Abstract
Solitary inflammatory cap polyp (ICP) is a rare rectal condition characterized by distinctive polypoid lesions with a “cap-like” appearance. It primarily occurs without underlying inflammatory bowel disease, making diagnosis and treatment challenging. We report the case of a 6-year-old female presenting with rectal bleeding and abdominal pain. Her medical history was unremarkable except for a recent episode of bloody diarrhea treated as amebiasis. Laboratory investigations revealed bicytopenia and hyponatremia. Colonoscopy identified a 1 × 1 cm sessile polyp in the rectum, which was excised and sent for histopathological analysis. The analysis confirmed ICP, characterized by polypoid rectal mucosa with crypt distortion, and inflammatory cell infiltration. The patient underwent successful polypectomy and received supportive care, leading to complete symptom resolution with no recurrence. This case highlights the importance of considering ICP in pediatric patients with rectal bleeding. Further studies are necessary to better understand the pathogenesis and establish standardized treatment guidelines for ICP.
Introduction
Solitary rectal inflammatory cap polyp (ICP) presents a significant clinical challenge due to its diverse clinical presentations and potential for misinterpretation. This uncommon entity, characterized by the presence of inflammatory rectal polyps, often develops in the context of long-standing inflammatory conditions such as ulcerative colitis or Crohn’s disease. However, it can also appear sporadically in the absence of underlying inflammatory bowel disease (IBD). The term “cap polyp” describes these polyps, which vary in size and form, because of their characteristic mushroom-like appearance. 1
ICP features polyps that are red and white capped, typically sessile, with elongated and only slightly twisted crypts, along with some surface erosion. Patients can have anywhere from one to hundreds of these polyps. 2 The rarity of the disease, combined with the challenges in diagnosing it, 3 contribute to the confusion with several other conditions. ICP predominantly affects females, with the highest incidence observed during their 50s. The age range of affected individuals spans from 5 to 90 years.4,5 Solitary rectal ICP can cause a range of symptoms, such as mucous discharge, rectal bleeding, and changes in bowel habits, even if they are benign. 6
The cause of ICP remains elusive. Williams et al 7 claimed that these polyps arise from chronic prolapse of the mucosa. This hypothesis is supported by the endoscopic features of ICP, their frequent occurrence within the left side of colon (particularly the rectum), and their concomitant presence with constipation—all characteristic findings of mucosal prolapse syndrome. Although ICP has been occasionally linked to colitis and colorectal cancer, it is plausible to infer that ICP may not be a direct result of prolapse injury. Rather, it could be a non-specific manifestation of an underlying inflammatory process. 8
Spontaneous resolution occurs only in a small percentage of cases. Unfortunately, there are no standardized clinical recommendations for the management of this condition. Therefore, optimal treatment strategies for the majority of patients with ICP have yet to be determined. Depending on the severity of symptoms, various treatment approaches may be considered, including polypectomy, medical therapy, or surgical resection of affected colonic segments. A number of ICP cases were successfully treated with anti-inflammatory drugs, antibiotics, and by clearing up any existing Helicobacter pylori (H. pylori) infections. 9 Here we present a case of solitary inflammatory cap polyp in a 6-year-old female child.
Clinical Presentation
A 6-year-old school-going child, previously healthy, presented 3 days ago with crampy lower abdominal pain of unknown etiology, with no aggravating, or relieving factors. She also experienced non-projectile vomiting of ingested material approximately 2 to 3 times per day, but no fever, chills, or rigors. She sought treatment at a nearby health center, where blood tests were performed and she was prescribed cephalexin and ibuprofen.
However, her condition has not improved. Three weeks before her current presentation, the patient experienced bloody diarrhea. After an investigation at that time, she was diagnosed with amebiasis and treated with metronidazole syrup, which relieved her symptoms. Her vaccination status is up-to-date according to the Ethiopian vaccine schedule. Currently attending KG-1, she is an academically proficient student. She has no past history of cough, shortness of breath, loss of consciousness, or abnormal body movements. Color changes in her urine or feces have also not been recorded.
Upon examination, her vital signs were within normal parameters. She exhibited appropriate weight and height for her age. The conjunctiva was pink, and the sclera was not icteric. No clinically significant lymph nodes were palpable. The chest exhibited clear and resonant sounds. Abdominal examination showed a flat abdomen with normal bowel sounds. The liver had a palpable border positioned 3 cm below the correct costal margin, with slight tenderness. The spleen was palpable 2 cm below the left costal margin, along the splenic growth line. There were no signs of fluid retention, and a per rectal examination revealed no abnormalities.
Following further investigations, a complete blood count revealed bicytopenia characterized by a white blood cell count of 3600 with neutrophilia (83%) and a platelet count of 34 000/microliter. Notably, her hemoglobin level was within normal limits at 14 gm/dl. Examination of her stool did not yield any evidence of ova or parasites. Serological testing showed negative results for hepatitis B and C viral markers. An assessment of serum electrolytes indicated hyponatremia with a low sodium level of 122 meq/l, while levels of other electrolytes including potassium, calcium, and chloride remained within normal ranges. The presence of hyponatremia in this patient could be attributed to several factors, including dilutional hyponatremia due to recent gastrointestinal fluid loss, potential electrolyte imbalance secondary to mild hepatosplenic schistosomiasis, or inadequate sodium intake during illness.
Abdominal ultrasound demonstrated a normal liver echotexture, however, the liver was mildly enlarged at 12.6 cm. Additionally, the portal vein wall was mildly thickened. A small amount of ascites was noted in the pelvis. The spleen was enlarged but exhibited normal echotexture. No bowel abnormalities or lymphadenopathy were detected. The ultrasound findings suggest mild hepatosplenic schistosomiasis with splenomegaly. The hepatosplenomegaly observed in this patient is likely due to mild hepatosplenic schistosomiasis, as suggested by the ultrasound findings of splenomegaly, mild ascites, and periportal thickening. Schistosomiasis can lead to chronic inflammation, periportal fibrosis, and subsequent portal hypertension, contributing to hepatosplenomegaly. Subsequently, colonoscopy showed 1 cm×1 cm sessile rectal polyp with surrounding mucosa showing whitish lesion (Figure 1a and b; https://files.fm/u/pxaf8cbtv2).
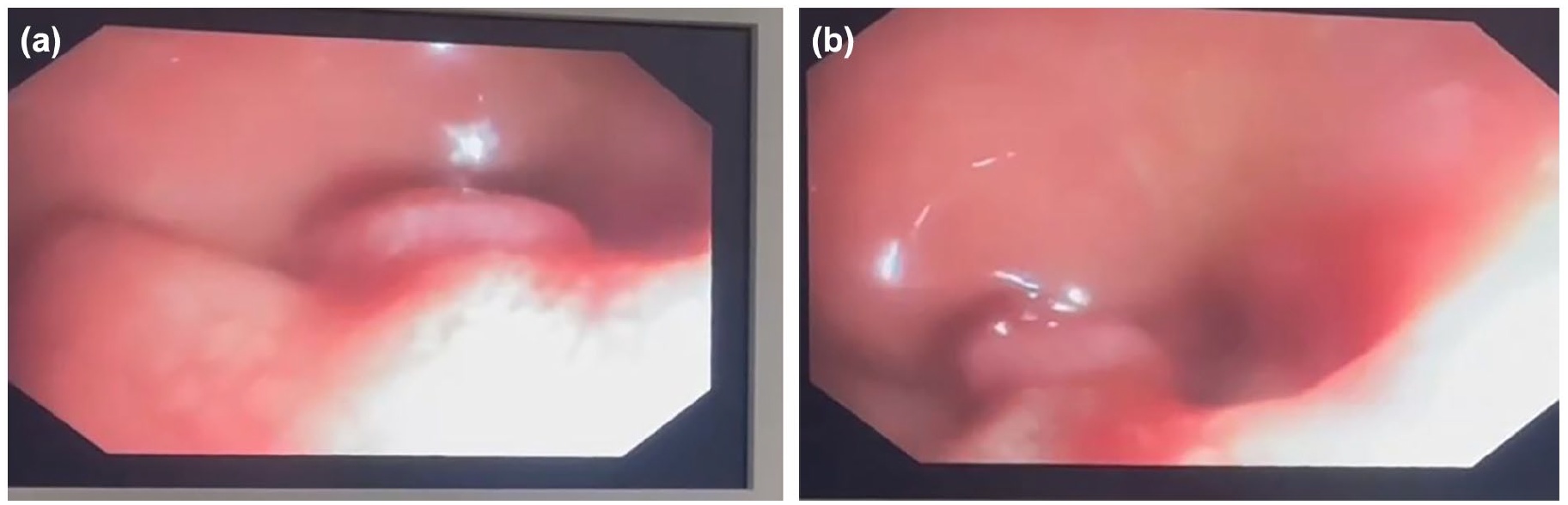
(a and b) Colonoscopy showing 1 cm × 1 cm sessile rectal polyp.
Therefore, polypectomy was done and the sample was subjected for histopathologic analysis. Additionally, supportive measures were given. Histopathologic analysis showed rectal mucosa in polypoid configuration, with surface erosion, crypt distortion / dilatation, and expanded lamina propria with neutrophilic and lymphoplasmacytic inflammation (Figure 2a-c). Finally the patient discharged from the hospital improved and appointed at pediatric chronic follow-up, and didn’t have any recurrence of symptoms on subsequent followup.

(a) (10×): colonoscopy biopsy (10×) showing rectal mucosa in polypoid configuration, with surface erosion, crypt distortion / dilatation and expanded lamina propria with neutrophilic and lymphoplasmacytic inflammation. (b) (20×): colonoscopy biopsy showing rectal mucosa in polypoid configuration, with surface erosion, crypt distortion/dilatation, and expanded lamina propria with neutrophilic and lymphoplasmacytic inflammation. (c) (40×): colonoscopy biopsy showing rectal mucosa with expanded lamina propria with neutrophilic and lymphoplasmacytic inflammation, dilated crypts, and reactive stromal cells.
Discussion
Rectal bleeding in children can arise from various etiologies, including infectious colitis, hemorrhoids, inflammatory bowel disease, and neoplastic lesions. While inflammatory cap polyps are rare, other uncommon causes of rectal bleeding include rectal mucosa-associated lymphoid tissue (MALT) lymphoma and juvenile polyps. MALT lymphoma of the rectum presents with gastrointestinal bleeding and can be identified through colonoscopy and biopsy.10 -12 Juvenile polyps, though benign, may cause significant rectal bleeding and require endoscopic removal. 10 Given the wide range of possible causes, careful diagnostic evaluation is crucial for effective management. Our case underscores the need to consider inflammatory cap polyps in the differential diagnosis of pediatric rectal bleeding and highlights the importance of colonoscopy for accurate diagnosis and treatment.
Colorectal polyps predominantly occur in the left side of the colon, especially in the rectum. However, they can occur in any segment of the colon. It has been noted that polyp formation initiates in the rectum and gradually move upward. Around 36% of individuals with ICP exhibit detectable rectal polyps during digital rectal examination. 13 However, the digital rectal examination in our patient was unremarkable. Proximal polyps generally are smaller in size compared to distal polyps. 14
Polyps have the potential to grow many centimeters in size, particularly in the rectum. Visual examination during colonoscopy can reveal polyps that appear flat, or may have a flat top and protrude as shown in this particular patient. The polyps may also resemble plaques due to the accumulation of multiple polyps. It is less common for the polyps to be pedunculated or semi-pedunculated. Typically, the polyps adhere to the colonic folds and are covered with a whitish fibrin cap. Removing the fibrin cap exposes a friable, dark red surface.14,15
ICP may manifest at any stage of life, with a higher prevalence among females. In most cases, individuals lack a familial history of polyposis. While ICP is occasionally asymptomatic, 16 typical presentations include bleeding per rectum (82%), mucoid type diarrhea (46%), and constipation or chronic straining. 13 A study from Singapore reported rectal bleeding in all patients of pediatric age group. 17 Mucoid diarrhea can be copious, causing incontinence and protein-losing enteropathy. This condition presents with loss of weight, deficiency of fat-soluble vitamin, and hypoalbuminemia, which can manifest as pitting edema in the lower extremities. Prolonged bloody diarrhea may result in anemia. Our patient presented with isolated rectal bleeding. 18
The quantity of polyps ranges from one to several hundred and is normally situated at the mucosal fold’s apex. These polyps exhibit diverse shapes, including flat, ulcerative or polypoid forms. The mucosa in between has been characterized as either normal or dotted with white spots. 14
The cap polyp has some distinct histological features: it shows elongated crypts, with the upper third expanding into cup-shaped or cystic structures. You’ll also notice thinning or erosion of the surface epithelium. On most polyps, there’s a cap over the eroded area made up of mucus, inflammatory cells, sloughed epithelial cells, and fibrin. In this patient histopathology showed rectal mucosa in polypoid configuration, with surface erosion, crypt distortion/dilatation, and expanded lamina propria with neutrophilic and lymphoplasmacytic inflammation. Only the larger polyps showed smooth muscle fibers extending from the muscularis mucose into the epithelium, while the muscularis mucose in the surrounding colon stayed intact. In rare instances, colitis cystica profunda can occur alongside the polyps or in areas of the colon that don’t have polyps. 6
Although sharing overlapping histologic features with mucosal prolapse, ICP should not be misdiagnosed as such. Given ICP’s similarity to other entities, it is often mistaken for polyposis syndromes, including Serrated Polyposis Syndrome (SPS), Cronkhite-Canada Syndrome (CCS), Juvenile Polyposis Syndrome (JPS), Cowden Syndrome (CS), or Inflammatory Bowel Disease (IBD) accompanied by pseudopolyps. ICP can be differentiated from SPS specifically, as most of the lesions in SPS are sessile serrated polyps (SSP). 19
During colonoscopy, polyps are often seen as flat, protruding, or flat-topped formations that may resemble plaques due to the aggregation of multiple polyps. More rarely, they may be pedunculated or semi-pedunculated. These polyps tend to ride over the colonic folds and are often covered by a whitish fibrin cap as shown in the endoscopic finding. Removing this cap reveals a friable surface with a dark red hue. The mucosal area between polyps is generally normal, although it may sometimes have a few white specks. A less severe version of the condition has been noted, showing red and swollen patches on the mucous membranes spread across the colon.14,15,20,21
Some ICP cases just get better on their own. Management guidelines for cap polyposis are lacking, and optimal ICP therapy for most patients is still undetermined. Some folks think we should treat ICP like a general reaction to an infection. Oiya et al 9 recently reported ICP resolution after eradicating H. pylori, This observation has been replicated in various case reports and small series, primarily in Japan.15,22 Shimizu et al 8 postulated that ICP might arise from Escherichia coli (E. coli) infection. An alternative hypothesis is that another infectious agent susceptible to H. pylori antibiotic regimens contributes to ICP. Despite antibiotic treatment, complete resolution of ICP has not been consistently observed, and recurrence after initial remission has been reported. 23 Reports have emerged regarding the utilization of metronidazole in the management of cap polyposis with the aim of mitigating inflammation. The observed response of ICP to metronidazole administration potentially stems from its anti-inflammatory properties rather than its antimicrobial effects. 8
ICP is treatable with therapies that address prolapse (eg, avoiding straining)), 18 steroids, 23 infliximab, 24 endoscopic or surgical resection.3,25 While corticosteroid treatment (such as, 40 mg/day of prednisolone initially) has shown to resolve polyps and symptoms within 1-2 months, early relapse is common. 23 Infliximab, a TNF alpha inhibitor, demonstrated significant efficacy after either 1 injection or 4 infusions over 8 weeks in numerous patients.24,26 However, treatment failures have been reported, characterized by the absence of infliximab’s inhibitory effect on TNF alpha levels in the tissue. 27 However, the small number of patients treated and the unclear natural history of ICP make it tough for these reports to identify which patients might resist therapy.
In practice, the first step in treatment is all about relieving symptoms to ease any excessive straining and tackle constipation. For patients like this one, a polypectomy is usually suggested. If someone tests positive for H. pylori and still has symptoms, we might think about eradicating the bacteria. After that, treatment could involve anti-inflammatory medications like steroids or infliximab, with the objective attaining remission and steering clear of surgery. Surgery is an option if these therapies fail. In patients who undergo localized resection of the diseased area, up to 37% experience recurrence of ICP in the colon. 8
To sum it up, ICP is still a condition we don’t fully understand, and we’re still in the dark about its natural progression and clear treatment guidelines. Colonoscopy with polypectomy is a readily available treatment option, considering the common location of CAP in the rectum. For patients dealing with recurring CAP polyposis who aren’t improving with medications, surgery and polypectomy are the main management principles. 28
Conclusion
Inflammatory cap polyps of the rectum are distinct clinical entities demanding meticulous assessment and management. Despite their relative rarity, they pose considerable health risks, necessitating timely identification and suitable intervention. This patient presented with lower abdominal pain and rectal bleeding. Subsequently, he was diagnosed with ICP and managed with polypectomy. Ongoing research endeavors aim to elucidate the pathogenesis, natural evolution, and optimal treatment approaches for inflammatory cap polyps, ultimately enhancing outcomes for afflicted patients.
Footnotes
Abbreviations
IBD: Inflammatory Bowel Disease
ICP: Inflammatory cap polyp
Ethical Considerations
The institution review board does not require ethical clearance for reporting case report.
Consent for Publication
Written informed consent was obtained from the parent for the publication of this case report.
Author Contributions
The case report was conceived, written, and edited by all writers. It is agreed that each author is responsible for every part of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Any other details on the case report are welcome to be provided by the manuscript's authors.
