisions.
Footnotes
Author Note
This manuscript is original, has not been published previously, and is not under consideration for publication elsewhere.
Ethical Considerations
Ethics approval for this case report was obtained from the Institutional Review Board (IRB) of the Vietnam University of Traditional Medicine (IRB-VN01.014). Written informed consent was obtained from the patient’s parents for the publication of the clinical details and accompanying images.
Consent to Participate
Written informed consent to participate was obtained from the patient’s parents.
Consent for Publication
Written informed consent for publication of the clinical details and accompanying images was obtained from the patient’s parents. Identifying information has been removed to protect patient confidentiality. The written consent is retained by the authors and is available upon request by the journal.
Author Contributions
KVN and AVTN supervised the clinical management of the case. TMH conceptualized the report, contributed to the methodology and drafted the initial manuscript. KVN, TVC and AVTN contributed to data interpretation, and critical revision of the manuscript. All authors approved the final version and agreed to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of the Use of Generative AI
During the preparation of this work the authors used ChatGPT (OpenAI) in order to assist with language editing and improve clarity, as English is not the first language of all authors. Following the use of this tool, the authors reviewed and edited the content thoroughly and hereby declare full responsibility for the authenticity of the contents of this publication.
